Abstract
Purpose
To compare the effectiveness of intraarticular (IA) multiple and single platelet-rich plasma (PRP) injections as well as hyaluronic acid (HA) injections in different stages of osteoarthritis (OA) of the knee.
Methods
A total of 162 patients with different stages of knee OA were randomly divided into four groups receiving 3 IA doses of PRP, one dose of PRP, one dose of HA or a saline injection (control). Then, each group was subdivided into two groups: early OA (Kellgren–Lawrence grade 0 with cartilage degeneration or grade I–III) and advanced OA (Kellgren–Lawrence grade IV). The patients were evaluated before the injection and at the 6-month follow-ups using the EuroQol visual analogue scale (EQ-VAS) and International Knee Documentation Committee (IKDC) subjective scores. Adverse events and patient satisfaction were recorded.
Results
There was a statistically significant improvement in the IKDC and EQ-VAS scores in all the treatment groups compared with the control group. The knee scores of patients treated with three PRP injections were significantly better than those patients of the other groups. There was no significant difference in the scores of patients injected with one dose of PRP or HA. In the early OA subgroups, significantly better clinical results were achieved in the patients treated with three PRP injections, but there was no significant difference in the clinical results of patients with advanced OA among the treatment groups.
Conclusion
The clinical results of this study suggest IA PRP and HA treatment for all stages of knee OA. For patients with early OA, multiple (3) PRP injections are useful in achieving better clinical results. For patients with advanced OA, multiple injections do not significantly improve the results of patients in any group.
Level of evidence
I.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The incidence of articular cartilage pathology is increasing because of the increase in sports activities and the prominence of physical activities in all age groups [6, 31]. Because of the continued increase in the mean age of the active population, OA is the most common degenerative joint disorder found in elderly individuals, and it has a significant effect on society [3, 9].
OA is a major cause of pain and disability and is detrimental to quality of life. Many non-invasive treatment options have been recommended to relieve symptoms and extend the quality of life and years of athletic activity for those with OA [13]. The treatment usually begins with non-steroidal anti-inflammatory drugs (NSAIDs), which have potential side effects that limit their use and lack clear data about their clinical therapeutic potency [5, 33]. Topical agents are widely used clinically for short-term use and are not effective for severe OA [33]. Oral supplements, such as glucosamine and chondroitin sulphate, have not been shown to be clearly effective for the treatment of OA and cannot be considered the most ideal treatment agents for OA [33]. Intraarticular (IA) steroid injections have short-term effects on knee pain and disability and negative effects on knee structures [23, 24]. The IA administration of hyaluronic acid (HA) has yielded favourable results in many studies and has been widely applied in clinical practice. Synovial fluid, a lubricant for articular surfaces, reduces surface stress and plays an important role in the movement of chondronutritive substances from the synovia [20, 21]. Finally, although many treatment options are available, none are considered ideal for OA. As these treatment options do not change the natural course of the disease, we investigated the clinical effects of a new method with promising results for altering the natural history of OA and compared it with a traditional treatment method.
Platelet-rich plasma (PRP) is an autologous blood product that contains an increased concentration of platelets and is used in orthopaedic and sports medicine practices to treat bone, tendon and ligament injuries [15, 28]. Additionally, PRP injections represent a treatment option for cartilage injuries and OA [8, 13]. Despite the promising preclinical results and wide clinical interest in orthopaedic and sports medicine, many unanswered questions regarding the clinical application and efficacy of PRP remain. There is a lack of clarity regarding the number and frequency of injections for proper effectiveness as well as the ideal treatment for different stages of gonarthrosis (from cartilage injury to advanced OA). Randomized clinical trials in limited numbers have compared PRP with HA and sham treatments and have demonstrated that IA PRP injection improves clinical signs [4, 18, 25]. To our knowledge, there is no prospective randomized study in the literature comparing single and multiple PRP injections and HA injections in the knee for pathologies ranging from early degeneration to OA.
The primary aim of this study was to explore the clinical effects of PRP for OA and to compare the results with a traditional IA HA injection therapy. The secondary aim of this study was to explore the ideal number of PRP injections required for different stages of OA.
