Abstract
Introduction and hypothesis
This study evaluated factors and their prevalence associated with urinary (UI) and fecal (FI) incontinence during and after a woman’s first pregnancy.
Methods
Nulliparous Chinese women with no UI or FI before pregnancy were studied with a standardized questionnaire for UI and FI from early pregnancy until 12 months after childbirth. Maternal characteristics and obstetric data were analyzed using descriptive analysis, independent sample t test, chi-square test, and logistic regression.
Results
Three hundred and twenty-eight (74.2 %) women completed the study. The prevalence of antenatal UI increased with gestation. Overall, 192 (58.5 %), 60 (18.3 %), and 76 (23.1 %) had normal vaginal delivery, instrumental delivery, and cesarean section, respectively. Twelve months after delivery, prevalence of stress urinary incontinence (SUI) and urge urinary incontinence (UUI) was 25.9 % [95 % confidence interval (CI) 21.5–30.6] and 8.2 % (95 % CI 5.2–11.2), respectively. In those who delivered vaginally, the prevalence was 29.7 % and 9.1 %, respectively. Prevalence of FI was 4.0 % (95 % CI 1.9–6.1). On logistic regression, vaginal delivery [odds ratio (OR) 3.6], antenatal SUI (OR 2.8), and UUI (OR 2.4) were associated with SUI. Antenatal UUI (OR 6.4) and increasing maternal body mass index (BMI) at the first trimester (OR 1.2) were associated with UUI. Antenatal FI was associated with FI (OR 6.1).
Conclusions
The prevalence of SUI, UUI, and FI were 25.9 %, 8.2 %, and 4.0 %, respectively, 12 months after delivery. Vaginal delivery, antenatal SUI, and UUI were associated with SUI; antenatal UUI and increasing maternal BMI at the first trimester were associated with UUI. Antenatal FI was associated with FI. Pregnancy, regardless of route of delivery and obstetric practice, had an effect on UI and FI.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
There appears to be a relationship between vaginal delivery (VD) and/or pregnancy and pelvic floor disorders [1–7]. Urinary incontinence (UI), a pelvic floor disorder, is common, with an estimated prevalence of 16–60 % during pregnancy and 16–34 % after spontaneous and instrumental VD [1–4]. Fecal incontinence (FI) is reported by 0.7–22 % of women soon after delivery and subsequently up to 3–4 years later [2, 5–7]. However, Burgio et al. reported that there was no difference in parity between perimenopausal continent and incontinent women [8]. Those who delivered vaginally without an anal sphincter tear and those delivered by cesarean section (CS) prior to labor were not completely protected from the development of new FI [9]. The precise relationship between pregnancy and the development of UI and FI remains to be definitively elucidated.
Most studies have focused on the effect of VD and mainly studied symptoms during the third trimester of pregnancy and at 6 weeks to 3 months after delivery. There is limited information on the antenatal period [1, 5]. Furthermore, the majority of women in previous studies have been Caucasian. Differences in pelvic connective tissue quality between Chinese and Caucasian women have previously been suggested [10, 11]. This ethnic difference has recently been emphasized by the documentation that Asian women have significantly less pelvic organ mobility, both during and after pregnancy, compared with Caucasians [12]. Past reports have also been characterized by differences in obstetric practice, including differing rates of CS, epidural analgesia, and prevalence of episiotomy, which was about 20 % in some studies compared with >70 % in primiparous women in our locality [13–15]. All these factors may influence the prevalence of UI and FI during and after pregnancy. We report a longitudinal study of UI and FI in women that commenced early in their pregnancy through to 1 year after delivery. The aim was to assess the prevalence of UI and FI in women who had no UI or FI prior to pregnancy. The study started in early pregnancy and continued after first delivery and used standard questionnaires. Potential factors that may affect the prevalence of UI or FI, such as maternal characteristics, obstetric parameters, and outcomes, were also studied.
