Abstract
Summary
Spinal cord injury (SCI) causes rapid and marked bone loss. The present study demonstrates that low-intensity vibration (LIV) improves selected biomarkers of bone turnover and gene expression and reduces osteoclastogenesis, suggesting that LIV may be expected to benefit to bone mass, resorption, and formation after SCI.
Introduction
Sublesional bone is rapidly and extensively lost following spinal cord injury (SCI). Low-intensity vibration (LIV) has been suggested to reduce loss of bone in children with disabilities and osteoporotic women, but its efficacy in SCI-related bone loss has not been tested. The purpose of this study was to characterize effects of LIV on bone and bone cells in an animal model of SCI.
Methods
The effects of LIV initiated 28 days after SCI and provided for 15 min twice daily 5 days each week for 35 days were examined in female rats with moderate severity contusion injury of the mid-thoracic spinal cord.
Results
Bone mineral density (BMD) of the distal femur and proximal tibia declined by 5 % and was not altered by LIV. Serum osteocalcin was reduced after SCI by 20 % and was increased by LIV to a level similar to that of control animals. The osteoclastogenic potential of bone marrow precursors was increased after SCI by twofold and associated with 30 % elevation in serum CTX. LIV reduced the osteoclastogenic potential of marrow precursors by 70 % but did not alter serum CTX. LIV completely reversed the twofold elevation in messenger RNA (mRNA) levels for SOST and the 40 % reduction in Runx2 mRNA in bone marrow stromal cells resulting from SCI.
Conclusion
The findings demonstrate an ability of LIV to improve selected biomarkers of bone turnover and gene expression and to reduce osteoclastogenesis. The study indicates a possibility that LIV initiated earlier after SCI and/or continued for a longer duration would increase bone mass.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Immobilization due to bed rest, space flight, or paralysis results in atrophy of skeletal muscle and loss of bone in immobilized body regions. In most cases, immobilization-related bone loss results from the combined effects of increased resorption and reduced formation. Spinal cord injury (SCI) results in an extreme form of immobilization that results in substantial muscle atrophy and bone loss. Muscle atrophy after SCI is associated with diminished strength, power, and muscle fatigue resistance [1–3]. Alterations in bone metabolism after SCI include a reduced bone formation rate, as well as marked acceleration of bone resorption associated with increased osteoclast surface and increased potential of bone marrow progenitors for osteoclastic differentiation [1–3]. Bone loss after SCI is relatively unique in its localization to the sublesional regions with the greatest declines observed at the distal femur and proximal tibia, where bone mineral density (BMD) may be reduced by more than 50 % within a few years after SCI [1, 2, 4–7]. Consequently, fractures of the distal femur and proximal tibia are the most common fractures in individuals with SCI [8–11]. In humans with SCI, BMD declines rapidly at all sublesional boney sites at rates as high as 1 % per week for the first year after injury [1, 2, 4]. In studies of SCI rats, loss of trabecular bone in the proximal tibia approaches 70 % at 3 weeks after a complete spinal cord transection [12] and over 60 % at the distal femoral metaphysis at 10 days after severe spinal cord contusion [13].
The observation that low intensity high-frequency mechanical vibration (LIV) increases bone mass and bone formation in healthy sheep [14] and mice [15] has generated interest in the possibility that LIV might also reduce or reverse pathological remodeling of bone. A growing body of evidence in laboratory animals and humans supports this concept. In clinical studies, LIV has been found to improve BMD in children with disabilities [16, 17] and post-menopausal women [18–21]. Anabolic effects of LIV do not appear to require gravitational loading [22, 23]. LIV has also been found to exert beneficial effects on the pool of bone marrow mesenchymal stem cells capable of osteoblastic differentiation. Numbers of bone marrow cells with the potential to differentiate into osteoblast-like cells when cultured in vitro are reduced by immobilization and normalized by LIV [24]. In some studies, muscle has also been suggested to be beneficially affected by LIV, as exemplified by finding that in normal mice, whole body LIV increased soleus muscle fiber cross sectional area [15].
Clinical studies have demonstrated beneficial effects of LIV for individuals with SCI including improvements in walking function [25], muscle blood flow [26], and spasticity [27, 28]. LIV may be expected to slow or reverse bone loss after severe immobilization due to conditions such as SCI [2, 4, 5, 29] and can be administered through the supine body in a manner that would permit significant skeletal loading [30]. The possible benefits of LIV to individuals with SCI are supported by findings of a case report of a single subject with motor-incomplete SCI in whom vibration appeared to improve BMD [31]. However, it remains uncertain whether LIV can reverse of attenuate structural and functional changes in bone and muscle after SCI.
The objectives of this study were to characterize effects of whole body LIV on muscle and bone in a rat model of SCI. We reasoned that effects of LIV might be enhanced by skeletal loading and thus conducted these investigations using an animal model of moderate severity contusion SCI in which sufficient motor function had returned by 28 days after the injury that animals were capable of weight bearing. We examined the effects of the application of LIV after an SCI on BMD, three-dimensional architecture of metaphysical trabecular bone, serum markers of bone metabolism, and differentiation potential of bone marrow progenitors, and on mass of paralyzed skeletal muscle and biomarkers of neuromuscular activation. Because little is known about the effects of a moderate severity contusion SCI on bone, we also examined the changes in bone, bone metabolic markers, and bone marrow progenitors resulting from this form of SCI. We expected that LIV would reduce bone resorption, stimulate bone formation, and correct abnormalities in osteoblastogenic or osteoclastogenic potential of marrow precursors in animals with SCI. We also expected that LIV would increase the mass of skeletal muscle and increase biomarkers of neuromuscular activity in skeletal muscle in animals with SCI.