It was hypothesized that treatment with PRP would lead to improvements in knee scores due to the release of GFs and bioactive molecules that would possibly affect the degenerated knee.
Materials and methods
This study was designed as a double-blind, randomized, placebo-controlled trial with 4 groups and 4 treatment methods (the PRP3 group, receiving 3 IA PRP injections; the PRP1 group, receiving 1 PRP injection and 2 saline injections; the HA group, receiving 3 HA injections and the control group, receiving 3 saline injections). Volunteer participants were blinded, subjected to a standardized IA injection protocol and assessed by EuroQol visual analogue scale (EQ-VAS) which is a simple, validated and commonly used patient-administered method that assesses pain intensity and International Knee Documentation Committee (IKDC) subjective scores before the treatment and at the 6-month follow-up. IKDC subjective evaluation form is a commonly used form that detects improvement in function and symptoms for knee disorders. The form has three domains: knee symptoms including seven items; sports and daily activities with ten items; and current knee function with one item. Scores for each item are summed to give a total score. The total score is calculated as (sum of items)/(maximum possible score) × 100, to give a total score of 100. Possible score ranges 0–100, where 100 means with the absence of symptoms and no limitation for daily or sporting activities. The adverse effects and patient satisfaction were recorded as well [11].
Patient selection
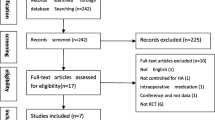
From January to September 2013, 250 consecutive patients (152 women and 98 men) who had a history of chronic (>4 months) pain or swelling radiographically documented grades I to IV gonarthrosis (graded according to the Kellgren–Lawrence classification scale for tibiofemoral joint degeneration) were enrolled. The exclusion criteria included previous lower extremity surgery, systemic disorders (diabetes, rheumatic diseases, severe cardiovascular diseases, haematological diseases, infections), patients with generalized OA, patients undergoing anticoagulant or antiaggregant therapy, the use of NSAIDs in the 5 days before injection, patients with haemoglobin values less than 11 g/dL and platelet values less than 150,000/mm3.
The radiographic examination of the volunteers included radiographs of the lower limbs under loading and roentgenograms of the affected knee in the anteroposterior (full extension) and lateral (30-degree flexion) positions. For the patients classified as grade 0 based on the Kellgren–Lawrence classification, magnetic resonance imaging (MRI) was performed to assess chondral degeneration. The patients without cartilage changes on MRI were excluded. In the patients with bilateral symptoms, only the side with significant symptoms was taken into consideration. The injections were postponed for the randomization.
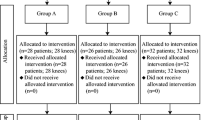
When the number reached a total of 182 participants who met the inclusion criteria, the patients were randomly assigned into four groups by a computer-derived protocol. Then, the patients were returned to the clinic for the injections. There were 46 patients in the PRP3 group and 45 patients in the PRP1 group. The 46 patients receiving 3 HA injections formed the HA group, and the remaining 45 patients were in the control group. Then, to determine the effect of the OA grade on the knee scores, each group was subdivided into two groups according to the Kellgren–Lawrence classification: early OA (Kellgren–Lawrence grade 0 with cartilage degeneration or grade I to III) and advanced OA (Kellgren–Lawrence grade IV) (Fig. 1).
The group assignments were only accessible to the study assistant and were concealed from the patients and the researchers in the study. A total of 162 of the 182 participants were prospectively evaluated at 6-month follow-ups. The mean age was 53.5 ± 13.4 (ranging from 27 to 84), and the mean body mass index was 29.1 ± 4.3 (ranging from 19.8 to 38). Ninety (55.6 %) of the patients were women and 72 (44.3 %) were men. Of the patients who were not evaluated, six declined treatment with injection therapy during the first clinical meeting, two underwent total knee replacement and 14 were lost to follow up. The randomization ensured that the baseline characteristics of the four groups were comparable with respect to age, gender and body mass index (BMI) (Table 1).
