Abstract
Background
Nodal pelvic/retroperitoneal recurrent prostate cancer (PCa) after primary therapy can be treated with salvage lymph node dissection (salvage-LND) in order to delay disease progression and offer cure for a subset of patients. Whether adjuvant radiotherapy (ART) in affected regions improves the outcome by elimination of residual tumour burden remains unclear.
Methods
A total of 93 patients with exclusively nodal PCa relapse underwent choline-positron-emission tomography-computed-tomography-directed pelvic/retroperitoneal salvage-LND; 46 patients had surgery only and 47 patients received ART in regions with proven lymph node metastases. In case of subsequent prostate specific antigen (PSA) progression, different imaging modalities were performed to confirm next relapse within or outside the treated region (TR). Mean follow-up was 3.2 years.
Results
Lymphatic tumour burden was balanced between the two groups. Additional ART resulted in delayed relapse within TR (5-year relapse-free rate 70.7 %) versus surgery only (5-year relapse-free rate 26.3 %, p < 0.0001). In both treatment arms, time to next relapse outside the TR was almost equal (median 27 months versus 29.6 months, p = 0.359). With respect to the detection of the first new lesion, regardless if present within or outside the TR, 5 years after the treatment 34.3 % of patients in the group with additional ART were free of relapse, versus 15.4 % in the surgery only group (p = 0.0122). ART had no influence on the extent of PSA reduction at latest follow-up compared to treatment with surgery only.
Conclusion
ART after salvage-LND provides stable local control in TR and results in overall significant improved next-relapse-free survival, compared to patients who received surgery only in case of nodal PCa-relapse.
Zusammenfassung
Hintergrund
Das nodal positive Prostatakarzinom(PCa)-Rezidiv nach Primärtherapie kann durch eine Salvage-Lymphadenektomie (Salvage-LND) therapiert werden. Der Krankheitsprogress wird aufgehalten und selektionierte Patienten erhalten eine zweite Chance auf eine Kuration. Ob eine adjuvante Strahlentherapie (ART Eradikation von verbleibenden Tumorzellen in der betreffenden Region) die Tumorfreiheitsrate verbessert, ist ungeklärt.
Material und Methoden
Insgesamt 93 Patienten mit einem ausschließlich nodalen PCa-Rezidiv wurden nach Diagnostik mittels Cholin-Positronenemissionstomographie/Computertomographie einer Salvage-LND unterzogen; 46/93 Patienten wurden ausschließlich operiert, 47/93 Patienten erhielten zusätzlich eine ART. Im Fall einer PSA-(prostataspezifisches-Antigen)-Progression wurde durch bildgebende Verfahren das nächste Rezidiv innerhalb oder außerhalb der behandelten Region (TR) diagnostiziert. Der mittlere Beobachtungszeitraum lag bei 3,2 Jahren.
Ergebnisse
Die Anzahl der Lymphknotenmetastasen war in den zwei Gruppen gleichverteilt. Eine zusätzliche ART bewirkte ein verzögertes Auftreten von neuen Metastasen in der TR (metastasenfreies 5-Jahres-Überleben 70,7 %) im Vergleich zur alleinigen Salvage-LND (metastasenfreies 5-Jahres-Überleben 26,3 %; p < 0,0001). Der Zeitpunkt bis zur Diagnose von Metastasen außerhalb der TR war in beiden Behandlungsarmen nicht signifikant unterschiedlich (27 Monate versus 29,6 Monate; p = 0,359). Unabhängig von der Lokalisation der neuen Metastasen lag das metastasenfreie 5-Jahres-Überleben bei Patienten mit einer Kombinationstherapie bei 34,3 %, bei Patienten mit alleiniger Operation hingegen bei 15,4 % (p = 0,0122). Die zusätzliche Strahlentherapie verursachte, verglichen mit einer alleinigen Operation, keinen signifikanten Unterschied bezüglich der PSA-Wert-Reduktion am Ende des Beobachtungszeitraums.