Methods
Animals
Female Sprague Dawley rats 3 months of age (Harlan Laboratories) were housed in temperature and humidity controlled rooms and provided with a 12:12-h day to night cycle. Animals were fed standard rat chow ad libitum. All procedures with experimental animals were approved by the Institutional Animal Care and Use Committee of the University of Miami and were in conformance with the National Institutes of Health Guide.
Experimental design
Rats were randomly assigned to one of the following groups: SCI without whole body LIV (SCI, N = 14), SCI with whole body LIV (LIV, N = 13), or sham SCI consisting of a laminectomy only (Sham, N = 8). A moderate severity SCI at the interspace between the ninth and tenth thoracic vertebra was produced using a New York University (MASCIS) impactor by dropping a 10 g weight from a height of 12.5 mm as described in detail elsewhere [32]. Control animals underwent only a laminectomy.
LIV was initiated 28 days after SCI. Starting with the LIV at 28 days post-injury was chosen based on following considerations: (i) we believed that gravitational loading might enhance any benefits of LIV, and animals have regained the ability to weight bear at 28 days as reflected by the mean Basso, Beattie, and Bresnahan (BBB) scores at this time for SCI animals (SCI-LIV 9.42 ± 0.31; SCI-no vibration 9.46 ± 0.25). This type of partial functional recovery in moderate severity SCI animals at 28 days post-injury has similarly demonstrated by other studies [33, 34]; (ii) This lag time prior to starting vibration may be somewhat comparable to that which occurs in a rehabilitation setting, where several weeks may pass before starting rehabilitation therapy after injury; and (iii) We recently demonstrated that when begun on day 29 after SCI and continued for 28 days, anabolic steroid nandrolone was able to reduce bone loss after SCI through the regulation of Wnt signaling pathway [35].
Animals randomized to the LIV group underwent 35 days of treatment performed twice daily for 15 min each session for 5 days each week (Fig. 1a). Animals with SCI were divided among groups that received LIV and a group that was placed directly on the vibrating platform (Soloflex®) for the same period of time without activating it (Fig. 1b). The vibration device was programmed in order to achieve frequency of vibration within a range of about 40 Hz (0.3 g) because these parameters were similar to those used in studies by Field-Fote and coworkers [27] and others [36, 37]. In particular, Xie et al. showed that whole body vibration at 45 Hz (0.3 g) for 15 min a day inhibited trabecular bone resorption and increased bone formation in the skeleton of grown mice [36]. Thirty-minute sessions were chosen based on the hope that these would be tolerable and of a duration that might be clinically feasible. Twice daily sessions were chosen because of information in animals and cells that response to LIV was greater with twice daily dosing in work from Rubin and coworkers, and we reasoned that this paradigm might enhance any beneficial effect in our animal model [38]. Characteristics of the vibration provided were determined with an iPad and the Vibration app from Diffraction Limited Design, LLC. The iPad was placed in each chamber with the plate activated. The observed frequency of vibration was approximately 37 Hz, and the mean acceleration across the four chambers was 0.238 g 0 to peak (range 0.161 to 0.273 g 0 to peak).
The control animals post laminectomy were also placed on the platform with activation in a manner identical to the SCI animals. The SCI animals were assigned to groups based on BBB score [39] at 4 weeks to prevent any confounding influence that might arise from unequal distributions of function among the groups (SCI-LIV 9.42 ± 0.31; SCI-no vibration 9.46 ± 0.25). To provide LIV intervention, animals were placed in chambers of a plexiglass box; a paper towel was placed on the bottom of each chamber. The box contained four chambers, and one rat was placed into each chamber. Rats were placed in chambers in a random order from one session to the next.
Animals were anesthetized (3 % isoflurane, 70/30 % N2O/O2) at 65 days post-SCI for blood collection, muscle, and bone harvesting. Blood was collected by intraventricular puncture, allowed to clot at room temperature, and centrifuged; serum was then removed and stored at −20 °C. Muscles were isolated by careful dissection, weighed, and flash frozen in liquid nitrogen. Hindlimbs were freed from the pelvis by cutting ligaments and connective tissues at the hip. Left hindlimbs were placed into sterile tubes containing ice-cold Minimum Essential Alpha Medium and kept at 4 °C until processing for isolation of bone marrow cells. Right hindlimbs were immersed in 4 % paraformaldehyde overnight after which fixative was drained and replaced with 70 % ethanol in water.
Dual energy X-ray absorptiometry
Areal bone mineral density (BMD) was measured using a small animal dual energy X-ray absorptiometer (DXA) (Lunar Piximus, Inside Out Sales, Fitchburg, WI) as previously described [40, 41]. Hindlimbs were positioned on the DXA platform with the knee flexed at an angle of 135°, and DXA images were acquired with Lunar Piximus software. The instrument was calibrated with a phantom following the procedures recommended by the manufacturer on each day of use. The metaphysis of the distal femur and proximal tibia were selected as regions of interest (ROI). The coefficient of variation for the repeated measurements for the ROI was approximately 1.5 %.
