Abstract
Hepatocellular carcinoma (HCC) is a global problem constituting the second leading cause of cancer deaths worldwide, thereby necessitating an accurate and cost-effective solution for managing care. Ultrasound is well poised to address this need due to its low cost, portability, safety, and excellent temporal resolution. The role of ultrasound for HCC screening has been well established and supported by multiple international guidelines. Similarly, contrast-enhanced ultrasound (CEUS) can be used for the characterization of focal liver lesions in high-risk populations, and standardized criteria for CEUS have been established by the American College of Radiology Liver Imaging Reporting & Data System (LI-RADS). Following HCC identification, CEUS can also be highly beneficial in treatment planning, delivery, and monitoring HCC response to locoregional therapies. Specific advantages of CEUS include providing real-time treatment guidance and improved diagnostic performance for the detection of residual tumor viability or recurrence, thereby identifying patients in need of retreatment substantially earlier than contrast-enhanced CT and MRI. This review provides a primer on ultrasound and CEUS for the screening and characterization of HCC, with an emphasis on assessing tumor response to locoregional therapies.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Ultrasound plays a central role in HCC (hepatocellular carcinoma) screening in at-risk populations. It is supported by the majority of liver societies including the American Association for the Study of Liver Diseases (AASLD) and the ACR appropriateness criteria [1]. With the guidance of the Ultrasound Liver Imaging and Reporting Data System (US LI-RADS), there are now specific recommendations for the technical performance of the exam and the reporting of findings which are connected to management recommendations [2]. Once a focal lesion is detected in an at-risk patient, CEUS can then be used to characterize the lesion and confirm HCC. Similarly, CEUS LI-RADS provides guidance on enhancement patterns and contrast kinetics for the characterization of HCC and other liver observations [2].
Following locoregional treatment of HCC, gray-scale ultrasound can provide information limited to size and morphology of tumor, with Doppler ultrasound and its variations (power Doppler, microvascular perfusion, etc.) providing some clue as to tumor viability. However, the use of CEUS places ultrasound on a similar footing as CT/MR and allows for assessment of tumor viability and treatment response via contrast enhancement. In this way, ultrasound may provide “one-stop” shopping for HCC detection, characterization, and treatment response assessment in a non-invasive, accessible, and cost-effective way with a low-risk profile for allergies, nephrotoxicity, and other issues associated with CT/MRI contrast agents.
This paper will briefly explore the role of ultrasound in focal liver observation detection and characterization. The focus of the paper, however, will be on the use of CEUS in locoregional treatment response. Principles of ultrasound contrast enhancement and differences between CEUS and MR/CT in assessing locoregional treatment response such as percutaneous ablation, transarterial chemoembolization, and radioembolization will be discussed.
The role of ultrasound and CEUS for HCC screening and characterization
HCC is a global problem constituting the second leading cause of cancer deaths worldwide, thereby, necessitating an accurate and cost-effective solution for detection of such a deadly disease [3]. Furthermore, HCC is the fastest-rising cause of cancer mortality in the United States [4]. Fortunately, HCC is a disease that can be positively impacted by screening and surveillance, fulfilling criteria put forth by the World Health Organization [4]. Target at-risk screening populations exist consisting of people with cirrhosis of any cause, some non-cirrhotic patients with hepatitis B, and patients with current or prior history of HCC. Additionally, the trajectory of disease severity can be halted with early detection. A screening modality with reasonable accuracy that is accessible and cost-effective exists in ultrasound. Most importantly, however, ultrasound is the only modality that has been shown to reduce disease-specific mortality in large-scale randomized controlled trials [5]. Notably, the largest randomized controlled trial studied 18,816 patients with hepatitis B virus or chronic hepatitis in Shanghai and found that screening and surveillance with ultrasound and serum alpha-fetoprotein every six months reduced mortality by 37% despite poor patient compliance [6]. Similar reductions have been seen in other trials [7]. Based on this and other data, many of the liver societies, including the American Association for the Study of Liver Diseases (AASLD), European Study of Liver Diseases (EASLD), and Asian Pacific Association for the Study of the Liver (APASL), advocate ultrasound for HCC screening and surveillance in at-risk patients as a clinically proven means of reducing mortality in an accessible and cost-effective way. Indeed, MRI and CT can be used for HCC screening as well, particularly in select high-risk patients who may have limitations in screening with ultrasound. However, MRI and CT HCC screening have not been proven to reduce mortality in randomized controlled trials and may not be global solutions due to specificity, cost, and accessibility.
Although ultrasound has been advocated for HCC screening, no standardized or recommended means of performing or reporting the exams previously existed until recently. In 2014, US LI-RADS was created to provide a standardized lexicon, technical recommendations, and a reporting guide for findings, which are tied to management recommendations (Fig. 1 [8]). The technical recommendations of US LI-RADS provide suggestions for equipment and patient parameters as well as a list of suggested sonographic windows, imaging techniques, and images to assure complete coverage of the liver. These recommendations attempt to increase the quality and reproducibility of the exam and ultimately, the accuracy of ultrasound screening and surveillance. Once the exam is performed, US LI-RADS provides a category score of 1, 2, or 3 reflecting findings (termed observations) and suggested management. A category 1 exam contains no observations suspicious for HCC and places the patient at routine screening in six months. Category 2 exams have a subthreshold observation, usually less than 1 cm for which a 3–6-month follow-up exam is suggested, while category 3 exams have an observation for which a contrast-enhanced exam (either CEUS, CT, or MRI) is required for further evaluation. LI-RADS also provides a visualization score meant to convey the expected sensitivity of ultrasound for visualization of the liver and detection of HCC. Here, visualization score A is an exam with little limitation, B is moderate limitation, and C is severe limitation for the detection of HCC (Fig. 1. Category scores 1,2,3 and visualization scores A,B,C). Examples of these classifications are provided in Fig. 2. In this way, exams are technically optimized for HCC detection, exams are assessed for the adequacy of visualization of HCC, and ultimate findings are connected to further management.

Reproduced with permission from the American College of Radiology (ACR) [8]
American College of Radiology US LI-RADS criteria describing reporting criteria for HCC screening in high-risk populations.
