Abstract
Juvenile hormone (JH) has important functions that regulate insect life. In adult individuals, it induces gonadotropic and behavioral changes. Manipulating JH levels helps to understand how it influences insect physiology. The effects of JH on Epiponini swarm-founding wasps have shown contrasting results, affecting reproduction, chemical compound expression, behavior, and age polyethism. In this study, we investigated whether JH affects reproductive physiology and production of fertility cues in a swarm-founding wasp species Polybia occidentalis in an age-controlled experimental setup. We treated newly emerged females with methoprene (JH analogue) and precocene-I (JH inhibitor) to determine their effects on ovary activation and cuticular hydrocarbon (CHC) expression. Furthermore, we compared the chemical profiles of treated workers with those of queens. Our results show that methoprene and precocene-I affected the CHC production in P. occidentalis. Additionally, females treated with methoprene were chemically more similar to queens than precocene- and acetone-treated females. Methoprene affected ovarian status (increasing ovary activation). These results suggest that different levels of JH reflect changes in chemical and reproductive traits in P. occidentalis females. Furthermore, we point out the existence of fertility cues in this Epiponini wasp.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Juvenile hormone (JH) is a lipid hormone from the sesquiterpenoid class of terpenes, found in diverse natural products. It is synthesized by the Corpora allata, a pair of glands localized in the insect brains (Nijhout 1994; Hartfelder. 2000; Huang 2020). JH has important functions in the regulation of adult life in social Hymenoptera (Hartfelder 2000; Huang 2020). In social wasps, there is evidence that JH regulates age polyethism and acts as a gonadotropic agent, increasing ovarian activation and controlling cuticular hydrocarbon (CHCs) expression, which in turn acts as a fertility cue (O’Donnell and Jeanne 1993; Oi et al. 2015a; Oliveira et al. 2017; Walton et al. 2020).
Manipulating JH levels (using JH analogue methoprene and anti-JH precocene) helps to understand how JH influences insect physiology, especially given the diversification of associated functions across different insect lineages (Slamá 1971; Robinson 1992; Ramaseshadri et al. 2012; Kesltrup et al. 2014a, 2014b). Methoprene is a JH analogue that triggers similar effects as pure JH in the physiology and behavior of insects. On the other hand, precocene is a compound that acts as an anti-JH, destroying the Corpora allata and consequently inhibiting JH production (Bowers 1976; O'Donnell and Jeanne 1993; Robinson and Vargo 1997; Giray et al. 2005; Burns et al. 2007; Gotoh et al. 2008). Social insect workers treated with methoprene are known to upregulate the production of queen-like chemical profiles, and in contrast, precocene-treated workers express worker-like chemical profiles (Oliveira et al. 2017; Oi et al. 2021a,b). Some specific CHCs act as queen signals in different social insect species and the comparison of their chemical profiles reveal that specific CHCs are conserved across different social insect lineages (Van Oystaeyen et al. 2014; Kather and Martin 2015; Holman 2018). Indeed, the JH has the function of controlling some features of solitary ancestral species, such as reproduction and the production of fertility cues (Oi et al. 2015b). In social wasps, JH furthermore displays a gonadotropic role in females by increasing ovary activation and oviposition (Tibbetts and Sheehan 2012—Polistes metricus; Kelstrup et al., 2014b—Synoeca surinama; Oi et al., 2021a—Vespula vulgaris; Prato et al. 2021—Polybia occidentalis; Ferreira et al. 2022—Mischocyttarus cerberus and Mischocyttarus cassununga), and is associated with colony ontogeny and pheromone regulation (Oliveira et al. 2017—V. vulgaris; Walton et al. 2020—Polistes fuscatus; Oi et al. 2021b—Polistes dominula and Polistes satan; reviewed in Tibbetts et al. 2020; Ferreira et al. 2022—M. cerberus and M. cassununga). Additionally, JH is a pleiotropic hormone, meaning that the endocrine system regulates two or more features in the same individual (Dantzer and Swanson 2017; Oliveira et al. 2017; Oi et al. 2020; Oi et al. 2021a, b).
JH may not have a conserved role in Epiponini swarm-forming wasps. Previous studies demonstrated that JH regulates reproduction in S. surinama but not in Polybia micans (Kelstrup et al. 2014a, b). In P. occidentalis, it was reported that JH influences ovarian status, behavior, and chemical profile (O’Donnell and Jeanne 1993; Prato et al. 2021), and we confirmed that JH influenced age polyethism (Prato et al. 2021). Additionally, the morphological caste differentiation evolved several times in the Epiponini to different degrees (Noll and Wenzel 2008; Noll et al. 2020) and it is likely to be under JH influence. It is thus not clear how JH acts on Epiponini wasps. This poorly studied yet species-rich tribe presents a wide phylogenetic range of biological characteristics. For instance, morphological castes and ovarian activation in workers are present in some species (Protopolybia and Chartergellus, respectively) but not in others (Synoeca) (Noll et al. 2020). The nests of swarm-forming wasps are polygynic and, in some species, queens and workers express morphological and physiological differences (Noll et al. 2020). For instance, in P. occidentalis, workers organize themselves in an age-polyethism system, which means that newly emerged females perform tasks within the nest, while older workers perform high-cost tasks, such as foraging (Jeanne 1991; Jeanne et al. 1992; O’Donnell and Jeanne 1992). Newly emerged females exhibit ovary activation which decreases as they age (O’Donnell 2001). Another group of females, namely the intermediaries, show ovarian activation, but are not inseminated (Noll and Zucchi 2000). The number of queens varies depending on the size and stage of the nest, and workers often eliminate queens during nest development (Forsyth 1978). In summary, these biological characteristics provide an interesting opportunity for intra-genus comparative studies and to understand how the endocrine system can affect the morphological development type (post-imaginal and pre-imaginal) (Kelstrup et al. 2014a).
