Abstract
If permission of full active and passive extension immediately after an anterior cruciate ligament (ACL) reconstruction will increase the post-operative laxity of the knee has been a subject of discussion. We investigated whether a post-operative rehabilitation protocol including active and passive extension without any restrictions in extension immediately after an ACL reconstruction would increase the post-operative anterior–posterior knee laxity (A–P laxity). Our hypothesis was that full active and passive extension immediately after an ACL reconstruction would have no effect on the A–P laxity and clinical results up to 2 years after the operation. Twenty-two consecutive patients (14 men, 8 women, median age 21 years, range 17–41) were included. All the patients had a unilateral ACL rupture and no other ligament injuries or any other history of previous knee injuries. The surgical procedure was identical in all patients and one experienced surgeon operated on all the patients, using the bone-patellar tendon-bone autograft. The post-operative rehabilitation programme was identical in both groups, except for extension training during the first 4 weeks post-operatively. The patients were randomly allocated to post-operative rehabilitation programmes either allowing (Group A, n=11) or not allowing [Group B (30 to −10°), n=11] full active and passive extension immediately after the operation. They were evaluated pre-operatively and at 6 months and 2 years after the reconstruction. To evaluate the A–P knee laxity, radiostereometric analysis (RSA) and KT-1000 arthrometer (KT-1000) measurements were used, range of motion, Lysholm score, Tegner activity level, the International Knee Documentation Committee (IKDC) evaluation system and one-leg-hop test quotient were used. Pre-operatively, the RSA measurements revealed side-to-side differences in Group A of 8.6 mm (2.3–15.4), median (range) and in Group B of 7.2 mm (2.2–17.4) (n.s.). The corresponding KT-1000 values were for Group A, 2.0 mm (0–8.0) and Group B, 4.0 mm (0–10.0) (n.s.). At 2 years, the differences between the two groups were minimal, regardless of the method that had been used. The RSA measurements in Group A were 2.7 mm (0–10.7) and in Group B 2.8 (−1.8 to 9.5). The KT-1000 values were for Group A, 1.0 mm (−1.5 to 3.5), and for Group B, 0.5 mm (−1.0 to 4.0), without any significant differences between the groups. Nor did the Lysholm score, Tegner activity level, IKDC or one-leg-hop test differ. Early active and passive extension training, without any restrictions in extension, immediately after an ACL reconstruction using bone-patellar tendon-bone graft did not increase post-operative knee laxity up to 2 years after the ACL reconstruction.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rupture of the anterior cruciate ligament (ACL) is a common injury during both sports and leisure time activities [3, 27].
A well-planned post-operative rehabilitation protocol is as important for the final outcome after an ACL reconstruction as the surgery itself. Early joint motion is also beneficial when it comes to avoiding capsular contractions and reducing swelling and pain. Post-operative immobilisation of the knee may contribute to limited range of motion (ROM), muscular hypotrophy and inferior knee function [37]. Rehabilitation protocols aim to restore ROM, strength, co-ordination and full function as soon as possible, without damaging the graft [18, 35, 36, 38].
To our knowledge, there are no randomised studies with an adequate follow-up, which have used such an accurate method as the radiostereometric analysis (RSA) measuring technique to study the influence of immediate and active extension on the anterior–posterior knee laxity (A–P laxity) of the knee, after an ACL reconstruction. Despite this many rehabilitation programme, include early full active and/or passive extension after an ACL reconstruction.
In 1990, Shelbourne and Nitz [35] presented their accelerated post-operative rehabilitation protocols after an ACL reconstruction, in which they allowed immediate full active extension of the knee and emphasised early aggressive rehabilitation. In 1995, Shelbourne et al. [34] presented 2–6 years follow-up after ACL reconstruction with autogenous patellar tendon graft and participation in accelerated rehabilitation programme. Three years later Muneta et al. [26] presented the outcome after an ACL reconstruction, with multi-strand semitendinosus tendon, in which they emphasised early aggressive rehabilitation.
Several studies have compared the effect of open kinetic chain (OKC) and closed kinetic chain (CKC) [7, 23–25, 42] and most of the post-operative rehabilitation protocols used today include a combination of both methods. However, only a few studies have addressed the question of whether early active and passive extension immediately after the ACL reconstruction [35] affects A–P laxity of the knee.
