Abstract
The goal of this study was to examine the use of a constructed wetland to mitigate the ecological impacts of simulated diazinon runoff from agricultural fields into receiving waters, via 48 h aqueous and sediment bioassays using Hyalella azteca. Aqueous animal 48 h survival varied temporally and spatially in conjunction with measured diazinon concentrations. Sediment H. azteca survival varied temporally and spatially in conjunction with measured diazinon concentrations, but less than aqueous exposures, confirming that sediment bound diazinon was less bioavailable than aqueous diazinon.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Constructed wetlands used to mitigate pesticide runoff from agricultural fields into receiving systems (e.g., lakes, rivers, streams) have been successful in reducing the concentrations of non-point source pollutants (Moore et al., 2000; Moore et al., 2002). Such wetlands also have important functions in enhancing the water quality and ecological values (Moore et al., 2002) and different phases (i.e., aqueous and sediment) have separate roles, as either sinks or sources, in determining the effectiveness of wetlands in mitigating pesticide toxicity. For these reasons, the importance of elucidating the potential effectiveness of wetlands in reducing pesticide toxicity to aquatic biota exposed to these different phases needs to be addressed.
The organophosphate insecticide diazinon (O, O-diethyl O-[2-isopropyl-6-methyl-4pyrimidinyl] phosphothiate) was used as a model contaminant in a constructed wetland designed to mitigate runoff from an agricultural field. Diazinon is used on a variety of agricultural crops such as fruit trees, corn, and tobacco, as well as domestically in homes and gardens for pest control (e.g., fire ants) (Burkepile et al., 2000). Approximately 6 million kg of diazinon are applied annually in the United States and, as a result, it contributes to the non-point-source contamination of aquatic environments (Banks et al., 2003).
This study examined the use of a constructed wetland to mitigate the ecological impacts of simulated diazinon runoff from agricultural fields into receiving aquatic systems by using standard 48 h aqueous and sediment bioassays with the freshwater test organism, Hyalella azteca. H. azteca (order: Amphipoda) is a freshwater crustacean that is an epibenthic detritivore, closely associated with surficial sediments. H. azteca occurs in wetlands and lakes throughout much of North America and is an important food source for birds, fish, amphibians, and larger invertebrates (de March, 1981).
Materials and Methods
The constructed wetland used was designed for the mitigation of agricultural contaminant runoff (e.g., sediment, pesticides, nutrients). The wetland was divided into three cells and included a sediment retention pond (SRP), primary cell (1° cell), and a secondary cell (2° cell) located adjacent to Beasley Lake in Sunflower County, Mississippi, USA. The constructed wetland was dosed with 160 μg/L diazinon (active ingredient) as Diazinon 4E® and 403,000 μg/L sediment (as a carrier) simulating a single 1.3 cm rainfall event and runoff from a 14 ha agricultural field. A pump was used to deliver water from Beasley Lake to the constructed wetland at a rate of 3,800 L/min for the duration of the four-hour simulated runoff event. Two liters of water and one liter of sediment were collected from each wetland cell 8 h, 48 h, 7 d, 15 d, and 27 d after initial dosing. Samples were preserved on ice and transported to the United States Department of Agriculture Agricultural Research Service (USDA-ARS) National Sedimentation Laboratory, Oxford, Mississippi for biological and chemical analysis.
Twenty-two aqueous (six from the University of Mississippi field station and 16 from the constructed wetland) and 17 wetland sediment samples were analyzed for diazinon. Analytical chemistry was conducted according to Bennett et al. (2000) using a Hewlett-Packard 6890 gas chromatograph equipped with dual HP 7683 ALS autoinjectors. Upon arrival, 1 L aliquot of aqueous samples were subsampled, extracted by sonification with reagent-grade KCl and 100 ml pesticide-grade ethyl acetate, dried over anhydrous sodium sulfate, subjected to cleanup by silica gel column chromatography, and concentrated to 1 mL for analysis (Cooper et al. 2003). Sediment samples were dried, ground, pre-wetted with ultrapure water followed by the addition of ethyl acetate. The mixture was sonificated and centrifuged (2000–2500 g). The extract was concentrated to near dryness using a nitrogen evaporator and the solvent was exchanged into hexane. The quantification levels for aqueous and sediment analyses were 0.01 μg/L and 0.1 μg/kg, respectively. Mean extraction efficiencies based on fortified samples were >90% for water and sediment.
