Abstract
Objective
Nordalbergin is a coumarin extracted from Dalbergia sissoo DC. To date, the biological effects of nordalbergin have not been well investigated. To investigate the anti-inflammatory responses and the anti-oxidant abilities of nordalbergin using lipopolysaccharide (LPS)-activated macrophages and LPS-induced sepsis mouse model.
Materials and methods
Production of nitrite oxide (NO), prostaglandin E2 (PGE2), pro-inflammatory cytokines (tumor necrosis factor (TNF)-α, interleukin (IL)-6 and IL-1β), reactive oxygen species (ROS), tissue damage and serum inflammatory markers, and the activation of the NLRP3 inflammasome were examined.
Results
Our results indicated that nordalbergin reduced the production of NO and pro-inflammatory cytokines in vitro and ex vivo. Nordalbergin also suppressed iNOS and cyclooxygenase-2 expressions, decreased NF-κB activity, and attenuated MAPKs signaling pathway activation by decreasing JNK and p38 phosphorylation by LPS-activated J774A.1 macrophages. Notably, nordalbergin diminished NLRP3 inflammasome activation via repressing the maturation of IL-1β and caspase-1 and suppressing ROS production by LPS/ATP- and LPS/nigericin-activated J774A.1 macrophages. Furthermore, nordalbergin exhibited protective effects against the infiltration of inflammatory cells and also inhibited the levels of organ damage markers (AST, ALT, BUN) by LPS-challenged mice.
Conclusion
Nordalbergin possesses anti-inflammatory effects in macrophage-mediated innate immune responses, alleviates ROS production, decreases NLRP3 activation, and exhibits protective effects against LPS-induced tissue damage in mice.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sepsis is a life-threatening, multi-organ dysfunction with diverse manifestations [1], which is marked by tissue infiltration of inflammatory cells in the last stage and increases persistently in varying amounts during the processes [2]. There were approximately 48.9 million reported cases of sepsis globally, and 11 million of these patients died to the condition, representing 19.7% of all deaths worldwide that year [3]. Macrophages play an essential role in all phases of sepsis with their ubiquitous presence and extensive effects on immune homeostasis and the inflammatory process [4]. Nevertheless, overreactive inflammation is associated with dysregulated host response leading to systemic effects, with sepsis being one of the most lethal outcomes.
Lipopolysaccharides (LPS), the outer membrane of Gram-negative bacteria, are the most common microbial mediator in sepsis [5] through recognition by Toll-like receptor 4 (TLR4) on host innate immune cells and can trigger its downstream signals to activate mitogen-activated protein kinase (MAPK) and transcription factor NF-κB [6], leading to the production of pro-inflammatory mediators (e.g. nitric oxide (NO) and prostaglandin E2 (PGE2)) and cytokines (e.g. tumor necrosis factor-α (TNF-α) and interleukin (IL)-6) [7, 8]. Dysregulation of MAPK and NF-κB signaling pathways can lead to the excessive and damaging inflammation characteristic of sepsis [9], whereas inhibition of MAPK and NF-κB signaling pathways represents a promising therapeutic strategy for sepsis by reducing the excessive inflammatory response [10].
NLRP3 inflammasome contains a signaling sensor (NLRP3), a protein adaptor (an apoptosis-associated speck-like protein with a caspase recruitment domain, ASC), and an effector protein (pro-caspase-1) [11]. Activation of the NLRP3 inflammasome is a two-signal process: the first signal (priming) triggers NLRP3 and pro-IL-1β expression via the NF-κB signaling pathway; while the second signal (activation) completes the full activation and formation of the NLRP3 inflammasome [12]. The NLR family pyrin domain containing 3 (NLRP3)/caspase-1/IL-1 axis has emerged as an important signaling pathway of the innate immune system and has been demonstrated to be associated with the development of sepsis [13, 14]. Deletion of NLRP3 in mice has been demonstrated to increase survival of polymicrobial sepsis [15, 16]. Moreover, sepsis induces reactive oxygen species (ROS) production that not only causes oxidative damage to tissues and organelles but also promotes NLRP3 inflammasome activation [14, 17]. Therefore, inhibition of ROS and NLRP3 inflammasome activation has been considered as a strategic approach to managing sepsis by addressing key drivers of the inflammatory response [17].
