Abstract
Pulmonary vein stenosis (PVS) in children is a morbid disease and limited progress has been made in improving outcomes for this heterogenous group of patients. Evaluation is currently limited to imaging techniques that fail to provide an adequate overview of the intraluminal and luminal pathology perpetuating our limited understanding of this condition. Optical coherence tomography (OCT) is an imaging modality which provides intraluminal profiling with microstructural detail through optical reflective technology. We sought to evaluate whether its use was technically feasible in pediatric PVS and whether the imaging data provided potentially useful outputs for clinical utility. Eleven patients were prospectively selected from our cardiac catheterization for OCT evaluation of their pulmonary veins (PV) during elective catheterization for PVS. Measurements were taken both pre and post intervention using both manual and automated tools. Stent morphology was characterized. Eleven patients had evaluation of 34 pulmonary veins, with 7 patients having more than one assessment, for a total of 25 overall catheterizations. Most patients were female (75%). Median age at cardiac catheterization was 35 months (range 5–45 months). Median weight of subjects was 10.6 kg (3.7–14.2) with a median BSA documented at 0.505 m2 (0.21–0.57). Median number of pulmonary veins involved was 3, (range 1–5 veins) and median contrast volume of 2.9 mL/kg (0.7–3.7) given. Median radiation dose (DAP) was 6095 µGy·cm2 (1670–12,400). Median number of previous cardiac catheterizations was 7 (range 1–11). All of the vessels with a diameter < 5 mm were adequately visualized. Of all the OCT images acquired, in 15 vessels (44%) contrast was used to clear the vessels from blood as an angiogram was required at the time, in the other 19 vessels (56%), saline was used with adequate imaging. There were no complications related to OCT. OCT is technically feasible to use in pediatric patients without any directly related complications. It provides intraluminal anatomy in children with both native and treated pulmonary venous stenosis when vessel size is less than 5 mm.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pulmonary venous stenosis is a complex disorder that affects a small subset of infants and the congenital heart disease population. Disease may be native [1], post-operative, or associated with other disorders such as chronic lung disease of prematurity. Disease may be focal, segmental, or diffuse and venous distribution may be isolated or affect several veins bilaterally [2]. This cohort of patients continue to suffer poor outcomes with respect to long-term survival though we have seen some recent improvements in short-to-medium term survival, particularly, as we have progressed towards more aggressive targeted treatment in the cardiac catheterization laboratory [3, 4]. We continue to have a limited understanding of the evolution of this condition and struggle to define those most likely to benefit from various treatments. It is also difficult to predict those likely to develop aggressive restenosis after balloon angioplasty or stent implantation. Our limited understanding is perpetuated by the inherent difficulty in evaluating intraluminal disease in these small vessels which are precariously accessed by catheterization via the left atrium [5].
Current treatment strategies involve surgical management and catheter-based treatments including balloon angioplasty and stenting [4, 6,7,8]. These treatments may be paralleled with pharmaceutical strategies including antithrombotic, anticoagulation, and systemic anti-proliferative agents like sirolimus [9,10,11].
Focused imaging evaluation is currently limited to echocardiography [12], Computed Tomography Angiogram (CTA) & cardiac Magnetic Resonance Imaging (cMRI) [13,14,15] as well as angiographic evaluation performed during invasive cardiac catheterization [2, 16,17,18]. These modalities provide an overview of disease distribution and anatomical profiling which can assist with treatment planning and decision making. The intraluminal profile of disease, including both the surface and subsurface characteristics of these vessels, is poorly appreciated on all of the above imaging evaluations. Our current limited understanding of the pathological basis of this disease process is based on histological and pathological specimens from surgical tissue and postmortem evaluation [19, 20]. Preliminary reports of intravascular ultrasound assessment of the pulmonary veins in children have been published and present some interesting findings with respect to the intraluminal profile of disease [21]. Ultrasound assessment is, however, limited in some instances in its ability to produce images of diagnostic resolution thus leaving questions with respect to this disease substrate and potential baseline luminal wall abnormalities in this population.
Optical coherence tomography is a novel imaging technology based on broadband low coherence infrared light combined with interferometry which can produce high-resolution images of opaque human tissues. Its use has been established in adult coronary evaluation facilitating the acquisition of surface and subsurface tissue characteristics [22]. This has facilitated the evaluation of complex coronary plaques and stent vessel relationships and has facilitated more informed practice around specific interventions for complex lesions and decision making [23, 24].