Schlussfolgerung
Eine zusätzliche ART bewirkt eine stabile lokale Tumorkontrolle in der behandelten Region und resultiert in einem signifikant verlängerten metastasenfreien Überleben verglichen mit einer alleinigen Salvage-LND beim nodalen Prostatakarzinomrezidiv.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Radical prostatectomy (RP) and radiotherapy (RT) are the standard treatment options for clinically localized prostate cancer (PCa) [10, 13, 23, 33]. However, recurrence after surgical removal of the prostate is well described. According to the level of prostate-specific antigen (PSA), Gleason score and TNM stage, biochemical relapse after RP ranges from 10–53 %, most of them (95 %) occurring within the first 5 years [4, 6, 12, 32]. Different recurrence patterns are known; (1) evidence of local recurrence in the prostatic fossa; (2) locoregional pelvic lymph node metastases (LNM), (3) distant metastases (LNM, soft tissue, bone) and (4) a combination of local and distant recurrence [14]. Choline positron emission tomography/computed tomography (PET/CT) has been shown to be a valuable tool to localize recurrent PCa, especially to detect lymph node (LN) involvement at the regional level with high accuracy [19, 27]. Salvage-RT is the mainstay therapy in the setting of recurrence of tumour in prostatic fossa, offering the potential of cure [3, 22, 26, 31]. Conversely, patients with distant metastases and/or regional limited pelvic LNM are treated with antihormonal therapy (AHT) regardless of the exact site of recurrence. AHT causes serious side effects and is of limited benefit due to the development of castration-resistant PCa and therefore serves merely as palliative therapy [1, 15].
Recently our group demonstrated that salvage lymph node ablation, either done by surgery or high precision RT, is an effective alternative strategy instead of AHT. Salvage lymph node dissection (salvage-LND) is able to defer cancer progression and offers for a subset of patients the chance of cure [17, 28, 29, 34]. However the role of adjuvant radiotherapy (ART) after salvage-LND has been shown to be feasible but its impact on local control, disease progression and pattern of failure has not been evaluated in detail. The rationale for ART is to eliminate possible residual tumour cells after choline-PET/CT-directed surgery. Although choline PET/CT has high specificity for detection of LNM, the lesion-based sensitivity of choline PET/CT is limited due to limited spatial resolution of PET/CT scanners [19, 27].
The aim of the current retrospective analysis was to evaluate the influence of ART after choline-PET/CT-directed salvage-LND concerning local tumour control (next relapse in treated region) and time to detection of new lesions outside of the treated region (TR) versus surgery only.
Materials and methods
Patients
A total of 93 patients with PCa recurrence (PSA > 0.2 ng/ml after RP, PSA 2 ng/ml above the nadir after RT in two consecutive measurements) and a choline-PET/CT positive for LNM ([11C]-choline or [18F]-fluoroethylcholine PET/CT) were treated with pelvic and/or retroperitoneal salvage-lymphadenectomy at Freiburg Hospital from 2005–2013. Inclusion criteria were verification of biochemical recurrence, presence of [11C]-choline- or [18F]-fluoroethylcholine-PET/CT-positive LNM (regardless of number) without detectable bone or visceral metastases, Charlson Comorbidity Index ≤ 2, age < 80 years. Antihormonal therapy, if administered beforehand, had to be discontinued for at least 8 weeks. All patients underwent additional bone scintigraphy to confirm exclusion of skeletal metastases. Regular PSA follow-up periods after salvage-LND were every 3 months, in case of rising PSA level every 8 weeks. Choline PET/CT, CT, MRI or bone scintigraphy was performed in case of PSA progression (two consecutive PSA-level rises). A new relapse within the TR was defined by the detection of at least one new lesion in the field of surgery or surgery plus ART, respectively. Relapse outside the TR was defined by the detection of at least one new lesion outside the field of surgery or surgery plus ART, respectively. ART was not performed for the following reasons in 46 patients: either because patients refused treatment (19/46), attending physicians did not follow our recommendation (12/46), the radiation port was considered too large so that the risk of toxicity would have been disproportionately high according to the individual situation (5/46), previous pelvic radiotherapy had been performed (5/46), or in case of rapid cancer progression (5/46).
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975 (in its most recently amended version). All patients gave signed written consent with respect to this experimental therapy and retrospective analysis. The local review board reviewed and approved the study (No. 135/12_130160). Follow-up of overall 93 patients was a mean 38.9 months (SD 29.3) and a median of 28.7 months.