MicroCT
To evaluate trabecular architecture of the distal femur, microCT was performed on fixed bones, as described previously [40] using a Scanco μCT scanner and a 16-μm voxel size. Image reconstruction and 3D quantitative analysis were performed using software provided by Scanco. Scans were initiated at the growth plate and moved proximally for a total of approximately 300 slices. A region of interest consisting of 100 slices beginning 0.5 mm proximal to the growth plate and continuing in a proximal direction were included in the analysis. Standard nomenclature and methods for bone morphometric analysis were employed [42].
Culture and differentiation of bone marrow progenitors
Culture and differentiation of bone marrow progenitors was assessed as previously described [40]. Briefly, cells were flushed from the marrow cavity with α-MEM and seeded into tissue culture wells in this medium. To culture osteoclasts (OC), cells were cultured for 2 days in α-MEM supplemented with macrophage colony-stimulating factor (M-CSF, 5 ng/ml), after which non-adherent cells were collected, purified by centrifugation in Ficoll-Plus (GE Life Sciences), then seeded into wells and cultured in α-MEM supplemented with M-CSF (30 ng/ml) and RANKL (60 ng/ml) for 4–6 days. Osteoclasts were identified by staining for tartrate-resistant acid phosphatase (TRAP) using a kit (Sigma-Aldrich, St. Louis, MO). Recruitment of marrow stromal cells to the osteoblast lineage was assessed at 10 days of culture by staining for alkaline phosphatase (AP) using a kit (Sigma-Aldrich); cells were cultured in α-MEM supplemented with15 % preselected fetal calf serum (Hyclone, Logan, UT) and ascorbic acid-3-phosphoate (1 mM).
Isolation of total RNA and real-time PCR (qPCR)
Procedures for qPCR were performed as previously described [43]. qPCR was performed with an ABI Via7 thermal cycler using ABI Taqman 2X PCR mix and ABI Assay on Demand qPCR primers, except for regulator of calcineurin (RCAN) 1.4 which was detected using an ABI Assay by Design primer and probe set. The real time PCR strategy for RCAN1.4 focused on detecting the unique boundary between exons 4 and 5 in RCAN 1.4. Changes in expression were calculated using the 2−∆∆Ct method [44] using 18S RNA as the internal control.
Serum levels of CTX and osteocalcin
Serum CTX and osteocalcin were determined by EIA using commercially available kits according the manufacturers recommended procedures. CTX was assayed using a RatLaps kit from Immunodiagnostic Systems (Fountain Hills, AZ). For osteocalcin, an Osteocalcin EIA kit from Biomedical Technologies Inc. (Stoughton, MA) was used. All samples were assayed in duplicate.
Statistics
Data were expressed as mean ± SD. The number N for each group is noted in the legend of each figure. The statistical significance of differences among means was tested using one-way ANOVA followed by a Newman–Keuls test post hoc to examine the significant differences between individual pairs of means. Differences were considered significant at P < 0.05. Statistical calculations were performed using Prism 4.0c (Graphpad Software, La Jolla, CA).
Results
Tricep muscle weights were not significantly different between the three groups (Fig. 2a). Gastrocnemius mass was significantly reduced after SCI by approximately 13 % and was not changed by LIV (Fig. 2b).
Effects of LIV on muscle and bone. a and b Mass of triceps and gastrocnemius muscles in each group are shown. c BMD at the distal femur and proximal tibia were assessed by DXA. Data are expressed as mean ± SD. Sham, N = 8; SCI, N = 14; SCI-LIV, N = 13. **p < 0.01 and ***p < 0.001 for group comparisons as indicated
Prior work in humans and rodents suggests that most of the bone loss [2, 45] after SCI is trabecular and that trabecular bone loss is most extensive at the proximal tibia and distal femur. Consistent with these prior reports, we found that areal BMD was reduced by approximately 5 % at the distal femoral and proximal tibial metaphyses in all SCI animals (n = 27). The loss of BMD was similar for the left and right hindlimb (data not shown). LIV did not significantly alter areal BMD at these sites (Fig. 2c). Assessment of bone architecture by microCT analysis of the distal femoral metaphysis in the SCI only animals (n = 14) demonstrated that SCI reduced bone volume/total volume (BV/TV), trabecular number (Tb.N), trabecular thickness (Tb.Th), and connectivity density (Conn.D) by 23, 9, 13, and 3.5 %, respectively; SCI increased Tb.Sp and structure model index (SMI) by 20 and 36.7 %, respectively. LIV (n = 13) did not significantly alter these SCI-mediated changes in trabecular bone architecture (Fig. 3).
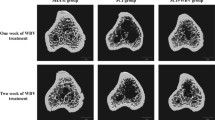
MicroCT examination of three-dimensional trabecular architecture. a Representative images are displayed of trabecular architecture at the distal femoral metaphysis from the respective groups (Sham, SCI, and SCI-LIV). b Plots of the structural parameters: relative bone volume (BV/TV [%]), trabecular thickness (Tb.Th), trabecular number (Tb.N), trabecular spacing (Tb.Sp), connectivity density (Conn.D), and structure model index (SMI). Data are expressed as mean ± SD. N = 4 to 5 animals per group. *p < 0.05 and **p < 0.01 for group comparisons as indicated
Serum levels of CTX were significantly (p < 0.05) elevated by 30 % in the SCI group and were not different when comparing SCI and SCI-LIV groups (Fig. 4a). An osteoclastogenesis assay using bone marrow precursors from femora and tibia revealed a twofold increase in TRAP+ multinucleated cells (CFU-oc) present in cultures of bone marrow cells from SCI rats compared to Sham-SCI rats (Fig. 4b). The number of osteoclasts in cultures from SCI-LIV animals was significantly (p < 0.05) reduced by approximately 70 % compared to that of SCI animals (Fig. 4b).