We surmised that due to the small caliber of pulmonary veins, similar in dimension to coronary arteries, that this invasive imaging modality may have useful application in the evaluation of both diseased and treated pulmonary venous stenosis in children.
Here, we report our experience with the use of OCT in a small cohort of patients and its feasibility to be used in pediatric patients with pulmonary vein stenosis.
Materials and Materials
We performed a single-center retrospective review of all patients who underwent pulmonary venous assessment with OCT from October 2020 to April 2021. The objectives were to determine the feasibility of performing OCT in pediatric pulmonary veins, evaluate if whether OCT can visualize the lumen, vessel walls, and stent characteristics (when applicable). The secondary objective was to identify any adverse events related directly to OCT. This study was approved by the institutional review board with a waiver of informed consent due to its retrospective nature.
OCT was performed at the time of cardiac catheterization at the discretion of the operator without a standard protocol. No patients had a cardiac catheterization for the sole purpose of performing OCT. Indications for cardiac catheterization included clinical symptoms, echocardiographic findings, and/or CTA findings of new or worsening pulmonary vein stenosis.
OCT was performed with a Dragonfly Optis Imaging catheter (Abbott Vascular, Santa Clara, CA, USA) into the pulmonary vein of interest. The Dragonfly catheter was advanced over a 0.014″ wire via either a 5Fr or 6Fr Judkins Right (JR) guide catheter (Terumo©) or a 4Fr long sheath. Layered contrast (2 mL)/saline (5 mL) was used to displace blood (which causes infra-red scatter attenuating the signal and limiting image acquisition) from the vascular area of interest, only when an angiogram was required. Seven mL of saline was used instead of contrast when an angiogram was not required. Imaging acquisition was achieved through automatic pullback, the system has two modes for image acquisition: “Survey” mode (rapid pullback of 75 mm over 2.1 s at 180 frames per second) and “High Resolution” mode (54 mm segment at the same frame rate over 3 s). Modes were selected as per operator’s preference (Table 1).
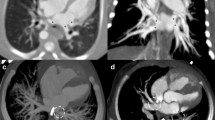
First, the veins were categorized as native veins or post stent implantation. Then these veins were divided based on adequate or inadequate visualization, this referred to the ability to see the vascular lumen and wall architecture enough to do measurements. For patient with prior stents in the vessel, we evaluated the ability to identify stent strouts and the stent lumen. A subgroup of the stented vessels was evaluated to identify which ones could obtain a 3D reconstruction of the stent (Figs. 1 and 2).
Native and stented pulmonary vein images. A OCT acquisition of normal pulmonary venous lumen (red) with OCT catheter (green) on a wire sitting at 9 o’clock. Intimal and medial/adventitial layer labeled with white and red arrows, respectively (LUPV: left upper pulmonary vein, OCT: optical coherence tomography). B OCT acquisition with wire artifact depicted with red dashed lines at 9 o’clock, stent struts individually labeled in green producing peripheral optical artifact (dashed yellow lines), red arrow depicting the diameter of the neo-lumen, and white arrow depicting the diameter of the true stent lumen. There is significant neo-intimal proliferation present (RUPV: right upper pulmonary vein, OCT: optical coherence tomography). C OCT acquisition in a stented LLPV distal from the more peripheral lesion. The resolution is more limited peripheral from the OCT catheter, however, post processing of the image in the zoomed window demonstrates the stent in white and the vessel wall in red allowing identification, localization, and quantification of stent mal-apposition. D Demonstrates a tributary branch vessel (red asterisk) merging with a stented pulmonary vein with stent struts crossing the confluence, wire artifact is noted at 12 o’clock. The OCT catheter is placed co-axially with the stented vessel of interest
Three-dimensional (3D) reconstructions of the region of interest were also post processed to evaluate the distribution of disease and its relationship to stent struts (Fig. 2). The OCT OS automatically registered stent anatomy through struct identification technology and performed automated measurements of area and diameter. These were repeated manually, and neo-intimal thickness was measured at the largest diameter from stent to neo-lumen (Fig. 3).