Forty-eight-hour static, nonrenewal, aqueous toxicity tests using Hyalella azteca obtained from cultures maintained at the USDA-ARS National Sedimentation Laboratory were conducted according to the modified United States Environmental Protection Agency (USEPA, 1994) protocol for conducting aqueous reference toxicity tests. Animals passing through a 425 μm stainless-steel mesh sieve, but retained by a 250 μm stainless-steel mesh sieve (approximately 4-5 d old) were collected for the experiment. Triplicate aqueous exposures each consisted of 200 mL of wetland sample water and three, 2-cm-diameter Norway maple leaf discs as a substrate and food (Moore et al., 1998; Burkepile et al., 2000). Ten H. azteca were placed in each exposure chamber. Similarly, 48 h sediment bioassays were conducted using the modified USEPA (1994) protocol as per the aqueous tests using 40 g wet sediment and 160 mL overlying water. Overlying water, free from diazinon (below the detection limit of 0.01 μg/L) and other priority pollutants, was obtained from the University of Mississippi Field Station, filtered to remove particulate matter using MFS 0.45 μm polymembrane filters, and hardness and alkalinity adjusted with NaHCO3 and CaCl to values between 60–80 mg/L as CaCO3 (Deaver and Rodgers, 1996; Burkepile et al., 2000). Toxicity tests were conducted in a Powers Scientific animal growth chamber with a 16:8 h photoperiod at 20 ± 1°C. Mean measured physical and chemical water characteristics were: temperature, 20–21°C, pH, 7.1–7.7, dissolved oxygen, 6.6–7.7 mg/L, conductivity, 48–242 μmhos/cm, hardness, 30–74 mg/L as CaCO3, and alkalinity, 34–47 mg/L as CaCO3 (APHA 1998). The bioassay endpoint measured was 48 h survival.
H. azteca 48 h aqueous and sediment survival data were analyzed using descriptive statistics and one-way analysis of variance (ANOVA) with Tukey’s multiple range test. If data failed parametric assumptions, a Kruskal-Wallis one-way ANOVA on ranks was utilized with Dunn’s multiple range test. Data analysis was conducted using SigmaStat® v.2.03 statistical software (SPSS, 1997).
Results and Discussion
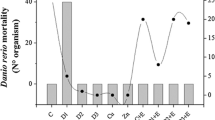
Chemical analysis revealed spatial and temporal variation in diazinon concentrations within aqueous and sediment samples among all three wetland cells (Table 1). All wetland cells (SRP, 1° cell, finishing cell) had measurable amounts of diazinon within the aqueous phase 8 h after dosing. However, diazinon was not detected in the sediment within the sediment retention pond, representative of water flow conditions within the constructed wetland and adsorption of diazinon by sediment as the water flow slowed. Aqueous and sediment diazinon concentrations typically decreased with increasing time periods due in part to material degradation, desorption, and advection. By 27 d, SRP and the area closest to the injection point, the sediment retention pond. Whereas, the greatest aqueous and sediment diazinon concentrations occurred within the primary wetland cell and, to a lesser extent, the finishing wetland cell. Such a pattern shows a rapid flow of water and associated pesticide out of the initial wetland cell and slower movement into the remaining two cells. Similar spatial and temporal patterns of transfer/transformation were observed for two other organophosphate insecticides, chlorpyrifos and methyl parathion, within aqueous and sediment phases in constructed wetlands (Moore et al., 2002; Schulz et al., 2003).