Dalbergia sissoo DC., commonly known as Indian rosewood or Sheesham, is a traditional medicinal plant with a long history of use for its analgesic, anti-nociceptive, anti-inflammatory, and immuno-modulatory properties [18]. Oral administration of the extract of Dalbergia sissoo leaves in rats possessed significant anti-inflammatory activity without any side effect on gastric mucosa [19]. Nordalbergin, a coumarin extracted from the bark of Dalbergia sissoo DC., has been demonstrated to stimulate the differentiation of HL-60 cells, which are human promyelocytic leukemia cells [19]. Our latest research has revealed that nordalbergin possesses anti-neuroinflammatory properties, effectively reducing the inflammatory reactions in microglia [20]. Nevertheless, the anti-inflammatory activity of nordalbergin in endotoxemia has not been investigated. Hence, the purpose of this research was to investigate the anti-inflammatory capabilities of nordalbergin on macrophages stimulated by LPS, using both in vitro and ex vivo approaches, along with an LPS-induced endotoxemia model in mice.
Materials and methods
Ethical statement
All procedures involving animal care and handling were conducted in compliance with the ethical guidelines set by the Committee on the Ethics of Animal Experiments at Kaohsiung Medical University, under the permit number 110,193.
Cell culture
J774A.1 murine macrophages and L-929 murine fibroblast cells were obtained from the Bioresource Collection and Research Center (Hsinchu, Taiwan). These cells were grown in Dulbecco’s Modified Eagle’s Medium (DMEM) for the macrophages and Roswell Park Memorial Institute (RPMI)-1640 medium for the fibroblasts. Both cell culture media were supplemented with 10% (v/v) heat-inactivated fetal bovine serum, along with 100 U/mL of penicillin and 100 U/mL of streptomycin (Life Technologies, USA). The cells were maintained in a 5% CO2 humidified environment at 37 °C and subcultured every 2 to 3 days.
NO and cell viability assays
NO production and cell viability were detected by the Griess reagent assay and the 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay respectively, following our previous study [7]. Cells were plated in 96-well plates (1 × 105 cells/well) overnight and then pretreated with different concentrations of nordalbergin (5, 10, 15, and 20 µM) for 1 h, followed by LPS treatment (1 µg/mL, from E. coli. O111:B4, Sigma Aldrich, USA) for 24 h. The treatment time and doses of nordalbergin were referred to previous study [20]. The supernatant of the cell culture was then collected and analyzed for the levels of nitrites using the Griess reagent assay following the manufacturer’s protocol (Sigma Aldrich, USA). The detection of nitrite is then determined as a colored azo-dye product of the Griess reaction that absorbs visible light at 540 nm measuring by a microplate spectrometer. For the cell viability, MTT reagent (Sigma-Aldrich, USA) was added to the cells at a final concentration of 0.5 mg/mL and incubated at 37 °C for 4 h. Viable cells with active metabolism converted MTT into purple formazan crystals, and the formazan crystals were then solubilized using 0.04 M HCl in isopropanol, and absorbance was measured at 570 nm using a microplate spectrometer.
Determination of cytokines production
An enzyme-linked immunosorbent assay (ELISA) was conducted to measure the production of PGE2, IL-6, TNF-α, and IL-1β. In brief, cells were seeded in 96-well plates at a density of 1 × 105 cells/well and incubated overnight. Subsequently, the cells were pretreated with different concentrations of nordalbergin (5, 10, 15, and 20 µM) for 1 h, followed by LPS treatment for 24 h. cell culture supernatant was harvested, and the levels of PGE2, IL-6 and TNF-α were examined by ELISA according to the manufacturer’s protocols (Thermo Scientific, USA). For the measurement of IL-1β, the major pro-inflammatory cytokine secreted by NLRP3 inflammaome activation, cells were pretreated with various concentrations of nordalbergin (5, 10, 15, and 20 µM) for 1 h, treated with 1 µg/mL LPS for 5 h, and then treated with either 5 mM ATP (Cayman Chemical, USA) or 10 µM nigericin (Cayman Chemical, USA) for 30 min. The treatment time and doses of nordalbergin were referred to previous study [20]. The levels of IL-1β in the cell culture supernatant were measured using ELISA kits according to the manufacturer’s protocols (Thermo Scientific, USA).