It is also believed that there are two post-operative periods during which the ACL graft and its fixations are most vulnerable. The first period starts immediately after the reconstruction [24–26, 30], while the second begins when the graft becomes weaker due to graft revitalisation, until it reaches its weakest point, approximately 12 weeks after the reconstruction [26, 30].
Our aim was to study whether permitting active and passive extension, without any restrictions in extension, immediately after ACL reconstruction would increase the post-operative A–P laxity and subsequently lead to an inferior clinical outcome.
Our hypothesis was that permitting active and passive extension, without any restrictions, immediately after an ACL reconstruction would not have any negative effect on the A–P laxity, producing similar clinical results up to 2 years after the operation.
Patients and methods
The study was approved by the Human Ethics Committee at Göteborg University. All the patients gave their informed consent.
Patients with a unilateral traumatic ACL rupture with remaining insufficiency and painful “giving-way” episodes were included. The criteria were that they should have no history of injury or any other symptoms on the opposite knee. Presence of meniscal tears from the index injury which could be treated with a partial resection at the arthroscopy was accepted.
Exclusion criteria were multiple knee ligament injuries, a history of previous knee injuries or knee surgery before the index injury, meniscal tears that underwent repair or knee problems on the opposite side.
Twenty-two consecutive patients (14 men, 8 women) were included.
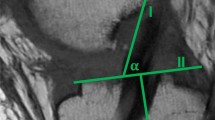
Arthroscopy was performed in all patients (injured knee), confirming the diagnosis. During the same session, the tantalum markers (diameter=0.8 mm) were inserted percutaneously in both the injured and the intact knee. To ascertain correct positioning, fluoroscopy was used during insertion. For the RSA measurements, at least three markers in each segment, located in a 3D triangle are needed. To ensure sufficient marker scatter and stability, four to five tantalum markers were implanted into the distal femur and proximal tibia on both the injured and the intact side (Fig. 1).
The tantalum markers can be seen in distal femur and proximal tibia in this lateral view of stereoradiograph. Cage markers are visible outside the knee. Two ceiling-mounted radiographic tubes, one anterior–posterior and one lateral, connected to two separated generators, were used to obtain simultaneous exposures, Table 2
The ACL reconstruction procedure was identical in all patients. The median age at the ACL reconstruction was 21 (16–41) years. The median time period between the index injury and the ACL reconstruction was 16 (4–45) weeks.
The operation was performed by one experienced surgeon, using a patellar tendon autograft. A standard arthroscopic one-incision technique was used in all patients. The width of the graft was 8–10 mm, depending on the size of the patellar tendon. A small notch plasty was performed to avoid graft impingement. The graft was placed in approximately the 10.30 (right knee) or 1.30 (left knee) position in the posterior intercondylar notch.
The fixation was performed using metallic interference screws, Cannu-flex silk screws 20 mm from at both ends, 7 mm diameter in femur and 7–9 mm in tibia (Acufex Ltd, Naples, FL, USA).
Before the operation the patients were randomised to one of two rehabilitation programmes using sealed, opaque envelopes. The patients were randomly allocated to post-operative rehabilitation programmes either allowing (Group A, n = 11) or not allowing (Group B, restricted motion between 30 and −10°, n = 11) active and passive extension, immediately after the operation.
There were no differences between the two groups in terms of age, time from injury to surgery and injured side. The male:female ratio was 5:6 in Group A and 9:2 in Group B (Table 1).
The patients were evaluated pre-operatively, at 6 months and 2 years after the operation. To evaluate the A–P knee laxity RSA [5, 6, 13–15] and KT-1000 measurements were used. Clinical tests included ROM, Lysholm score, Tegner activity level, the International Knee Documentation Committee (IKDC) evaluation system and one-leg-hop test were used. All the patients were examined at follow-up by independent observers. Radiographers at the Department of Radiology performed the RSA examinations and dedicated technicians performed measurements of the digital radiographs. The clinical tests and KT-1000 measurements were done by a physical therapist, who did not participate in the surgical procedure or the final evaluation of data.