Materials and methods
This was a prospective observational study of nulliparous Chinese women recruited in a tertiary obstetric unit during the first trimester (9–12 weeks of gestation) from August 2009 to September 2010. An experienced research assistant explained the study to women, who were asked if they had UI, FI, or prolapse symptoms within 1 year before the pregnancy. Written informed consent was obtained. Inclusion criteria were nulliparous women of Chinese ethnicity with a singleton pregnancy who had no known UI, FI, or prolapse symptoms prior to pregnancy. Exclusion criteria were <18 years, non-Chinese ethnicity, and those who refused to participate. Participants were seen in the first (9–12 weeks of gestation), second (26–28 weeks of gestation), and third (35–38 weeks of gestation) trimesters and were followed up at 8 weeks, 6 months, and 12 months after delivery. At the first visit, demographic data were obtained, and maternal height and weight were measured; thereafter, only maternal body weight was measured. Symptoms and severity of pelvic floor disorders, including UI and FI, were studied using validated Chinese Pelvic Floor Distress Inventory (PFDI) during each visit [16]. A response of “yes” to Urinary Distress Inventory Stress Subscale questions 20–22 regarding urine leakage related to coughing, sneezing, or laughing, and physical exercise such as walking, running, aerobics, lifting, or bending over were regarded as presence of stress urinary incontinence (SUI). A response of “yes” to question 19 regarding urine leakage associated with a feeling of urgency was regarded as the presence of urge urinary incontinence (UUI). Mixed UI (MUI) was defined as the presence of both SUI and UUI; and any UI was defined as the presence of either SUI or UUI. In addition, a response of “yes” to Colorectal Anal Distress Inventory Incontinence Subscale questions 38–40 regarding lose stool beyond one’s control if the stool is well formed or loose or liquid were regarded as symptoms of FI with solid or loose stool, respectively. Losing gas from the rectum beyond one’s control is regarded as flatus incontinence. The terminology used in this manuscript follows the International Urogynecologic Association (IUGA)/International Continence Society (ICS) joint report on terminology for female pelvic floor dysfunction [17]. Women were also asked whether symptoms, if present, bothered them. Investigators were blinded to the PFDI data until the study was completed.
Women were managed according to departmental protocols, which were formulated according to principles of evidence-based medicine. These protocols are detailed on the departmental Intranet. Monthly audits are conducted of all deliveries, and deviations from protocol are discussed. Data on obstetric outcomes were obtained from the computerized central medical records when the women were discharged from hospital. Delivery mode was defined as VD, including normal VD (NVD), instrumental delivery, CS, elective CS (ElCS) prior to onset of labor, and emergency CS (EmCS) performed after the onset of labor. After delivery, all women attended a postnatal talk before discharge and were taught how to do pelvic floor exercise (PFE). It was left to them to do these exercises according to their own needs. At each postnatal visit, they were asked if they had performed PFE and if so, how frequently. They were regarded as performing PFE if they practiced it at least two times per week.
Sample size calculation
Assuming the prevalence of UI is 25 % after VD, with an accepted error of 5 %, a sample of 263 participants would be required. In addition, we estimated that only 75 % of participants would achieve a VD and that there would be a dropout rate of 20 %, implying that in all we would need to recruit 438 study participants.
Statistical analysis
Descriptive analysis was used to study the prevalence of UI and FI during and after delivery. Indices measured in two groups were compared using the independent sample t test. For comparison of frequencies, chi-square test or a two-sided Fisher exact test was used, and chi-square test for trend was used where appropriate. Logistic regression was used to assess the impact of factors, e.g., maternal age, body mass index (BMI) at first trimester or at 12 months after delivery, antenatal SUI or UUI, and delivery mode on the likelihood of SUI, UUI, and FI at 12 months after delivery. P < 0.05 was considered statistically significant.