Protein precipitation from cell supernatants
Protein precipitation from the cell supernatant was performed by the methanol-chloroform precipitation method following a previous study [21]. Briefly, cell supernatants were collected and the protein was precipitated by methanol and chloroform, followed by drying the samples at 55℃ for 5–10 min. Subsequently, the transparent protein pellets were reconstituted with 1x SDS. In the last step, equal volumes (15 µL protein/lane) of the resuspended proteins were analyzed using western blotting.
Western blot analysis
Cells were lysed by radioimmunoprecipitation assay (RIPA) buffer containing protease and phosphatase inhibitor cocktails (Sigma Aldrich, USA) and then quantified by BCA assay kit (Thermo Scientific, USA). Briefly, equal amounts of protein were separated using sodium dodecyl-sulfate polyacrylamide gel electrophoresis and then transferred to polyvinylidene fluoride membranes (EMD Millipore, USA). Subsequently, the membranes were probed overnight with primary antibodies at 4 °C and then incubated with horseradish peroxidase-conjugated secondary antibodies for 1 h at room temperature. Protein bands were visualized using chemiluminescent detection, carried out with a Bio-Rad ChemiDoc XRS+ system, and quantified using Image Lab software version 5.1 (Bio-Rad Laboratories, Inc., USA).
NF-κB promoter reporter assay
J-blue cells, an NF-κB promoter reporter cell line, was derived from J774A.1 cells as described previously [7]. Briefly, J-blue cells were cultured in 96-well plates (1 × 105 cells/mL) overnight, followed by adding different concentrations of nordalbergin (5,10,15, and 20 µM) for 1 h and then primed with LPS for 18 h. The supernatant from the cell culture was collected, and the activity of secreted embryonic alkaline phosphatase (SEAP) present in the medium was measured using the QUANTI-Blue medium (InvivoGen, USA). The optical density at 655 nm was then evaluated and analyzed using an ELISA reader.
ROS detection
The detection of intracellular ROS was analyzed by CM-H2DCFDA staining (Invitrogen, USA) following previous study [22]. Briefly, a total of 5 × 105 cells were seeded in 6-well plates and cultured overnight. Subsequently, the cells were pretreated with different concentrations of nordalbergin (10 and 20 µM) for 1 h and then treated with 1 µg/mL LPS for 24 h. This was followed by a treatment with ATP (5 mM) or nigericin (10 µM) for 30 min. The cells were then washed with phosphate-buffered saline (PBS), collected in round-bottom tubes, centrifuged at 1,400 x g for 5 min, and the supernatant was discarded. The cells were stained with 5 µM CM-H2DCFDA at 37 °C for 20 min, washed twice with PBS, and subsequently, ROS production was quantified using a Cytomics FC500 flow cytometer (Beckman Coulter Inc., USA).
Immunofluorescence staining
The co-localization of ASC and caspase-1 was analyzed by immunofluorescence staining according to our previous study [7]. Primary antibodies against caspase-1 (sc-56,036, Santa Cruz, USA) and ASC (sc-22,514-R, Santa Cruz, USA) were used for staining. To visualize the cell nuclei, DAPI staining (Invitrogen, USA) was employed, and the stained cells were observed under a confocal laser microscope (Leica, USA). Subsequently, the fluorescence intensity in the captured images was quantified using Imaris 9.6 Image Analysis Software (Oxford Instruments, UK).
Isolation of peritoneal exudate macrophages (PEM) and bone marrow-derived macrophages (BMDM)
Female C57BL/6 mice, aged between 6 and 8 weeks, were acquired from the National Lab Animal Center (Taipei, Taiwan). The methods of PEM and BMDM isolation were followed by previous study [23]. Briefly, mice were intraperitoneally injected with 1 mL sterile 3% thioglycollate medium (Sigma Aldrich, USA) for 4 days. Cells in the peritoneal cavity were harvested using sterile ice-cold PBS and then cultured in RPMI-1640 medium at a concentration of 1 × 106 cells/mL for 4 h. Subsequently, non-adherent cells were washed away by PBS, and the adherent peritoneal cells were used as PEM. For the BMDM isolation, bone marrow cells were extracted from the femur and tibia of each mouse, cultured in RPMI medium (containing 10% FBS, 1% penicillin/streptomycin), and added 15% L929 conditioned medium to induce macrophage differentiation for 7 days. Afterward, the adherent cells were used as BMDM [23].