PRP preparation
The PRP specimens were collected as described by Filardo et al. [8] from all the participants. A total of 150 mL of venous blood (collected in a bag containing 21 mL of sodium citrate) was collected under aseptic conditions from the antecubital vein. Additionally, a peripheral blood count was obtained. To collect 20 mL of PRP, two centrifugations (the first at 1500 rpm for 6 min and the second at 3500 rpm for 12 min) were performed. The PRP unit was divided into 4 small units of 5 mL each: 1 unit was sent to the laboratory for a platelet count as well as concentration and bacteriological tests, 1 unit was used for injection within 2 h, and the remaining 2 units were stored at −30 °C.
There was no significant difference in the total number of platelets per millilitre between the PRP groups (n.s.) (PRP3 group: concentration factor of 5.2× (1118,000 µL); PRP1 group: concentration factor of 5.3× (1152,000 µL); n.s.).
The injections were administered every 7 days in all the groups. In the PRP3 and PRP1 groups, 1 mL of CaCl2 was added to activate the platelets. For the second and third injections in the PRP3 group, the samples were thawed in a dry thermostat at 37 °C for 30 min before administration.
In the HA group, 39 patients were treated with a high molecular weight HA preparation [30 mg/2 mL, Orthovisc (Anika Therapeutics Inc, Woburn, Massachusetts, USA)]. The treatment consisted of 3 injections of 2 mL once weekly.
Treatment procedure and follow-up
The skin was sterilely dressed, and each injection was administered by an unblinded physician using the superolateral approach with a 22-g needle. The knee was immobilized for 10 min after the injection, and the patient was discharged after a 1-h observation with instructions to use cold therapy on the affected area for pain relief. Physical activity was not limited; however, NSAIDs were not allowed during the follow-up period. Paracetamol was prescribed for discomfort.
The patients were evaluated before the injection and at the 6-week, 3-month and 6-month follow-ups by the clinician who was blinded to the patients and content of the injections. The EQ-VAS (as recommended by the ICRS evaluation package) and IKDC subjective scores were used for the clinical evaluation. Adverse effects were recorded, and patient satisfaction (satisfied, partially satisfied, not satisfied) at the end of 6 months was evaluated.
All the participants provided written informed consent, and the study was approved by the Inonu University, Turgut Ozal Medical School, Malatya, Turkey Ethics Committee (2013-171).
Statistical analysis
GPower software was used for the sample size estimation. A sample size of 140 individuals in total (35 per arm) was proposed to give 80 % power to detect an effect size of 0.8 (one-tail) between groups for continuous outcome variables. Anticipating protocol violators and early discontinuations of 25 %, it was projected that 175 patients should be included in the study.
The data were reported as the means ± standard deviations (SDs) or frequencies. Normality was confirmed using the Shapiro–Wilk test. The quantitative data were compared by one-way analysis of variance (ANOVA) followed by the Bonferroni test when the variances were homogenous or the Tamhane T2 test when the variances were non-homogenous. The qualitative data were analysed using Pearson’s Chi-square test. In each group, the knee scores at the 6-week, 3-month and 6-month follow-ups were compared with repeated measures one-way ANOVA followed by the Bonferroni test, and p < 0.05 values were considered significant. All the analyses were conducted with IBM SPSS software, v. 22.0 for Windows.
Results
Six patients left during the first treatment because they were unable to tolerate injection therapy and were therefore excluded from the study for not beginning the treatment protocol. There was a statistically significant improvement in the IKDC and EQ-VAS scores in all the treatment groups compared with the control group at the 6-month follow-up (p < 0.05). Further analysis showed that the PRP3 group had significantly better results than the PRP1 and HA groups (PRP3 vs. PRP1, p = 0.001, and PRP3 vs. HA, p = 0.001). However, no significant differences were found between the PRP1 and HA groups (PRP1 vs. HA, n.s.). The results are summarized in Table 2 and Fig. 2.