Ethics approval
Ethics approval for the study was granted by the local institute. (CRE -2009.257)
Results
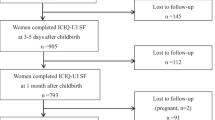
Four hundred and seventy-four (474) women were invited during the first trimester of their pregnancy to participate in the study; 32 declined. Therefore, 442 women were recruited; 328 (74.2 %) completed the study. A total of 114 (25.8 %) withdrew from the study; there was a second trimester miscarriage, 32 delivered in another hospital, and 15 were lost to follow-up after delivery. Among the rest, 29 were followed up to 8 weeks only and 29 up to 6 months after delivery. Eight were pregnant again by the time of their 1-year follow-up. For those completing the study, mean age was 30.6 ± 3.8 years. Mean BMI at first trimester was 21.0 ± 2.8 kg/m2. There was no difference in maternal characteristics between those completing the study and those who delivered in other hospitals. There were also no significant differences between those completing the study and those who failed to follow-up after delivery regarding maternal age, BMI, gestation at delivery, delivery mode, infant birth weight, and head circumference.
The following analysis included only those who completed the study. The prevalence of UI and FI during pregnancy is shown in Table 1. There was no significant difference in antenatal prevalence of UI and FI between those completing the study and those who failed to follow-up or who were delivered in other hospitals. The prevalence of SUI, UUI, and MUI was significantly increased with more advanced gestation (X 2 for trend 67.4, 19.6, and 20.2 respectively, all P < 0.005); this increasing trend was not observed for any type of FI. Women who reported antenatal SUI or UUI were significantly older than those without these symptoms (31.9 ± 3.9 vs 29.7 ± 3.5 years, respectively, P < 0.001; and 31.8 ± 3.8 vs 30.4 ± 3.9, respectively, P = 0.012), but this was not associated with antenatal FI. Maternal weight and BMI of women who reported antenatal FI were significantly greater than those without the symptom (22.4 ± 3.2 vs 20.9 ± 2.7 kg, respectively, P = 0.008; 56.1 ± 8.3 vs 52.3 ± 7.4 kg/m2, respectively, P = 0.01). Weight gain during pregnancy was not associated with antenatal UI or FI.
Overall, 192 (58.5 %), 60 (18.3 %), 13 (4 %), and 63 (19.2 %) women had NVD, instrumental delivery (46 ventous extraction (VE) and 14 forceps delivery (FrD)), ElCS, and EmCS, respectively. Mean gestation at delivery was 39.2 ± 1.8 weeks, and mean birth weight was 3.09 ± 0.47 kg. Intrapartum epidural analgesia was used by 24 (9.4 %) of those having VD; left mediolateral episiotomy was made in 242 (96 %), 94.8 % of NVD, and all instrumental deliveries. In those having VD, 39 and ten, respectively, had a first- or second-degree vaginal tear. There was no third- or fourth-degree vaginal tears documented. Among those who had EmCS, three of eight women suffered secondary arrest of labor at cervical dilatation 8 cm and presenting part at the ischial spine level; one woman had labor arrested at the second stage, with presenting part at the ischial spine level. No instrumental VD was attempted in this woman.
The prevalence of UI and FI after delivery is also shown in Table 1; prevalence according to delivery mode is shown in Table 2. The prevalence of SUI and UUI was 25.9 % [95 % confidence interval (CI) 21.5–30.6] and 8.2 % (95 % CI 5.2–11.2) at 12 months after delivery. Respective prevalence in VD was 29.7 % (95 % CI 24.1–35.3) and 9.1 % (95 % CI 5.6–12.7). The prevalence of SUI, UUI, and any UI at 8 weeks, 6 months, and 12 months after delivery were all significantly lower than that found in the third trimester of pregnancy. However, there was no difference in prevalence of mixed UI at 12 months after delivery when compared with the third trimester. Twenty-seven (44.2 %) women who reported SUI at 8 weeks reported that their symptoms had resolved by 12 months. Of the 85 who reported SUI at 12 months, 33 (38.8 %) had reported such symptoms since 8 weeks after delivery. Only 11 (12.9 %) and two (7.4 %) of those who reported SUI and UUI at 12 months regarded their symptoms as moderate or severe. The prevalence of FI of solid or liquid or loose stool at 12 months was 4.0 % (95 % CI 1.9–6.1), and the majority reported FI of liquid or loose stool. Only one reported that the FI bothered her to a moderate degree. At 6 and 12 months after delivery, 62 (19.9 %) and 55 (16.8 %) women respectively reported that they performed PFE. There were significantly more women performing PFE if they reported SUI at 12 months after delivery [22 (22.8 %) vs 33 (14.3 %), p = 0.01), but there was no difference if they reported UUI or FI.