Choline PET/CT and image interpretation
Depending on the availability of the radionuclides, whole body [11C]-choline/[18F]-fluoroethylcholine PET/CT were performed using routine in-house acquisition protocols including orally and intravenous contrast-enhanced CT. Board certified nuclear medicine specialists and radiologists: HCR (7 years’ experience in reading choline PET/CT and experience in nuclear medicine, 14 years’ experience in diagnostic radiology), GW (5 years’ experience in reading choline PET/CT and experience in nuclear medicine) and TK (6 years’ experience in diagnostic radiology) independently evaluated PET/CT studies, bone scans, CT and MRI scans using a PACS workstation (AGFA HealthCare, Aachen, Germany). Different findings were resolved by consensus reading.
Salvage lymph node dissection
According to the affected regions directed by choline PET/CT (pelvic left/right and/or retroperitoneal) salvage-LND was performed. Salvage-LND included complete removal of lymphatic and fatty tissue of pelvic or retroperitoneal regions. All salvage-LNDs were performed by the same surgeon (WSS).
Histopathology of LNM
LNs were laminated longitudinally; a central slice was completely embedded in paraffin. Hematoxylin and eosin staining was used for primary diagnosis in morphologically clear cases. If required, immunohistochemistry (cytokeratins, PSA. DAKO, Glostrup, Denmark) was used to verify macro- or micrometastases.
Adjuvant radiotherapy
Pelvic and/or retroperitoneal great vessels served as guidance to define the clinical target volume (CTV). An approximately 8–10 mm margin around the vessels was drawn to define the CTV. Planning target volume (PTV) was 5–7 mm around the CTV. The cranial border of the retroperitoneal LN region was the renal vessels and the inferior border the aortic bifurcation. The cranial border of a pelvic region (left/right) was the aortic bifurcation. The external and internal iliac contours, including obturator node-subregion, were connected on each slice, carving out bowel loops and bladder. Presacral LN were included (inferior to S3), while the posterior border was the anterior sacrum and anterior border approximately 10 mm anterior to the anterior sacral bone. The inferior border of the pelvic CTV was the top of the femoral heads, which represent the bony landmark for the inguinal ligament. Bowel, bladder and bone had been carved out in every slice to fulfill the following dose constraints: small bowel V45 Gy < 250 ml, V50 Gy < 100 ml; rectum V50 Gy < 50 %, bladder V55 Gy < 50 % [9, 20]. Volumes of prior irradiation were excluded. LN regions were treated five times a week with 1.8 Gy/fraction up to a mean dose of 49.7 Gy with 3D conformal irradiation or intensity-modulated radiation therapy (IMRT; Table 2). The patients’ positioning was controlled using cone-beam CT (image-guided radiotherapy, IGRT). Due to dose constraints in adjacent normal tissues (small bowel, colon) the shrinking field technique in terms of dose escalation > 45 Gy to involved LN regions was performed in 11 of 47 cases. If imaging suggested presence of local recurrence (12 of 47 patients), based on MRI and choline PET/CT findings, the prostate fossa received dose escalation (mean 69.2 Gy). Linear accelerators with 6 and 10 MV photons were used equipped with electronic portal imaging.
Statistical analysis
Descriptive statistics was done by calculating means, medians and standard deviations (SD). Kaplan–Meier method was used to estimate time to next relapse within and/or outside the TR and to estimate cancer-specific survival. To analyze predictors for next relapse in the TR, univariate and multivariate Cox regression analysis was performed. Continuous variables were compared with a two-sided unpaired t-test. The χ2 test was used for analyzing contingency tables. The level of significance was indicated by a p-value < 0.05. PSA reduction in percent was calculated as follows: 100 – [PSA (ng/ml) at end of follow up × 100/PSA (ng/ml) at salvage-LND]. All statistics were done with SPSS v19 (IBM Corp. Armonk, NY, USA).
Results
Detailed information about primary PCa stage from 93 patients are shown in Table S1. Data from 93 choline-PET/CT-directed salvage-LNDs are presented in Table 1. Variables from salvage-LND obtained from patients with surgery only (n = 46) and those who had additional ART (n = 47) are listed separately. Data about 47 ARTs with respect to age of patients, regions, total dose and concurrent RT of the prostatic fossa are given in Table 2. Figure Sup.1A shows a representative choline PET/CT from a patient with LNM in the right obturator region in September 2012. Figure Sup.1B shows a choline PET/CT of the same patient in October 2013 after salvage-LND and ART of the pelvic regions. A representative IMRT-boost plan is shown in Figure Sup.1C. This patient received local boost irradiation up to a total dose of 56.6 Gy.