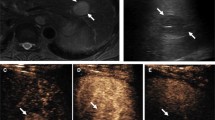
US LI-RADS Category and Visualization Scores. Figure 2A–D Category Scores. a US-1 Negative: No ultrasound evidence of HCC. Here, a benign cyst is shown with the arrow. b US-2 Subthreshold. Observation is detected that may warrant short-term ultrasound surveillance. The arrow points to a subcentimeter echogenic observation only seen on the high-resolution linear array surface images. c, d Positive: Observation is detected that may warrant multiphase contrast-enhanced imaging. Here, a solid hypoechoic mass is detected on screening ultrasound marked by the arrow. Figure 2e–g Visualization Scores. e Visualization A has no or minimal limitations. f Visualization B has moderate limitations due to extensive coarsened echotexture from cirrhosis. g Visualization C has severe limitations due to a very limited sonographic window
Once a focal liver lesion is detected by ultrasound, it can be successfully characterized by CEUS. Ultrasound contrast agents consist of gas bubbles of different composition typically stabilized by a phospholipid or protein envelope that is injected intravenously, small enough to transverse the pulmonary vasculature, thereby reaching the target organ vasculature. Ultrasound contrast agents differ from CT/MR agents in that they are true intravascular contrast agents without deposition into the extravascular space. This has implications in the enhancement patterns of some lesions, most notably cholangiocarcinoma. Additionally, many benign masses such as hemangiomas can be conclusively identified by CEUS, thereby obviating the need to biopsy [9]. The CEUS characteristics of various lesions are comprehensively described in CEUS LI-RADS, a guideline which strives to provide guidance for technical performance, interpretation, and reporting of CEUS exams (Fig. 3 [9]). In CEUS, lesion diagnosis is based not only on the morphologic appearance of contrast enhancement but also on the contrast kinetics. HCC typically will appear as hyper-enhancing in the early arterial phase, with mild washout that occurs later and less profoundly than is seen with other malignancies such as metastases or cholangiocarcinoma (See Fig. 4 for example). Several studies have validated the use of CEUS in HCC characterization. A meta-analysis of 53 studies from 1998–2016 by Zhang, et al. shows a pooled sensitivity of 85% and a specificity of 91% [10]. A recent study by Huang, et al. concentrating on small HCCs less than 2.0 cm showed a sensitivity of 73.3% and a specificity of 97.1%, which compares favorably to sensitivities and specificity of MRI in HCC characterization [11]. Consequently, while ultrasound is a favorable tool for the identification of suspicious masses in high-risk populations, CEUS can be used for the characterization and identification of HCC.

Reproduced with permission from the ACR [9]
American College of Radiology CEUS LI-RADS criteria describing reporting criteria for the characterization of focal liver lesions in high-risk populations.
Example images showing the use of CEUS LI-RADS. (Left) B-mode image demonstrating an indeterminate liver lesion (arrows). (Center) CEUS shows hyperenhancement in the mass (arrows) relative to the adjacent liver during the arterial phase. (Right) Late and mild washout is observed within the mass (arrow) in the portal phase. Using CEUS LI-RADS criteria, this mass is classified as CEUS LR-5
CEUS assessment of response to locoregional therapy
Following identification and characterization, a variety of locoregional therapies are available for the treatment of HCC and can be used for palliative care, locoregional tumor control, or bridge to transplant. While the level of established evidence varies by therapy, CEUS has inherent properties that can be advantageous relative to CE-MRI/CT for monitoring response to locoregional therapy. The portability and real-time nature of CEUS enable treatment guidance at the patient bedside, and CEUS can provide real-time feedback on tumor viability. Lack of concerns with ionizing radiation and renal contrast contraindications combined with low cost make CEUS a practical alternative worldwide as described above. Finally, the higher temporal resolution and limited artifacts generated by embolic material also provide the potential for earlier (within minutes to days) detection of viable tumor, resulting in much earlier retreatment, if required [12]. However, it should also be pointed out that many of the limitations of conventional ultrasound still apply to CEUS. The modality may be user and reader dependent, and both radiologist and sonographer training may vary by institution. Patient body habitus, bowel gas, and lesion size and location within the liver may limit imaging access in some patients. Finally, CEUS is not as readily available as MRI but its acceptance and availability are rapidly increasing. Despite these limitations, evidence continues to emerge on the potential benefit of CEUS for evaluating locoregional therapies. This evidence is discussed in more detail below.
Ablation
Thermal ablation is a preferred non-surgical treatment method for HCC patients with Barcelona Clinic Liver Cancer (BCLC) stage 0-A disease and for the treatment of a variety of other solid tumors in the liver [13]. It is primarily performed using microwave ablation (MWA) or radiofrequency ablation (RFA), although success has also been demonstrated with laser and cryoablation [14]. The procedure is generally performed under ultrasound or CT guidance, in which real-time feedback of the ablation cavity can be provided. There is no uniform consensus on follow-up schedules, although 1-month follow-up with CE-MRI or CT is generally used, with 3–6-month interval follow-up thereafter to monitor for tumor recurrence [15, 16].
CEUS can be successfully used to improve real-time image guidance during thermal liver ablation due to its ability to delineate real-time tumor perfusion dynamics [17, 18]. The technique was demonstrated to improve electrode placement, especially in patients with lesions that are incompletely visualized on routine gray-scale sonography, resulting in improved treatment outcomes [19]. In patients with HCC poorly imaged by gray-scale ultrasound selected for radiofrequency ablation, Minami et al. demonstrated complete tumor ablation after a single treatment in 95.2% of the patients treated using CEUS-guided ablation versus 32% in patients where the treatment was guided by routine gray-scale ultrasound alone [20]. Similar findings were presented by Yan et al. with a tumor recurrence rate of 16% in patients treated with CEUS-guided ablation compared to 48% in patients treated with ablation under routine ultrasound guidance [21]. In addition, patients treated with CEUS-guided MWA demonstrated significantly better progress-free survival [17].
For treatment assessment, the vast majority of studies with CEUS have been performed within the first week or first month after RFA. These studies have demonstrated exceptional sensitivity of CEUS, often exceeding 80% with near 100% specificity (Table 1). This diagnostic performance is generally equivalent or often better than CECT or MRI performed within the same timeline (Table 1). In addition, due to its exceptional temporal and spatial resolution and relative portability, CEUS can be used for immediate assessment of RFA treatment effect while the patients are still in the interventional suite, allowing immediate retreatment and substantial reduction in incomplete ablation rates after initial treatment [22,23,24]. In addition, CEUS performed at the end of ablation can be used to evaluate for immediate post-procedural complications, such as hemorrhage and hepatic infarction [22]. However, some limitations of CEUS may affect its diagnostic accuracy when performed immediately or within the first 30 min after the ablation. In these instances, post-ablative reactive hyperemia may mask viable enhancing tumor and its irregular profile may falsely indicate the persistence of tumor [25]. Additionally, gas bubbles produced during the ablation may obscure portions of the tumors, limiting visualization [26]. As a result, CEUS performed immediately post treatment may have sensitivity in detecting incomplete ablation as low as 33% due to difficulty in differentiating viable tumor from post-procedural reactive hyperemia and is generally not recommended for this early post-treatment [27, 28].