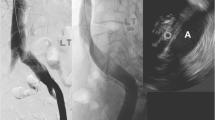
A OCT acquisition of distal RUPV in patient with significant proximal stenosis, white asterisk depicts 2 large collateral vessels entering the lumen (red asterisk), there is wire artifact seen (green arrow) (OCT: optical coherence tomography, RUPV: right upper pulmonary vein). B Stented (red) RUPV s/p cutting balloon angioplasty with Boston Scientific Wolverine (3 mm) which contains 3 atherotomes. The neo-lumen is represented in white. The thickness of the atherotome impression is demonstrated in the zoomed window with a small red arrow in comparison to the thickness of the neo-intima (large red arrow) (RUPV: right upper pulmonary vein). C and D Paired angiography with OCT acquisition. There are two wires placed in two distal tributary branches of the RUPV. The OCT catheter and its supporting wire are sitting in the main RUPV (red) and a second wire sits in a tributary branch (green). The OCT image demonstrates the 2 branches coalescing with both wires producing wire artifact (depicted in white)
Further data collection involved the collection of patient demographics, cardiac diagnosis, hemodynamic and angiographic data as well as index and prior procedural data. Continuous data were reported as median (range) or mean (SD) and categorical data were reported as frequencies.
Results
Thirty-four pulmonary veins in 11 patients during 25 cardiac catheterizations underwent OCT evaluation. Patient demographics, clinical data, procedural data, and OCT data are presented in Table 2. Most patients were female (75%). Median age at cardiac catheterization was 35 months (range 5–45 months). Median weight of subjects was 10.6 kg (3.7–14.2) with a median BSA documented at 0.505 m2 (0.21–0.57).
All veins accepted the Dragonfly catheter and there were no OCT-related adverse events. Pre- and post-intervention assessment were performed in 20 pulmonary veins (59%). Six patients had more than one OCT assessment 8 to 12 weeks after the index evaluation for comparative assessment. One patient had cardiac catheterization performed every 6 weeks for 6 months, this allowed 4 separate times when OCT was used for pulmonary vein luminal evaluation.
Median number of pulmonary veins involved was 3 (range 1–5 veins), and median contrast volume of 2.9 mL/kg (0.7–3.7) given. Median radiation dose (DAP) was 6095 µGy·cm2 (1670–12,400). Median number of previous cardiac catheterizations was 7 (range 1–11). Median procedural time was 190 min (139–236) with a median fluoroscopy time of 64.4 min (35.8–97). 46% of patients had history prematurity. There was no significant difference in procedural times, radiation doses, and contrast dose for each individual patient between the cardiac catheterization when OCT was used and prior catheterizations.
All patients admitted electively for cardiac catheterization were discharged within 48 h of admission.
Pulmonary veins assessed by OCT are summarized in Fig. 4. All of the vessels with a diameter ≤ 5 mm were adequately visualized. In vessels with diameter larger than 5 mm, there was not enough blood clearance with direct injection or wedge injection, to identify the vessel wall or the vessel lumen diameter. Of all the OCT images acquired, in 15 vessels (44%) contrast was used to clear the vessels from blood as an angiogram was required at the time, in the other 19 vessels (56%), saline was used with adequate imaging.
A and B Pre- and post-angioplasty images of a patient with an LLPV stent and critical neo-intimal proliferation (white arrow), the stent is demonstrated in red and the neo-lumen in white. The zoom windows demonstrate both attached and mobile thrombus within the neo-lumen (white asterisk) after successful balloon angioplasty (LLPV: left lower pulmonary vein). C and D Aggressive neo-intimal proliferation (green arrow) in a stented (red) RUPV. The neo-lumen is depicted in white both pre and post angioplasty with a significant lumen gain demonstrated. Ragged edges of the neo-intima are seen adjacent the white line in the post-angioplasty image
High resolution mode was used in 8 pulmonary veins, Survey Mode was used in 24 pulmonary veins and Stationary acquisition (no pullback done) was done in 2 pulmonary veins. Of the veins/stents with luminal diameter < 5 mm, only 37% of the veins were visualized when using the High resolution mode; when compared with 100% of the veins that were visualized when using the Survey Mode. The vessels evaluated with the stationary acquisition were all > 5 mm.
Anatomical description of the most relevant OCT images is described in Figs. 1 and 5.
Advanced stent analysis features with yellow windows demonstrating the vessel tissue characterization and “fly through” with 3D reconstructed stent (A) and reconstructed image of the stent morphology without related tissue (B). Green windows demonstrate the live OCT image with automatic stent strut identification software labeling struts adjacent to the intimal layer (white dots). The struts create significant sono-artifact which is usual. Red windows in A, B demonstrate L mode mapping of the diameter variability throughout the long axis of the vessel with stent vessel apposition and expansion auto-registration and automatic identification of zones of area loss (target zones). White windows in A, B represent standard reconstructed L mode images of the vessel with superimposed stent imaging. Legend: (OCT: optical coherence tomography)
Discussion
We present a novel application of optical coherence tomography (OCT) and the first overview of the practical approach and technical feasibility of pediatric pulmonary venous assessment using OCT. In this study, OCT was successfully performed in patients under 5 years of age, hemodynamically fragile, and with severe congenital heart disease without adverse events.