Univariate and multivariate proportional hazard regression model predicting the risk of next relapse within the treated region after different local treatments (surgery only versus surgery with ART) is shown in Table 3.
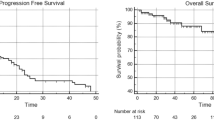
Figure 1a shows the Kaplan–Meier estimation of time to next relapse within the TR after either salvage-LND only or salvage-LND with ART. Median time to detectable next relapse in the TR after salvage-LND was 10 months. Probability for 5-year next-relapse-free survival in the TR after salvage-LND and ART was 70.7 versus 26.3 % for surgery only.
a Time to next relapse detected within the treated region (TR) from 46 patients who received salvage lymph node dissection (salvage-LND) only compared to 47 patients who received salvage lymph node dissection with adjuvant radiotherapy (salvage-LND and ART). b Time to next relapse detected outside the TR from 46 patients who received salvage-LND only compared to 47 patients who received salvage-LND combined with ART. c Time to next relapse detected either within or outside the TR from 46 patients who received salvage-LND only. d Time to next relapse detected either within or outside the TR from 47 patients who received salvage-LND and ART. e Time to next relapse, regardless if detected within or outside the TR, from 46 patients who received salvage-LND only compared to 47 patients who received salvage-LND with ART. Time to next relapse, regardless if detected within or outside the TR from all 93 patients. f Cancer-specific survival of 46 patients who received salvage-LND compared to 47 patients who received salvage-LND and ART and cancer-specific survival from overall 93 patients
Median time to next relapse outside of the TR was not significantly different between patients who received surgery only (29.6 months) and those who were treated with additional ART (27.0 months; p-value = 0.359; Fig. 1b).
Regarding the 46 patients who received salvage-LND only, median time to detection of next relapse within TR (9.9 months) or outside TR (29.6 months) was not significantly different (p = 0.425; Fig. 1c).
Time to next relapse with the focus on the location of the lesions from patients who had combination therapy is shown in Fig. 1d. Median time to detection of new lesions outside the TR was 27 months, whereas the 5-year next-relapse-free survival in the TR was 70.7 % (Fig. 1d).
Time to next relapse, regardless within or outside the TR, was significant different between the two cohorts (p = 0.0122). In patients with additional ART, the next relapse was detected 17 months later than in the cohort of patients with surgery only (1 month for salvage-LND vs. 27 months for salvage-LND and ART; Fig. 1e).
Time to next relapse of all 93 patients taken together was a median 21.6 months (Fig. 1e). Median cancer-specific survival was not significantly different between the two treatment arms (p = 0.819; Fig. 1f). Cancer-specific survival for all 93 patients was a median 102.2 months (Fig. 1f).
Frequency of LNM in pelvic regions was similar in the two cohorts (salvage-LND only 61 %, salvage-LND and ART 70 %, p-value = 0.388; Table 2). Regions (pelvic, retroperitoneal, pelvic and retroperitoneal) of histological proven LNM from patients who had salvage-LND only are shown in Fig. 2a. The regions of the leading lesions of the next relapse in 33 cases after salvage-LND only are shown in Fig. 2b; 40 % (13/33) of those patients again exhibited a relapse in pelvic regions with LNM.
a Illustration of the distribution (pelvic, retroperitoneal, pelvic and retroperitoneal) of histologically proven lymph node metastases (LNM) obtained from salvage lymph node dissection (salvage-LND) only. Numbers in coloured circles in the according regions represent LNM distribution from patient 1–46. Pie chart shows the overall regional distribution of LNM. b Illustration of lesion distribution of 33 events of next relapses after salvage-LND detected by imaging. Numbers in coloured circles indicate the leading lesion of patients 1–46. Pie chart shows the overall distribution of next relapse after salvage-LND. *Combinations: pelvic LNM and/or retroperitoneal LNM and/or bone metastases and/or miscellaneous M1 LNM. c Illustration of the distribution (pelvic, retroperitoneal, pelvic and retroperitoneal) of histologically proven LNM obtained from salvage-LND followed by adjuvant radiotherapy (ART). Numbers in coloured circles in the according regions represent LNM distribution from patients 1–47. Pie chart shows the overall distribution of lesions prior to salvage LND and ART. d Illustration of lesion distribution of 23 events of next relapses after salvage-LND and ART. Numbers in coloured circles indicate the leading lesions of patients 1–47. Pie chart shows the overall distribution of next relapses after surgery and radiotherapy. †Combinations: pelvic LNM and/or retroperitoneal LNM and/or bone metastases and/or miscellaneous M1 LNM
The locations of histologically proven LNM from 47 patients who received additional ART are shown in Fig. 2c. The pattern of next relapse after surgery and ART in 23 cases is shown in Fig. 2d. Only 4 % developed recurrent pelvic LNM again and 9 % pelvic and retroperitoneal LNM again. The proportion of patients with a new recurrent pelvic LNM was significantly different (p-value = 0.0009) between the two cohorts: surgery only 57.6 % (19/33) vs. combination therapy 13 % (3/23).