Hyalella azteca 48 h survival in aqueous exposures varied temporally and, to a lesser extent, spatially in conjunction with measured diazinon concentrations (Tables 1 and 2). Limited pretreatment (time 0 h) aqueous survival occurred due to the very soft nature of the natural wetland water (Table 2). Aqueous hardness and alkalinity ranged from 17.1–51.3 mg/L as CaCO3 and 34.2–51.3 mg/L as CaCO3, resulting in suboptimal survival rates. Grapentine and Rosenberg (1992) observed H. azteca to be in low abundance or absent from lakes with lower calcium concentrations (<2 mg/L). However, clear patterns of exposure effects were observed. Aqueous survival decreased significantly, compared to time 0 h, in all three wetland cells (SRP, 1° cell, 2° cell) up to 7 d after diazinon dosing. By 15 d post-dosing, SRP showed a decrease in toxicity (13% survival), and by 27 d water from SRP showed a further nominal decrease in toxicity with the 2° cell increasing from 0% to 3% survival. The limited initial (0 h) survival due to water softness contributed to lower than expected observed survival with diazinon concentrations of <1 μg/L. Reported H. azteca 48 h LC50s ranging from 15–22 μg diazinon/L (Giddings et al., 1996; Werner and Nagel, 1997; Burkepile et al., 2000) are much greater than the concentrations effects observed in the current study (≥ 0.1 μg diazinon/L).
H. azteca survival in diazinon-contaminated sediment also varied temporally and spatially in conjunction with measured diazinon concentrations (Table 1 and 2), but to a much lesser extent than aqueous exposures confirming that sediment bound diazinon was less bioavailable than aqueous diazinon. Similar results were observed with another organophosphate insecticide, chlorpyrifos, where sediment 10–d LC50 values were 100–600 times greater than aqueous 10 d LC50 values for Chironomus tentans (Ankley et al., 1994). Patterns of animal survival showed no significant temporal changes in SRP or 1° wetland cells. The 2° cell showed a significant decrease in survival 48 h after dosing, although by 7 d and thereafter survival was greater than 80%. Although 1° cell had the greatest measured sediment diazinon concentrations 8 h, 7 d, and 15 d after dosing, relatively high H. azteca survival is associated with higher total organic carbon (TOC) in these sediments. Sediment from SRP had only 1–1.5% TOC, 1° cell had 2–5% TOC and 2° cell had 1.8–2% TOC. The influence of TOC on the bioavailability of insecticides with low water solubility to Hyalella azteca has been documented previously (Nebeker et al., 1989; Ankley et al., 1994). Thus the higher TOC in the 1° cell sediment mitigated the greater diazinon concentrations. Significant spatial variation only occurred 48 h after dosing, showing decreasing survival from SRP to 1° cell and 2° cell. Again, spatial H. azteca responses coincide with measured diazinon concentrations as seen temporally, with exceptions associated with differences in sediment TOC.
Based upon the responses of Hyalella azteca to aqueous and sediment diazinon contamination, these data indicate that sediment diazinon can move from a contaminant sink during initial pesticide influx to a source of diazinon contamination affecting nontarget aquatic organisms for days to weeks after entering a constructed wetland. Further studies are needed to elucidate the relationship between aqueous and sediment phases in pesticide contamination within aquatic systems and associated effects on nontarget aquatic organisms.