Vaginal delivery was associated with a higher prevalence of SUI and any UI after delivery. It was also associated with a higher prevalence of UUI and MUI at 8 weeks but not at 6 or 12 months after delivery. However, delivery mode was not associated with any type of FI. Factors affecting SUI and UUI are listed in Table 3. Direct logistic regression was performed to assess the impact on the likelihood of SUI of six factors found to be significant on univariable analysis. The full model was statistically significant, [n = 328, x 2 (6) = 53.3, P < 0.005]. Only three factors made a statistically significant contribution to the model. The strongest predictor was VD [odds ratio (OR) 3.6], followed by antenatal SUI (OR 2.8), and antenatal UUI (OR 2.4) (Table 4). Direct logistic regression was also performed to assess the impact of four factors on the likelihood of UUI [n = 328, x 2 (4) = 31.7, P < 0.005)] The strongest predictor of UUI at 12 months after delivery was antenatal UUI (OR 6.4) and maternal BMI at first trimester of pregnancy (OR 1.2) (Table 4).
Only maternal BMI at the first trimester (23.1 ± 4.1 vs 21.0 ± 2.7, P < 0.008), antenatal FI (45.5 % vs 7.1 %, P = 0.001), and antenatal SUI (53.8 % vs 24.9 %, P = 0.046) were associated with FI of solid, or liquid or loose stool at 12 months after delivery. All women who reported FI of solid, or liquid or loose stool at 12 months and who delivered vaginally had an episiotomy. Operative VD or perineal tear were not associated with FI. Logistic regression indicated that only antenatal FI increased the likelihood of FI at 12 months after delivery, with an OR of 6.1 (95 % CI 1.75–21.5, P = 0.005).
Discussion
Pelvic floor disorders, including UI and FI, have been shown to be associated with childbirth. However, they could be due to pregnancy, labor, and/or the delivery process. A longitudinal study recruiting only women with no known UI, FI, or prolapse symptoms prior to pregnancy is the best way to elucidate the relationship between incontinence and pregnancy and childbirth. This prospective longitudinal observational study had an adequate sample to study the prevalence of UI and FI. There were no statistical differences between women completing the study and those who did not. The default rate was considered acceptable or lower compared with other large-scale studies, especially in view of the fact that we were able to follow-up the women 1 year after delivery [7, 13]. The obstetric characteristic of the study population, such as age, BMI, rate of intrapartum epidural analgesia, delivery mode, and episiotomy, were comparable with monthly or annual data at our institution and other published data and the audit report of Hong Kong College of Obstetricians and Gynaecologists. It is unlikely that the study sample was significantly biased, and our results can be generalized to Chinese women with similar maternal characteristics and delivering in institutions with similar obstetric practice [14, 18, 19].
The prevalence of antenatal UI during first, second, and third trimester of pregnancy was similar to the respective prevalence of 8.3 %, 32 %, and 35 % in nulliparous Caucasian women who were continent before pregnancy [7]. The prevalence of antenatal SUI and UUI at different trimesters was also similar. In Solans-Domenech et al.’s and our study, about 52–53 % of women remained continent [7]. Brown et al. reported only 46 % of women remained continent by 32 weeks of gestation, and it is expected that the continence rate will be even lower by the late third trimester of pregnancy [3]. However, the prevalence of antenatal MUI was higher in our study, and it was also significantly more prevalent at the third trimester compared with the first and second trimesters. Zhu et al. reported the prevalence of UI, SUI, UUI, and MUI to be 26.7 %, 18.6 %, 2.0 %, and 4.3 %, respectively, in Chinese women during late pregnancy [4]; their prevalence was lower than in our finding. This could be due to a younger population—26.4 years in their study. Age was confirmed to be a risk factor for antenatal UI in both studies.