Histopathology and biochemistry examination
C57BL/6 mice were treated with nordalbergin (5 or 10 mg/kg body weight) or an equivalent volume of the vehicle (dimethyl sulfoxide, DMSO) for 1 h, followed by an intraperitoneal injection of LPS (50 mg/kg body weight) for 5 h. The mice were euthanized, and the tissues of the lung and liver were subjected to hematoxylin and eosin (H&E) staining. In addition, blood samples were drawn to measure the levels of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and blood urea nitrogen (BUN), utilizing the Fuji Dri-Chem 3500i Biochemistry Analyzer (Fujifilm Ltd, Japan).
Statistical analysis
The experimental results are presented as mean ± standard deviation (SD). All data shown were performed from three independent experiments and each was conducted in triplicate. Statistical evaluations were performed using one-way ANOVA and subsequent Tukey post-hoc tests through GraphPad Prism 9 (USA) for comparing multiple groups. A p-value of less than 0.05 was considered statistically significant.
Results
Nordalbergin decreases the production of NO, attenuates the expression of COX-2 and inducible nitric oxide synthase (iNOS), and represses the secretion of proinflammatory cytokines by LPS-activated J774A.1 cells
Previous study showed that 20 µM nordalbergin effectively reducing the LPS-induced inflammatory reactions in microglia [20]. To test the anti-inflammatory effects of nordalbergin on J774A.1 cells, we first examined the cytotoxic effects by MTT assay. As shown in Fig. 1A, nordalbergin, at concentrations up to 20 µM, showed no effect on cell survival; thus, these safe doses were employed in the subsequent experiments. NO is produced by iNOS and plays an important role in immune defense and modulation of inflammation. Endotoxemic shock is a systemic inflammatory response that is associated with excessive NO production and leads to tissue damages [24]. The impact of nordalbergin on NO production in LPS-activated macrophages was investigated using the Griess reagent assay to measure NO levels. As shown in Fig. 1B, nordalbergin significantly suppressed the production of NO by LPS-activated J774A.1 cells. COX-2 is one of the cyclooxygenases and its activity can be regulated by NO, as described previously [25]; moreover, the products of COX-2 could also affect iNOS expression [26]. PGE2, an critical inflammatory synthesized by COX-2, also contributes to inflammation and pain through promoting vasodilation, increases vascular permeability, and enhances the pain response [27]. The findings of our experiments revealed that nordalbergin notably reduced the expression of COX-2 and iNOS by LPS-activated J774A.1 cells (Fig. 1C-E). Furthermore, our experimental data indicated that nordalbergin substantially reduced the levels of the proinflammatory mediator PGE2 and cytokines TNF-α and IL-6 by LPS-activated J774A.1 cells (Fig. 1F-H), suggesting that nordalbergin might have potential in attenuating the production of proinflammatory mediators NO and PGE2 and the secretion of proinflammatory cytokines TNF-α and IL-6 during LPS-induced endotoxemia.
Effects of nordalbergin on cell viability, NO production, and proinflammatory cytokine secretion (PGE2, TNF-α and IL-6) as well as iNOS and COX-2 expression by LPS-activated J774A.1 cells. Cells were pre-treated with various concentrations (0, 5, 10, 15 and 20 µM) of nordalbergin for 1 h and then primed with LPS (1 µg/mL) for 24 h. (A) Cell viability was examined by MTT assay. (B) The production of NO was analyzed using Griess reagent assay. Expression of iNOS, COX-2 and β-actin (loading control) was measured by western blot. Representative images are shown in (C). The quantified results of (D) NOS2/β-actin and (E) COX-2/β-actin are shown and were performed from three independent experiments. The secretion of pro-inflammatory cytokines (F) PGE2, (G) TNF-α, and (H) IL-6 was measured by ELISA. Values are presented as means ± SD, and the significant differences are indicated as *p < 0.05, **p < 0.01, and ***p < 0.001
Nordalbergin suppresses JNK and p38 phosphorylations and decreases NF-κB activation by LPS-activated macrophages
Mitogen-activated protein kinase (MAPK) signaling pathways regulate a variety of biological processes and can be classified into three subfamilies: extracellular-regulated kinase (ERK1/2), C-Jun N-terminal kinase (JNK) and p38 MAPK [7]. To investigate the effect of nordalbergin on MAPK signaling pathways in LPS-activated macrophages, J774A.1 cells were pre-treated with different concentrations of nordalbergin (0, 5, 10, 15, and 20 µM) for 1 h and then primed with 1 µg/mL LPS for 2 h. The levels of MAPK-related proteins were assessed using Western blot analysis. As shown in Fig. 2A-D, nordalbergin significantly attenuated the phosphorylation of JNK and p38, but not ERK 1/2. Moreover, J-blue cells were pre-treated with different concentrations of nordalbergin (0, 5, 10, 15, and 20 µM) for 1 h, followed by 1 µg/mL LPS treatment for 18 h. The activity of NF-κB promoter was detected by NF-κB promoter activity assay. As shown in Fig. 2E, the experimental results demonstrated a substantial reduction in NF-κB promoter activity by LPS-activated macrophages under nordalbergin treatment.