Rehabilitation
The rehabilitation programmes, either with or without restricted active and passive extension between 30 and −10° was started immediately after the operation. The patients were supervised by a physiotherapist and trained three times a week during the first 4 weeks.
Rehabilitation brace
The same model of rehabilitation brace was used in both groups. This brace allowed 10° of hyperextension according to a radiographic examination of one of the author’s knee.
Weeks 0–2
Full weight-bearing was allowed in both groups immediately post-operatively and crutches were used for 10 days in both groups. The same model of rehabilitation brace was used in both groups, either without (Group A) or with (Group B) restricted active and passive extension between 30 and −10°. The training programme, including active and passive extension exercises, CKC, was started immediately post-operatively in both groups. One example is an active knee extension and flexion exercises sitting on the floor and muscle contraction exercises for both Hamstrings and Quadriceps.
Weeks 3–4
The two groups still used their rehabilitation brace. The training programme continued with or without restricted extension according to the initial protocol. CKC exercises for Hamstrings and Quadriceps were continued. Gait, stationary biking, proprioception and balance training was started. OKC training was started in both groups.
The brace was removed in the end of week 4 in both groups.
Weeks 5–6
Active and passive extension without any restrictions was now allowed in both groups. Isokinetic concentric and eccentric OKC quadriceps training was initiated in week 6, and isokinetic hamstrings training as well.
Weeks 7–12
Functional exercises, for example stair walking and skip the rope exercises were started. Slideboard exercises were initiated in week 12.
Weeks 13–17
Straight ahead jogging was permitted on an even surface. Eccentric and concentric muscle training was continued with increased weight and speed.
Weeks 18–24
Sport specific training. Jogging on an uneven surface and with 90–360° turns were initiated.
Week 25
The patient was allowed to return to sports activities, if the muscle strength was 90% of the intact leg or more.
Anterior–posterior laxity
Anterior–posterior laxity was measured using RSA and KT-1000. The difference in A–P laxity between the injured and the intact knee is presented (side-to-side difference).
Radiostereometric analysis
Radiostereometric analysis has been used to evaluate the laxity and kinematics of ACL-injured knees for more than a decade [6, 10, 13–15, 17, 19–21, 32]. It has mainly been used to quantify the effect of an ACL rupture, and to measure the A–P laxity during the follow-up after ACL surgery. As a tool for measuring skeletal and implant motions, RSA is accurate and precise down to 0.1 mm and 0.1–0.3° [4, 29] and it is accurate and precise when it comes to measuring A–P laxity for repeated testing over time [9]. When the effect of external forces is studied, the repeatability decreases due to several factors, such as variations in muscular tension and inconsistencies in the application of external forces [5, 6, 14, 20, 21].
RSA examinations
All the patients were examined in a radiographic laboratory specifically designed to perform RSA examinations of joints (Fig. 2a). The RSA measurements were made by one experienced examiner from the radiographic department. Two ceiling-mounted radiographic tubes, one anterior–posterior and one lateral, connected to two separated generators, were used to obtain simultaneous exposures. The patients were examined in the supine position with the knee in a Plexiglas calibration cage [32] (Fig. 2b). The distal femur was fixed with an adjustable frame to minimise femoral movements. Anterior and posterior loads were applied approximately 7 cm distal to the joint line (Fig. 2b). We used the same set-up as that previously described by Brandsson et al. [6] and Kärrholm et al. [21].
The following positions were tested:
-
extended knee (0°)
-
30° of flexion
-
30° of flexion with an anterior traction of 150 N
-
30° of flexion with a posterior pushing force of 80 N
The mean intra-articular displacement of the two tips of the intercondylar eminence along an anterior–posterior axis of the knee represented the A–P laxity. The femoral markers were used as fixed reference segments. The median value (range) of the “mean errors of rigid body fitting” and condition numbers representing marker stability and scatter were 0.047 mm (0.0–0.236) and 107 (40–318). During the pre-operative examinations, both the injured and the non-injured side were examined. At follow-up, the post-operative side-to-side differences in displacement were based on the pre-operative measurements of the intact knee, i.e. the baseline examination. Measurements of digital radiographs and computations of three-dimensional co-ordinates [28] were performed using a software package (UMRSA 5.0, RSA Biomedical, Umeå, Sweden).