Here, we focus on the effects that JH triggers in Epiponini wasps: induction of ovarian activation, acceleration of age polyethism, and cuticular chemical production. We investigate whether JH is responsible for regulating ovary activation and the production of CHCs in P. occidentalis females kept under controlled conditions. This contrasts from our previous study (Prato et al. 2021), which was conducted in the field and focused on the influence of JH on the division of tasks, Studies have indeed demonstrated that experiments conducted under field and laboratory conditions affect differently the traits under investigation such as differences in physiology, behavior, gene expression, and development time (Calisi and Bentley 2009; Campbell et al. 2009; Jandt et al. 2015). We thus performed our experiments in the laboratory to control for the influence of age, interaction among workers, and food intake. Furthermore, we investigated whether JH is responsible for upregulating the production of CHCs that may be linked with ovary activation, and thus might represent key compounds to inform fertility status in the species. To do this, we compared the chemical profile of treated workers with those of fertile queens. We hypothesized that reproductive and chemical traits of P. occidentalis are directly affected by their circulating levels of JH. We predicted that females treated with methoprene would express higher levels of ovary activation and their cuticular surface would become more queen-like. Conversely, we predicted the opposite effects for females treated with precocene that is low levels of ovarian activation and chemical profile contrasting from queen-like.
Materials and methods
Study site and experiments
The experiments were conducted in the Laboratório de Comportamento e Ecologia de Insetos Sociais at the Universidade de São Paulo (USP), Ribeirão Preto Campus (21°09′50.7"S, 47°51′32.1"W) between August and October 2016. Two nests of P. occidentalis were collected in the field and kept under laboratory conditions to sample newly emerged females. The newly emerged females were treated topically on the abdomen with either a single dose of 1 µL of a solution at 5 µg/µl of methoprene (MT) Pestanal® (SUPELCO, analytical standard) (25 individuals) or a single dose of 1 µL of a solution at 5 µg/µl of precocene-I (PC) (SIGMA-ALDRICH) (25 individuals) diluted in acetone. The solvent group (25 individuals) was treated with acetone (AC) (2 µl of acetone). The choice of solution concentration was based on previous toxicity tests (Prato et al. 2021). The females from each group were paint-marked with non-toxic ink (Magic®) and kept in a plastic box with sucrose solution, mealworm larvae, and a piece of the nest for 10 days. We chose 10 days based on a previously published work, which reported that at a later age, almost all the females of P. occidentalis have filamentous, not activated ovaries (O’Donnell 2001). After 10 days, the treated females were euthanized in the temperature freezer for posterior extraction of CHCs, and dissection to check their ovarian activation. For comparison, six queens (Q) were collected from a third nest belonging to the same region to analyze the CHCs, ovarian status, and insemination.
CHCs analyses
Cuticular hydrocarbons were extracted in hexane solvent (Macron Fine Chemicals, 95% n-Hexane) by immersing the body for two minutes. The solvent from the extract was left to evaporate in a flow chamber for 24 h. The vial with the extract was resuspended in 50 μL of hexane and 2 μL of this solution was injected (Splitless mode) in a Gas Chromatography—Mass Spectrometer (GC–MS) (Shimadzu, model QP2010), using a DB-5MS column (length 30 m), with the injector temperature at 280 ºC and the helium carrier gas flow set at 1 mL/min. The oven temperature started at 150 ºC, rising by 7 ºC/min until 260 ºC, and was kept at this temperature for 5 min. After, the temperature was further increased to 310 ºC at a 5 ºC/min rate and held for 10 min. An external alkane standard solution C21–C40 (Fluka analytical) was used to identify the linear hydrocarbons. The methylated compounds were identified based on the mass spectrometric fragmentation patterns (ion and molecular mass) (Carlson et al. 1998).
Reproductive trait categorization
The ovaries of treated females were dissected in saline solution and categorized into two types, activated or non-activated (Noll and Zucchi 2000). The ovary of the queens were categorized as activated (mature oocytes ready for laying), shown in Fig. 1a. Treated females were categorized in the following way: activated ovary of workers (small cells and oocytes and few mature oocytes) (Fig. 1b), and non-activated ovary of workers (filamentous ovarioles) (Fig. 1c).
