Abstract
Although specific binding of 20-hydroxyecdysone (20E) and its analogs (ecdysteroids) to the ecdysone receptor ligand-binding domain (EcR-LBD) in insects has been well documented, information on the EcR-ligand binding in Helicoverpa armigera is limited. Hence, an attempt has been made to screen effective natural plant-based agonists from a library of 25 non-azadirachtin neem limonoids and was compared with the commercially available insecticide, tebufenozide, through in silico approach. Results indicated that six compounds, namely nimbolide, azadirone, nimolinone, meliacinol, nimbocinol, azadiradione, efficiently docked with the active site of H. armigera EcR-LBD. The binding energies of top-ranked six molecules ranged from −10.54 to −12.22 kcal/mol, which was superior to the third-generation insect growth regulator (IGR), tebufenozide RH5992. Two factors are especially important in binding: (1) the residues Cys 508 and Asn 504, which are the most common in hydrogen-bonding interactions and (2) hydrophobic pocket residues—Asn 504, Met 507, Val 416, Tyr 408 and Thr 343. We also recognized one aromatic ring, 3–7 vicinal acceptors and 1–3 distal hydrophobic groups as minimum pharmacophoric feature. A significant correlation coefficient of 0.6823 was observed supporting positively the docking studies. These data could help in the application of natural compounds as alternatives to chemicals in pest management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Helicoverpa armigera is one of the world’s most devastating pest species feeding on more than 100 major agricultural crops (Sharma, 2001) and is found in Asia, Europe, Africa and Australia causing an estimated damage between US $ 2 and 5 billion annually (Lammers and MacLeod, 2007; Tay et al., 2013). H. armigera has developed resistance to synthetic insecticides (Gunning and Easton, 1994; Srinivas et al., 2004) due to its biological characteristics such as polyphagy, high mobility and fecundity, facultative diapause and high population build-up (Fitt, 1989; Yang et al., 2013).
The ecdysone receptor complex is the key element, which enacts the ecdysteroid-induced physiological and morphological changes during insect moulting regulated by ecdysteroid hormones like 20-hydroxyecdysone (20E) and its analogs that bind to the ligand-binding domain of the ecdysone receptor (Jayachandran et al., 2013). The ecdysone receptor (EcR) belongs to the nuclear hormone receptor superfamily that functions as a ligand-activated transcription factor. The basic structure of EcR consists of five modular domains referred to as A/B (transcriptional activation domain), C (DNA-binding domain; DBD), D (hinge region), E (ligand-binding domain; LBD) and F (not well-defined region) (Thummel, 1995). It is a heterodimer of two proteins, EcR-LBD (which contains the active site for ecdysteroids) and the ultraspiracle protein (USP), which is a homologue of the retinoid X receptor (RXR; Oro et al., 1990).
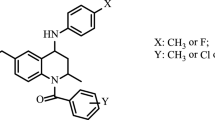
The moulting process is initiated by a number of transcription factors in the nuclear receptor superfamily. This results in the up-regulation of several late genes in the hormone pathway and help in mediating the moulting process (Zheng et al., 2010). Since these receptors are limited to invertebrates, they have been exploited as an attractive target for insecticide development (Graham, 2002; Palli et al., 2005). The ecdysteroid agonists, such as tebufenozide (RH-5992), mimic the natural function of the endogenous insect moulting hormone 20-hydroxyecdysone (20E), inducing premature lethal moulting in larval stages and aborting reproduction in adults, especially in Lepidoptera and Coleoptera (Nagata et al., 2005).
Azadirachta indica A. Juss (Meliaceae), commonly known as “neem”, yields more than 300 bioactive chemical compounds such as terpenoids, limonoids, flavonoids, amino acids and carbohydrates (Alland et al., 2005). Limonoids from the neem tree have attracted considerable research attention in recent years owing to their wide range of bioactivities such as insect anti-feedant, anti-microbial, anti-cancer, anti-malarial, anti-inflammatory, antioxidant, anti-proliferative effects, cytotoxic and growth regulatory properties (Murugan et al., 1998; Nanduri et al., 2003; Setzer and Setzer, 2003; Kumar et al., 2008). Apart from azadirachtin, several non-azadirachtin limonoids (NAL) inhibit feeding in some specific insect pests, yet have not been given enough importance (Koul et al., 1996). These studies have provided impetus for screening other neem compounds in detail to identify potential phytochemicals that could be used in commercial formulations (Koul et al., 2003). Bioinsecticides being less hazardous to the environment and human health can serve as the best alternative for pest management (Murray and Isman, 2006).