KT-1000 arthrometer test
A standard KT-1000 (MEDmetric Corp., San Diego, CA, USA) was used [32]. One experienced observer, physiotherapist, performed all the measurements [2]. Both legs were placed on the thigh support with the knee in 30° of flexion. The arms of the patient were placed along the side of the body and the patient was instructed to relax. Before each test, the instrument was calibrated to zero. The intact knee was always tested first. The median value of three measurements for each knee was registered, using a force of 139 N.
Range of motion
Range of motion was recorded on both sides pre-operatively and at each follow-up. A standard hand-held goniometer was used. Values were rounded off to the nearest increment of 5°. The extension measurements were performed with the patient in the supine position and flexion was measured when the patient slid his/her heel as close to the buttocks as possible without any help from the arms.
Clinical tests
The ROM, the Lysholm score, Tegner activity level [41], the IKDC evaluation system [12] and one-leg-hop test were performed pre-operatively and at each follow-up.
During the one-leg-hop test, the patient jumped and landed on the same foot with his/her hands behind his/her back. The longest hop of three attempts was registered. A quotient (%) was calculated between the intact and the injured knee [11, 40].
Statistical methods
All the values are presented as the median and range. The Mann–Whitney U-test was used in the independent comparison of the two groups for non-parametric data and Wilcoxon‘s-signed-rank test was used to evaluate changes in parameters over time. A P-value of less than 0.05 was regarded as statistically significant.
Results
All the randomised patients in Group A and Group B completed their participation in the study.
Anterior–posterior laxity
Pre-operatively, the side-to-side difference using RSA was 8.6 mm (2.3–15.4) in Group A and 7.2 mm (2.2–17.4) in Group B, and using the KT-1000 2.0 mm (0–8.0) in Group A and 4.0 mm (0–10.0) in Group B (Tables 2 and 3), without any statistical differences between Groups A and B (n.s.).
At the 6-month follow-up, the side-to-side difference using RSA was 3.4 mm (−0.6 to 11.5) in Group A and 3.4 mm (−3.3 to 7.8) in Group B, and using the KT-1000, 0 mm (−3.0 to 1.5) in Group A and 1.5 mm (−0.5 to 4.5) in Group B (Tables 2 and 3), without any statistical differences between Groups A and B (n.s.).
At the 2-year follow-up, the side-to-side difference using RSA was 2.7 mm (0–10.7) in Group A and 2.8 mm (−1.8 to 9.5) in Group B, and using the KT-1000 1.0 mm (−1.5 to 3.5) in Group A, and 0.5 mm (−1.0 to 4.0) in Group B (Tables 2 and 3), without any differences between Groups A and B (n.s.).
Both groups displayed a significant reduction in A–P laxity between the pre-operative examination and the 2-year follow-up for both the RSA and KT-1000 measurements (Tables 2 and 3), in Group A (RSA P = 0.005 and KT-1000 P = 0.0096), and in Group B (RSA P = 0.005 and KT-1000 P = 0.004).
Neither patients in Group A nor Group B had any motion problems in terms of restricted knee flexion or extension at 2-year follow-up. The ROM did not differ between them pre-operatively, after 6 months and 2 years. The ROM data is presented in Tables 4 and 5.
The clinical results in terms of the Lysholm score, Tegner activity level, one-leg-hop test and IKDC did not differ between the two groups (Table 5).
Five patients had a partial meniscal tear, two medial in Group A and one medial and two laterals in Group B. These tears were addressed with a partial resection at the time of the index operation.
Discussion
Our hypothesis that permitting active and passive extension training without any restrictions in extension immediately after an ACL reconstruction, using bone-patellar tendon-bone graft, could be done without jeopardising the A–P laxity of the knee was verified.
Our finding has clinical relevance when it comes to the choice of post-operative rehabilitation after an ACL reconstruction, using bone-patellar tendon-bone graft. According to our findings permission of full active and passive extension training immediately after an ACL reconstruction did not increase the post-operative A–P laxity.
The current trend in the post-operative management of ACL reconstruction is early active extension training after the ACL reconstruction. Our aim was to as accurately as possible evaluate the A–P laxity in the knee after an ACL reconstruction.