LIV protected against SCI-induced osteoclastogenesis in ex vivo cultures, but had no effect on serum CTX. a Serum CTX levels are shown. b Representative images of cultured osteoclasts and numbers of multinucleated TRAP+ cells in ex vivo cultures are shown. Data are expressed as mean ± SD. N = 8 for sham group, N = 14 for SCI group, and N = 13 for SCI-LIV group in a and N = 3 to 4 animals per group for b. *p < 0.05 and ***p < 0.001 for the comparison of the indicated pairs of groups
Serum osteocalcin was significantly (p < 0.05) reduced by SCI by 20 % and was increased by LIV to levels that were at least equal to those for the Sham group (Fig. 5a). The values for CFU-f were significantly reduced for the SCI group and not significantly altered by LIV (Fig. 5b). Expression of SOST messenger RNA (mRNA) in these cultured cells was increased twofold for SCI but decreased with LIV to levels equal to or lower than those for Sham (Fig. 6a (a)). Cellular mRNA levels of Wnt signaling target genes Runx2, OPG, and Tcf7 were lower for SCI compared to Sham animals (Fig. 6a (b–d)); LIV significantly increased Runx2 levels to those at or above levels observed in Sham animals. In addition, LIV induced a non-significant change in mRNA levels for Tcf7.
LIV protected against SCI-related reduction of serum osteocalcin but not osteoblast differentiation in ex vivo cultures. a Serum osteocalcin levels in each group are shown. b Representative images of alkaline phosphatase-stained cells and quantitation of stained cells (CFU-f) are shown. Data are expressed as mean ± SD. N = 8 for sham group, N = 14 for SCI group, and N = 13 for SCI-LIV group in a and N = 3 to 4 per group for b. *p < 0.05 and ***p < 0.001 for the comparison of the indicated pairs of groups
Gene expression in ex vivo cultured osteoblasts and gastrocnemius muscle. a (a – d). The mRNA levels for SOST, Runx2, OPG, and Tcf7 were determined by real-time PCR in ex vivo cultured osteoblasts. Data are expressed as mean ± SD. N = 3 to 4 per group. a p = 0.05 and *p < 0.05 for the comparison of the indicated pairs of groups. b (a – b). The mRNA levels for PGC-1α and RCAN1.4 were determined by real-time PCR in gastrocnemius muscle. Data are expressed as mean ± SD. Sham, N = 8; SCI, N = 14; SCI-LIV, N = 13. *p < 0.05 for the comparison of the indicated pairs of groups
mRNA levels of PGC-1α and RCAN1.4 in gastrocnemius muscle were also examined. A marked and significant reduction in RCAN1.4 was observed after SCI, whereas the more modest changes in PGC-1α did not reach significance. LIV tended to raise PGC-1α and RCAN 1.4 expression (Fig. 6b), although these changes were not statistically different.
Discussion
Bone and muscle loss after moderate severity contusion SCI
The MASCIS moderate severity contusion injury employed in this study results in a brief initial period of several days of severely impaired function followed by gradual though partial, recovery of motor function that permits weight support and some stepping [39]. Mild persistent motor deficits are typically observed. This model of SCI was chosen for the current study because it ensured that LIV would be administered together with gravitational loading with the expectation that when combined with the forces of muscle contraction and gravity, LIV would have a greater likelihood to attenuate or reverse the deleterious effects of SCI on bone. In the present study, at 63 days after moderate severity contusion SCI, there was modest, though significant, atrophy of gastrocnemius muscle and a significant decrease in expression level of RCAN1.4. RCAN1.4 is transcribed through an alternative promoter that has several NFAT consensus sites; levels of this transcript reflect calcineurin signaling through NFAT [46] which, in turn, is exquisitely sensitive to neuromuscular activity [47, 48]. Thus, these data are consistent with expected lowering of post-injury gastrocnemius muscle neuromuscular activity below that of Sham animals. Taken together, these data indicate that even the mild decrease in motor function caused by a moderate severity contusion SCI is sufficient to induce significant, albeit small, deterioration of sublesional skeletal muscle.
Consistent with the reduction in muscle mass observed after contusion SCI, a small decrement in tibial and femoral BMD was observed, with much more extensive loss of trabecular bone. This result differs from that of Voor et al. who found no significant reduction in trabecular or cortical bone at 2–3 or 8 weeks after employing a similar contusion SCI model to the one used herein [33]. However, these authors reported significant declines in trabecular and cortical bone at the proximal tibial metaphysis in a more severe SCI model at 2–3 weeks post-injury [33]. Also, marked bone loss was observed 10 days after a severe contusion injury in another report [13].
The decline in serum osteocalcin observed in our SCI group is consistent with reduced bone formation, and it is in agreement with a report that bone formation rate assessed by dynamic histomorphometry is reduced at 10 days after severe contusion SCI [13]. The decrease in the osteoblastogenic potential of bone marrow stromal cells after SCI is notable because it occurred even though weight bearing had been restored, albeit possibly for too limited a time before sacrifice for the full effects of reloading of the skeleton to have developed. A decrease in CFU-f has also been reported in studies of spinal cord transected rats [35, 40]. The elevation of serum CTX and osteoclastogenesis in cultured bone marrow cells in the SCI group indicates that accelerated bone resorption persists even after weight bearing and partial return of motor function resume in rats with motor-incomplete SCI. It might be predicted that even in these mildly injured animals, bone loss will continue for some time, albeit at a reduced rate compared to the initial few days and weeks after injury or to animals with a severe contusion injury or complete transection.