CEUS can also be successfully used for the detection of residual tumor in patients with both primary and metastatic liver malignancies treated with MWA. Pregler et al. examined 30 consecutive patients with an untreated HCC lesion larger than or equal to 3 cm treated with percutaneous MWA. In all patients, both CEUS and CE-MRI were performed 24 h after the ablation with 6-week post-ablation MRI used as reference standard. The sensitivities and specificities for the detection of residual tumor on day 1 were 100% and 83% for CEUS and 87% and 67% for MRI [29]. Similar findings were presented in a larger study by Peng et al. who evaluated 231 lesions treated by MWA [30]. In their study, residual tumor detection with CEUS and CE-MRI 1 month after ablation demonstrated sensitivities of 86.5% and 84.6%; specificities of 98.3% and 98.9%; and overall accuracies of 95.7% and 95.7%, respectively, with no significant statistical differences in diagnostic performance between CEUS and CE-MRI. Examples of CEUS cases from HCCs following partial and complete MWA are provided in Figs. 5 and 6, respectively.
HCC patient after MWA ablation. a Late-arterial CT image demonstrates avascular ablation cavity (*) and a vague area of increased enhancement anterior to the ablation cavity (arrow) that was interpreted as AV-shunt. b Portal-venous CT image in the same patient demonstrates area of decreased enhancement anterior to the ablation cavity (arrow) that was interpreted as post-ablation changes with no evidence of recurrent or residual tumor. On ultrasound, the ablation cavity is well visualized on B-mode (circled). c Early arterial CEUS with avascular ablation cavity (*) similar to CT and a large area of arterial phase hyperenhancement (arrow), which on portal-venous CEUS image d demonstrated early washout (arrow), consistent with incomplete ablation and large residual tumor anterior to the ablation cavity
HCC patient after MWA ablation. a Pre-contrast T1-weighted MRI image demonstrates ablation cavity (*) expected post-ablation hemorrhagic necrosis. b Contrast-enhanced MRI image demonstrates avascular ablation cavity (*) with mild peripheral enhancement (arrow). c Early arterial CEUS demonstrates completely avascular ablation cavity (*), similar to MRI, with small area of central calcification (arrow) seen on corresponding B-mode image. No areas of arterial phase hyperenhancement (APHE) were detected in the surrounding liver parenchyma. d Portal venous CEUS image demonstrates no contrast washout surrounding the ablation cavity (arrow), confirming the absence of residual untreated tumor. Please, note far superior resolution of CEUS compared to CE-MRI
Recent developments in CEUS technology allow fusion of previously obtained CT/MRI/PET-CT in which the tumor is most conspicuous, with real-time CEUS used for guidance of ablation probe/antenna placement to improve lesion visualization, procedure guidance, and post-treatment assessment. This approach has shown to be useful for improving the treatment effectiveness rate up to 98.8–100% compared to 67.7–93.5% reported in previous studies under guidance by conventional ultrasound [31,32,33,34,35,36].
Transarterial embolization
Transarterial embolization is a catheter directed therapy in which embolic material with or without chemotherapy is deposited into the tumor vasculature via the hepatic artery and its branches [37]. It is recommended for HCC patients with BCLC stage B disease but can also be used in stage A patients not amenable to ablation or in stage C patients for downstaging or palliative care [38]. It is also used for patients with other primary and metastatic liver tumors. Treatment response is currently evaluated using CECT or CE-MRI 4 to 6 weeks post treatment [39]. However, CEUS has demonstrated several clear advantages over CT and MRI approaches and has been extensively validated in multiple clinical trials (Table 2).
Contrast-enhanced CT and MR imaging is generally obtained only after 4 weeks post transarterial chemoembolization (TACE) to reduce arterial phase hyperenhancement associated with post-ablation inflammation and hyperemia within the tumor and in the surrounding liver parenchyma on both CT and MRI and lipiodol artifacts on CT [40,41,42]. This typically results in a 6–8-week delay of retreatment in patients with incomplete ablation. However, initial TACE data demonstrated a return-to-normal liver function approximately 7 days post treatment [43], indicating retreatment via repeat TACE or percutaneous ablation is permissible much earlier. Unlike CT and MRI, CEUS demonstrates very minimal artifacts from lipiodol deposition and is able to reliably differentiate viable tumor from post-procedural inflammation. As a result, many studies have demonstrated an ability of CEUS to detect viable tumor substantially sooner than the current 4–6 weeks required to assess TACE treatment response with CT and MRI (Table 2).
Protocols for CEUS of TACE treatment response evaluation typically follow similar guidelines to those recommended for indeterminate liver imaging, as in CEUS LI-RADS. Continuous imaging is used with sonographic focus on looking for residual tumor enhancement during the arterial enhancement phase. Volumetric (3D and 4D) CEUS has been explored for locoregional embolization follow-up, but currently only recommended for research purposes. In liver masses completely treated by TACE, the treatment cavity should demonstrate well-defined margins on CEUS with no internal flow on any enhancement phase and no evidence of nodular peripheral enhancement.
Follow-up times range from days to up to two weeks and demonstrate similar diagnostic performance to CT and MRI imaging at 1 month [44,45,46,47,48,49,50]. Examples of CEUS 1–2 weeks post treatment in cases with and without viable tumor are presented in Figs. 7 and 8, respectively. Concordant findings are observed with CE-MRI 4–6 months post treatment, demonstrating how treatment planning can be initiated earlier using CEUS.
HCC patient after TACE. (Top): CEUS 1 week post treatment demonstrates the tumor (circled on B-mode) with peripheral nodular enhancement (arrow) in the arterial phase, suggesting the presence of viable tumor. (Bottom) Similar findings are demonstrated on CE-MRI 1 month post treatment, indicating a need for retreatment. These findings illustrate how retreatment can potentially be performed earlier when using CEUS for residual detection
HCC patient after TACE. (Top) CEUS 1 week post treatment demonstrates the tumor (circled on B-mode) with no internal enhancement (arrow) in the arterial phase with smooth margins indicating complete tumor embolization. (Bottom) Similar findings are demonstrated on CE-MRI 1 month post treatment, indicating the absence of viable tumor (arrow). These findings illustrate how patient management can be established earlier using CEUS as compared to MRI
Additionally, the improved temporal resolution of CEUS relative to CT and MRI allows detection of smaller areas of viable tumor not appreciated by other imaging modalities. While the specificity of CEUS is variable, several studies have demonstrated superior sensitivity of CEUS for detecting viable tumor relative to contrast-enhanced cross-sectional imaging [46, 50,51,52]. Volumetric and quantitative approaches using 3D and 4D CEUS may further improve this diagnostic performance, although these are still in their infancy [53, 54]. An example of CEUS acquired 1 month post treatment and the accompanying CE-MRI are provided in Fig. 9. Residual enhancement is evident within the tumor on CEUS, but not clearly appreciated on CE-MRI. Viable tumor in this case was later confirmed by tumor growth and retreatment angiography. While this example demonstrates clear clinical benefit, larger multi-center trials with blinded readers are still needed to directly compare modalities and are currently underway (see for example NCT# 02,764,801).