To our knowledge, there are no randomised studies with adequate follow-up after an ACL reconstruction, which have used such an accurate method as RSA to evaluate the effect of different training protocols on the A–P laxity of the knee.
In spite of the lack of randomised studies many rehabilitation programmes already include early full active and/or passive extension after an ACL reconstruction.
The principles of accelerated rehabilitation were described by Shelbourne and Nitz [35] and were further modified by Muneta et al. [26]. The results have been considered as uniformly good, especially in patients operated on using bone-patellar tendon-bone autograft. Muneta et al. [26] described less good results in patients operated on using hamstrings (multi-strand semitendinosus tendon), especially in terms of increased knee laxity at follow-up. A recent study has shown that the time to recovery is often longer than 6 months [1].
We choose to use a rehabilitation brace to adapt to clinical practice. One important problem with such braces is that they do not guarantee concordance with the true extension of the patient’s knee [22]. In the present study we found that the brace used could allow hyperextension to 10°, provided these conditions were found in the knee to be tested.
We used the RSA method and the conventional KT-1000 arthrometer. The first of these methods relies on skeletal markers and has high resolution and reproducibility. There are some disadvantages using RSA because it is an invasive method, and each patient and investigation will take longer time and need more resource, for instance a specially designed laboratory, is needed. Even if the radiation is lower than for an ordinary radiographic examination, the examinations add to the total radiation burden. The advantage with highly accurate measurements is that they can provide important information from a relatively small patient cohort and after a comparatively short period of time. This means that the numbers of patients exposed to a new unproven treatment or implant can be limited to a minimum.
The present study did not reveal any significant differences in terms of knee laxity, using either method. The recorded values differed between the RSA and the KT-1000. All patients in the study underwent an arthroscopy where the ACL rupture was verified. Ten of these 22 patients had a pre-operative side-to-side difference less than 3 mm using the KT-1000. This underlines one of the problems using the KT-1000. The results are similar compared with previous studies [16], which have shown that KT-1000 underestimates the side-to-side difference. Despite the fact that the results of the two methods were not numerically identical, both indicated that early active and passive extension training is safe and that it does not jeopardise the A–P laxity of the knee.
Besides the primary aim we studied some clinical parameters such as ROM, the Lysholm score, Tegner activity level, one-leg-hop test and IKDC. The reason for this was to show that these patients had a clinical status similar to other ACL studies with a clinical approach at the follow-up examinations.
We were not able to document any clinical superiority in the knee function using the early extension training protocol at the 6-month or 2-year follow-up.
Some studies have shown that aggressive extension immediately after an ACL reconstruction might increase the laxity of the knee [8], and some other studies have shown that females have more laxity in their knees than males after an ACL reconstruction [39]. In the randomising process it so happened that there were more females, who were allowed early extension. Despite this asymmetry, which theoretically could imply increased risk of graft elongation in the early extension group, no such effect was observed.
One interesting and potentially important finding was that more patients regained hyperextension (5–10°) of the injured knee in the early extension training group, compared with the traditional group. This finding is in line with the observation of Shelbourne and Nitz [35], who found that extension losses was deleterious in terms of the ability of patients to return to sports participation. Loss of extension also increased the risk of further surgical interventions due to symptomatic extension deficit. These researchers also noted that patients who started the closed- or open kinetic exercises early after the ACL reconstruction regained quadriceps strength earlier than those who were treated with a delayed protocol. The accelerated protocol was also shown to be beneficial in terms of less anterior knee pain [35].
We conclude that it appears to be safe, to start early active and passive extension training without restriction in extension immediately after the ACL reconstruction with a patellar tendon autograft.