The analysis of TRAP stained sections of trabecular bone from the animals in our study revealed that at 63 days after SCI, there is a decrease in osteoclast number in distal femur (Supplemental Figure 1). The reasons for this unexpected decrease are unclear but may be related to effects of reloading bone by partial weight bearing or to the extensive loss of trabecular bone (23 % decrease in BV/TV % as determined by microCT, Fig. 3b) that occurred after SCI. This event might be indicative of the low degree of osteoclast activity in low turnover state of bone loss at 2 months after SCI, which is different from the high degree of osteoclast activity in high turnover state of bone loss earlier after SCI. Consistent with this interpretation, at 10 days post-injury, the number of mature osteoclasts was increase by 330 % at the growth plate in the injured rats, suggesting the increased osteoclastic resorption [13].
Effects of LIV
The primary benefits of LIV treatment when instituted at 29 days post-injury were increased serum osteocalcin and Runx2 expression in cultured osteoblasts, accompanied by reduced SOST expression; in addition, LIV reduced osteoclastogenesis. Despite these highly encouraging biochemical and cell culture findings after 35 days of LIV, BMD was not significantly increased nor were measures of trabecular bone or serum CTX levels improved. Specifically, LIV did not alter BMD at the distal femur or proximal tibia, and trends for LIV to increase trabecular bone, while consistent among measures, were non-significant. Because muscle contraction is the largest source of mechanical loading of bone [49, 50], it is also important to note that LIV did not appreciably alter muscle weights or expression of PGC-1α or RCAN1.4, demonstrating that LIV did not appear to have benefits to skeletal muscle, at least in this model of SCI. We note that LIV treatment was initiated at 4 weeks after injury, when much of the bone loss expected after SCI would have already occurred [12, 13]. Whether instituting LIV at an earlier time after injury or providing treatment for a longer post-injury interval would produce more favorable results is worthy of consideration. LIV, as applied, did not reduce serum CTX levels, suggesting that there was no reduction in bone resorptive activity. The lack of effect of LIV on serum CTX was somewhat unexpected because other forms of mechanical loading through electrical stimulation of neuromuscular contractions rapidly reduce levels of this serum marker of bone resorption [51]. The persistently elevated bone resorptive activity during the application of LIV would be anticipated to have attenuated any anabolic effect of LIV on bone.
Although LIV did not alter the osteoblastic potential of bone marrow stromal cells in our study, a finding that is discrepant from the observation that LIV improved the ability of marrow stromal cells to undergo osteoblast differentiation after hind limb unloading [24], LIV nonetheless enhanced gene expression in osteoblasts cultured from marrow stromal cells. The most notable of these effects in osteoblasts was that LIV reversed the immobilization-induced increases in SOST expression. The SOST gene encodes sclerostin, a potent inhibitor of Wnt signaling that is almost exclusively expressed in osteocytes [52, 53]. Loss of function or deletion mutations of SOST result in high bone mass in humans [54, 55]. Expression levels of SOST are inversely related to skeletal loading [56]. Sclerostin has been found to stimulate osteocytes to release RANKL (receptor activator of NF-kB ligand) [57], a TNF family member responsible for differentiation and activation of osteoclasts [58]. It is possible that LIV reduced SOST expression in bone, thereby reducing RANKL expression and diminishing stimulation of the pool of hematopoietic cells capable of osteoclastic differentiation, a posit supported by the finding that LIV reduced the CFU-oc of bone marrow cells from SCI animals. LIV also significantly increased the expression of the Wnt/β-catenin responsive gene Runx2 and showed trends to upregulate that of two other Wnt-responsive genes, OPG and Tcf7, both of which were downregulated by SCI. Taken together, these findings suggest that LIV activates Wnt signaling in ex vivo cultured osteoblasts from unloaded bone after SCI, in part, through reduced SOST expression. If so, these changes would favor increased BMD by stimulating bone formation and reducing bone resorption.
Consistent with these cellular effects, LIV increased osteocalcin levels in serum, indicating greater rates of bone formation. However, as LIV was applied to the entire skeleton, our data do not permit an assessment of regional cellular responses. Our findings raise the interesting possibility that LIV provides sufficient bone loading to downregulate SOST expression, although specific examination of osteocyte SOST expression will be necessary to verify this possibility.
We found no effect of LIV on muscle atrophy after SCI or on expression of two activity-dependent genes, PGC-1α and RCAN1.4. While these findings suggest that there was no effect of LIV on muscle tissue, when interpreting these findings, it should be noted that improvements in neuromuscular function might increase muscle strength without any change in muscle volume or cross sectional area. Consistent with this interpretation in both normal and activity-restricted mice, vibration increased maximal torque of anterior crural muscles without significant changes in muscle size [59].
As a limitation of the study, we do not have specific measurement regarding transmission of vibration through the skeleton. However, data from Bauman and coworkers [30] suggests that efficient transmission of vibration through the skeleton is achieved in individuals with SCI with even partial loading of the skeleton accomplished by elevation of the head using a tilt table. In our study, LIV was initiated at a time when the ability to weight bear had returned suggesting that considerable transmission of vibrations through the skeleton did occur.