Effects of nordalbergin on JNK, p38 MAPK and NF-kB signaling pathways by LPS-activated J774A.1 cells. Cells were pretreated with various concentrations (10 and 20 µM) of nordalbergin for 1 h and then primed with LPS (1 µg/mL) for 2 h. Expressions of MAPK-associated proteins and β-actin (loading control) were analyzed by western blot. The intensities of bands of phospho-proteins were normalized to total proteins. Representative images are shown in (A). The quantified results of (B) phospho-p38/p38, (C) phospho-JNK/JNK, and (D) phospho-ERK/ERK are shown (n = 3). (E) J-Blue cells were pretreated with various concentrations (0, 5, 10, 15, and 20 µM) of nordalbergin for 1 h, following priming of LPS (1 µg/mL) for 18 h. Afterward, cell culture supernatant was collected and detected by SEAP activity assay. The analyzed results were from three independent experiments. Values are presented as means ± SD, and the significant differences are indicated as *p < 0.05, **p < 0.01, and ***p < 0.001
The MAPK and NF-κB pathways often interact and can amplify inflammatory responses [28]. These results suggest that the concurrent inhibition of JNK, p38 MAPK, and NF-κB pathways by nordalbergin can lead to a synergistic reduction in inflammatory mediator production, offering a robust anti-inflammatory effect.
Nordalbergin represses the production of IL-1β and suppresses the activation of NLRP3 inflammasome in J774A.1 cells activated by LPS/ATP and LPS/nigericin
To investigate the impact of nordalbergin on the activation of the NLRP3 inflammasome by LPS/ATP- and LPS/nigericin-activated macrophages, J774A.1 cells were pre-treated with nordalbergin at different concentrations (10 and 20 µM) for 1 h. Subsequently, the cells were primed with 1 µg/mL LPS for 5 h, followed by treatment with either ATP (5 mM) or nigericin (10 µM) for 30 min. Expression of NLRP3 inflammasome-related proteins in culture supernatant and cell lysates was measured by western blotting. Moreover, the production of IL-1β in culture supernatant was measured by ELISA. Our experimental results indicated that nordalbergin reduced the cleavage of both caspase-1 and IL-1β in macrophages activated by LPS/ATP and LPS/nigericin (Fig. 3A-G). Furthermore, nordalbergin led to a reduction in the secretion of cleaved caspase-1 and cleaved IL-1β into the culture supernatant in macrophages activated by LPS/ATP and LPS/nigericin (Fig. 3A and E). Additionally, the ELISA results demonstrated that nordalbergin significantly inhibited the production of IL-1β in macrophages activated by LPS/ATP and LPS/nigericin (Fig. 3D and H). The reduction in IL-1β levels can decrease the recruitment and activation of other immune cells, further mitigating the inflammatory cascade [29]. This helps in controlling the extent and duration of inflammation, protecting tissues from damage [29, 30].