References
Augustsson J, Thomée R, Karlsson J (2004) Ability of a new hop test to determine functional deficits after anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc 12:350–356
Ballantyne BT, French AK, Heimsoth SL, Kachingwe AF, Lee JB, Soderberg GL (1995) Influence of examiner experience and gender on interrater reliability of KT-1000 arthrometer measurements. Phys Ther 75:898–906
Bjordal JM, Arnöy F, Hannestad B, Strand T (1997) Epidemiology of anterior cruciate ligament injuries in soccer. Am J Sports Med 25:341–345
Bragdon CR, Malchau H, Yuan X, Perinchief R, Kärrholm J, Borlin N, Estok DM, Harris WH (2002) Experimental assessment of precision and accuracy of radiostereometric analysis for the determination of polyethylene wear in a total hip replacement model. J Orthop Res 20:688–695
Brandsson S, Karlsson J, Eriksson BI, Kärrholm J (2001) Kinematics after tear in the anterior cruciate ligament. Dynamic bilateral radiostereometric studies in 11 patients. Acta Orthop Scand 72:372–378
Brandsson S, Karlsson J, Swärd L, Kartus J, Eriksson BI, Kärrholm J (2002) Kinematics and laxity of the knee joint after anterior cruciate ligament reconstruction. Am J Sports Med 3:361–367
Bynum EB, Barrack RL, Alexander AH (1995) Open versus closed chain kinetic exercises after anterior cruciate ligament reconstruction—a prospective randomized study. Am J Sports Med 23:401–406
DeVita P, Hortobagyi T, Barrier J (1998) Gait biomechanics are not normal after anterior cruciate ligament reconstruction and accelerated rehabilitation. Med Sci Sports Exerc 30:1481–1488
Fleming BC, Peura GD, Abate JA, Beynnon BD (2001) Accuracy and repeatability of Roentgen stereophotogrammetric analysis (RSA) for measuring knee laxity in longitudinal studies. J Biomech 34:1355–1359
Fridén T, Ryd L, Lindstrand A (1992) Laxity and graft fixation after reconstruction of the anterior cruciate ligament. A roentgen stereophotogrammetric analysis of 11 patients. Acta Orthop Scand 63:80–84
Gauffin H, Tropp H, Odenrick P (1988) Effect of ankle disk training on postural control in patients with functional instability of the ankle joint. Int J Sports Med 9:141–144
Hefti F, Muller W, Jakob RP, Staubli HU (1993) Evaluation of knee ligament injuries with the IKDC form. Knee Surg Sports Traumatol Arthrosc 1:226–234
Jonsson H, Elmqvist L-G, Kärrholm J, Fugl-Meyer A (1992) Graft lengthening after surgery of anterior cruciate ligament. Roentgen stereophotogrammetry of 32 cases. Acta Orthop Scand 63:587–592
Jonsson H, Kärrholm J (1994) Three-dimensional knee joint movements during a step-up: evaluation after anterior cruciate ligament rupture. J Orthop Res 12:769–779
Jonsson H, Kärrholm J, Elmqvist LG (1989) Kinematics of active knee extension after tear of the anterior cruciate ligament. Am J Sports Med 17:792–802
Jonsson H, Kärrholm J, Elmqvist LG (1993) Knee laxity after cruciate ligament injury. The KT-1000 arthrometer versus roentgen stereophotogrammetry in 86 patients. Acta Orthop Scand 64:567–570
Jorn LP, Fridén T, Ryd L (1998) Simultaneous measurements of sagittal knee laxity with an external device and radiostereometric analysis. J Bone Joint Surg Br 80:169–172
Järvinen M, Natri A, Lehto M, Kannus P (1995) Reconstruction of chronic anterior cruciate ligament insufficiency in athletes using a bone-patellar tendon-bone autograft. A two-year follow up study. Int Orthop 19:1–6
Kärrholm J (1989) Roentgen stereophotogrammetry. Review of orthopaedic applications. Acta Orthop Scand 60:491–503
Kärrholm J, Brandsson S, Freeman MAR (2000) Changes of axial tibial rotation at the weight-bearing knee studied by RSA. J Bone Joint Surg Br 82:1201–1203
Kärrholm J, Selvik G, Elmqvist LG, Hansson LI, Jonsson H (1988) Three-dimensional instability of the anterior cruciate deficient knee. J Bone Joint Surg Br 70:777–783
Mikkelsen C, Cerulli G, Lorenzini M, Bergstrand G, Werner S (2003) Can a post-operative brace in slight hyperextension prevent extension deficit after anterior cruciate ligament reconstruction? A prospective randomized study. Knee Surg Sports Traumatol Arthrosc 11:318–321