Statistical analyses
All statistical analyses were performed using R software (version 4.0.2, R core team 2018). The area of the peaks in the chromatograms were transformed in relative amounts. The CHCs from the different treatments were compared using the Permanova test (Bray–Curtis distance). For that, we used the adonis function from the vegan package (Oksanen et al. 2013). To visually represent our multivariate chemical dataset and check for cluster formation based on their CHCs according to different treatments, a principal component analysis (PCA) was performed with the prcomp function of the stats package (Team RC et al. 2013). To identify the compounds that most contributed to the separation of the groups, a discriminant analysis (SIMPER) was performed using 999 permutations. We used the simper function from the vegan package (Oksanen et al. 2013). Furthermore, the main compounds that were important in the group separations (results from SIMPER analysis) were divided into three different chemical classes (linear alkanes, methyl alkanes, and alkenes). The classes of compounds and the individual compounds per group were analyzed using a Mann–Whitney test.
The Generalized Linear Model was done to analyze the ovarian status of the workers between the treatments. The ovary was the dependent variable, and the treatment and origin nest as the explanatory variables. A post hoc Tukey test was used to compare the treatments. Those statistical analyses used the lme4, emmeans, effects, and HH packages (R Development Core Team 2008; Bates et al. 2020).
Results
CH differences
The cuticle of P. occidentalis expressed a chemical profile that included 65 compounds, which ranged from 22 to 40 carbons chain length (Table 1 supplementary material). The differences between treated workers and queens were both qualitative and quantitative. The CHCs’ profiles from workers treated with methoprene and precocene were significantly different when compared to the solvent group (Permanova: F93,3: 38.66; p < 0.01) (Fig. 2; Table 2 supplementary material). The most important compounds that contributed to the separation of the treatments were methyl alkanes (66.7%), linear alkanes (21%), alkenes (8.8%), and unknown compounds (3.5%). The main compounds responsible for groups’ separation varied mainly between the chain lengths from n-C25 to n-C39.
Principal components analysis (PCA) based on the relative abundances of the chemical compounds present in their cuticle of Polybia occidentalis females”. Red acetone, Blue precocene, Green methoprene, and Yellow queens. Highlighted compounds: 3MeC27, n-C27, n-C29, MeC29 = 15-;13-;11-;9-7MeC29, MeC31 = 15-;13-;11-;9MeC31 and MeC39 = 15-;13MeC39
The relative abundances of each chemical compound class varied among the treated workers. The proportion of linear alkanes differed among all the treatments (p < 0.01), except acetone and precocene (Mann–Whitney—W = 356; p = 0.40) (Fig. 3a; Table 1). In this case, workers from the methoprene-treated group showed the highest proportion of linear alkanes (Table 3 supplementary material). The linear alkanes n-C27, n-C28, n-C29, and n-C31 were higher and statistically different in the methoprene-treated group when compared with the precocene and acetone treatment groups (Fig. 3b; Table 2). The proportion of methylated alkanes varied among all the treatments (Fig. 3a; Table 1). Considering methylated compounds only, there were also specific methylated compounds that were higher and statistically different in workers from the methoprene-treated group when compared with the two others (e.g., 13-;11-;9MeC27; 3MeC27; 3.13-;3.11diMeC27 and 3.15-;3.13-;3.11diMeC29) (Fig. 4; Table 3). Workers from the precocene-treated group presented a higher proportion of methylated compounds than the other treatments (Table 3 supplementary material). Workers from the methoprene-treated group expressed the highest proportion of alkenes and this class of compound varied among all the treatments, but it was not different between workers from the acetone and precocene-treated groups (W = 223.5; p = 0.08) (Fig. 3a; Table 1 and Table 3 supplementary material). Interestingly, the alkene z-C29-1 occurred in a higher proportion in workers from the methoprene-treated group when compared with precocene and acetone groups. The alkene z-C29-2 was exclusively present in workers from the methoprene group. Finally, some compounds remained unidentified due to their low-quality mass spectra. These compounds were likely methylated hydrocarbons and were present in workers from the methoprene and acetone groups (W = 515.5; p < 0.01) and appeared in a higher proportion in the acetone group (Fig. 3a; Table 1 and Table 3 supplementary material).