HCC patient after TACE. a CEUS 1 month post treatment demonstrates clear internal enhancement within the tumor (circled on B-mode) in the arterial phase and irregular enhancing margins on the right border of the treatment cavity (arrow), indicating viable tumor. b CE-MRI at 1 month demonstrates no clear evidence of residual tumor viability within the treatment cavity (arrow). Tumor viability was later confirmed by tumor growth and viable vascularity on angiography during retreatment. These findings show how CEUS can improve the diagnostic performance of MRI for monitoring response of HCC to TACE
The ability of CEUS to aid in TACE treatment planning has also been demonstrated. Advanced processing approaches using CEUS prior to treatment have been shown to be useful for predicting the likelihood of treatment response [55, 56]. These works employ artificial intelligence and characterization of vascular networks to identify tumors likely to fully take up and retain embolic material [57, 58]. Intra-procedural CEUS has also been used with arterial injection through the treating catheter do identify residual blood flow post deployment of the embolic material [57,58,59]. Finally, CEUS follow-up of previously treated lesions has also been used to identify the specific feeding vessels for aiding in retreatment planning, demonstrating good (85%) correlation with angiography findings at retreatment [60].
Radioembolization
Transarterial radioembolization (TARE) is recommended for patients with BCLC stage B disease but is also frequently used in patients with stage C disease at many institutions [61]. Aside from HCC, the technique has also been shown to be effective for other primary and metastatic liver tumors [62, 63]. This approach involves arterial injection of (20–40 µm) glass beads containing yttrium-90 (Y-90) into the tumor blood supply. The radioisotope undergoes a pure beta emission as it decays to stable Zirconium-90 with a half-life of 64 h [64]. Dosages vary by institutional protocols, tumor size, and treatment planning. Traditional dosages range from approximately 110–150 Gy [65], with reported response rates ranging from 25–80% [65,66,67,68]. However, recently the safety and benefits of radiation segmentectomy, in which one to two segments are treated with dosages > 190 Gy, have been demonstrated in patients with BCLC stages A-B disease [69, 70].
Treatment response of HCC to TARE is generally assessed using contrast-enhanced MRI. Follow-up times vary by institution, but generally are greater than 3 months after treatment to enable reduction of post-treatment inflammation and improve radiotherapy response assessment [71]. The use of LI-RADS for treatment response has been proposed but has yet to be fully validated, with early data demonstrating a propensity for equivocal findings [72]. The use of mRECIST has shown moderate interobserver (ĸ = 0.56) agreement, but this decreases in cases with heterogeneous enhancement prior to treatment [73]. These limitations in follow-up time, definitive diagnosis, and imperfect correlation to pathologic necrosis have generated interest in using CEUS to monitor TARE response.
Limited data on the use of CEUS to monitor response to TARE currently exist [12]. The ability of CEUS to monitor changes in tumoral perfusion following TARE has been noted as early as one week post treatment [74] with changes in tumoral vascularity and perfusion dynamics correlating well with longer term mRECIST CT or MRI findings 3–4 months post treatment [75]. An example of CEUS post TARE is provided in Fig. 10. Notice that the areas of patchy enhancement are clearly visible on CEUS, indicating viable tissue within the treatment cavity. Given the advantages in temporal resolution, cost, and accessibility described above, CEUS is well poised as a modality for monitoring HCC response to TARE. However, larger studies investigating diagnostic performance with longer patient follow-up time are still needed before clinical adoption is justified.
Incomplete HCC treatment after TARE. a pretreatment contrast-enhanced MRI shows a 6.8 cm HCC (circled) in segment 6/7. b MRI 4 months post TARE demonstrates patchy arterial phase contrast enhancement (arrow) within the treated HCC. c CEUS 4 months post TARE using dual imaging showing the tumor (circled) on B-mode and patchy arterial phase contrast enhancement within the viable residual tumor (arrow) with much greater resolution compared to MRI
Conclusions
The role of ultrasound in screening and CEUS characterization of HCC in high-risk populations is well established. Additionally, the inherent properties of CEUS (cost, safety, portability, and high temporal resolution) make it well poised as a first-line imaging modality for monitoring HCC response to locoregional therapy, providing assessment that can be performed within minutes to days after treatment, substantially sooner than with CECT and MRI and with similar if not potentially better accuracy.
References
Horowitz JM, Kamel IR, Arif-Tiwari H, Asrani SK, Hindman NM, Kaur H, McNamara MM, Noto RB, Qayyum A, Lalani T (2017) ACR Appropriateness Criteria(®) Chronic Liver Disease. J Am Coll Radiol 14:S391-s405. https://doi.org/10.1016/j.jacr.2017.08.045
American College of Radiology Committee on LI-RADS® (2018) CT/MRI LI-RADS® v2018 CORE. https://www.acr.org/-/media/ACR/Files/RADS/LI-RADS/LI-RADS-2018-Core.pdf?%20la=en. Accessed 01 January 2021
Ferlay J, Soerjomataram I, Ervik M, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F (2012) GLOBOCAN 2012: Estimated Cancer Incidence, Mortality and Prevalence Worldwide in 2012. International Agency for Research on Cancer. globocan.iarc.fr. Accessed 01 January 2021
National Cancer Institute Surveillance, Epidemiology, and End Results (SEER) Program (2009) SEER*Stat Database: Incidence-SEER Research Data 9 Registries. Accessed 01 January 2021