The extent of PSA reduction in percent after salvage-LND only or after salvage-LND and ART is shown in Fig. 3a, b. Overall, 30 of 46 (65.2 %) individuals after salvage-LND only and 33 of 47 (70.2 %) patients after combination therapy showed PSA reduction at the latest follow-up. The proportion of patients with PSA reduction under AHT at the end of follow-up was not significantly different between patients who were treated with surgery only (17/30; 56.7 %) and those who had combination therapy (63.6 %; p-value = 0.614).
Reduction of prostate-specific antigen (PSA) in percent at the end of follow-up compared to PSA level (ng/ml) at the time of salvage lymph node dissection for 46 patients who received surgery only (a) and those 47 men who received additional radiotherapy (b). Blue bars: no antihormonal therapy at the end of follow up (No AHT), orange bars: antihormonal therapy at the end of follow-up (AHT), red bars: chemotherapy at the end of follow-up (chemotherapy)
ART after salvage-LND resulted in no to mild toxicity (no grade 3 or 4 according to CTAE Criteria 4.0; [7]). The spectrum of adverse events and toxicity, respectively, at latest follow-up was almost equal to those before start of ART. A detailed description of toxicity, side effects and quality of life of this experimental approach was recently published by Jilg et al. [18].
Discussion
To the best of our knowledge this is the first study that analysed the effect of adjuvant radiotherapy after salvage-LND in case of nodal recurrent PCa in detail. We were able to demonstrate a “salvage of salvage strategy” due to the fact that about 45 % of our patients had prior to extended lymph node dissection and ART a previous salvage radiotherapy of the prostatic fossa because of a first biochemical relapse.
Removing of LNM in case of nodal relapse has been performed and studied at different sites in Europe, but subsequent adjuvant ART is even more experimental [17, 24, 30, 34]. However similar to the situation of primary high risk PCa presence of micrometastases in adjacent tissue right in the neighbourhood of resected primary cancer or LNM may be the origin of the next relapse. This consideration forms the base of this approach of ART after salvage-LND. Early RT in the setting of high-risk PCa after RP (pT3, R1) turned out to improve biochemical and clinical disease-free survival [2, 4, 13, 35, 36]. In addition adjuvant RT in primary node-positive PCa patients after RP resulted in excellent long-term outcome concerning biochemical recurrence-free survival and cancer-specific survival [8]. The role of pelvic irradiation versus prostate only in case of primary RT for aggressive advanced PCa remains controversial. Although the RTOG 9413 trial showed for whole pelvic RT a statistically significant benefit over prostate only (4-year progression-free-survival 54 vs. 47 %, p = 0.02), in the long-term analysis this advantage was no longer present [25]. Whether those previous results from RT/salvage-RT in different PCa stages can be translated into the ART of nodal recurrent PCa remains to be determined.
We were able to show that ART had a significant influence on local control (next relapse) in the treated region. Men who received ART after surgery exhibited robust local 5-year next-relapse-free survival of 70.7 % versus salvage-LND only with 26.3 % (Fig. 1a). Unfortunately we could not show that ART after salvage LND has an influence on the timepoint of detection of distant lesions (outside on the TR; Fig. 1b). It seems that distant metastatic placement had already taken place when ART was performed, but has not been detected by choline PET/CT. Consequently at a certain disease status, tumour progression seems irreversible in the majority of patients and may not be influenced by aggressive local therapy like surgery in combination with ART. In our univariate analysis we could show that ART and number of histological LNM were independent predictors for the next relapse within the treated region (Table 3). The fact that the number of histologically confirmed LNM was not significantly different in the two groups (Table 1) suggests that patients with high lymphatic, unfavourable tumour-burden were equally distributed in both groups, corroborating the crucial role of ART.