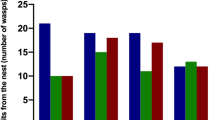
a Relative abundance of different compound classes (linear alkanes, methyl alkanes, and alkenes) found in Polybia occidentalis according to the group of treated workers (acetone, methoprene, and precocene) and queen. b Specific compounds of linear alkanes and methyl-alkane n-C27, 3MeC27, n-C28, n-C29, n-C31 found in Polybia occidentalis according to the group of treated workers (acetone, methoprene, and precocene) and queen. The compounds represented in “b” correspond to the ones that were previously demonstrated to act as queen pheromones in other social wasps (Van Oystaeyen et al. 2014; Oi et al. 2016). Different letters indicate statistical differences between groups of a given compound
Abundance of different methylated alkanes found in Polybia occidentalis workers according to the treatment groups. (1- 13-;11-;9MeC27; 2- 7MeC27; 3- 5MeC27; 4- 3MeC27; 5- 5.13diMeC27; 6- 3.13-;3.11diMeC27; 7- 3.7diMe27; 8- 4MeC28; 9- 4.14-;4.12diMeC28; 10- 9.13diMeC29; 11- 5.15-;5.13-;5.11diMeC29; 12- 3.15-;3.13-;3.11diMeC29) (The data are presented as mean and standard deviations). Different letters indicate statistical differences between treatments of a given compound
The chemical profile of queens was statistically different to the chemical profile of workers (Permanova: F98,4: 34.75; p < 0.01) (Fig. 2; Table 4 supplementary material). The classes of compounds varied among the treated workers and queens. The abundance of linear alkanes was significantly similar between workers treated with acetone and precocene, but they differed from methoprene and queens groups. These compounds were higher in abundance in workers treated with methoprene and queens, and they did not differ statistically from each other (W = 64; p = 0.59) (Fig. 3a; Table 4 and Table 5 supplementary material). The methylated alkanes appeared in a higher proportion in the workers treated with precocene, and this compound class varied between all the workers treated groups and queens (p ≤ 0.01), except in methoprene and queens (W = 56; p = 0.35) (Fig. 3a; Table 4 and Table 5 supplementary material). Finally, workers treated with methoprene were the ones who had the higher proportion of alkenes. We found in our samples CHCs that have already been highlighted in the literature as queen pheromones in other social wasps (n-C27, 3MeC27, n-C28, n-C29, n-C31) (Van Oystaeyen et al. 2014; Oi et al. 2016). We observed that these linear alkanes and 3-MeC27 appeared in higher proportions in workers treated with methoprene, which were statistically different when compared to other treated groups (Fig. 3b; Table 2 and 3, 4). Also, these compounds in workers treated with methoprene were statistically different in relation to the queen group, except the n-C29 (Fig. 3b; Table 5).
Reproductive traits
Workers belonging to different groups expressed different levels of ovary activation (Treatment—Anova Chisq = 22.42, p < 0.001***; Nest—Anova Chisq = 4.11, p = 0.042*). Workers treated with acetone expressed mostly non-activated ovaries (76% versus 24% of activated ovaries). The same trend was detected in workers treated with precocene, in which 88% of them expressed non-activated ovaries, while only 12% had activated ovaries. On the other hand, workers treated with methoprene exhibited a higher rate of ovary activation (65% of them), whereas only 35% had non-activated ovaries. The pairwise comparisons among the treatments were significant between workers treated with acetone and methoprene (estimate = 2.47, z ratio = 3.26, p = 0.001**) and workers treated with methoprene and precocene (estimate = − 2.98, z ratio = − 3.73, p = 0.0005***) (Fig. 5). However, workers treated with acetone and precocene did not differ from each other (estimate = − 0.51, z ratio = − 0.62, p = 0.529) (Fig. 5).
Discussion
Our results demonstrate that the JH analogue (methoprene) and the JH inhibitor (precocene) influenced the chemical profiles in treated workers of P. occidentalis. However, only the methoprene treatment influenced ovarian activation. These results are in line with previously published data for other social wasp species (Robinson and Vargo 1997; Oliveira et al. 2017; Oi et al. 2021a, b; Ferreira et al. 2022).
Workers of P. occidentalis treated with methoprene chemically resembled queens, but were consistently different to other two groups of workers. The most evident sign of chemical modulation in workers treated with methoprene is the increased proportion of some linear alkanes. In contrast, workers treated with precocene suffered a decrease in their proportion of linear alkanes and increased the level of methyl-alkane compounds when compared with the queen and other treated workers. The variation that we detected in the chemical profile of workers treated with methoprene and precocene probably occurred due to the treatments affecting their JH titers. Previous studies have shown that the topical administration of JH analogue is linked directly with the production of some long-chain hydrocarbons in social wasps (Oliveira et al. 2017; Oi et al. 2020, 2021b). Our results show an increase in this type of hydrocarbon. The linear alkanes (between C27 and C31) were the most important compounds responsible to separate workers treated with methoprene and queens from the acetone group. The four linear alkanes (n-C27, n-C28, n-C29, and n-C31) and one alkene (z-C29) in higher quantity may indicate putative fertility cue in P. occidentalis wasps and could potentially be candidates for queen pheromones in this species. Those compounds were previously reported for other wasps’ species (Sledge et al. 2004—P. dominulus; van Zweden et al. 2013—Dolichovespula saxonica; Van Oystaeyen et al. 2014—V. vulgaris; Oi et al. 2015a—V. vulgaris). Altogether, our results support the idea that queens and fertile females overproduce specific saturated hydrocarbons, which is shown in several lineages of Hymenoptera (Van Oystaeyen et al. 2014; Kather and Martin 2015). However, the difference in the chemical profiles encountered among the workers and queens can also be related to their different colonies of origin, since the CHCs have been reported to vary in individuals of the same species belonging to different nests (Dapporto et al. 2006; Tannure-Nascimento et al. 2007; Martin et al. 2008; Lorenzi and d’Ettorre 2020). On the other hand, queens and methoprene-treated workers had differences only in the alkenes class, which suggests that they are more chemically similar to each other. Nevertheless, our results need to be interpreted carefully as only two nests were used in the experiments, meaning that larger sample size may be more representative of the species.