The present study is focused on the screening of potent insect growth regulators by choosing twenty-five non-azadirachtin limonoids to test its insecticidal activity through molecular docking studies in H. armigera ecdysone receptor (HaEcR).
Materials and methods
Retrieval of HaEcR protein structure and preparation for docking
The 3D coordinates of the crystal structure of EcR bound to bisacylhydrazine compound BYI-06830 (PDB code: 3IXP) at a resolution of 2.85 Å were retrieved from protein data bank (PDB; Berman et al., 2007). Hetero atoms, ligands and water molecules were removed from the protein structure of EcR. Preparation of the target protein with AutoDock tools (ADT) involved the addition of polar hydrogens to the macromolecule, an essential step to correct the calculation of partial charge. Finally, Gasteiger charges were calculated for each atom of the macromolecule (Gasteiger et al., 1990). The charged protein was converted to the “PDBQT” format and read through AutoGrid.
Ligand retrieval and preparation for docking
Twenty-five limonoid compounds from A. indica possessing insecticidal activity were selected for the study on the basis of literature survey (Table 1; Fig. 1). 2D structures of these compounds were downloaded in SDF format from the PubChem database (https://pubchem.ncbi.nlm.nih.gov/; Bolton et al., 2008). These 2D structures were converted to 3D structures with the help of Open Babel, using PyRx software (http://pyrx.sourceforge.net/) and were energy minimized by mmff94 force field with an energy gradient of 0.05 (Halgren, 1996; Wolf, 2009).
Setting grid map parameters
Grid map was created with a 3D lattice of regularly spaced points, surrounding (entirely or partially) and centred on the active site on chain D of HaEcR-LBD (3IXP). AutoGrid programme was used to generate the grid maps. The grid dimensions were 46 Å × 48 Å × 64 Å with points separated by 0.375 Å with grid centre 5.838 × 65.407 × 12.076 to encompass entire active site.
Running AutoDock
Molecular docking was performed in AutoDock 4.2 (Morris et al., 1998; Huey et al., 2007) using a Genetic Algorithm–the Least Square (GA–LS), which performs a semi-flexible docking keeping the protein itself as rigid while the ligand as flexible. The parameters were set to 10 runs, population size to 150, maximum number of energy evaluations to 25 million and 27,000 generations, mutation rate of 0.02, crossover rate of 0.80 and the rest of the parameters were set to default values. The docking results were saved as “.dlg” file, and all the docked conformation outputs were viewed using inbuilt visualization features of AutoDock. For each ligand, the best docked conformation with the least energy and high stability was selected and the complex file was exported. The analysis and visualization of the binding interaction of protein and ligands were performed using Discovery Studio Visualizer V4.0 (Accelrys Software Inc.). The docking results were clustered on the basis of root mean square deviation (RMSD) and ranked on the basis of free energy of binding.
Validation of docking
Redocking of co-crystallized ligand was performed, and the bound ligand, BYI-06830 (bisacylhydrazine compound), found in the crystal structure was extracted and docked into the corresponding binding pocket. The ability of the ligand to reproduce the orientation and the position of the ligand in the bound form were determined.
Prediction of insecticide potency (Tice rule)
Bioavailability and bioactivity of compounds are considered as important parameters for development of potential insecticides. The insecticide-likeness of the potential compounds was predicted using Tice rule. According to Tice rule, insecticidal compounds should have: (a) molecular weight ≤500 g/mol, (b) number of hydrogen-bond donors ≤3, (c) number of hydrogen-bond acceptors ≤12, (d) partition coefficient (log P) ≤5 and (e) no. of rotatable bonds ≤12 (Tice, 2001). The molecular properties of investigated compounds were computed by using online Molinspiration cheminformatics software (www.molinspiration.com).