Wilson JM, Jungner YG (1968) Principles and practices of screening for disease. World Health Organization. https://apps.who.int/iris/handle/10665/37650. Accessed 01 January 2021
Zhang BH, Yang BH, Tang ZY (2004) Randomized controlled trial of screening for hepatocellular carcinoma. J Cancer Res Clin Oncol 130:417-22. https://doi.org/10.1007/s00432-004-0552-0
Yang B, Zhang B, Xu Y, Wang W, Shen Y, Zhang A, Xu Z (1997) Prospective study of early detection for primary liver cancer. J Cancer Res Clin Oncol 123:357-60. https://doi.org/10.1007/BF01438313
American College of Radiology Committee on LI-RADS® (2017) Ultrasound LI-RADS® v2017. https://www.acr.org/Clinical-Resources/Reporting-and-Data-Systems/LI-RADS/Ultrasound-LI-RADS-v2017. Accessed 15 January 2021
American College of Radiology Committee on LI-RADS® (2017) CEUS LI-RADS® v2017. https://www.acr.org/Clinical-Resources/Reporting-and-Data-Systems/LI-RADS/CEUS-LI-RADS-v2017. Accessed 15 January 2021
Zhang J, Yu Y, Li Y, Wei L (2017) Diagnostic value of contrast-enhanced ultrasound in hepatocellular carcinoma: a meta-analysis with evidence from 1998 to 2016. Oncotarget 8:75418-75426. https://doi.org/10.18632/oncotarget.20049
Huang JY, Li JW, Lu Q, Luo Y, Lin L, Shi YJ, Li T, Liu JB, Lyshchik A (2020) Diagnostic Accuracy of CEUS LI-RADS for the Characterization of Liver Nodules 20 mm or Smaller in Patients at Risk for Hepatocellular Carcinoma. Radiology 294:329-339. https://doi.org/10.1148/radiol.2019191086
Lyshchik A (2019) Specialty Imaging: Fundamentals of CEUS. Elsevier, Philadelphia
Forner A, Reig M, Bruix J (2018) Hepatocellular carcinoma. Lancet 391:1301-1314. https://doi.org/10.1016/S0140-6736(18)30010-2
Head HW, Dodd GD, 3rd (2004) Thermal ablation for hepatocellular carcinoma. Gastroenterology 127:S167-78. https://doi.org/10.1053/j.gastro.2004.09.031
Sala M, Llovet JM, Vilana R, Bianchi L, Solé M, Ayuso C, Brú C, Bruix J (2004) Initial response to percutaneous ablation predicts survival in patients with hepatocellular carcinoma. Hepatology 40:1352-60. https://doi.org/10.1002/hep.20465
Ayuso C, Rimola J, García-Criado A (2012) Imaging of HCC. Abdom Imaging 37:215-30. https://doi.org/10.1007/s00261-011-9794-x
Madsen HH, Rasmussen F (2011) Contrast-enhanced ultrasound in oncology. Cancer Imaging 11 Spec No A:S167–73. https://doi.org/10.1102/1470-7330.2011.9042
Piscaglia F, Nolsøe C, Dietrich CF, Cosgrove DO, Gilja OH, Bachmann Nielsen M, Albrecht T, Barozzi L, Bertolotto M, Catalano O, Claudon M, Clevert DA, Correas JM, D'Onofrio M, Drudi FM, Eyding J, Giovannini M, Hocke M, Ignee A, Jung EM, Klauser AS, Lassau N, Leen E, Mathis G, Saftoiu A, Seidel G, Sidhu PS, ter Haar G, Timmerman D, Weskott HP (2012) The EFSUMB Guidelines and Recommendations on the Clinical Practice of Contrast Enhanced Ultrasound (CEUS): update 2011 on non-hepatic applications. Ultraschall Med 33:33-59. https://doi.org/10.1055/s-0031-1281676
Huang DY, Yusuf GT, Daneshi M, Husainy MA, Ramnarine R, Sellars ME, Sidhu PS (2017) Contrast-enhanced US-guided Interventions: Improving Success Rate and Avoiding Complications Using US Contrast Agents. Radiographics 37:652-664. https://doi.org/10.1148/rg.2017160123
Minami Y, Kudo M, Kawasaki T, Chung H, Ogawa C, Shiozaki H (2004) Treatment of hepatocellular carcinoma with percutaneous radiofrequency ablation: usefulness of contrast harmonic sonography for lesions poorly defined with B-mode sonography. AJR Am J Roentgenol 183:153-6. https://doi.org/10.2214/ajr.183.1.1830153
Yan SY, Zhang Y, Sun C, Cao HX, Li GM, Wang YQ, Fan JG (2016) Comparison of real-time contrast-enhanced ultrasonography and standard ultrasonography in liver cancer microwave ablation. Exp Ther Med 12:1345-1348. https://doi.org/10.3892/etm.2016.3448
Chen MH, Yang W, Yan K, Zou MW, Solbiati L, Liu JB, Dai Y (2004) Large liver tumors: protocol for radiofrequency ablation and its clinical application in 110 patients--mathematic model, overlapping mode, and electrode placement process. Radiology 232:260-71. https://doi.org/10.1148/radiol.2321030821
Mauri G, Porazzi E, Cova L, Restelli U, Tondolo T, Bonfanti M, Cerri A, Ierace T, Croce D, Solbiati L (2014) Intraprocedural contrast-enhanced ultrasound (CEUS) in liver percutaneous radiofrequency ablation: clinical impact and health technology assessment. Insights Imaging 5:209-16. https://doi.org/10.1007/s13244-014-0315-7
Zhao X, Wang W, Zhang S, Liu J, Zhang F, Ji C, Li X, Gan W, Zhang G, Guo H (2012) Improved outcome of percutaneous radiofrequency ablation in renal cell carcinoma: a retrospective study of intraoperative contrast-enhanced ultrasonography in 73 patients. Abdom Imaging 37:885-91. https://doi.org/10.1007/s00261-011-9828-4
Francica G, Meloni MF, Riccardi L, de Sio I, Terracciano F, Caturelli E, Iadevaia MD, Amoruso A, Roselli P, Chiang J, Scaglione M, Pompili M (2018) Ablation treatment of primary and secondary liver tumors under contrast-enhanced ultrasound guidance in field practice of interventional ultrasound centers. A multicenter study. Eur J Radiol 105:96-101. https://doi.org/10.1016/j.ejrad.2018.05.030
Meloni MF, Andreano A, Zimbaro F, Lava M, Lazzaroni S, Sironi S (2012) Contrast enhanced ultrasound: Roles in immediate post-procedural and 24-h evaluation of the effectiveness of thermal ablation of liver tumors. J Ultrasound 15:207-14. https://doi.org/10.1016/j.jus.2012.09.001
Roccarina D, Garcovich M, Ainora ME, Riccardi L, Pompili M, Gasbarrini A, Zocco MA (2015) Usefulness of contrast enhanced ultrasound in monitoring therapeutic response after hepatocellular carcinoma treatment. World J Hepatol 7:1866-74. https://doi.org/10.4254/wjh.v7.i14.1866