For the early OA subgroups, significantly better results were found for all the treatment groups compared with the control group (p < 0.005). PRP3 showed a significant improvement compared with the PRP1 and HA groups (p = 0.001), and there were no significant differences between the PRP1 and HA groups (n.s.) (Tables 3, 4).
For the advanced OA subgroups, significantly better results were achieved for all the treatment groups compared with the control group (p < 0.05). However, there were no significant differences in the knee scores between the advanced OA subgroups of the PRP3, PRP1 and HA groups at the 6-month follow-up (n.s.) (Tables 3, 4).
At the end of 6 months, 76.9 % of the patients were satisfied, 12.8 % were partially satisfied, and 10.3 % were not satisfied in the PRP3 group. In the PRP1 group, 72.7 % of the patients were satisfied, 18.2 % were partially satisfied, and 9.1 % were not satisfied. In the HA group, 64.1 % of the patients were satisfied, 23.1 % were partially satisfied, and 12.8 % were not satisfied at the end of the procedure. In the control group, 5 % of the patients were satisfied, 15 % were partially satisfied, and 80 % were not satisfied with the IA injection procedure. According to these results, patients in the PRP3, PRP1 and HA groups were more satisfied than those in the control group; however, the differences between the three groups were not significant (n.s.).
Discussion
The most important finding of the present study was that multiple PRP injections resulted in better clinical results, particularly in patients with early OA. However, there was no difference in the results between treatment methods in the patients with advanced OA. One of the important results of this study was that a single dose of PRP or HA did not have a superior effect on the patients with early or advanced OA.
Synovial fluid viscoelasticity that results from HA is essential for normal joint function and acts as a lubricant and shock absorber [1]. Some clinical studies and meta-analyses have demonstrated satisfactory results with IA HA treatment, whereas others found no differences compared with placebo groups [2, 10, 14, 30]. In this study, significant improvements were observed in the HA group, suggesting that IA HA treatment is an effective treatment for patients with knee degeneration. However, the significant decrease in knee scores at the 6-month follow-up confirms that the treatment is only effective for a short time, as described by Bellamy et al. [2].
One area of cartilage regeneration research is focused on PRP, which is the plasma component in whole blood that is processed to contain a higher concentration of platelets with growth factors [16, 17]. Many in vitro studies have suggested that PRP may act as a stem cell growth promoter and chondrogenic differentiator [29, 32]. Animal studies have shown different PRP treatment results. In a study that utilized an anterior cruciate ligament-transected rabbit model, the authors reported that PRP-treated rabbits had significantly decreased progression of OA [26].
A few prospective studies have been designed to evaluate the effectiveness of PRP on knee degeneration and have obtained statistically significant improvements in all the clinical scores at the end of therapy [7]. However, a limitation of these studies was the lack of a control group. In contrast to improved results, some prospective studies have concluded that PRP does not affect outcomes [12, 22]. In a randomized, double-blind study with a control group, Patel et al. [25] concluded that single or double PRP injections in knees with mild or moderate OA produced improved scores compared with those resulting from saline injections. However, the patients treated with two injections may have known that they were not in the control group. We tried to avoid such bias by administering an equal number of injections to all the patients. To our knowledge, only a few prospective studies were designed to evaluate the effectiveness of PRP and the superiority of HA and PRP treatment for knee degeneration [4, 13, 27]. These well-designed studies concluded that PRP injections showed better clinical results than HA injections; however, there are limitations in the studies. These studies did not have control groups and did not include randomization, except the study by Cerza et al. [4]. To address these limitations, a control group was included into the study. Injections were postponed, and patients were randomly divided into groups prior to injection therapy; then, patients were asked to return for injection therapy.
Although a significant decrease in knee scores was recorded within 6 months following the treatment, it has been hypothesized that IA PRP and HA treatment would be beneficial in patients with all stages of OA. In patients with early OA, significantly better clinical results were obtained with multiple PRP injections; it is hypothesized that multiple PRP injections for these patients would yield an effective treatment method. Unlike previous studies [13, 25], this study has revealed no significant difference between single PRP injections and HA injections in patients with early OA.