Correlation between experimental IC50 and docking results
The experimental inhibitory concentration (IC50 in µg/ml) values of gedunin (50.8), salanin (74.5), nimbolide (1,000), nimbocinol (250.8), azadiradione (249.3) and tebufenozide (20) were collected from the available literature (Rochanakij et al., 1985; Carlson, 2000; Koul et al., 2003, 2004) and were correlated with the docking score. The data were prepared as mean ± standard error mean (SEM), and linear regression analysis was performed considering experimental IC50 (µg/ml) values and binding energy (kcal/mol) as independent and dependent variables, respectively, by using SPSS ver.20 software. P < 0.0001 was considered to be significant.
Results and discussion
HaEcR-ligand-binding domain
From the several crystal structures determined, it was observed that the DNA-binding domain (DBD) and LBD are highly conserved in insect EcRs (Iwema et al., 2009). DBD usually involves in hormone response element (HRE) recognition (Thomson et al., 2009) and the LBD in receptor dimerization, ligand recognition and cofactor interactions (Nakagawa and Henrich, 2009). Apart from these, the LBD also contains the ligand-binding pocket (LBP), which binds ecdysteroids as well as certain non-steroidal EcR agonists such as the DAH-based insecticides (Tohidi-Esfahani et al., 2011). It is now known that EcR and USP form a heterodimer, which functions as the natural ecdysone receptor. Although USP is an essential partner for the high affinity binding of ecdysteroids, the ligand-binding pocket resides in the EcR protein and the presence of ligand class enhances heterodimer stability for transcription (Graham et al., 2007a, 2007b). HaEcR has very high identity to other lepidopteran EcRs (Spodoptera exigua 90 %, Plodia interpunctella 83 % and Plutella xylostella 80 %), but less with Aedes albopictus and Drosophila pseudoobscura (72 % identity; Jayachandran et al., 2013). The bisacylhydrazine insecticidal compound, tebufenozide (RH5992), binds in the Lepidoptera EcR cavity that overlaps the pocket occupied by bound ecdysteroids (Tohidi-Esfahani et al., 2011). The structures of the nuclear receptors constantly provided valuable information about ligand recognition and the activation mechanism of nuclear receptors. Studies building homology models based on a comparison of the EcR-LBD with known crystal structures have been employed to determine the three-dimensional (3D) structure of the EcR-LBD (Wurtz et al., 2000), and docking studies have also been carried out to simulate how a candidate ligand binds to a receptor (Kasuya et al., 2003).
Docking of non-azadirachtin limonoids (NAL)
Limonoids of Meliaceae have complex structure with a very high degree of oxidation and rearrangement (Roy and Saraf, 2006). In silico docking, experiment was performed to evaluate the binding potential of non-azadirachtin limonoids from A. indica to the HaEcR-LBD. We selected 25 limonoid compounds and a non-steroidal ecdysone agonist (tebufenozide), as a reference (Table 1; Fig. 1). AutoDock run resulted in the energy scores between −2.34 and −12.22 kcal/mol. Out of 25 chosen non-azadirachtin limonoids, nimbolide (−12.22 kcal/mol), azadirone (−11.31 kcal/mol), nimolinone (−11.29 kcal/mol), meliacinol (−11.08 kcal/mol), nimbocinol (–10.73 kcal/mol) and azadiradione (−10.54 kcal/mol) were able to dock (higher binding affinity) better than the reference ligand (Tebufenozide- −10.46 kcal/mol) in the active site (Table 1). The best docked ligand molecules were selected based on the binding energy and good interaction with the active site’s residues. Lesser the inhibitory constant (Ki), higher the binding affinity of limonoids. Nimbolide (1.1 nM), azadirone (5.16 nM), nimolinone (5.33 nM), meliacinol (7.54 nM), nimbocinol (13.74 nM) and azadiradione (18.85 nM) came out to be the most promising hits with K in nanomolar range. All the top scorers along with their binding energies and no. of hydrogen bonds are listed in Table 1. The top score NAL ligand values include steric and H-bonding intermolecular function, stronger receptor ligand binding, lipophilic interactions, polar attractive/repulsive interactions, solvation of the protein and ligand, entropy term for the ligand and binding free energy. The order of ligands based on docking score is nimbolide > azadirone > nimolinone > meliacinol > nimbocinol > azadiradione > tebufenozide. Azadirone and azadiradione shared one hydrogen bond with Cys 508, while meliacinol and nimbocinol moiety with Asn 504 (Table 1; Fig. 3).