Current practice with OCT in cardiovascular medicine is mostly limited to its utility in detailed coronary plaque assessment in adults where it guides therapeutic approaches [22,23,24]. There has been limited experience of the use of OCT in pediatrics and only a couple of publications describing its use outside the coronary scope in congenital heart disease [25,26,27]. OCT in adult pulmonary veins has been carried out as part of attempting to understand some of the morphological changes in patients with atrial fibrillation, some of whom have complex myofibrillar changes beginning at the venoatrial confluence [28, 31].
In pediatric practice, IVUS was been shown to be feasible and safe in pediatric patients with pulmonary vein stenosis [21] and can adequately visualize pulmonary vein lumen and walls. However, although OCT and IVUS may have similar applications, their technologies have clear differences. The axial and lateral resolutions of OCT are 70–90 µm which is enhanced from IVUS. The resolution of OCT (to 10–20 µm) is about tenfold higher than that of IVUS (to 100–150 µm), but the maximum depth of tissue penetration is lower with OCT (1–3 mm) than with IVUS (4–8 mm). More differences are included in Table 3.
One of the more relevant technical differences is related to the strong attenuation of light (for OCT) by blood, this is why blood must first be removed during an OCT examination to eliminate massive scattering of light by red blood cells. In the adult practice, this is performed using contrast. Due to the difficulty to clear the blood in these short pulmonary veins in pediatric population with elevated heart rates, the OCT “Survey Mode” was the most commonly used by our team due to the shortest acquisition time.
In our experience, contrast or saline can be used, delivered with a 5F or 6F Guide catheter or a 4F sheath positioned at the pulmonary vein ostia. We used contrast exclusively when an angiogram was required, then OCT interrogation can be obtained at the same time. This allows automatic registration of the OCT catheter, the intraluminal image, and the angiographic appearance of the vessel. Otherwise, our preference is to use saline solution and keep the contrast doses as low as possible in our already fragile population. There is still registration to catheter position with the live fluoroscopic acquisition and the intraluminal appearance. The quality of the images was not related to the use of contrast or saline in our experience. Co-registration with cine-angiography (Fig. 5C and D) facilitates angiographic labeling and targeting at the time of intervention.
The pulmonary veins that were not adequately visualized by OCT in our study were all larger than 5 mm or the High resolution mode was being used. The reason behind this is due to several factors that include heart rate of the patients being evaluated, the time of image acquisition, and the fact that the blood flow is towards the tip of the OCT catheter (and not the opposite as in the coronary arteries); all of these factors, intimately related to the time that the contrast or saline stays in the vessel to clear the blood. When the blood is not completely clear, the lumen is obscured, and the luminal walls cannot be identified.
For patients with vessels or stents larger than 5 mm, we obtained stationary acquisition. With this technique, we evaluated a 1 mm segment of a specific area of the vessel with a quick injection of saline. The acquisition is fast enough that works in larger vessels but is limited to an extremely small section. The information obtained was not felt to be clinically useful to the operator.
If the area of interest is the pulmonary vein ostia, a pulmonary wedge angiogram can be performed if a second venous access is available. This would allow antegrade flow of contrast in levophase to the pulmonary vein and clears the blood enough for a good quality image. The 3D reconstruction tools and intraluminal assessment with OCT are relevant for small patients for procedural planning, especially for stent placement in very short segments of pulmonary veins (Fig. 2).
Pulmonary veins in children fall within the appropriate size range and wall thickness for OCT image evaluation, particularly in patients with diseased vessels [29, 30]. Established luminal geometric assessment in OCT evaluation has been really useful in our practice and includes minimal lumen area (mm2), lumen obstruction (%), stent apposition, stent expansion, minimal stent cross sectional area (mm2), lumen gain (%), late lumen loss (%), and restenosis quantification (%). Geometric assessment of the entire short and long axis (L-mode) of the pulmonary veins may be relevant to qualify the exact area of narrowing requiring treatment, identify targets for healthy vessel diameters as well as for decision making on balloon or stent sizing and design. Strut analysis can decipher stent vessel relationships [22] allowing adequate stent vessel apposition relationships in several of our patients (Fig. 1). Relationships of the stent to the native vessel and its intima in pulmonary venous stenosis may assist in identifying those most likely to suffer from aggressive restenosis [31].