Patel et al. [25] concluded that a single dose of PRP is as effective as a double dose. Our study confirms this conclusion only for patients with advanced OA, as no difference between treatment methods has been found. Multiple injections are unnecessary for patients with advanced OA. These results may provide guidance with respect to treatment protocols because there is currently no consensus regarding treatment methods.
The better clinical results observed in patients with a lower degree of cartilage injury could be explained by a high response to GFs by less degenerated joints with a higher percentage of living and vital cells. Despite the significantly better results in the advanced OA group compared with the control group, the lack of a significant difference between the treatment options for this group supports this theory. In this study, patient scores in the control group worsened over time, indicating that patients with OA need to be treated with effective methods to avoid discomfort and further disability.
PRP and HA may influence joint homoeostasis by reducing synovial membrane hyperplasia and modulating the cytokine level. This mechanism temporarily leads to an improved clinical outcome without affecting the cartilage tissue structure [19]. Despite declining clinical outcomes, the significantly better results associated with multiple IA PRP injections in patients with early OA suggest that further experimental studies should be conducted on this issue. The differences between multiple and single PRP injections with respect to the effects on cellular mechanism should be clearly explored. Even if multiple injections showed no different effects from those of single injections at the cellular level, the clinical efficacy of multiple injections is obvious. Determining the most appropriate time for additional injections is important in the planning of future treatments.
This study has some limitations, including the short-term follow-up period. The patients that have been included into this study have been evaluated at a maximum of 6-month follow-up as the viscosupplementation therapy could only be repeated after some time, and a different treatment option might have been needed for OA patients. In this study, a high number of patients were lost (20 of 182 participants) at follow-up. However, the 162 remaining patients were still acceptable according to our power analysis, which required a sample size of 140 individuals in total and 35 per arm. Additionally, longer follow-ups without additional treatment could have led to a loss of patients from the study. Radiographic follow-up methods may be used to evaluate cartilage degeneration; however, this evaluation could not be conducted due to ethical issues and financial costs. Another weakness of this study is that image guidance was not used to ensure the location of the needle in the knee joint. Ideally, the present investigation would have been conducted through a multicentre study; however, it has been predicted that it would be difficult to optimize the treatment protocols for all the centres. Not optimizing the treatment protocols would affect the reliability of the study and would have decreased the value of the study; thus, we performed a single-centre study.
Conclusion
The clinical results indicate that IA PRP and HA treatment is suggested for all stages of knee OA. For patients with early OA, multiple (3) PRP injections are useful in achieving better clinical results. For patients with advanced OA, multiple injections are unnecessary and do not significantly affect patient knee scores.
References
Balazs EA, Denlinger JL (1993) Viscosupplementation: a new concept in the treatment of osteoarthritis. J Rheumatol Suppl 39:3–9
Bellamy N, Campbell J, Robinson V, Gee T, Bourne R, Wells G (2006) Viscosupplementation for the treatment of osteoarthritis of the knee. Cochrane Database Syst Rev 2:CD005321