Two-dimensional plot of docked poses and binding interactions of Helicoverpa armigera ecdysone receptor ligand-binding pocket (HaEcR-LBD) to the non-steroidal ecdysone agonist, tebufenozide (a) and non-azadirachtin limonoid compounds nimbolide (b), azadirone (c), nimolinone (d), meliacinol (e), nimbocinol (f) and azadiradione (g)
The docking results showed that ligands bind to the hydrophobic core of the EcR-LBD, which consist of hydrophobic amino acids (Cys, Leu, Val, Ile, Phe and Met), lipophobic amino acids (Thr and Tyr) and hydrophilic amino acids (Lys, Gly, Thr and Asp). Three polar amino acid residues (Tyr 408, Asn 504 and Cys 508) are implicated in the hydrogen-bond interaction network with the ligands. In the six top-scored NAL compounds, nine amino acids (Leu, Trp, Thr, Met, Asn, Gln, Tyr, Ser and Val) make up the binding cavity including the reference ecdysteroid agonist, tebufenozide, indicating the significant roles of these residues in binding. The six top-most scorers comprised five amino acids (Phe, Ile, Gln, Asp and Cys) in the binding pocket of the HaEcR-LBD (Fig. 3b–g), which were not present in HaEcR-LBD–tebufenozide complex (Fig. 3a).
The electrostatic bond and van der Waals interactions between the binding pocket of HaEcR-LBD and the ligands are presented in Table 1 and Fig. 3. Nimbolide seemed to be the most potent hit having −12.22 kcal/mol as binding energy. This compound was bound tightly to the binding site by electrostatic bonds (Leu 420, Tyr 403, Met 380/507, Val 416, Gln 503, Thr 343, Asn 504, Cys 508) and van der Waals interactions (Asp 419, Met 413, Tyr 408, Leu 500/511, Phe 336, Ile 339, Trp 526). One side of the binding site was completely hydrophobic with residues like Met 380, Leu 420 and Val 416 while lipophobic residues are concentrated in the same site including Gln 503 and Tyr 403/408 (Fig. 3b). Azadirone interacts with lipophobic (Thr 343 and Tyr 408) and hydrophobic (Cys 508, Leu 511/518/522, Ile 339, Met 507/381 and Phe 336) residues (Fig. 3c). Nimolinone moiety occupied a hydrophobic pocket of Cys 508, Leu 500/522, Val 416, Phe 408 and Met 507/381/413 with only one hydrogen bond between Cys 508 with cyclic carbonyl oxygen, while the remaining part is buried inside a region rich with amino acids having low hydropathy index (Asn 504 and Thr 340; Fig. 3d). The moiety of meliacinol contains hydrophobic, lipophobic, hydrophilic residues along with low hydropathy index (Fig. 3e). In all top six structures, one aromatic ring, 3–7 vicinal acceptors and 1–3 distal hydrophobic groups were observed, which could be the minimum pharmacophoric feature. Superimposed graphical representation of the six best docked compounds with EcR-LBD active site is shown in Fig. 4.