Du J, Li HL, Zhai B, Chang S, Li FH (2015) Radiofrequency ablation for hepatocellular carcinoma: utility of conventional ultrasound and contrast-enhanced ultrasound in guiding and assessing early therapeutic response and short-term follow-up results. Ultrasound Med Biol 41:2400-11. https://doi.org/10.1016/j.ultrasmedbio.2015.05.004
Pregler B, Beyer LP, Wiesinger I, Nießen C, Jung EM, Stroszczynski C, Wiggermann P (2016) Microwave ablation of large HCC lesions: Added value of CEUS examinations for ablation success control. Clin Hemorheol Microcirc 64:483-490. https://doi.org/10.3233/CH-168113
Qu P, Yu X, Liang P, Cheng Z, Han Z, Liu F, Yu J (2013) Contrast-enhanced ultrasound in the characterization of hepatocellular carcinomas treated by ablation: comparison with contrast-enhanced magnetic resonance imaging. Ultrasound Med Biol 39:1571-9. https://doi.org/10.1016/j.ultrasmedbio.2013.03.030
Monsky W, Keravnou C, Averkiou M (2019) Contrast-enhanced ultrasound to ultrasound fusion during microwave ablation: feasibility study in a perfused porcine liver model. J Ultrasound 22:323-335. https://doi.org/10.1007/s40477-019-00366-5
Park HJ, Lee MW, Rhim H, Cha DI, Kang TW, Lim S, Song KD, Lim HK (2015) Percutaneous ultrasonography-guided radiofrequency ablation of hepatocellular carcinomas: usefulness of image fusion with three-dimensional ultrasonography. Clin Radiol 70:387-94. https://doi.org/10.1016/j.crad.2014.12.003
Xu EJ, Lv SM, Li K, Long YL, Zeng QJ, Su ZZ, Zheng RQ (2018) Immediate evaluation and guidance of liver cancer thermal ablation by three-dimensional ultrasound/contrast-enhanced ultrasound fusion imaging. Int J Hyperthermia 34:870-876. https://doi.org/10.1080/02656736.2017.1373306
Mauri G, Gennaro N, De Beni S, Ierace T, Goldberg SN, Rodari M, Solbiati LA (2019) Real-Time US-(18)FDG-PET/CT Image Fusion for Guidance of Thermal Ablation of (18)FDG-PET-Positive Liver Metastases: The Added Value of Contrast Enhancement. Cardiovasc Intervent Radiol 42:60-68.https://doi.org/10.1007/s00270-018-2082-1
Clevert DA, Paprottka PM, Helck A, Reiser M, Trumm CG (2012) Image fusion in the management of thermal tumor ablation of the liver. Clin Hemorheol Microcirc 52:205-16.https://doi.org/10.3233/CH-2012-1598
Turtulici G, Orlandi D, Corazza A, Sartoris R, Derchi LE, Silvestri E, Baek JH (2014) Percutaneous radiofrequency ablation of benign thyroid nodules assisted by a virtual needle tracking system. Ultrasound Med Biol 40:1447-52. https://doi.org/10.1016/j.ultrasmedbio.2014.02.017
Gonsalves CF, Brown DB (2009) Chemoembolization of hepatic malignancy. Abdom Imaging 34:557-65.https://doi.org/10.1007/s00261-008-9446-y
Bruix J, Llovet JM (2002) Prognostic prediction and treatment strategy in hepatocellular carcinoma. Hepatology 35:519-24.https://doi.org/10.1053/jhep.2002.32089
Brown DB, Nikolic B, Covey AM, Nutting CW, Saad WE, Salem R, Sofocleous CT, Sze DY (2012) Quality improvement guidelines for transhepatic arterial chemoembolization, embolization, and chemotherapeutic infusion for hepatic malignancy. J Vasc Interv Radiol 23:287-94.https://doi.org/10.1016/j.jvir.2011.11.029
Yan FH, Zhou KR, Cheng JM, Wang JH, Yan ZP, Da RR, Fan J, Ji Y (2002) Role and limitation of FMPSPGR dynamic contrast scanning in the follow-up of patients with hepatocellular carcinoma treated by TACE. World J Gastroenterol 8:658-62. https://doi.org/10.3748/wjg.v8.i4.658
Iqbal SI, Stuart KE (2018) Assessment of tumor response in patients receiving systemic and nonsurgical locoregional treatment of hepatocellular cancer. In: Goldberg RM (ed). UpToDate. Waltham, MA
Imaeda T, Yamawaki Y, Seki M, Goto H, Iinuma G, Kanematsu M, Mochizuki R, Doi H, Saji S, Shimokawa K (1993) Lipiodol retention and massive necrosis after lipiodol-chemoembolization of hepatocellular carcinoma: correlation between computed tomography and histopathology. Cardiovasc Intervent Radiol 16:209-13.https://doi.org/10.1007/BF02602962
Varela M, Real MI, Burrel M, Forner A, Sala M, Brunet M, Ayuso C, Castells L, Montañá X, Llovet JM, Bruix J (2007) Chemoembolization of hepatocellular carcinoma with drug eluting beads: efficacy and doxorubicin pharmacokinetics. J Hepatol 46:474-81.https://doi.org/10.1016/j.jhep.2006.10.020
Minami Y, Kudo M, Kawasaki T, Kitano M, Chung H, Maekawa K, Shiozaki H (2003) Transcatheter arterial chemoembolization of hepatocellular carcinoma: usefulness of coded phase-inversion harmonic sonography. AJR Am J Roentgenol 180:703-8.https://doi.org/10.2214/ajr.180.3.1800703
Morimoto M, Shirato K, Sugimori K, Kokawa A, Tomita N, Saito T, Imada T, Tanaka N, Nozawa A, Numata K, Tanaka K (2003) Contrast-enhanced harmonic gray-scale sonographic-histologic correlation of the therapeutic effects of transcatheter arterial chemoembolization in patients with hepatocellular carcinoma. AJR Am J Roentgenol 181:65-9.https://doi.org/10.2214/ajr.181.1.1810065
Kono Y, Lucidarme O, Choi SH, Rose SC, Hassanein TI, Alpert E, Mattrey RF (2007) Contrast-enhanced ultrasound as a predictor of treatment efficacy within 2 weeks after transarterial chemoembolization of hepatocellular carcinoma. In: J Vasc Interv Radiol, United States, pp 57–65.https://doi.org/10.1016/j.jvir.2006.10.016
Xia Y, Kudo M, Minami Y, Hatanaka K, Ueshima K, Chung H, Hagiwara S, Inoue T, Ishikawa E, Kitai S, Takahashi S, Tatsumi C, Ueda T, Hayaishi S, Maekawa K (2008) Response evaluation of transcatheter arterial chemoembolization in hepatocellular carcinomas: the usefulness of sonazoid-enhanced harmonic sonography. Oncology 75 Suppl 1:99-105.https://doi.org/10.1159/000173430
Shaw CM, Eisenbrey JR, Lyshchik A, O'Kane PL, Merton DA, Machado P, Pino L, Brown DB, Forsberg F (2015) Contrast-enhanced ultrasound evaluation of residual blood flow to hepatocellular carcinoma after treatment with transarterial chemoembolization using drug-eluting beads: a prospective study. J Ultrasound Med 34:859-67. https://doi.org/10.7863/ultra.34.5.859