The prevalence of antenatal FI of formed stool was similar to other studies, but the prevalence of FI of liquid or loose stool and flatus incontinence was higher in our study [7]. We identified increasing maternal weight and higher BMI as risk factors, but not age or weight gain during pregnancy; age >35 years and excessive weight gain during pregnancy were associated with FI in a previous study [7]. It should be pointed out that our antenatal population had a substantially lower BMI than those reported in Caucasian populations.
The prevalence of postpartum UI and different types of UI after the first delivery was similar to the pooled prevalence of UI 25.5 %, SUI 12 %, and UUI 3 % from previous Caucasian studies, though most reported the prevalence for 6 weeks to 6 months after delivery [2, 7, 20–22]. The reported prevalence by Yang et al. of incontinence in Chinese women was lower than in our study, which could be due to a high CS rate of 55 % compared with 23 % in our study [6]. There was a trend of increasing SUI prevalence from 8 weeks to 12 months after delivery, though it did not reach statistical significance. There was no difference in the prevalence of UI, SUI, UUI, or antenatal SUI and UUI in women completing the study compared with those who did not follow-up at 6 or 12 months. Therefore, this could not account for the trend. There was also no difference in the proportion of women performing PFE at 6 months or 12 months after delivery to explain the trend. There is scant information in the published literature to compare with our 12-month data. Arya et al. showed UI decreased significantly from 2 weeks to 1 year in normal VD and VE groups but not in the forceps delivery group; Serati et al. showed UI was 27 % and 23 % at 6 and 12 months, respectively, after delivery [23, 24].
VD, including normal and instrumental delivery; antenatal SUI; and UUI were risk factors for SUI at 12 months, whereas antenatal UUI and higher maternal BMI at the first trimester were factors for UUI at 12 months after delivery. Many previous studies also reported VD as a risk factor for postpartum UI or SUI [4, 6, 19, 25]. However, other obstetric factors, such as duration of labor and second-stage, infant’s birth weight, and episiotomy, were not associated with SUI or UUI.
Antenatal UI was also found to be a risk factor for postnatal UI, and the risk of UI was higher among women who had CS or VD than among nulliparous women [4, 7, 22, 26, 27]. These findings suggest that the pathophysiology of UI, SUI, or UUI begins during pregnancy, before the onset of labor or delivery [26]. Our findings only identified greater maternal age as being associated with both antenatal SUI and UUI. This factor cannot be altered, and indeed, the proportion of pregnant women of advanced maternal age is increasing [14]. Maternal BMI, identified as a risk factor for postnatal UI in other studies, was not a risk factor in the our study, which may be due to a much lower BMI of women in this study [6, 20].
The prevalence of postnatal FI with formed stool or liquid/loose stool was 0.3 % and 4 %, respectively, which is similar to the reported prevalence by Chaliha et al., but it was higher than those reported by Solans-Domenech et al. and Yang et al. [6, 7, 21]. The prevalence remained the same during the 12 months after delivery, whereas that of flatus incontinence decreased (Table 1). Only one risk factor, antenatal FI, was identified. The effect of episiotomy on postnatal FI remains controversial [6, 28, 29]. There was limited local data for comparison, and further research is needed. This is of great interest because of the high prevailing rates of episiotomy in local nulliparous women and the potential higher prevalence of FI in women of Asian ethnicity found in a previous study [29]. Only one woman in our study reported that the symptoms bothered her to any great degree. Currently, the decision to perform an episiotomy rests with the person conducting the delivery. Although the right mediolateral episiotomy is usual in many places, left mediolateral episiotomy is practiced locally. We continue to have a high episiotomy rate because we continue to observe a very low rate of third- and fourth-degree tears compared with Asian women delivering in places where there is a far more restrictive policy on episiotomy [30].