Mikkelsen C, Werner S, Eriksson E (2000) Closed kinetic chain alone compared to combined open and closed kinetic chain exercises for quadriceps strengthening after anterior cruciate ligament reconstruction with respect to return to sports: a prospective matched follow-up study. Knee Surg Sports Traumatol Arthrosc 8:337–342
Morrisey MC, Drechsler WI, Morrisey D, Knight PR, Armstrong P, McAuliffe T (2002) Effects of distally fixated versus nondistally fixated leg extensor resistance training on knee pain in the early period after anterior cruciate ligament reconstruction. Phys Ther 82:35–43
Morrisey MC, Hudson ZL, Drechsler WI, Coutts FJ, Knight PR, Ki JB (2000) Effects of open versus closed kinetic chain training on knee laxity in the early period after anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc 8:343–348
Muneta T, Sekiya I, Ogiuchi T, Yagishita K, Yamamoto H, Shinomiy K (1998) Effects of aggressive early rehabilitation on the outcome of anterior cruciate ligament reconstruction with multi-strand semitendinosus tendon. Int Orthop 22:352–356
Nielsen AB, Yde J (1989) Epidemiology and traumatology of injuries in soccer. Am J Sports Med 17:803–807
Noyes FR, Mangine RE, Barber S (1987) Early knee motion after open and arthroscopic anterior cruciate ligament reconstruction. Am J Sports Med 15:149–160
Onsten I, Berzins A, Shott S, Sumner DR (2001) Accuracy and precision of radiostereometric analysis in the measurement of THR femoral component translations: human and canine in vitro models. J Orthop Res 19:1162–1167
Rodeo SA, Arnoczky SP, Torzilli PA, Hidaka C, Warren RF (1993) Tendon healing in a bone tunnel. A biomechanical and histological study in the dog. J Bone Joint Surg Am 75:1795–1803
Roos H, Karlsson J (1998) Anterior cruciate ligament instability and reconstruction. Review of current trends in treatment. Scand J Med Sci Sports 8:426–431
Selvik G (1974, 1989) Roentgen stereophotogrammetry. A method for the study of the kinematics of the skeletal system. Thesis, Lund: University of Lund Sweden, Reprint: Acta Orthop Scand (Suppl. 232)
Sernert N, Kartus J, Köhler K, Ejerhed L, Karlsson J (2001) Evaluation of the reproducibility of the KT-1000 arthrometer. Scand J Med Sci Sports 11:120–125
Shelbourne KD, Klootwyk M, Wilckens J, Decarlo M (1995) Ligament stability two to six years after anterior cruciate ligament reconstruction with autogenous patellar tendon graft and participation in accelerated program. Am J Sports Med 23:575–579
Shelbourne KD, Nitz P (1990) Accelerated rehabilitation after anterior cruciate ligament reconstruction. Am J Sports Med 18:292–299
Shelbourne KD, Patel DV (1996) Rehabilitation after autogenous bone-patellar tendon-bone ACL reconstruction. Instr Course Lect 45:263–273
Shelbourne KD, Patel DV (1999) Treatment of limited motion after anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc 7:85–92
Shelbourne KD, Rowdon GA (1994) Anterior cruciate ligament injury. The competitive athlete. Sports Med 17:132–140
Shino K, Inoue M, Horibe S, Nakamura H, Ono K (1987) Measurement of anterior instability of the knee. A new apparatus for clinical testing. J Bone Joint Surg Br 69:608–613
Stark J (1850) Two cases of ruptured crucial ligaments of the knee joint. Edinb Med Surg 74:267–271
Tegner Y, Lysholm J, Lysholm M, Gillquist J (1986) A performance test to monitor rehabilitation and evaluate anterior cruciate ligament injuries. Am J Sports Med 14:156–159
Yack HJ, Collins CE, Whieldon TJ (1993) Comparison of closed and open kinetic chain exercise in the anterior cruciate ligament-deficient knee. Am J Sports Med 21:49–54
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Isberg, J., Faxén, E., Brandsson, S. et al. Early active extension after anterior cruciate ligament reconstruction does not result in increased laxity of the knee. Knee Surg Sports Traumatol Arthrosc 14, 1108–1115 (2006). https://doi.org/10.1007/s00167-006-0138-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-006-0138-2