The current analysis shows that ART has no significant influence on PSA response and PSA reduction (Fig. 3a, b) at the end of follow-up, respectively; this is in concordance with the findings of our group obtained by a previous analysis of a subset of both cohorts [17]. Furthermore, this confirms the observation that PSA reaches its limit as a valid marker in progressive advanced PCa [21]. Interestingly in our former analysis ART did not have a significant influence on overall clinical progression (detection of new metastases regardless of the location [17]). Probably due to the currently larger number of patients and due to the subclassification into next relapse within and outside the TR we have now been able to show that ART in the lymph node dissected region is able to provide tumour control.
Several groups investigated the efficacy of salvage irradiation of recurrent LNM [5, 11, 16, 28]. Picchio et al. [28] for example showed that 20 of 47 patients who received helical tomography with simultaneous integrated boost RT exhibited metabolic response confirmed by choline PET/CT. Because of inhomogeneity and different endpoints it is difficult to compare results from irradiation alone versus surgery only or combination of salvage-LND with ART.
Limitations
The number of analysed patients per treatment group was relatively low. Our analysis is based on a single centre experience and data acquisition including retrospective evaluation of two treatment strategies; only a treatment-arm with ART is missing. Inhomogeneity of the whole cohort concerning intermitted AHT and salvage RT prior to salvage-LND was present.
Conclusion
ART after salvage-LND provides stable local control in the TR and results in overall significantly improved next-relapse-free survival, compared to surgery only. Prerequisites of such interdisciplinary salvage therapy concepts are modern functional imaging techniques like PET/CT, standardized surgery approaches with thorough experience in salvage surgery and modern radiotherapy concepts including IGRT and IMRT. Prospective randomized trials are warranted to confirm these initial encouraging results in order to show that such interdisciplinary salvage therapy strategies really have the potential to prolong initiation of merely palliative AHT and offer the chance of cure to a subset of patients with nodal recurrent PCa.
Abbreviations
- ART:
-
Adjuvant radiotherapy
- AHT:
-
Antihormonal therapy
- CI:
-
Confidence interval
- CT:
-
Computed tomography
- CTV:
-
Clinical target volume
- IGRT:
-
Image-guided radiotherapy
- IMRT:
-
Intensity-modulated radiotherapy
- LN:
-
Lymph nodes
- LNM:
-
Lymph node metastases
- MRI:
-
Magnetic resonance imaging
- PET/CT:
-
Positron emission tomography/computed tomography
- PCa:
-
Prostate cancer
- PSA:
-
Prostate-specific antigen
- PTV:
-
Planning target volume
- Salvage-LND:
-
Salvage lymph node dissection
- SD:
-
Standard deviation
- Sup:
-
Supplement
- TR:
-
Treated region
References
Benson RC (1993) Total androgen blockade: the United States experience. Eur Urol 24:72–76
Bolla M, Van Poppel H, Tombal B et al (2012) Postoperative radiotherapy after radical prostatectomy for high-risk prostate cancer: long-term results of a randomised controlled trial (EORTC trial 22911). Lancet 380:2018–2027
Bottke D, Bartkowiak D, Schrader M et al (2012) Radiotherapy after radical prostatectomy: immediate or early delayed? Strahlenther Onkol 188:1096–1101
Briganti A, Joniau S, Gandaglia G et al (2013) Patterns and predictors of early biochemical recurrence after radical prostatectomy and adjuvant radiation therapy in men with pT3N0 prostate cancer: implications for multimodal therapies. Int J Radiat Oncol Biol Phys 87:960–967
Casamassima F, Masi L, Menichelli C et al (2011) Efficacy of eradicative radiotherapy for limited nodal metastases detected with choline PET scan in prostate cancer patients. Tumori 97:49–55
Coleman CN, Beard CJ, Kantoff PW et al (1994) Rate of relapse following treatment for localized prostate cancer: a critical analysis of retrospective reports. Int J Radiat Oncol Biol Phys 28:303–313