Busija L, Bridgett L, Williams SR, Osborne RH, Buchbinder R, March L, Fransen M (2010) Osteoarthritis. Best Pract Res Clin Rheumatol 24:757–768
Cerza F, Carnì S, Carcangiu A, VaI Di, Schiavilla V, Pecora A, BiG De, Ciuffreda M (2012) Comparison between hyaluronic acid and platelet-rich plasma, intra-articular infiltration in the treatment of gonarthrosis. Am J Sports Med 40:2822–2827
Clegg DO, Reda DJ, Harris CL, Klein MA, O’Dell JR, Hooper MM, Bradley JD, Bingham COr, Weisman MH, Jackson CG, Lane NE, Cush JJ, Moreland LW, Schumacher HRJ, Oddis CV, Wolfe F, Molitor JA, Yocum DE, Schnitzer TJ, Furst DE, Sawitzke AD, Shi H, Brandt KD, Moskowitz RW, Williams HJ (2006) Glucosamine, chondroitin sulfate, and the two in combination for painful knee osteoarthritis. N Engl J Med 354:795–808
Curl WW, Krome J, Gordon ES, Rushing J, Smith BP, Poehling GG (1997) Cartilage injuries: a review of 31,516 knee arthroscopies. Arthroscopy 13:456–460
Filardo G, Kon E, Buda R, Timoncini A, Di Martino A, Cenacchi A, Fornasari PM, Giannini S, Marcacci M (2011) Platelet-rich plasma intra-articular knee injections for the treatment of degenerative cartilage lesions and osteoarthritis. Knee Surg Sports Traumatol Arthrosc 19:528–535
Filardo G, Kon E, Di Martino A, Di Matteo B, Merli ML, Cenacchi A, Fornasari PM, Marcacci M (2012) Platelet-rich plasma vs hyaluronic acid to treat knee degenerative pathology: study design and preliminary results of a randomized controlled trial. BMC Musculoskelet Disord 13:229
Grogan SP, D’Lima DD (2010) Joint aging and chondrocyte cell death. Int J Clin Rheumtol 5:199–214
Housman L, Arden N, Schnitzer TJ, Birbara C, Conrozier T, Skrepnik N, Wei N, Bockow B, Waddell D, Tahir H, Hammond A, Goupille P, Sanson BJ, Elkins C, Bailleul F (2014) Intra-articular hylastan versus steroid for knee osteoarthritis. Knee Surg Sports Traumatol Arthrosc 22(7):1684–1692
Irrgang JJ, Anderson AF, Boland AL, Harner CD, Kurosaka M, Neyret P et al (2001) Development and validation of the International Knee Documentation Committee subjective knee form. Am J Sports Med 29:600–613
Kesikburun S, Tan AK, Yilmaz B, Yaşar E, Yazicioğlu K (2013) Platelet-rich plasma injections in the treatment of chronic rotator cuff tendinopathy: a randomized controlled trialwith 1-year follow-up. Am J Sports Med 41:2609–2616
Kon E, Mandelbaum B, Buda R, Filardo G, Delcogliano M, Timoncini A, Fornasari PM, Giannini S, Marcacci M (2011) Platelet-rich plasma intra-articular injection versus hyaluronic acid viscosupplementation as treatments for cartilage pathology: from early degeneration to osteoarthritis. Arthroscopy 27:1490–1501
Lo GH, LaValley M, McAlindon T, Felson DT (2003) Intra-articular hyaluronic acid in treatment of knee osteoarthritis: a meta-analysis. JAMA 290:3115–3121
Lopez-Vidriero E, Goulding KA, Simon DA, Sanchez M, Johnson DH (2010) The use of platelet rich plasma in arthroscopy and sports medicine: optimizing the healing environment. Arthroscopy 26:269–278
McCarrel T, Fortier L (2009) Temporal growth factor release from platelet-rich plasma, trehalose lyophilized platelets, and bone marrow aspirate and their effect on tendon and ligament gene expression. J Orthop Res 27:1033–1042
Mehta V (2010) Platelet-rich plasma: a review of the science and possible clinical applications. Orthopedics 33:111
Mei-Dan O, Carmont MR, Laver L, Mann G, Maffulli N, Nyska M (2012) Platelet-rich plasma or hyaluronate in the management of osteochondral lesions of the talus. Am J Sports Med 40:534–541
Mix KS, Sporn MB, Brinckerhoff CE, Eyre D, Schurman DJ (2004) Novel inhibitors of matrix metalloproteinase gene expression as potential therapies for arthritis. Clin Orthop Relat Res 427:S129–S137