Conclusions
Significant loss of BMD occurred at the distal femur and proximal tibia at 63 days after a moderate severity contusion SCI in rats that was associated with a substantial decline of trabecular bone mass and structure. When initiated 29 days after a contusion injury and continued for 35 days, LIV elevated serum osteocalcin levels and partially corrected the increased osteoclastogenesis in ex vivo cultures and defects in the gene expression of bone marrow cells undergoing osteoblastic differentiation. Although some weight bearing and locomotion had recovered in these SCI animals at the time of LIV, bone resorption continued and was associated with increased osteoclastic potential of bone marrow cells. Whether LIV initiated earlier after SCI and/or continued for a longer duration would have increased bone mass is a possibility that could be addressed in future studies.
References
Dudley-Javoroski S, Shields RK (2008) Muscle and bone plasticity after spinal cord injury: review of adaptations to disuse and to electrical muscle stimulation. J Rehabil Res Dev 45:283–296
Qin W, Bauman WA, Cardozo C (2010) Bone and muscle loss after spinal cord injury: organ interactions. Ann N Y Acad Sci 1211:66–84
Biering-Sorensen B, Kristensen IB, Kjaer M, Biering-Sorensen F (2009) Muscle after spinal cord injury. Muscle Nerve 40:499–519
Qin W, Bauman WA, Cardozo CP (2010) Evolving concepts in neurogenic osteoporosis. Curr Osteoporos Rep 8:212–218
Bauman WA, Cardozo C (2013) Spinal cord injury: pathophysiology and clinical issues. In: Rosen C (ed) Primer on the metabolic bone diseases and disorders of bone metabolism, 8th edn. American Society for Bone and Mineral Research, Washington D.C., pp 1018–1027
Giangregorio L, McCartney N (2006) Bone loss and muscle atrophy in spinal cord injury: epidemiology, fracture prediction, and rehabilitation strategies. J Spinal Cord Med 29:489–500
Dauty M, Perrouin Verbe B, Maugars Y, Dubois C, Mathe JF (2000) Supralesional and sublesional bone mineral density in spinal cord-injured patients. Bone 27:305–309
Logan WC Jr, Sloane R, Lyles KW, Goldstein B, Hoenig HM (2008) Incidence of fractures in a cohort of veterans with chronic multiple sclerosis or traumatic spinal cord injury. Arch Phys Med Rehabil 89:237–243
Garland DE, Adkins RH, Kushwaha V, Stewart C (2004) Risk factors for osteoporosis at the knee in the spinal cord injury population. J Spinal Cord Med 27:202–206
Akhigbe T, Chin AS, Svircev JN, Hoenig H, Burns SP, Weaver FM, Bailey L, Carbone L (2013) A retrospective review of lower extremity fracture care in patients with spinal cord injury. J Spinal Cord Med
Morse LR, Battaglino RA, Stolzmann KL, Hallett LD, Waddimba A, Gagnon D, Lazzari AA, Garshick E (2009) Osteoporotic fractures and hospitalization risk in chronic spinal cord injury. Osteoporos Int 20:385–392
Jiang SD, Jiang LS, Dai LY (2007) Changes in bone mass, bone structure, bone biomechanical properties, and bone metabolism after spinal cord injury: a 6-month longitudinal study in growing rats. Calcif Tissue Int 80:167–175
Morse L, Teng YD, Pham L, Newton K, Yu D, Liao WL, Kohler T, Muller R, Graves D, Stashenko P, Battaglino R (2008) Spinal cord injury causes rapid osteoclastic resorption and growth plate abnormalities in growing rats (SCI-induced bone loss in growing rats). Osteoporos Int 19:645–652
Rubin C, Turner AS, Bain S, Mallinckrodt C, McLeod K (2001) Anabolism. Low mechanical signals strengthen long bones. Nature 412:603–604
Xie L, Rubin C, Judex S (2008) Enhancement of the adolescent murine musculoskeletal system using low-level mechanical vibrations. J Appl Physiol 104:1056–1062
Ward K, Alsop C, Caulton J, Rubin C, Adams J, Mughal Z (2004) Low magnitude mechanical loading is osteogenic in children with disabling conditions. J Bone Miner Res 19:360–369
Reyes ML, Hernandez M, Holmgren LJ, Sanhueza E, Escobar RG (2011) High-frequency, low-intensity vibrations increase bone mass and muscle strength in upper limbs, improving autonomy in disabled children. J Bone Miner Res 26:1759–1766
Rubin C, Recker R, Cullen D, Ryaby J, McCabe J, McLeod K (2004) Prevention of postmenopausal bone loss by a low-magnitude, high-frequency mechanical stimuli: a clinical trial assessing compliance, efficacy, and safety. J Bone Miner Res 19:343–351
Beck BR, Norling TL (2010) The effect of 8 mos of twice-weekly low- or higher intensity whole body vibration on risk factors for postmenopausal hip fracture. Am J Phys Med Rehabil 89:997–1009
Von Stengel S, Kemmler W, Bebenek M, Engelke K, Kalender WA (2011) Effects of whole-body vibration training on different devices on bone mineral density. Med Sci Sports Exerc 43:1071–1079