References
American Public Health Association (APHA) (1998) Standard methods for the examination of water and wastewater. 20th ed. Washington, DC
Ankley GT, Call DJ, Cox JS, Kahl MD, Hoke RA, Kosian PA (1994) Organic carbon partitioning as a basis for predicting the toxicity of chlorpyrifos in sediments. Environ Toxicol Chem 13:621–626
Banks KE, Wood SH, Matthews C, Thuesen KA (2003) Joint acute toxicity of diazinon and copper to Ceriodaphnia dubia. Environ Toxicol Chem 22:1562–1567
Bennett ER, Moore MT, Cooper CM, Smith S Jr (2000) Method for the simultaneous extraction and analysis of two current use pesticides, atrazine and lambda-cyhalothrin, in sediment and aquatic plants. Bull Environ Contam Toxicol 64:825–833
Burkepile DE, Moore MT, Holland MM (2000) Susceptibility of five nontarget organisms to aqueous diazinon exposure. Bull Environ Contam Toxicol 64:114–121
Deaver E, Rodgers JH Jr (1996) Measuring bioavailable copper using anodic stripping voltammetry. Environ Toxicol Chem 15:1925–1930
de March BGE (1981) Hyalella azteca (Saussure). In: Lawrence SG (ed.) Manual for the Culture of Selected Freshwater Invertebrates. Canadian Spec Publ Fish Aquat Sci 54:61–77
Cooper C, Smith S Jr, Moore M (2003) Surface water, ground water and sediment quality in three oxbow lake watersheds in the Mississippi delta agricultural region: pesticides. Int J Ecol Environ Sci 29:171–284
Giddings JM, Biever RC, Annunziato MF, Hosmer AJ (1996) Effects of diazinon on large outdoor pond microcosms. Environ Toxicol Chem 15:618–629
Grapentine LC, Rosenberg DM (1992) Responses of the freshwater amphipod Hyalella azteca to environmental acidification. Can J Fish Aquat Sci 49:52–64
Moore MT, Huggett DB, Gillespie WB Jr, Rodgers JH Jr, Cooper CM (1998) Comparative toxicity of chlordane, chlorpyrifos, and aldicarb to four aquatic testing organisms. Arch Environ Contam Toxicol 34:152–157
Moore MT, Rodgers JH Jr, Cooper CM, Smith S Jr (2000) Constructed wetlands for mitigation of atrazine-associated agricultural runoff. Environ Poll 110:393–399
Moore MT, Schulz R, Cooper CM, Smith S Jr, Rodgers JH Jr (2002) Mitigation of chlorpyrifos runoff using constructed wetlands. Chemosphere 46:827–835
Nebeker AV, Schuytema GS, Griffis WL, Barbitta JA, Carey LA (1989) Effect of sediment organic carbon on survival of Hyalella azteca exposed to DDT and endrin. Environ Toxicol Chem 8:705–718
Schulz R, Moore MT, Bennett ER, Farris JL, Smith S Jr, Cooper CM (2003) Methyl parathion toxicity in vegetated and nonvegetated wetland mesocosms. Environ Toxicol Chem 22:1262–1268
Statistical Package for the Social Sciences (SPSS), Inc. (1997) SigmaStat for Windows version 2.03
USEPA (1994) Methods for measuring the toxicity and bioaccumulation of sediment associated contaminants with freshwater invertebrates. EPA 600/R-94/024 Washington, DC
Werner I, Nagel R (1997) Stress proteins HSP60 and HSP70 in three species of amphipods exposed to cadmium, diazinon, dieldrin and fluoranthene. Environ Toxicol Chem 16:2393–2403
Acknowledgments
Appreciation is extended to L. Arbuckle and R. W. Steinriede for sample collection and bioassay assistance as well as Erin Bennett, Kristie Willett, and Lloyd Southwick for reviewing earlier versions of the manuscript. Mention of equipment, computer programs, or a pesticide does not constitute an endorsement for use by the US Department of Agriculture nor does it imply pesticide registration under FIFRA as amended. All programs and services of the USDA are offered on a nondiscriminatory basis without regard to race, color, national origin, religion, sex, marital status, or handicap.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Smith, S., Lizotte, R.E. & Moore, M.T. Toxicity Assessment of Diazinon in a Constructed Wetland Using Hyalella azteca . Bull Environ Contam Toxicol 79, 58–61 (2007). https://doi.org/10.1007/s00128-007-9215-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-007-9215-6