Workers treated with methoprene increased ovarian activation when compared to precocene and acetone treatments; this result is not surprising as JH has been shown to have a gonadotropic effect in other social insects (Bloch et al. 2000; Shorter and Tibbetts 2009; Tibbetts et al. 2013; Kelstrup et al. 2014b; Amsalem et al. 2014a; Oliveira et al. 2017; Walton et al. 2020; Oi et al. 2020; Oi et al. 2021a, b; Prato et al. 2021; Ferreira et al. 2022). However, in the species A. mellifera and P. micans, JH does not influence ovarian activation but does control the chemical signaling and age polyethism (Fluri et al. 1981; Robinson 1992; Hartfelder 2000; Kelstrup et al. 2014a). It is possible that the JH acts as a pleiotropic hormone in P. occidentalis affecting multiple traits, such as behavioral maturation, fertility, and chemical signaling. However, we cannot discard the hypothesis that in our study, the CHCs changed as a by-product of ovary activation that was caused by hormonal treatments.
In summary, our results provide experimental evidence that the reproduction and the CHCs’ production are under JH hormonal control in P. occidentalis. These changes in CHCs may act not only in the chemical communication among workers linked to age polyethism (Prato et al. 2021) but also in the communication between queens and workers. This study helps to unveil functions associated with JH in the evolution of the Polybia lineage. The presence of some queen-characteristic linear alkanes, that were also detected to be upregulated by reproductive workers (treated with methoprene) indicate the existence of fertility cues in this species. Future studies conducting bioassays with the queen-characteristic compounds are necessary to test their role in the division of reproductive labor in colonies of P. occidentalis.
References
Amsalem E, Malka O, Grozinger C, Hefetz A (2014) Exploring the role of juvenile hormone and vitellogenin in reproduction and social behavior in bumble bees. BMC Evol Biol 14:45. https://doi.org/10.1186/1471-2148-14-45
Bates D, Maechler M, Bolker B, Walker S, Christensen RHB, Singmann H, Dai B, Scheipl F, Grothendieck G, Green P, Fox J, Bauer A, Krivitsky PN (2020) lme4: Linear Mixed-Effects Models using 'Eigen' and S4. https://cran.r-project.org/web/packages/lme4/index.html (accessed 01 December 2020).
Bloch G, Borst DW, Huang ZY, Robinson GE, Cnaani J, Hefetz A (2000) Juvenile hormone titers, juvenile hormone biosynthesis, ovarian development and social environment in Bombus terrestris. J Insect Physiol 46:47–57. https://doi.org/10.1016/S0022-1910(99)00101-8
Bowers WS, Tomihisa O, Cleere JS, Marsella PA (1976) Discovery of insect anti- juvenile hormones in plants: plants yield a potential fourth generation insecticide. Science 193:542–547. https://doi.org/10.1126/science.986685
Burns SN, Meer RKV, Teal PEA (2007) Mating flight activity as dealation factors for red imported fire ant (Hymenoptera: Formicidae) female alates. Ann Entomol Soc Am 100:257–264. https://doi.org/10.1603/0013-8746(2007)100[257:MFAADF]2.0.CO;2
Calisi RM, Bentley GE (2009) Lab and field experiments: are they the same animal? Horm Behav 56(1):1–10. https://doi.org/10.1016/j.yhbeh.2009.02.010 (Epub 2009 Mar 9 PMID: 19281813)
Campbell DL, Weiner SA, Starks PT, Hauber ME (2009) Context and control: behavioural ecology experiments in the laboratory. Ann Zool Fenn 46:112–123. https://doi.org/10.5735/086.046.0204
Carlson DA, Bernier UR, Sutton BD (1998) Elution patterns from capillary GC for methyl-branched alkanes. J Chem Ecol 24:1845–1865. https://doi.org/10.1023/A:1022311701355
Cuvillier-Hot V, Cobb M, Malosse C, Peeters C (2001) Sex, age and ovarian activity affect cuticular hydrocarbons in Diacamma ceylonense, a queenless ant. J Insect Physiol 47(4–5):485–493. https://doi.org/10.1016/S0022-1910(00)00137-2