Mladenovic Z, Saurel AS, Berenbaum F, Jacques C (2014) Potential role of hyaluronic acid on bone in osteoarthritis: matrix metalloproteinases, aggrecanases, and RANKL expression are partially prevented by hyaluronic acid in interleukin 1-stimulated osteoblasts. J Rheumatol 41:945–954
Mongkhon JM, Thach M, Shi Q, Fernandes JC, Fahmi H, Benderdour M (2014) Sorbitol-modified hyaluronic acid reduces oxidative stress, apoptosis and mediators of inflammation and catabolism in human osteoarthritic chondrocytes. Inflamm Res 63:691–701
Morishita M, Ishida K, Matsumoto T, Kuroda R, Kurosaka M, Tsumura N (2014) Intraoperative platelet-rich plasma does not ımprove outcomes of total knee arthroplasty. J Arthroplasty 29(12):2337–2341
Nakazawa F, Matsuno H, Yudoh K, Watanabe Y, Katayama R, Kimura T (2002) Corticosteroid treatment induces chondrocyte apoptosis in an experimental arthritis model and in chondrocyte cultures. Clin Exp Rheumatol 20:773–781
Ostergaard M, Halberg P (1998) Intra-articular corticosteroids in arthritic disease: a guide to treatment. BioDrugs 9:95–103
Patel S, Dhillon MS, Aggarwal S, Marwaha N, Jain A (2013) Treatment with platelet-rich plasma is more effective than placebo for knee osteoarthritis: a prospective, double-blind, randomized trial. Am J Sports Med 41:356–364
Saito M, Takahashi KA, Arai Y, Inoue A, Sakao K, Tonomura H, Honjo K, Nakagawa S, Inoue H, Tabata Y, Kubo T (2009) Intraarticular administration of platelet- rich plasma with biodegradable gelatin hydrogel microspheres prevents osteoarthritis progression in the rabbit knee. Clin Exp Rheumatol 27:201–207
Say F, Gürler D, Yener K, Bülbül M, Malkoc M (2013) Platelet-rich plasma injection is more effective than hyaluronic acid in the treatment of knee osteoarthritis. Acta Chir Orthop Traumatol Cech 80:278–283
Sundman EA, Cole BJ, Fortier LA (2011) Growth factor and catabolic cytokine concentrations are influenced by the cellular composition of platelet-rich plasma. Am J Sports Med 39:2135–2140
van Buul G, Koevoet WL, Kops N, Bos PK, Verhaar JA, Weinans H, Bernsen MR, van Osch G (2011) Platelet-rich plasma releasate inhibits inflammatory processes in osteoarthritic chondrocytes. Am J Sports Med 39:2362–2370
van der Weegen W, Wullems JA, Bos E, Noten H, van Drumpt RA (2015) No difference between intra-articular injection of hyaluronic Acid and placebo for mild to moderate knee osteoarthritis: a randomized, controlled, double-blind trial. J Arthroplasty 30(5):754–757
Widuchowski W, Widuchowski J, Trzaska T (2007) Articular cartilage defects: study of 25,124 knee arthroscopies. Knee 14:177–182
Xie X, Wang Y, Zhao C, Guo S, Liu S, Jia W, Tuan RS, Zhang C (2012) Comparative evaluation of MSCs from bone marrow and adipose tissue seeded in PRP-derived scaffold for cartilage regeneration. Biomaterials 33:7008–7018
Zhang W, Moskowitz RW, Nuki G, Abramson S, Altman RD, Arden N, Bierma-Zeinstra S, Brandt KD, Croft P, Doherty M, Dougados M, Hochberg M, Hunter DJ, Kwoh K, Lohmander LS, Tugwell P (2008) OARSI recommendations for the management of hip and knee osteoarthritis, Part II: OARSI evidence-based, expert consensus guidelines. Osteoarthritis Cartilage 16:137–162
Acknowledgments
We thank Irfan Kuku from Inonu University, Turgut Ozal Medical Center, Department of Haemotology, Malatya, Turkey, and Yusuf Turkoz from Inonu University, Turgut Ozal Medical Center, Department of Biochemistry, Malatya, Turkey.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Rights and permissions
About this article
Cite this article
Görmeli, G., Görmeli, C.A., Ataoglu, B. et al. Multiple PRP injections are more effective than single injections and hyaluronic acid in knees with early osteoarthritis: a randomized, double-blind, placebo-controlled trial. Knee Surg Sports Traumatol Arthrosc 25, 958–965 (2017). https://doi.org/10.1007/s00167-015-3705-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-015-3705-6