von Stengel S, Kemmler W, Engelke K, Kalender WA (2011) Effects of whole body vibration on bone mineral density and falls: results of the randomized controlled ELVIS study with postmenopausal women. Osteoporos Int 22:317–325
Garman R, Gaudette G, Donahue LR, Rubin C, Judex S (2007) Low-level accelerations applied in the absence of weight bearing can enhance trabecular bone formation. J Orthop Res 25:732–740
Judex S, Lei X, Han D, Rubin C (2007) Low-magnitude mechanical signals that stimulate bone formation in the ovariectomized rat are dependent on the applied frequency but not on the strain magnitude. J Biomech 40:1333–1339
Ozcivici E, Luu YK, Rubin CT, Judex S (2010) Low-level vibrations retain bone marrow’s osteogenic potential and augment recovery of trabecular bone during reambulation. PLoS ONE 5:e11178
Ness LL, Field-Fote EC (2009) Whole-body vibration improves walking function in individuals with spinal cord injury: a pilot study. Gait Posture 30:436–440
Herrero AJ, Menendez H, Gil L, Martin J, Martin T, Garcia-Lopez D, Gil-Agudo A, Marin PJ (2011) Effects of whole-body vibration on blood flow and neuromuscular activity in spinal cord injury. Spinal Cord 49:554–559
Ness LL, Field-Fote EC (2009) Effect of whole-body vibration on quadriceps spasticity in individuals with spastic hypertonia due to spinal cord injury. Restor Neurol Neurosci 27:621–631
Murillo N, Kumru H, Vidal-Samso J, Benito J, Medina J, Navarro X, Valls-Sole J (2011) Decrease of spasticity with muscle vibration in patients with spinal cord injury. Clin Neurophysiol 122:1183–1189
Bauman WA, Schnitzer TJ, Chen D (2010) Management of osteoporosis after spinal cord injury: what can be done? Point/counterpoint. PM R 2:566–572
Asselin P, Spungen AM, Muir JW, Rubin CT, Bauman WA (2011) Transmission of low-intensity vibration through the axial skeleton of persons with spinal cord injury as a potential intervention for preservation of bone quantity and quality. J Spinal Cord Med 34:52–59
Davis R, Sanborn C, Nichols D, Bazett-Jones DM, Dugan EL (2010) The effects of whole body vibration on bone mineral density for a person with a spinal cord injury: a case study. Adapt Phys Act Q: APAQ 27:60–72
Pinzon A, Marcillo A, Quintana A, Stamler S, Bunge MB, Bramlett HM, Dietrich WD (2008) A re-assessment of minocycline as a neuroprotective agent in a rat spinal cord contusion model. Brain Res 1243:146–151
Voor MJ, Brown EH, Xu Q, Waddell SW, Burden RL, Burke DA, Magnuson DS (2012) Bone loss following spinal cord injury in a rat model. J Neurotrauma 29:1676
Wirth F, Schempf G, Stein G, Wellmann K, Manthou M, Scholl C, Sidorenko M, Semler O, Eisel L, Harrach R, Angelova S, Jaminet P, Ankerne J, Ashrafi M, Ozsoy O, Ozsoy U, Schubert H, Abdulla D, Dunlop SA, Angelov DN, Irintchev A, Schonau E (2013) Whole-body vibration improves functional recovery in spinal cord injured rats. J Neurotrauma 30:453–468
Sun L, Pan J, Peng Y, Wu Y, Li J, Liu X, Qin Y, Bauman WA, Cardozo C, Zaidi M, Qin W (2013) Anabolic steroids reduce spinal cord injury-related bone loss in rats associated with increased Wnt signaling. J Spinal Cord Med 36:616–622
Xie L, Jacobson JM, Choi ES, Busa B, Donahue LR, Miller LM, Rubin CT, Judex S (2006) Low-level mechanical vibrations can influence bone resorption and bone formation in the growing skeleton. Bone 39:1059–1066
Wysocki A, Bulter M, Shamliyan T, Kane R (2011) Whole-body vibration therapy for osteoporosis. AHRQ Publication No. 11(12)-EHC083-EF
Sen B, Xie Z, Case N, Styner M, Rubin CT, Rubin J (2011) Mechanical signal influence on mesenchymal stem cell fate is enhanced by incorporation of refractory periods into the loading regimen. J Biomech 44:593–599
Basso DM, Beattie MS, Bresnahan JC (1996) Graded histological and locomotor outcomes after spinal cord contusion using the NYU weight-drop device versus transection. Exp Neurol 139:244–256
Qin W, Sun L, Cao J, Peng Y, Collier L, Wu Y, Creasey G, Li J, Qin Y, Jarvis J, Bauman WA, Zaidi M, Cardozo C (2013) The central nervous system (CNS)-independent anti-bone-resorptive activity of muscle contraction and the underlying molecular and cellular signatures. J Biol Chem 288:13511–13521
Cardozo CP, Qin W, Peng Y, Liu X, Wu Y, Pan J, Bauman WA, Zaidi M, Sun L (2010) Nandrolone slows hindlimb bone loss in a rat model of bone loss due to denervation. Ann N Y Acad Sci 1192:303–306
Parfitt AM, Drezner MK, Glorieux FH, Kanis JA, Malluche H, Meunier PJ, Ott SM, Recker RR (1987) Bone histomorphometry: standardization of nomenclature, symbols, and units. Report of the ASBMR Histomorphometry Nomenclature Committee. J Bone Miner Res 2:595–610
Wu Y, Zhao J, Zhao W, Pan J, Bauman WA, Cardozo CP (2012) Nandrolone normalizes determinants of muscle mass and fiber type after spinal cord injury. J Neurotrauma 29:1663–1675