Superimposed structure of docked poses of the six non-azadirachtin limonoid compounds along with ecdysone agonist, tebufenozide, at binding pocket of Helicoverpa armigera ecdysone receptor (HaEcR-LBD) with colour codes a tebufenozide in red (reference), b nimbolide in green, c azadirone in yellow, d nimolinone in light blue, e meliacinol in purple, f nimbocinol in pink and g azadiradione in deep blue (Color figure online)
Criteria for avoidance of local minima and false-positives
Graham et al. (2007a, b) have experimented on the recombinant EcR-LBD and found that tebufenozide, a commercially available insecticide, has differential binding affinity across the taxonomic orders and reflects the selective toxicity against lepidopteran pests. They have also been demonstrated to have no/low toxicity towards non-lepidopteran species as well as pollinators, predators and parasites (Dhadialla et al., 1998; Retnakaran et al., 2003). In order to avoid inaccuracy in the scoring function of docking study, non-steroidal ecdysteroid agonist—tebufenozide was docked against the protein HaEcR-LBD as a reference and the results were used as a benchmark. The rank of each non-azadirachtin limonoid compound was determined by the binding free energy of the lowest energy cluster. In all the cases, densely populated cluster coincided well with the lowest energy cluster resulted 2–3 clusters with a single conformation. The clusters possessed 99 % conformation with −8.56 kcal/mol average binding energy. The estimated free energy of binding should not be used as a sole criterion for the selection of ligand ranking. To avoid irrelevant local minima and minimize the false-positives, the following criteria’s were followed: (1) The non-azadirachtin limonoid ligand was bound inside the pocket in the HaEcR-LBD receptor; (2) the non-polar/polar atoms in the ligand docked were near the non-polar/polar atoms of the receptor; and (3) hydrogen bonding and hydrophobic interactions.
Validation of docking study and confirmation of bound ligand, BYI-06830
Docking procedures aim to identify correct poses of ligands in the binding pocket of a protein and to predict the affinity between the ligand and the protein. In other words, docking describes a process by which two molecules fit together in three-dimensional space. To check the validity of docking study, redocking with bound ligand (BYI-06830) to the EcR protein was performed. The RMSD of the result was determined to be 1.29 Å, which suggests the reliability of the molecular docking procedure (Fig. 2 ).
Tice rule and potency of ecdysteroid agonists
No violation of Tice rule was observed in nimbolide, nimolinone, nimbocinol and azadiradione, whereas azadirone and meliacinol violated the properties of LogP and molecular weight, respectively (Table 2). Nimbolide, nimolinone, nimbocinol and azadiradione are proved to be potent ecdysteroid agonists against H. armigera (Praveena and Sanjayan, 2011). Based on Tice rule, it can be presumed that nimbolide may be a potent inhibitor of HaEcR-LBD receptor through its ecdysteroid agonist activity. Further, in-depth laboratory and field studies are needed to support this claim.
Regression analysis between experimental IC50 and binding energy
A linear regression analysis was performed to examine whether the docking score of non-azadirachtin limonoids (gedunin, salanin, nimbolide, nimbocinol, azadiradione and tebufenozide) can be correlated with the experimental IC50 values of H. armigera. Docking score was predicted as gedunin (−10.44), salanin (−9.25), nimbolide (−12.22), nimbocinol (−10.73), azadiradione (−10.54) and tebufenozide (−10.43). The predicted binding energies (kcal/mol-AutoDock) were plotted against available experimental IC50 values from the literature (Rochanakij et al., 1985; Carlson, 2000; Koul et al., 2003, 2004). The experimental IC50 values of NAL showed a linear correlation (r = 0.8260) with the calculated binding energy of the chosen limonoids (y = 0.0023x − 4,170.5; F 1,22 = 47.239; P < 0.0001). The coefficient of determination (r 2 = 0.6823) shown a good fit with the statistical model, which is an acceptable value (SD = 0.5134) in such docking practice. These results suggest that AutoDock has performed well in predicting the binding energies and also rationalized the mechanism by which these ecdysteroid agonists work (Fig. 5).