Liu M, Lin MX, Lu MD, Xu ZF, Zheng KG, Wang W, Kuang M, Zhuang WQ, Xie XY (2015) Comparison of contrast-enhanced ultrasound and contrast-enhanced computed tomography in evaluating the treatment response to transcatheter arterial chemoembolization of hepatocellular carcinoma using modified RECIST. Eur Radiol 25:2502-11.https://doi.org/10.1007/s00330-015-3611-9
Watanabe Y, Ogawa M, Kumagawa M, Hirayama M, Miura T, Matsumoto N, Nakagawara H, Yamamoto T, Moriyama M (2020) Utility of Contrast-Enhanced Ultrasound for Early Therapeutic Evaluation of Hepatocellular Carcinoma After Transcatheter Arterial Chemoembolization. J Ultrasound Med 39:431-440.https://doi.org/10.1002/jum.15118
Kim HJ, Kim TK, Kim PN, Kim AY, Ko EY, Kim KW, Sung KB, Ha HK, Kim HC, Lee MG (2006) Assessment of the therapeutic response of hepatocellular carcinoma treated with transcatheter arterial chemoembolization: comparison of contrast-enhanced sonography and 3-phase computed tomography. J Ultrasound Med 25:477-86.https://doi.org/10.7863/jum.2006.25.4.477
Paul SB, Dhamija E, Gamanagatti SR, Sreenivas V, Yadav DP, Jain S, Shalimar, Acharya SK (2017) Evaluation of tumor response to intra-arterial chemoembolization of hepatocellular carcinoma: Comparison of contrast-enhanced ultrasound with multiphase computed tomography. Diagn Interv Imaging 98:253-260.https://doi.org/10.1016/j.diii.2016.09.002
Nam K, Stanczak M, Lyshchik A, Machado P, Kono Y, Forsberg F, Shaw CM, Eisenbrey JR (2018) Evaluation of Hepatocellular Carcinoma Transarterial Chemoembolization using Quantitative Analysis of 2D and 3D Real-time Contrast Enhanced Ultrasound. Biomed Phys Eng Express 4:035039. https://doi.org/10.1088/2057-1976/aabb14
Moschouris H, Malagari K, Marinis A, Kornezos I, Stamatiou K, Nikas G, Papadaki MG, Gkoutzios P (2012) Hepatocellular carcinoma treated with transarterial chemoembolization: Evaluation with parametric contrast-enhanced ultrasonography. World J Radiol 4:379-86.https://doi.org/10.4329/wjr.v4.i8.379
Oezdemir I, Wessner CE, Shaw C, Eisenbrey JR, Hoyt K (2020) Tumor Vascular Networks Depicted in Contrast-Enhanced Ultrasound Images as a Predictor for Transarterial Chemoembolization Treatment Response. Ultrasound Med Biol 46:2276-2286. https://doi.org/10.1016/j.ultrasmedbio.2020.05.010
Liu D, Liu F, Xie X, Su L, Liu M, Kuang M, Huang G, Wang Y, Zhou H, Wang K, Lin M, Tian J (2020) Accurate prediction of responses to transarterial chemoembolization for patients with hepatocellular carcinoma by using artificial intelligence in contrast-enhanced ultrasound. Eur Radiol 30:2365-2376. https://doi.org/10.1007/s00330-019-06553-6
Moschouris H, Malagari K, Kalokairinou M, Stamatiou K, Marinis A, Papadaki MG (2011) Contrast-enhanced ultrasonography with intraarterial administration of SonoVue for guidance of transarterial chemoembolization: an initial experience. Med Ultrason 13:296-301
Lekht I, Nayyar M, Luu B, Guichet PL, Ho J, Ter-Oganesyan R, Katz M, Gulati M (2017) Intra-arterial contrast-enhanced ultrasound (IA CEUS) for localization of hepatocellular carcinoma (HCC) supply during transarterial chemoembolization (TACE): a case series. Abdom Radiol (NY) 42:1400-1407. https://doi.org/10.1007/s00261-016-1016-0
Shiozawa K, Watanabe M, Ikehara T, Yamamoto S, Matsui T, Saigusa Y, Igarashi Y, Maetani I (2018) Efficacy of intra-arterial contrast-enhanced ultrasonography during transarterial chemoembolization with drug-eluting beads for hepatocellular carcinoma. World J Hepatol 10:95-104.https://doi.org/10.4254/wjh.v10.i1.95
Wessner CE, Shaw CM, Stanczak M, Forsberg F, Lyshchik A, Tan A, Eisenbrey JR (2020) Contrast-enhanced Ultrasound Identifies Patent Feeding Vessels in Transarterial Chemoembolization Patients With Residual Tumor Vascularity. Ultrasound Q 36:218-223.https://doi.org/10.1097/RUQ.0000000000000513
Bruix J, Sherman M (2011) Management of hepatocellular carcinoma: an update. Hepatology 53:1020-2.https://doi.org/10.1002/hep.24199
Memon K, Lewandowski RJ, Kulik L, Riaz A, Mulcahy MF, Salem R (2011) Radioembolization for primary and metastatic liver cancer. Semin Radiat Oncol 21:294-302. https://doi.org/10.1016/j.semradonc.2011.05.004
Hickey R, Lewandowski R, Salem R (2015) Yttrium-90 radioembolization is a viable treatment option for unresectable, chemorefractory colorectal cancer liver metastases: further evidence in support of a new treatment paradigm. Ann Surg Oncol 22:706-7.https://doi.org/10.1245/s10434-014-4165-9
Paul SB, Sharma H (2014) Role of Transcatheter Intra-arterial Therapies for Hepatocellular Carcinoma. J Clin Exp Hepatol 4:S112-21. https://doi.org/10.1016/j.jceh.2014.03.048
Andreana L, Isgrò G, Marelli L, Davies N, Yu D, Navalkissoor S, Burroughs AK (2012) Treatment of hepatocellular carcinoma (HCC) by intra-arterial infusion of radio-emitter compounds: trans-arterial radio-embolisation of HCC. Cancer Treat Rev 38:641-9. https://doi.org/10.1016/j.ctrv.2011.11.004
Salem R, Lewandowski RJ, Kulik L, Wang E, Riaz A, Ryu RK, Sato KT, Gupta R, Nikolaidis P, Miller FH, Yaghmai V, Ibrahim SM, Senthilnathan S, Baker T, Gates VL, Atassi B, Newman S, Memon K, Chen R, Vogelzang RL, Nemcek AA, Resnick SA, Chrisman HB, Carr J, Omary RA, Abecassis M, Benson AB, 3rd, Mulcahy MF (2011) Radioembolization results in longer time-to-progression and reduced toxicity compared with chemoembolization in patients with hepatocellular carcinoma. Gastroenterology 140:497-507.e2.https://doi.org/10.1053/j.gastro.2010.10.049
Carr BI (2004) Hepatic arterial 90Yttrium glass microspheres (Therasphere) for unresectable hepatocellular carcinoma: interim safety and survival data on 65 patients. Liver Transpl 10:S107-10. https://doi.org/https://doi.org/10.1002/lt.20036