OCT has brought to our practice all the known advantages of IVUS in pulmonary vein stenosis patients with significantly enhanced features that allow a high-definition image with 3D reconstructions and analysis of the wall vessel.
Limitations
This was a single-center study on a small number of complex patients and carried out with no standardized approach to acquisition. There is no normal data on OCT imaging in pediatric pulmonary venous disease nor does this disease bear similarity to adult coronary artery disease. The interpretation of imaging data obtained by OCT evaluation may be interpreted with caution and is not currently validated for diagnostic utility.
Conclusion
OCT is technically feasible and safe to use in pediatric patients for pulmonary veins evaluation. It provides intraluminal anatomy in children with both native and treated pulmonary venous stenosis when vessel size is less than 5 mm.
References
Sadr IM, Tan PE, Kieran MW, Jenkins KJ (2000) Mechanism of pulmonary vein stenosis in infants with normally connected veins. Am J Cardiol 86(5):577
Latson LA, Prieto LR (2007) Congenital and acquired pulmonary vein stenosis. Circulation 115(1):103–108
Vanderlaan RD, Rome J, Hirsch R, Ivy D, Caldarone CA (2020) Pulmonary vein stenosis: Treatment and challenges. J Thorac Cardiovasc Surg. 2020 Jun 27. https://www.jtcvs.org/article/S0022-5223(20)31796-7/abstract. Accessed 21 Dec 2020
Patel JD, Briones M, Jones S, Suthar D, Gray R, Petit CJ (2020) Medical and invasive management of congenital and acquired pulmonary vein stenosis. Curr Treat Options Pediatr 6(3):170–181
del Cerro MJ, Moledina S, Haworth SG, Ivy D, Dabbagh MA, Banjar H et al (2016) Cardiac catheterization in children with pulmonary hypertensive vascular disease: consensus statement from the pulmonary vascular research institute, pediatric and congenital heart disease task forces. Pulm Circ 6(1):118–125
Kalfa D, Belli E, Bacha E, Lambert V, di Carlo D, Kostolny M et al (2018) Outcomes and prognostic factors for postsurgical pulmonary vein stenosis in the current era. J Thorac Cardiovasc Surg 156(1):278–286
Cory MJ, Ooi YK, Kelleman MS, Vincent RN, Kim DW, Petit CJ (2017) Reintervention is associated with improved survival in pediatric patients with pulmonary vein stenosis. JACC Cardiovasc Interv 10(17):1788–1798
Esch JJ, Porras D, Bergersen L, Jenkins KJ, Marshall AC (2015) Systemic embolic complications of pulmonary vein angioplasty in children. Pediatr Cardiol 36(7):1357–1362
Callahan R, Esch JJ, Wang G, Ireland CM, Gauvreau K, Jenkins KJ (2020) Systemic sirolimus to prevent in-stent stenosis in pediatric pulmonary vein stenosis. Pediatr Cardiol 41(2):282–289
Callahan R, Kieran MW, Baird CW, Colan SD, Gauvreau K, Ireland CM et al (2018) Adjunct targeted biologic inhibition agents to treat aggressive multivessel intraluminal pediatric pulmonary vein stenosis. J Pediatr 198:29-35.e5
Pazos-López P (2016) Pulmonary vein stenosis: etiology, diagnosis and management. World J Cardiol 8(1):81
Koestenberger M, Friedberg MK, Nestaas E, Michel-Behnke I, Hansmann G (2016) Transthoracic echocardiography in the evaluation of pediatric pulmonary hypertension and ventricular dysfunction. Pulm Circ 6(1):15–29
Vyas HV, Greenberg SB, Krishnamurthy R (2011) MR imaging and CT evaluation of congenital pulmonary vein abnormalities in neonates and infants. Radiographics 32(1):87–98
Hassani C, Saremi F (2017) Comprehensive cross-sectional imaging of the pulmonary veins. Radiographics 37(7):1928–1954
Valsangiacomo ER, Levasseur S, McCrindle BW, MacDonald C, Smallhorn JF, Yoo S-J (2003) Contrast-enhanced MR angiography of pulmonary venous abnormalities in children. Pediatr Radiol 33(2):92–98
Barrera CA, Saul D, Rapp JB, Smith CL, White AM, Biko DM et al (2020) Diagnostic performance of CT angiography to detect pulmonary vein stenosis in children. Int J Cardiovasc Imaging 36(1):141–147