Many studies had been focused on determining the distribution, nature, and practical use of plant-derived ecdysteroid substances that have moulting inhibitory activity against H. armigera (Cohen et al., 1996; Dhadialla et al., 1998; Murugan et al., 1998; Retnakaran et al., 2003). Various NAL compounds existing in neem plant either jointly or independently contribute to behavioural efficacy (e.g., repellence and feeding deterrence) and physiological efficacy, and/or as acute toxicity and developmental disruption against H. armigera (Murray and Isman, 2006; Lammers and Macleod, 2007; Jaychandrana et al., 2013). Koul et al., 2003 have further shown that azadirachtin being the most active compound in neem is not synergized or influenced by any other limonoid, but other non-azadirachtin limonoids show synergism in specific combinations, which may be due to their different modes of action. In this study, the chemical interaction between the selected non-azadirachtin ligands (nimbolide, azadirone, nimolinone, meliacinol, nimbocinol and azadiradione) and the target protein (HaEcR-LBD) has been found to be good and has the best interaction scores. Similar to the identified ligands of phytochemical origin, it indicated that these limonoids are safer to the environment. Since permanent activation of EcRs by certain compounds has been reported (Wing et al., 1988; Dhadialla et al., 1998; Nakagawa, 2005) for inhibiting metamorphosis process, we hypothesize that these non-azadirachtin neem limonoid compounds, possessing a greater binding affinity than tebufenozide could also serve as an agonist against EcR, can be used for the development of potent insecticides.
Conclusion
In conclusion, the present study reports the insecticidal effect of six plant-based non-azadirachtin limonoids on HaEcR-LBD. Docking experiment suggested that these compounds showed higher interaction energies compared to the DBH-based insecticide, tebufenozide, where Cys 508 and Asn 504 involve in hydrogen-bonding interactions. Tice rule of insecticide likeliness also indicated that lead compounds can be potent candidates as ecdysteroid agonists. Linear regression analysis in terms of predicted binding energy and its experimental activities strongly suggests that these could be applied successfully in pest management programs against H. armigera.
References
Alland C, Moreews F, Boens D, Carpentier M, Chiusa S, Lonquety M, Renault N, Wong Y, Cantalloube H, Chomilier J, Hochez J, Pothier J, Villoutreix BO, Zagury JF, Tufféry P (2005) RPBS: a web resource for structural bioinformatics. Nucleic Acids Res 33:44–49
Berman HM, Henrick K, Nakamura H, Markley J, Bourne PE, Westbrook J (2007) Realism about PDB. Nat Biotechnol 25:845–846
Bolton EE, Wang Y, Thiessen PA, Bryant SH (2008) Integrated platform of small molecules and biological activities. Annu Rep Comput Chem 4:217–241
Carlson GR (2000) Tebufenozide: a novel caterpillar control agent with unusually high target selectivity. Green chemical syntheses and processes. In: ACS symposium series. pp 8–17. doi:10.1021/bk-2000-0767.ch002
Cohen E, Quistad GB, Casida JE (1996) Cytotoxicity of nimbolide, epoxyazadiradione and other limonoids from neem insecticide. Life Sci 58:1075–1081
Dhadialla TS, Carlson GR, Le DP (1998) New insecticides with ecdysteroidal and juvenile hormone activity. Annu Rev Entomol 43:545–569
Fitt GP (1989) The ecology of Heliothis in relation to agroecosystems. Annu Rev Entomol 34:17–53
Gasteiger J, Rudolph C, Sadowski J (1990) Automatic generation of 3D-atomic coordinates for organic molecules. Tetrahedron Comput Methodol 3:537–547
Graham LD (2002) Ecdysone-controlled expression of transgenes. Expert Opin Biol Ther 2:525–535
Graham LD, Johnson WM, Pawlak-Skrzecz A, Eaton RE, Bliese M, Howell L, Hannan GN, Hill RJ (2007a) Ligand binding by recombinant domains from insect ecdysone receptors. Insect Biochem Mol Biol 37:611–626