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408
Bauman WA, Cardozo C (2013) Immobilization osteoporosis. In: Marcus R, Nelson D, Rosen CJ (eds) Osteoporosis, 4th edn. Academic
Ni YG, Berenji K, Wang N, Oh M, Sachan N, Dey A, Cheng J, Lu G, Morris DJ, Castrillon DH, Gerard RD, Rothermel BA, Hill JA (2006) Foxo transcription factors blunt cardiac hypertrophy by inhibiting calcineurin signaling. Circulation 114:1159–1168
Bassel-Duby R, Olson EN (2006) Signaling pathways in skeletal muscle remodeling. Annu Rev Biochem 75:19–37
Schiaffino S (2010) Fibre types in skeletal muscle: a personal account. Acta Physiol (Oxf) 199:451–463
Schoenau E (2005) From mechanostat theory to development of the “functional muscle-bone-unit”. J Musculoskelet Neuronal Interact 5:232–238
Rittweger J, Beller G, Ehrig J, Jung C, Koch U, Ramolla J, Schmidt F, Newitt D, Majumdar S, Schiessl H, Felsenberg D (2000) Bone-muscle strength indices for the human lower leg. Bone 27:319–326
Arija-Blazquez A, Ceruelo-Abajo S, Diaz-Merino MS, Godino-Duran JA, Martinez-Dhier L, Florensa-Vila J (2013) Time-course response in serum markers of bone turnover to a single-bout of electrical stimulation in patients with recent spinal cord injury. Eur J Appl Physiol 113:89–97
Poole KE, van Bezooijen RL, Loveridge N, Hamersma H, Papapoulos SE, Lowik CW, Reeve J (2005) Sclerostin is a delayed secreted product of osteocytes that inhibits bone formation. FASEB J 19:1842–1844
Ke HZ, Richards WG, Li X, Ominsky MS (2012) Sclerostin and Dickkopf-1 as therapeutic targets in bone diseases. Endocr Rev 33:747–783
Brunkow ME, Gardner JC, Van Ness J, Paeper BW, Kovacevich BR, Proll S, Skonier JE, Zhao L, Sabo PJ, Fu Y, Alisch RS, Gillett L, Colbert T, Tacconi P, Galas D, Hamersma H, Beighton P, Mulligan J (2001) Bone dysplasia sclerosteosis results from loss of the SOST gene product, a novel cystine knot-containing protein. Am J Hum Genet 68:577–589
Balemans W, Patel N, Ebeling M, Van Hul E, Wuyts W, Lacza C, Dioszegi M, Dikkers FG, Hildering P, Willems PJ, Verheij JB, Lindpaintner K, Vickery B, Foernzler D, Van Hul W (2002) Identification of a 52 kb deletion downstream of the SOST gene in patients with van Buchem disease. J Med Genet 39:91–97
Robling AG, Niziolek PJ, Baldridge LA, Condon KW, Allen MR, Alam I, Mantila SM, Gluhak-Heinrich J, Bellido TM, Harris SE, Turner CH (2008) Mechanical stimulation of bone in vivo reduces osteocyte expression of Sost/sclerostin. J Biol Chem 283:5866–5875
Wijenayaka AR, Kogawa M, Lim HP, Bonewald LF, Findlay DM, Atkins GJ (2011) Sclerostin stimulates osteocyte support of osteoclast activity by a RANKL-dependent pathway. PLoS ONE 6:e25900
Kearns AE, Khosla S, Kostenuik PJ (2008) Receptor activator of nuclear factor kappaB ligand and osteoprotegerin regulation of bone remodeling in health and disease. Endocr Rev 29:155–192
McKeehen JN, Novotny SA, Baltgalvis KA, Call JA, Nuckley DJ, Lowe DA (2013) Adaptations of mouse skeletal muscle to low-intensity vibration training. Med Sci Sports Exerc 45:1051–1059
Acknowledgments
This work was supported by the Veterans Health Administration, Rehabilitation Research and Development Service (B9212-C and B0687-R), Biomedical Laboratory Research and Development Service (BX000521), the Department of Defense (#SC090504), and The Miami Project to Cure Paralysis. We wish to thank Drs. Edelle Field-Fote and Mark Nash for critical reading of the manuscript.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 18 kb)
Supplemental Figure 1
Histomorphometric analysis of bone at 63 days after a moderate mid-throacic contusion SCI. A. Representative sections of trabecular bone from the femoral metaphysis immunostained for TRAP. Note the reddish areas of TRAP+ staining on trabecular surfaces representing osteoclasts. B. Histomorphometric quantification of osteoclast numbers and surface: osteoclast surface per bone surface (Oc.S/Bs %); surface eroded by osteoclasts per bone surface (ES /Bs%), and number of osteoclasts per tissue area of interest (N. Oc/ T. Ar, Oc/mm2). Data are expressed as mean ± SEM. N = 5–6 per group. Significance of differences was determined using one-way ANOVA with a Newman–Keuls test post hoc. **P < 0.01 versus the indicated group; NS. No statistic significance. (GIF 277 kb)
Rights and permissions
About this article
Cite this article
Bramlett, H.M., Dietrich, W.D., Marcillo, A. et al. Effects of low intensity vibration on bone and muscle in rats with spinal cord injury. Osteoporos Int 25, 2209–2219 (2014). https://doi.org/10.1007/s00198-014-2748-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-014-2748-8