da Silva RC, Prato A, Oi CA, Turatti ICC, Do Nascimento FS (2020) Dominance hierarchy, ovarian activity and cuticular hydrocarbons in the primitively eusocial wasp Mischocyttarus cerberus (Vespidae, Polistinae, Mischocyttarini). J Chem Ecol 46(9):835–844. https://doi.org/10.1007/s10886-020-01206-1
Dantzer B, Swanson EM (2017) Does hormonal pleiotropy shape the evolution of performance and life history traits? Integr Comp Biol 57(2):372–384. https://doi.org/10.1093/icb/icx064
Dapporto L, Fondelli L, Turillazzi S (2006) Nestmate recognition and identification of cuticular hydrocarbons composition in the swarm founding paper wasp Ropalidia opifex. Biochem Syst Ecol 34(8):617–625. https://doi.org/10.1016/j.bse.2006.04.002
Ferreira HM, da Silva RC, do Nascimento, FS, Wenseleers, T, Oi, CA, (2022) Reproduction and fertility signalling under joint juvenile hormone control in primitively eusocial Mischocyttarus wasps. Chemoecology. https://doi.org/10.1007/s00049-022-00370-y
Fluri P, Sabatini AG, Vecchi MA, Wille H (1981) Blood juvenile hormone, protein and vitellogenin titres in laying and non-laying queen honeybees. J Api Res 20:221–225. https://doi.org/10.1080/00218839.1981.11100500
Forsyth A (1978) Studies on the behavioral ecology of polygynous social wasps. Ph.D. Thesis, Harvard University, Cambridge
Giray T, Giovanetti M, West-Eberhard MJ (2005) Juvenile hormone, reproduction, and worker behavior in the Neotropical social wasp Polistes canadensis. Proc Natl Acad Sci USA 102:3330–3335. https://doi.org/10.1073/pnas.0409560102
Gotoh H, Cornette R, Koshikawa S, Miura T (2008) Effects of precocenes on the corpora allata and the JH titer in the damp-wood termite Hodotermopsis sjostedti (Isoptera: Termopsidae). Sociobiology 52:345
Hartfelder K (2000) Insect juvenile hormone: from “status quo” to high society. Braz J Med Biol Res 33:157–177. https://doi.org/10.1590/S0100-879X2000000200003
Holman L (2018) Queen pheromones and reproductive division of labor: a meta-analysis. Behav Ecol 29(6):1199–1209. https://doi.org/10.1093/beheco/ary023
Huang ZY (2020) Juvenile Hormone. In: Starr CK (ed) Encyclopedia of Social Insects. Springer International Publishing, Cham, pp 1–3
Jandt JM, Thomson JL, Geffre AC, Toth AL (2015) Lab rearing environment perturbs social traits: a case study with Polistes wasps. Behav Ecol 26(5):1274–1284. https://doi.org/10.1093/beheco/arv082
Jeanne RL (1991) Polyethism. In: Ross KG, Matthews RW (eds) The Social Biology of Wasps. Cornell University Press, Ithaca, NY, pp 389–425
Jeanne RL, Williams NM, Yandell BS (1992) Age polyethism and defense in a tropical social wasp (Hymenoptera: Vespidae). J Lnsect Behav 5(2):211–227. https://doi.org/10.1007/BF01049290
Kather R, Martin SJ (2015) Evolution of cuticular hydrocarbons in the hymenoptera: a meta-analysis. J Chem Ecol 41(10):871–883. https://doi.org/10.1007/s10886-015-0631-5
Kelstrup HC, Hartfelder K, Nascimento FS, Riddiford LM (2014a) Reproductive status, endocrine physiology and chemical signaling in the Neotropical, swarm-founding eusocial wasp Polybia micans. J Exp Biol 217:2399–2410. https://doi.org/10.1242/jeb.096750
Kelstrup HC, Hartfelder K, Nascimento FS, Riddiford LM (2014b) The role of juvenile hormone in dominance behavior, reproduction and cuticular pheromone signaling in the caste-flexible epiponine wasp synoeca surinama. Frontiers in Zool 11:78. https://doi.org/10.1186/s12983-014-0078-5
Kelstrup HC, Hartfelder K, Esterhuizen N, Wossler TC (2017) Juvenile hormone titers, ovarian status and epicuticular hydrocarbons in gynes and workers of the paper wasp Belonogaster longitarsus. J Insect Physiol 98:83–92. https://doi.org/10.1016/j.jinsphys.2016.11.014
Lorenzi MC, d’Ettorre P (2020) Nestmate recognition in social insects: what does it mean to be chemically insignificant? Front Ecol Evol 7:488. https://doi.org/10.3389/fevo.2019.00488
Martin SJ, Helanterä H, Drijfhout FP (2008) Colony-specific hydrocarbons identify nest mates in two species of Formica ant. J Chem Ecol 34(8):1072–1080. https://doi.org/10.1007/s10886-008-9482-7