Graham LD, Pilling PA, Eaton RE, Gorman JJ, Braybrook C, Hannan GN, Pawlak-Skrzecz A, Noyce L, Lovrecz GO, Lu L, Hill RJ (2007b) Purification and characterization of recombinant ligand-binding domains from the ecdysone receptors of four pest insects. Protein Expr Purif 53:309–324
Gunning RV, Easton CS (1994) Endosulphon resistance in Helicoverpa armigera (Hübner) (Lepidoptera: Noctuidae) in Australia. J Aust Entomol Soc 33:9–12
Halgren TA (1996) Merck molecular force field. V. Extension of MMFF94 using experimental data, additional computational data and empirical rules. J Comput Chem 17:616–641
Huey R, Morris GM, Olson AJ, Goodsell DS (2007) A semiempirical free energy force field with charge-based desolvation. J Comput Chem 28:1145–1152
Isman MB (2006) Botanical insecticides, deterrents, and repellents in modern agriculture and an increasingly regulated world. Annu Rev Entomol 51:45–66
Iwema T, Chaumot A, Studer RA, Robinson-Rechavi M, Billas IM, Moras D, Laudet V, Bonneton F (2009) Structural and evolutionary innovation of the heterodimerization interface between USP and the ecdysone receptor EcR in insects. Mol Biol Evol 26:753–768
Jayachandran B, Hussain M, Asgari S (2013) Regulation of Helicoverpa armigera ecdysone receptor by miR-14 and its potential link to baculovirus infection. J Invertebr Pathol 114:151–157
Kasuya A, Sawada Y, Tsukamoto Y, Tanaka K, Toya T, Yanagi M (2003) Binding mode of ecdysone agonists to the receptor: comparative modeling and docking studies. J Mol Model 9:58–65
Koul O, Shankar JS, Kapil RS (1996) The effect of neem allelochemicals on nutritional physiology of larval Spodoptera litura. Entomol Exp Appl 79:43–50
Koul O, Multani JS, Daniewski WM, Singh G, Berlozecki S (2003) 6β-Hydroxygedunin from Azadirachta indica its potentiation effects with some non-azadirachtin limonoids in neem against lepidopteran larvae. J Agric Food Chem 51:2937–2942
Koul O, Multani JS, Goomber S, Daniewski WM, Berlozecki S (2004) Activity of some non-azadirachtin limonoids from against Azadirachta indica lepidopteran larvae. Aust J Entomol 43:78–84
Kumar NS, Murugan K, Zhang W (2008) Additive interaction of Helicoverpa armigera Nucleopolyhedro virus and Azadirachtin. Biocontrol 53:869–880
Lammers JW, Macleod A (2007) Report of a pest risk analysis: Helicoverpa armigera (Hübner, 1808) http://www.fera.defra.gov.uk/plants/plantHealth/pestsDiseases/documents/helicoverpa.pdf. Accessed 10 Aug 2014
Morris GM, Goodsell DS, Halliday RS, Huey R, Hart WE, Belew RK, Olson AJ (1998) Automated docking using a Lamarckian genetic algorithm and an empirical binding free energy function. J Comput Chem 19:1639–1662
Murugan K, Jeyabalan D, Kumar NS, Babu R, Sivaramakrishnan S, Nathan SS (1998) Antifeedant and growth-inhibitory properties of neem limonoids against the cotton bollworm, Helicoverpa armigera (Hübner). Int J Trop Insect Sci 18:157–162
Nagata S, Maruyama T, Ohira T, Nagasawa H (2005) Cloning and characterization of ecdysone receptor and ultraspiracle cDNAs from Spodoptera litura. Ann N Y Acad Sci 1040:417–419
Nakagawa Y (2005) Nonsteroidal ecdysone agonists. Vitam Horm 73:131–173
Nakagawa Y, Henrich VC (2009) Arthropods nuclear receptors and their role in moulting. FEBS J 276:6128–6157
Nanduri S, Thunuguntla SS, Nyavanandi VK, Kasu S, Kumar PM, Ram PS, Rajagopal S, Kumar RA, Devi DS, Rajagopalan R, Venkateswarlu A (2003) Biological investigation and structure-activity relationship studies on azadirone from Azadirachta indica A Juss. Bioorg Med Chem Lett 13:4111–4115
Oro AE, McKeown M, Evans RM (1990) Relationship between the product of the Drosophila ultraspiracle locus and the vertebrate retinoid X receptor. Nature 347:298–301
Palli SR, Hormann RE, Schlattner U, Lezzi M (2005) Ecdysteroid receptors and their applications in agriculture and medicine. Vitam Horm 73:59–100