Only the prevalence and distress as a result of UI and FI were studied. Frequency and severity, which may also be important information for obstetricians, midwives, and pregnant women, was not studied. A longer-term follow-up study would be valuable to assess the trend of UI and FI and the potential effect of future pregnancies or deliveries on these problems.
In conclusion, the prevalence of SUI, UUI, and FI of formed or liquid stool at 12 months after delivery were 25.9 %, 8.2 %, and 4.0 %, respectively. Vaginal delivery (operative or spontaneous) and antenatal SUI and UUI were associated with SUI; antenatal UUI and higher maternal BMI at the first trimester were associated with UUI. Antenatal FI was associated with postnatal FI. These findings suggest pregnancy, regardless of delivery and obstetric practices, has an effect on UI and FI.
References
Thom DH, Rortveit G (2010) Prevalence of postpartum urinary incontinence: a systematic review. Acta Obstet et Gynecol 89:1511–1522
Burgio KL, Zyczynski H, Locher JL, Richter HE, Redden DT, Wright KC (2003) Urinary incontinence in the 12-month postpartum period. Obstet Gynecol 102:1291–1298
Brown SJ, Donath S, MacArthur C, McDonald EA, Krastev AH (2010) Urinary incontinence in nulliparous women before and during pregnancy: prevalence, incidence, and associated risk factors. Int Urogynecol J 21:193–202
Zhu L, Li L, Lang J, Xu T (2012) Prevalence and risk factors for peri- and postpartum urinary incontinence in primiparous women in China: a prospective longitudinal study. Int Urogynecol J 23:563–572
Schraffordt ES, Vervest HAM, Oostvolgel HJM (2003) Anorectal symptoms after various modes of vaginal delivery. Int Urogynecol J Pelvic Floor Dysfunct 14:244–249
Yang X, Zhang HX, Yu HY, Gao XL, Yang HX, Dong Y (2010) The prevalence of fecal incontinence and urinary incontinence in primiparous postpartum Chinese women. Eur J Obstet Gynecol Reprod Biol 152:214–217
Solans-Domenech M, Sanchez E, Espuna-Pons M, Pelvic Floor Research Group (2010) Urinary and anal incontinence during pregnancy and postpartum. Obstet Gynecol 115:618–628
Burgio KL, Matthews KA, Engel BT (1991) Prevalence, incidence and correlates of urinary incontinence in healthy middle-aged women. J Urol 146:1225–1229
Borello-France D, Burgio KL, Richter H, Zyczynski H, Fitzgerald MP, Whitehead W et al (2006) Fecal and urinary incontinence in primiparous women. Obstet Gynecol 108:863–872
Zacharin RF (1977) “A Chinese anatomy” – the pelvic supporting tissues of the Chinese and Occidental female compared and contrasted. Aust NZJ Obstet Gynaec 17:1–11
Howard D, DeLancey JO, Tunn R, Ashton Miller JA (2000) Racial differences in the structure and function of the stress urinary continence mechanism. Obstet Gynecol 95:713–717
Dietz HP (2003) Do Asian women have less pelvic organ mobility than Caucasians? Int Urogynecol J Pelvic Floor Dysfunct 14:250–253
Shek K, Dietz H (2010) Intrapartum risk factors for levator trauma. BJOG 117:1485–1492
Hong Kong College of Obstetricians & Gynaecologists (2004) Territory-wide Obstetrics and Gynaecology Audit. Hong Kong College of Obstetricians and Gynaecologists Web. http://hkcog.obg.cuhk.edu.hk/public/docs/audit/audit_og_2004.asp. Accessed 22 October 2012.