Ou P, Marini D, Celermajer DS, Agnoletti G, Vouhé P, Sidi D et al (2009) Non-invasive assessment of congenital pulmonary vein stenosis in children using cardiac-non-gated CT with 64-slice technology. Eur J Radiol 70(3):595–599
Imnadze G, Kranig W, Grove R, Wolff E, Thale J (2014) The ABC of a simple method for pulmonary vein angiography. Indian Pacing Electrophysiol J 14(1):53–59
Kovach AE, Magcalas PM, Ireland C, McEnany K, Oliveira AM, Kieran MW et al (2017) Paucicellular fibrointimal proliferation characterizes pediatric pulmonary vein stenosis: clinicopathologic analysis of 213 samples from 97 patients. Am J Surg Pathol 41(9):1198–1204
Pogoriler JE, Kulik TJ, Casey AM, Baird CW, Mullen MP, Jenkins KJ et al (2016) Lung pathology in pediatric pulmonary vein stenosis. Pediatr Dev Pathol 19(3):219–229
Callahan R, Jenkins KJ, Gauthier Z, Gauvreau K, Porras D (2020) Preliminary findings on the use of intravascular ultrasound in the assessment of pediatric pulmonary vein stenosis. Catheter Cardiovasc Interv 97:4
Bezerra HG, Costa MA, Guagliumi G, Rollins AM, Simon DI (2009) Intracoronary optical coherence tomography: a comprehensive review. JACC Cardiovasc Interv 2(11):1035–1046
Räber L, Mintz GS, Koskinas KC, Johnson TW, Holm NR, Onuma Y et al (2018) Clinical use of intracoronary imaging. Part 1: guidance and optimization of coronary interventions: an expert consensus document of the European Association of Percutaneous Cardiovascular Interventions. Eur Heart J 39(35):3281–3300
Ijsselmuiden AJJ, Zwaan EM, Oemrawsingh RM, Bom MJ, Dankers FJWM, de Boer MJ et al (2018) Appropriate use criteria for optical coherence tomography guidance in percutaneous coronary interventions. Neth Heart J. 26(10):473–483
McGovern E, Hosking MCK, Balbacid E, Voss C, Berger F, Schubert S et al (2019) Optical coherence tomography for the early detection of coronary vascular changes in children and adolescents after cardiac transplantation: findings from the International Pediatric OCT Registry. JACC Cardiovasc Imaging 12(12):2492–2501
Hill JA, Mahadevaiah G, Jenkins MW (2015) Optical coherence tomography imaging of the patent ductus arteriosus: first known uses in congenital heart disease. Catheter Cardiovasc Interv Off J Soc Card Angiogr Interv 85(2):278–281
Homma Y, Hayabuchi Y, Ono A, Kagami S (2018) Pulmonary artery wall thickness assessed by optical coherence tomography correlates with pulmonary hemodynamics in children with congenital heart disease. Circ J Off J Jpn Circ Soc 82(9):2350–2357
Lye TH, Iyer V, Marboe CC, Hendon CP (2019) Mapping the human pulmonary venoatrial junction with optical coherence tomography. Biomed Opt Express 10(2):434–448
Robida A (1989) Diameters of pulmonary veins in normal children-an angiocardiographic study. Cardiovasc Intervent Radiol 12(6):307–309
Sairanen H, Louhimo I, Tolppanen EM (1986) Pulmonary vein diameter in normal children. Pediatr Cardiol 6(5):259–261
Nagoshi R, Shinke T, Otake H, Shite J, Matsumoto D, Kawamori H et al (2013) Qualitative and quantitative assessment of stent restenosis by optical coherence tomography: comparison between drug-eluting and bare-metal stents. Circ J Off J Jpn Circ Soc 77(3):652–660
Acknowledgements
Abbott Vascular for supporting this initiative.
Funding
N/A.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Jenny E. Zablah and Gareth Morgan are consultants/speakers for Abbott.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zablah, J.E., O’Callaghan, B., Shorofsky, M. et al. Technical Feasibility on the Use of Optical Coherence Tomography in the Evaluation of Pediatric Pulmonary Venous Stenosis. Pediatr Cardiol 43, 1054–1063 (2022). https://doi.org/10.1007/s00246-022-02824-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00246-022-02824-0