Kirchner T, Marquardt S, Werncke T, Kirstein MM, Brunkhorst T, Wacker F, Vogel A, Rodt T (2019) Comparison of health-related quality of life after transarterial chemoembolization and transarterial radioembolization in patients with unresectable hepatocellular carcinoma. Abdom Radiol (NY) 44:1554-1561. https://doi.org/10.1007/s00261-018-1802-y
Lewandowski RJ, Gabr A, Abouchaleh N, Ali R, Al Asadi A, Mora RA, Kulik L, Ganger D, Desai K, Thornburg B, Mouli S, Hickey R, Caicedo JC, Abecassis M, Riaz A, Salem R (2018) Radiation Segmentectomy: Potential Curative Therapy for Early Hepatocellular Carcinoma. Radiology 287:1050-1058. https://doi.org/10.1148/radiol.2018171768
Arndt L, Villalobos A, Wagstaff W, Cheng B, Xing M, Ermentrout RM, Bercu Z, Cristescu M, Shah A, Wedd J, Majdalany BS, Magliocca JF, Sellers MT, Kokabi N (2020) Evaluation of Medium-Term Efficacy of Y90 Radiation Segmentectomy vs Percutaneous Microwave Ablation in Patients with Solitary Surgically Unresectable < 4 cm Hepatocellular Carcinoma: A Propensity Score Matched Study. Cardiovasc Intervent Radiol
Venkatanarasimha N, Gogna A, Tong KTA, Damodharan K, Chow PKH, Lo RHG, Chandramohan S (2017) Radioembolisation of hepatocellular carcinoma: a primer. Clin Radiol 72:1002-1013. https://doi.org/10.1016/j.crad.2017.07.021
Aslam A, Do RKG, Chernyak V, Mediratta-Lala M (2020) LI-RADS imaging criteria for HCC diagnosis and treatment: emerging evidence. Curr Hepatlogy 19:437-447. https://doi.org/10.1007/s11901-020-00546-6
Seyal AR, Gonzalez-Guindalini FD, Arslanoglu A, Harmath CB, Lewandowski RJ, Salem R, Yaghmai V (2015) Reproducibility of mRECIST in assessing response to transarterial radioembolization therapy in hepatocellular carcinoma. Hepatology 62:1111-21. https://doi.org/10.1002/hep.27915
Eisenbrey JR, Forsberg F, Wessner CE, Delaney LJ, Bradigan K, Gummadi S, Tantawi M, Lyshchik A, O'Kane P, Liu JB, Intenzo C, Civan J, Maley W, Keith SW, Anton K, Tan A, Smolock A, Shamimi-Noori S, Shaw CM (2020) US-triggered Microbubble Destruction for Augmenting Hepatocellular Carcinoma Response to Transarterial Radioembolization: A Randomized Pilot Clinical Trial. Radiology:202321. https://doi.org/10.1148/radiol.2020202321
Delaney LJ, Forsberg F, Wessner CE, Machado P, Lyshchik A, O’Kane P, Gummadi S, Tan A, Shaw CM, Eisenbrey JR (2019) Predicting long term HCC response to radioembolization using contrast-enhanced ultrasound 1–2 weeks post treatment. IEEE INTERNATIONAL ULTRASONICS SYMPOSIUM (IUS). Glasgow, United Kingdom, pp 2129–2132. https://doi.org/10.1109/ULTSYM.2019.8926264
Meloni MF, Goldberg SN, Livraghi T, Calliada F, Ricci P, Rossi M, Pallavicini D, Campani R (2001) Hepatocellular carcinoma treated with radiofrequency ablation: comparison of pulse inversion contrast-enhanced harmonic sonography, contrast-enhanced power Doppler sonography, and helical CT. AJR Am J Roentgenol 177:375-80. https://doi.org/10.2214/ajr.177.2.1770375
Choi D, Lim HK, Lee WJ, Kim SH, Kim YH, Lim JH (2003) Early assessment of the therapeutic response to radio frequency ablation for hepatocellular carcinoma: utility of gray scale harmonic ultrasonography with a microbubble contrast agent. J Ultrasound Med 22:1163-72.https://doi.org/10.7863/jum.2003.22.11.1163
Wen YL, Kudo M, Zheng RQ, Minami Y, Chung H, Suetomi Y, Onda H, Kitano M, Kawasaki T, Maekawa K (2003) Radiofrequency ablation of hepatocellular carcinoma: therapeutic response using contrast-enhanced coded phase-inversion harmonic sonography. AJR Am J Roentgenol 181:57-63. https://doi.org/10.2214/ajr.181.1.1810057
Kim CK, Choi D, Lim HK, Kim SH, Lee WJ, Kim MJ, Lee JY, Jeon YH, Lee J, Lee SJ, Lim JH (2005) Therapeutic response assessment of percutaneous radiofrequency ablation for hepatocellular carcinoma: utility of contrast-enhanced agent detection imaging. Eur J Radiol 56:66-73. https://doi.org/10.1016/j.ejrad.2005.03.023
Dill-Macky MJ, Asch M, Burns P, Wilson S (2006) Radiofrequency ablation of hepatocellular carcinoma: predicting success using contrast-enhanced sonography. AJR Am J Roentgenol 186:S287-95. https://doi.org/10.2214/AJR.04.1916
Lu MD, Yu XL, Li AH, Jiang TA, Chen MH, Zhao BZ, Zhou XD, Wang JR (2007) Comparison of contrast enhanced ultrasound and contrast enhanced CT or MRI in monitoring percutaneous thermal ablation procedure in patients with hepatocellular carcinoma: a multi-center study in China. Ultrasound Med Biol 33:1736-49. https://doi.org/10.1016/j.ultrasmedbio.2007.05.004
Salvaggio G, Campisi A, Lo Greco V, Cannella I, Meloni MF, Caruso G (2010) Evaluation of posttreatment response of hepatocellular carcinoma: comparison of ultrasonography with second-generation ultrasound contrast agent and multidetector CT. Abdom Imaging 35:447-53. https://doi.org/10.1007/s00261-009-9551-6
Bo XW, Xu HX, Sun LP, Zheng SG, Guo LH, Lu F, Wu J, Xu XH (2014) Bipolar radiofrequency ablation for liver tumors: comparison of contrast-enhanced ultrasound with contrast-enhanced MRI/CT in the posttreatment imaging evaluation. Int J Clin Exp Pathol 7:6108-16
Funding
NIH R01s CA215520, CA238241, CA194307.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
JRE: Equipment, contrast agent, and research support from GE Healthcare. Equipment support from Siemens Healthineers. Equipment support from Canon Medical Systems. Drug and speaker fees from Lantheus Medical Imaging. Royalties from Elsevier. HG: None. ES: None. AL: GE Healthcare—research support, consulting, and speaker fees; Siemens—research support; Canon Medical—research support; Bracco Diagnostics—research support, advisory board member; Elsevier—book royalties.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Eisenbrey, J.R., Gabriel, H., Savsani, E. et al. Contrast-enhanced ultrasound (CEUS) in HCC diagnosis and assessment of tumor response to locoregional therapies. Abdom Radiol 46, 3579–3595 (2021). https://doi.org/10.1007/s00261-021-03059-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-021-03059-y