Nijhout H (1994) Insect Hormones. Princeton University Press
Noll FB, Wenzel JW (2008) Caste in the swarming wasps: ‘queenless’ societies in highly social insects. Biol J Linn Soc Lond 93:509–522. https://doi.org/10.1111/j.1095-8312.2007.00899.x
Noll FB, Zucchi R (2000) Increasing caste differences related to life cycle progression in some Neotropical swarm-founding polygynic polistine wasps (Hymenoptera Vespidae Epiponini). Ethol Ecol Evol 12:43–65. https://doi.org/10.1080/03949370.2000.9728322
Noll FB, da Silva M, Soleman RA, Lopes RB, Grandinete YC, Almeida EA, Carpenter WJW, JM, (2020) Marimbondos: systematics, biogeography, and evolution of social behaviour of neotropical swarm-founding wasps (Hymenoptera: Vespidae: Epiponini). Cladistics 37(4):423–441. https://doi.org/10.1111/cla.12446
O’Donnell S, Jeanne RL (1992) Life-long patterns of forager behaviour in a tropicalswarm-founding wasp: Effects of specialization and activity level on longevity swarm-founding wasp: Effects of specialization and activity level on longevity. Anim Behav. https://doi.org/10.1016/S0003-3472(05)80314-8
O’Donnell S, Jeanne RL (1993) Methoprene accelerates age polyetism in workers of a social wasp (Polybia occidentalis). Physiol Entomo 18:189–194. https://doi.org/10.1111/j.1365-3032.1993.tb00467.x
O’Donnell S (2001) Worker age, ovary development, and temporal polyethism in the swarm-founding wasp Polybia occidentalis (Hymenoptera: Vespidae). J Insect Behav 14:201–213. https://doi.org/10.1023/A:1007837727984
Oi CA, Van Oystaeyen A, Oliveira RC, Millar JG, Verstrepen KJ, van Zweden JS, Wenseleers T (2015a) Dual effect of wasp queen pheromone in regulating insect sociality. Curr Biol 25:1638–1640. https://doi.org/10.1016/j.cub.2015.04.040
Oi CA, van Zweden JS, Oliveira RC, Van Oystaeyen A, Nascimento FS, Wenseleers T (2015b) The origin and evolution of social insect queen pheromones: Novel hypotheses and outstanding problems. BioEssays. https://doi.org/10.1002/bies.201400180
Oi CA, Millar JG, van Zweden JS, Wenseleers T (2016) Conservation of queen pheromones across two species of vespine wasps. J Chem Ecol 42(11):1175–1180. https://doi.org/10.1007/s10886-016-0777-9
Oi CA, Brown RL, da Silva RC, Wenseleers T (2020) Reproduction and signals regulating worker policing under identical hormonal control in social wasps. Sci Rep 10:18971. https://doi.org/10.1038/s41598-020-76084-4
Oi CA, Ferreira HM, da Silva RC, Bienstman A, Nascimento FSD, Wenseleers T (2021a) Effects of juvenile hormone in fertility and fertility-signaling in workers of the common wasp Vespula vulgaris. PLoS ONE 16:e0250720. https://doi.org/10.1371/journal.pone.0250720
Oi CA, da Silva RC, Stevens I, Ferreira HM, Nascimento FS, Wenseleers T (2021b) Hormonal modulation of reproduction and fertility signaling in Polistine wasps. Curr Zool. https://doi.org/10.1093/cz/zoab026/617119
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin PR, O’hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Oksanen MJ (2013) Package ‘vegan.’ Community Ecology Package, Ver-Sion 2:1–295
Oliveira RC, Vollet-Neto A, Oi CA, Van Zweden JS, Nascimento F, Brent CS, Wenseleers T (2017) Hormonal pleiotropy helps maintain queen signal honesty in a highly eusocial wasp. Sci Rep 7:1654. https://doi.org/10.1038/s41598-017-01794-1
Prato A, da Silva RC, Assis DS, Mateus S, Hartfelder K, Nascimento FSD (2021) Juvenile hormone affects age polyethism, ovarian status, and cuticular hydrocarbon profile in workers of a Polybia occidentalis wasp. J Exp Biol. https://doi.org/10.1242/jeb.240200
R Development Core Team. (2008) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. http://www.Rproject.org (accessed 18 January 2017).
Ramaseshadri P, Farkaš R, Palli SR (2012) Recent progress in juvenile hormone analogs (JHA) research, in: Dhadialla, T.S. (Ed.), Adv Insect Physiol. 43, pp. 353–436. Academic Press.