Gossett DR, Su RD (2008) Episiotomy practice in a community hospital setting. J Reprod Med 53:803–808
Chan SS, Cheung RY, Yiu AK, Li JC, Lai BP, Choy KW et al (2011) Chinese validation of Pelvic Floor Distress Inventory (PFDI) and Pelvic Floor Impact Questionnaire (PFIQ). Int Urogynecol J 22:1305–1312
Haylen BT, de Ridder D, Freeman RM, Swift SE, Berghmans B, Lee J et al (2010) An International Urogynecologic Association (IUGA)/International Continence Society (ICS) joint report on the terminology for female pelvic floor dysfunction. Int Urogynecol J 21:5–26
Chan SS, Cheung RY, Yiu KW, Lee LL, Pang AW, Choy KW et al (2012) Prevalence of levator ani muscle injury in Chinese women after first delivery. Ultrasound Obst Gynecol 39:704–709
Leung TY, Leung TN, Sahota DS, Chan OK, Chan LW, Fung TY et al (2008) Trends in maternal obesity and associated risks of adverse pregnancy outcomes in a population of Chinese women. BJOG 115:1529–1537
Glazener CMA, Herbison GP, MacArthur C, Lancashire R, McGee MA, Grant AM et al (2006) New postnatal urinary incontinence: obstetric and other risk factors in primiparae. BJOG 113:208–217
Chaliha C, Kalia V, Stanton SL, Monga A, Sultan AH (1999) Antenatal prediction of postpartum urinary and fecal incontinence. Obstet Gynecol 94:689–694
Brown S, Gartland D, Donath S, MacArthur C (2011) Effects of prolonged second stage, method of birth, timing of caesarean section and other obstetric risk factors on postnatal urinary incontinence: an Australian nulliparous cohort study. BJOG 118:991–1000
Arya LA, Jackson ND, Myers DL, Verma A (2001) Risk of new onset urinary incontinence after forceps and vacuum delivery in primiparous women. Am J Obstet Gynecol 185:1318–1323, discussion 1323-1324
Serati M, Salvatore S, Khullar V, Uccella S, Bertelli E, Ghezzi F et al (2008) Prospective study to assess risk factors for pelvic floor dysfunction after delivery. Acta Obstet Gynecol Scand 87:313–318
Ekstrom A, Altman D, Wiklund I, Larsson C, Andolf E (2008) Planned cesarean section versus planned vaginal delivery: comparison of lower urinary tract symptoms. Int Urogynecol J 19:459–465
Groutz A, Helpman L, Gold R, Pauzner D, Lessing JB, Gordon D (2007) First vaginal delivery at an older age: does it carry an extra risk for the development of stress urinary incontinence? Neurourol Urodynam 26:779–782
Rortveit G, Daltveit AK, Hannestad YS, Hunskaar S, Norwegian EPINCONT Study (2003) Urinary incontinence after vaginal delivery or cesarean section. N Engl J Med 348:900–907
Casey BM, Schaffer JK, Bloom SL, Heartwell SF, McIntire DD, Leveno KJ (2005) Obstetric antecedents for postpartum pelvic floor dysfunction. AJOG 192:1655–1662
MacArthur C, Glazener C, Lancashire R, Herbison P, Wilson D, Grant A (2005) Faecal incontinence and mode of first and subsequent delivery: a six-year longitudinal study. BJOG 112:1075–1082
Dahlen H, Homer C (2008) Perineal trauma and postpartum perineal morbidity in Asian and non-Asian primiparous women giving birth in Australia. J Obstet Gynecol Neonatal Nurs 37:455–463
Acknowledgements
This study obtained grants from Health and Health Service Research Fund from Department of Health Bureau of Government of Hong Kong S. A. R.
Details of ethics approval
Ethics approval for the study was granted by the Joint Chinese University of Hong Kong — New Territories East Cluster Clinical Research Ethics Committee (CRE -2009.257) on 17 July 2009.
Funding
This study obtained grants from Health and Health Service Research Fund from Department of Health Bureau of Government of Hong Kong S. A. R.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chan, S.S.C., Cheung, R.Y.K., Yiu, K.W. et al. Prevalence of urinary and fecal incontinence in Chinese women during and after their first pregnancy. Int Urogynecol J 24, 1473–1479 (2013). https://doi.org/10.1007/s00192-012-2004-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00192-012-2004-8