Praveena A, Sanjayan KP (2011) Inhibition of acetylcholinesterase in three insects of economic importance by linalool, a monoterpene phytochemical. In: Dunston PA (ed) Insect pest management, a current scenario, entomology research unit. St. Xavier’s College, Palayamkottai, pp 340–345
Retnakaran A, Krell P, Feng Q, Arif B (2003) Ecdysone agonists: mechanism and importance in controlling insect pests of agriculture and forestry. Arch Insect Biochem Physiol 54:187–199
Rochanakij S, Thebtaranonth Y, Yenjai C, Yuthavong Y (1985) Nimbolide, a constituent of Azadirachta indica, inhibits Plosmodium falciparum in culture. Southeast Asian J Trop Med Public Health 16:66–72
Roy A, Saraf S (2006) Limonoids: overview of significant bioactive triterpenes distributed in plants kingdom. Biol Pharm Bull 29:191–201
Setzer WN, Setzer MC (2003) Plant-derived triterpenoids as potential antineoplastic agents. Mini Rev Med Chem 3:540–556
Sharma HC (2001) Cotton bollworm/legume pod borer, Helicoverpa armigera (Hubner) (Noctuidae: Lepidoptera): biology and management. Crop Protection Compendium, CABI Oxon
Srinivas R, Udikeri SS, Jayalakshmi SK, Sreeramulu K (2004) Identification of factors responsible for insecticide resistance in Helicoverpa armigera. Comp Biochem Physiol C Toxicol Pharmacol 137:261–269
Tay WT, Soria MF, Walsh T, Thomazoni D, Silvie P, Behere GT, Anderson C, Downes S (2013) A brave new world for an old world pest: Helicoverpa armigera (Lepidoptera: Noctuidae) in Brazil. PLoS One. doi:10.1371/journal.pone.0080134
Thomson SA, Baldwin WS, Wangl YH, Kwon G, LeBlanc GA (2009) Annotation, phylogenetics, and expression of the nuclear receptors in Daphnia pulex. BMC Genomics 10:1–14
Thummel CS (1995) From embryogenesis to metamorphosis: the regulation and function of Drosophila nuclear receptor superfamily members. Cell 83:871–877
Tice CM (2001) Selecting the right compounds for screening: does Lipinski’s Rule of 5 for pharmaceuticals apply to agrochemicals? Pest Manag Sci 57:3–16
Tohidi-Esfahani D, Lawrence MC, Graham LD, Hannan GN, Simpson AM, Hill RJ (2011) Isoforms of the heteropteran Nezara viridula ecdysone receptor: protein characterisation, RH5992 insecticide binding and homology modelling. Pest Manag Sci 67:1457–1467
Wing KD, Slawecki RA, Carlson GR (1988) RH-5849, nonsteroidal ecdysone agonist: effects on larval Lepidoptera. Science 241:470–472
Wolf LK (2009) Digital briefs: new software and websites for the chemical enterprise. C&EN 87:31
Wurtz JM, Guillot B, Fagart J, Moras D, Tietjen K, Schindler M (2000) A new model for 20-hydroxyecdysone and dibenzoylhydrazine binding: a homology modeling and docking approach. Protein Sci 9:1073–1084
Yang Y, Li Y, Wu Y (2013) Current status of insecticide resistance in Helicoverpa armigera after 15 years of Bt cotton planting in China. J Econ Entomol 106:375–381
Zheng WW, Yang DT, Wang JX, Song QS, Gilbert LI, Zhao XF (2010) Hsc70 binds to ultraspiracle resulting in the upregulation of 20-hydroxyecdsone-responsive genes in Helicoverpa armigera. Mol Cell Endocrinol 315:282–291
Acknowledgments
We are thankful to the Department of Biotechnology, New Delhi, Govt. of India for their financial assistance in the form of Research Fellowship to RPY and KSI and for the Bioinformatics Infrastructure Facility (No. BT/BI/12/060/2012 (NERBIF-MUA).
Conflict of interest
The authors declare that there is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yadav, R.P., Syed Ibrahim, K., Gurusubramanian, G. et al. In silico docking studies of non-azadirachtin limonoids against ecdysone receptor of Helicoverpa armigera (Hubner) (Lepidoptera: Noctuidae). Med Chem Res 24, 2621–2631 (2015). https://doi.org/10.1007/s00044-015-1320-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-015-1320-1