Robinson GE (1992) Regulation of division of labor in insect societies. Annu Rev Entomol 37:637–665. https://doi.org/10.1146/annurev.en.37
Robinson GE, Vargo EL (1997) Juvenile hormone in adult eusocial Hymenoptera: gonadotropin and behavioral pacemaker. Arch Insect Biochem Physiol 35:559–583. https://doi.org/10.1002/(SICI)1520-6327(1997)35:4%3c559::AID-ARCH13%3e3.0.CO;2-9
Shorter JR, Tibbets EA (2009) The effect of juvenile hormone on temporal polyethism in the paper wasp Polistes dominulus. Insectes Soc 56:7–13. https://doi.org/10.1007/s00040-008-1026-1
Slama K (1971) Insect juvenile hormone analogues. Annu Rev Biochem 40:1079–1102
Sledge MF, Trinca I, Massolo A, Boscaro F, Turillazzi S (2004) Variation in cuticular hydrocarbon signatures, hormonal correlates and establishment of reproductive dominance in a polistine wasp. J Insect Physiol 50:73–83. https://doi.org/10.1016/j.jinsphys.2003.10.001
Tannure-Nascimento IC, Nascimento FS, Turatti IC, Lopes NP, Trigo JR, Zucchi R (2007) Colony membership is reflected by variations in cuticular hydrocarbon profile in a Neotropical paper wasp, Polistes satan (Hymenoptera, Vespidae). Genet Mol Res 6(2):390–396
Team RC, Team MRC, Suggests MASS, Matrix S (2013) Package “Stats” RA Lang Environment Stat Comput. R Foundation for Statistical Computing, Vienna
Tibbetts EA, Sheehan MJ (2012) The effect of juvenile hormone on Polistes wasp fertility varies with cooperative behavior. Horm Behav 61:559–564. https://doi.org/10.1016/j.yhbeh.2012.02.002
Tibbetts EA, Vernier C, Jinn J (2013) Juvenile hormone influences precontest assessment behaviour in Polistes dominulus paper wasps. Anim Behav 85:1177–1181. https://doi.org/10.1016/j.anbehav.2013.03.003
Tibbetts EA, Laub EC, Mathiron AG, Goubault M (2020) The challenge hypothesis in insects. Horm Behav 123:104533. https://doi.org/10.1016/j.yhbeh.2019.05.002
Van Oystaeyen A, Oliveira RC, Holman L, van Zweden JS, Romero C, Oi CA, d’Ettorre P, Khalesi M, Billen J, Wäckers F, Millar JG, Wenseleers T (2014) Conserved class of queen pheromones stops social insect workers from reproducing. Science 343:287–290. https://doi.org/10.1126/science.1244899
Van Zweden JS, Bonckaert W, Wenseleers T, d’Ettorre P (2013) Queen signaling in social wasps. Evolution 68(4):976–986. https://doi.org/10.1111/evo.12314
Walton A, Tumulty JP, Toth AL, Sheehan MJ (2020) Hormonal modulation of reproduction in Polistes fuscatus social wasps: Dual functions in both ovary development and sexual receptivity. J Insect Physiol 120:103972. https://doi.org/10.1016/j.jinsphys.2019.103972
Acknowledgements
We would like to thank the anonymous reviewers, Dr Sidnei Mateus for helping to collect the nests, Dr Ayrton Vollet Neto for helping with the hormone dilutions, and Iona Cunningham-Eurich and Dr Emeline Favreau for proofing the manuscript. This study was funded by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq—Process: 142285/2018-8, 405082/2018-5, and 307702/2018-9), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001 (RCS) and Fundação de Amparo à Pesquisa do Estado de São Paulo (Fapesp) under Grant APS [proc. nº 2016/11887-4], RCS [proc. nº 2018/22461-3], and FSN [proc. nº 2018/10996-0]. Funding was provided from Research Foundation Flanders to CAO (postdoctoral fellowship FWO-12V6318N, GOF8319N, and research grant FWO-1513219N).
Funding
Conselho Nacional de Desenvolvimento Científico e Tecnológico, 142285/2018-8, Amanda Prato, 307702/2018-9, Fabio Santos do Nascimento, 405082/2018-5, Fabio Santos do Nascimento, Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, 01, Rafael Carvalho da Silva, Fundação de Amparo à Pesquisa do Estado de São Paulo, 2016/11887-4, Amanda Prato, 2018/10996-0, Fabio Santos do Nascimento, 2018/22461-3, Rafael Carvalho da Silva, Research Foundation Flanders to CAO, FWO-12V6318N, Cintia Akemi Oi, GOF8319N, Cintia Akemi Oi, FWO-1513219 N, Cintia Akemi Oi.
Author information
Authors and Affiliations
Contributions
Conceptualization [Amanda Prato and Fabio S. Nascimento]; methodology [Amanda Prato and Fabio S. Nascimento]; formal analysis and investigation [Amanda Prato, Rafael Carvalho da Silva, Cintia Akemi Oi, and Izabel Cristina C. Turatti]; writing—original draft preparation [Amanda Prato and Fabio S. Nascimento]; writing—review and editing [Amanda Prato, Rafael Carvalho da Silva, Cintia Akemi Oi, and Fabio S. Nascimento].
Corresponding author
Ethics declarations
Conflict of interest
No competing interests declared.
Additional information
Communicated by Thomas Schmitt.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Prato, A., da Silva, R.C., Oi, C.A. et al. Juvenile hormone regulates reproductive physiology and the production of fertility cues in the swarm-founding wasp Polybia occidentalis. Chemoecology 32, 171–180 (2022). https://doi.org/10.1007/s00049-022-00376-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00049-022-00376-6