Ctcaev4.0 N (2009) National Cancer Institute common terminology criteria for adverse events v4.0 NCI, NIH, DHHS. NIH publication # 09-7473
Da Pozzo LF, Cozzarini C, Briganti A et al (2009) Long-term follow-up of patients with prostate cancer and nodal metastases treated by pelvic lymphadenectomy and radical prostatectomy: the positive impact of adjuvant radiotherapy. Eur Urol 55:1003–1011
Fiorino C, Alongi F, Perna L et al (2009) Dose-volume relationships for acute bowel toxicity in patients treated with pelvic nodal irradiation for prostate cancer. Int J Radiat Oncol Biol Phys 75:29–35
Guckenberger M, Lawrenz I, Flentje M (2014) Moderately hypofractionated radiotherapy for localized prostate cancer: long-term outcome using IMRT and volumetric IGRT. Strahlenther Onkol 190:48–53
Guerrero Urbano T, Khoo V, Staffurth J et al (2010) Intensity-modulated radiotherapy allows escalation of the radiation dose to the pelvic lymph nodes in patients with locally advanced prostate cancer: preliminary results of a phase I dose escalation study. Clin Oncol (R Coll Radiol) 22:236–244
Han M, Partin AW, Zahurak M (2003) Biochemical (prostate specific antigen) recurrence probability following radical prostatectomy for clinically localized prostate cancer. J Urol 169:517–523
Heidenreich A, Bastian PJ, Bellmunt J et al (2014) EAU guidelines on prostate cancer. Part 1: screening, diagnosis, and local treatment with curative intent-update 2013. Eur Urol 65:124–137
Heidenreich A, Bastian PJ, Bellmunt J et al (2014) EAU guidelines on prostate cancer. Part II: treatment of advanced, relapsing, and castration-resistant prostate cancer. Eur Urol 65:467–479
Higano CS (2003) Side effects of androgen deprivation therapy: monitoring and minimizing toxicity. Urology 61:32–38
Jereczek-Fossa BA, Beltramo G, Fariselli L et al (2012) Robotic image-guided stereotactic radiotherapy, for isolated recurrent primary, lymph node or metastatic prostate cancer. Int J Radiat Oncol Biol Phys 82:889–897
Jilg CA, Rischke HC, Schultze-Seemann W (2012) Salvage lymph node dissection with adjuvant radiotherapy for choline-PET-CT positive lymph nodes in patients with nodal recurrence of prostate cancer. J Urol 2012 Dec;188(6):2190–2197. doi:10.1016/j. juro.2012.08.041. (Epub 2012 Oct 18)
Jilg CA, Leifert A, Schnell D et al (2014) Toxicity and quality of life after choline-PET/CT directed salvage lymph node dissection and adjuvant radiotherapy in nodal recurrent prostate cancer. Radiat Oncol 9:178
Jilg CA, Schultze-Seemann W, Drendel V et al (2014) Detection of lymph node metastasis in patients with nodal prostate cancer relapse using F/C-choline positron emission tomography/computerized tomography: influence of size of nodal tumor infiltration and accuracy related to lymph node regions. J Urol 2014 Feb 8. pii: S0022-5347(14)00119-0. doi:10.1016/j.juro.2013.12.054. (Epub ahead of print)
Lawton CA, Michalski J, El-Naqa I et al (2009) RTOG GU radiation oncology specialists reach consensus on pelvic lymph node volumes for high-risk prostate cancer. Int J Radiat Oncol Biol Phys 74:383–387
Leibowitz-Amit R, Templeton AJ, Omlin A et al (2014) Clinical variables associated with PSA response to abiraterone acetate in patients with metastatic castration-resistant prostate cancer. Ann Oncol 25:657–662
Lohm G, Lutcke J, Jamil B et al (2014) Salvage radiotherapy in patients with prostate cancer and biochemical relapse after radical prostatectomy: long-term follow-up of a single-center survey. Strahlenther Onkol 190:727–731
Lu-Yao GL, Yao SL (1997) Population-based study of long-term survival in patients with clinically localised prostate cancer. Lancet 349:906–910
Martini T, Mayr R, Trenti E et al (2012) The role of C-choline-PET/CT-guided secondary lymphadenectomy in patients with PSA failure after radical prostatectomy: lessons learned from eight cases. Adv Urol 2012:601572
Millar J, Boyd R, Sutherland J (2008) An update of the phase III trial comparing whole pelvic to prostate only radiotherapy and neoadjuvant to adjuvant total androgen suppression: updated analysis of RTOG 94-13, with emphasis on unexpected hormone/radiation interactions: in regard to Lawton et al. (Int J Radiat Oncol Biol Phys 2007;69:646–655.). Int J Radiat Oncol Biol Phys 71:316 (author reply 316)
Pasquier D, Ballereau C (2008) Adjuvant and salvage radiotherapy after prostatectomy for prostate cancer: a literature review. Int J Radiat Oncol Biol Phys 72:972–979
Picchio M, Briganti A, Fanti S et al (2011) The role of choline positron emission tomography/computed tomography in the management of patients with prostate-specific antigen progression after radical treatment of prostate cancer. Eur Urol 59:51–60
Picchio M, Berardi G, Fodor A et al (2014) C-choline PET/CT as a guide to radiation treatment planning of lymph-node relapses in prostate cancer patients. Eur J Nucl Med Mol Imaging 41:1270–1279
Rigatti P, Suardi N, Briganti A (2011) Pelvic/retroperitoneal salvage lymph node dissection for patients treated with radical prostatectomy with biochemical recurrence and nodal recurrence detected by [11C]choline positron emission tomography/computed tomography. Eur Urol 60:935–943
Rinnab L, Mottaghy FM, Blumstein NM (2007) Evaluation of [11C]-choline positron-emission/computed tomography in patients with increasing prostate-specific antigen levels after primary treatment for prostate cancer. BJU Int 100:786–793
Rischke HC, Knippen S, Kirste S et al (2012) Treatment of recurrent prostate cancer following radical prostatectomy: the radiation-oncologists point of view. Q J Nucl Med Mol Imaging 56:409–420
Sanpaolo P, Barbieri V, Genovesi D (2014) Biologically effective dose and definitive radiation treatment for localized prostate cancer: treatment gaps do affect the risk of biochemical failure. Strahlenther Onkol 190:732–738
Schiller K, Petrucci A, Geinitz H et al (2014) Impact of different setup approaches in image-guided radiotherapy as primary treatment for prostate cancer: a study of 2940 setup deviations in 980 MVCTs. Strahlenther Onkol 190:722–726
Suardi N, Gandaglia G, Gallina A et al (2014) Long-term outcomes of salvage lymph node dissection for clinically recurrent prostate cancer: results of a single-institution series with a minimum follow-up of 5 years. Eur Urol 2014 Feb 18. pii: S0302-2838(14)00130-4. doi:10.1016/j.eururo.2014.02.011. (Epub ahead of print)
Thompson IM, Tangen CM, Paradelo J et al (2009) Adjuvant radiotherapy for pathological T3N0M0 prostate cancer significantly reduces risk of metastases and improves survival: long-term followup of a randomized clinical trial. J Urol 181:956–962
Wiegel T, Bartkowiak D, Bottke D et al (2014) Adjuvant radiotherapy versus wait-and-see after radical prostatectomy: 10-year follow-up of the ARO 96-02/AUO AP 09/95 trial. Eur Urol. 2014 Mar 21. pii: S0302-2838(14)00247-4. doi:10.1016/j.eururo.2014.03.011. (Epub ahead of print)
Compliance with ethical guidelines
Conflict of interest
H.C. Rischke, W. Schultze-Seemann, G. Wieser, M. Krönig, V. Drendel, P. Stegmaier, T. Krauss, K. Henne, N. Volegova-Neher, D. Schlager, S. Kirste, A.-L. Grosu, and C.A. Jilg state that there are no conflicts of interest.
All studies on humans described in the present manuscript were carried out with the approval of the responsible ethics committee and in accordance with national law and the Helsinki Declaration of 1975 (in its current, revised form). Informed consent was obtained from all patients included in studies.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Rischke, H., Schultze-Seemann, W., Wieser, G. et al. Adjuvant radiotherapy after salvage lymph node dissection because of nodal relapse of prostate cancer versus salvage lymph node dissection only. Strahlenther Onkol 191, 310–320 (2015). https://doi.org/10.1007/s00066-014-0763-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-014-0763-5
Keywords
- Prostate cancer relapse
- Salvage lymph node dissection
- Adjuvant radiotherapy
- Lymph node metastases
- Salvage radiotherapy