Nordalbergin attenuates NLRP3 inflammasome activation by LPS-stimulated J774A.1 cells. Cells were pre-treated with various concentrations (10 and 20 µM) of nordalbergin for 1 h and then primed with LPS (1 µg/mL) for 5 h, followed by ATP (5 mM) or nigericin (10 µM) treatment for 30 min. Expressions of the NLRP3-related proteins in both culture supernatant and cell lysates, as well as β-actin (loading control), were analyzed by western blot. The intensities of bands of cleaved-form proteins were normalized to total-form proteins. Representative images are shown in (A, E) and the quantified results of (B, F) cleaved IL-1β/IL-1β and (C, G) cleaved caspase-1/ caspase-1 are shown and were performed from three independent experiments. (D, H) Cell culture supernatant was collected, and the examination of IL-1β secretion was analyzed by ELISA. Values are presented as means ± SD, and the significant differences are indicated as *p < 0.05, **p < 0.01, and ***p < 0.001
Nordalbergin reduces the co-localization of NLRP3 inflammasome components (ASC and caspase-1) in J774 A.1 cells stimulated by LPS/ATP
The examination of co-localization between NLRP3 inflammasome components (ASC and caspase-1) was conducted as it has been recognized as a marker for evaluating inflammasome activation [31]. To determine NLRP3 activation in terms of the co-localization of ASC and caspase-1, J774 A.1 cells were pre-treated with various concentrations (10 and 20 µM) of nordalbergin for 1 h. Subsequently, the cells were primed with 1 µg/mL LPS for 5 h, followed by treatment with 5 mM ATP for 30 min. As shown in Fig. 4A, our experimental results revealed that, in comparison to untreated cells, the co-localization of ASC and caspase-1 was enhanced by LPS/ATP treatment, signifying the activation of the NLRP3 inflammasome. Notably, nordalbergin significantly reduced the co-localization of ASC and caspase-1 in J774 A.1 cells activated by LPS/ATP (Fig. 4B). By disrupting the process of ASC and caspase-1 co-localization, nordalbergin effectively prevents the assembly and subsequent activation of the inflammasome, thus reducing the release of IL-1β.
Effects of Nordalbergin on the colocalization of NLRP3 inflammasome components (ASC and caspase-1). Cells were pre-treated with various concentrations (10 and 20 µM) of nordalbergin for 1 h and then primed with LPS (1 µg/mL) for 5 h, followed by treatment of ATP (5 mM) for 30 min. (A) Cells were stained with caspase-1 (green), ASC (red), and DAPI (blue) and then analyzed using confocal microscopy and Imaris software for confocal 3D image reconstruction. (B) The co-localization of caspase-1 and ASC signals was determined by the thresholds of the 2D histogram by Mander’s coefficient. Values are presented as means ± SD, and the significant differences are indicated as *p < 0.05, **p < 0.01 and ***p < 0.001
Nordalbergin inhibits the production of NO and proinflammatory cytokines in LPS-activated PEM and BMDM
According to the above experimental results (Fig. 1), nordalbergin demonstrated significant inhibitory effects on the release of inflammatory mediators (NO and PGE2) as well as pro-inflammatory cytokines (TNF-α, IL-6, and IL-1β) in J774A.1 cells. To further elucidate the anti-inflammatory activities of nordalbergin, we aimed to implement the primary-culture macrophages, PEM and BMDM into the ex vivo experimental design. Thioglycolate-elicited PEM and BMDM were pre-treated with different concentrations of nordalbergin (0, 5, 10, 15, and 20 µM) for 1 h, followed by 1 µg/mL LPS treatment for 24 h. MTT assay was initially performed to elucidate the possible cytotoxic effects of nordalbergin on PEM and BMDM. The results demonstrated that concentrations of nordalbergin up to 20 µM had no cytotoxic effects on LPS-activated PEM and LPS-activated BMDM (Fig. 5A and F). In addition, the experimental results indicated that nordalbergin significantly attenuated NO production in PEM and BMDM (Fig. 5B and G). Moreover, nordalbergin repressed the production of pro-inflammatory cytokines (TNF-α, IL-6, and IL-1β) in PEM (Fig. 5C-E) and BMDM (Fig. 5H-J) as well.
Effects of nordalbergin on peritoneal macrophages (PEM) and bone marrow-derived macrophages (BMDM). PEM (A-E) and BMDM (F-J) were pre-treated with various concentrations (0, 5, 10, 15, and 20 µM) of nordalbergin for 1 h and then primed with LPS (1 µg/mL) for 24 h. (A, F) Cell viability was analyzed by MTT assay. (B, G) The production of nitric oxide (NO) was measured by Griess reagent assay. ELISA was performed to measure the secretion of pro-inflammatory cytokines (C, H) TNF-α and (D, I) IL- 6. (E, J) Cells were pre-treated with various concentrations (0, 5, 10, 15, and 20 µM) of nordalbergin for 1 h and then primed with LPS (1 µg/mL) for 5 h, followed by ATP (5 mM) treatment for 30 min. Cell culture supernatant was collected and the production of IL-1β was detected by ELISA. The analyzed results were from three independent experiments. Values are presented as means ± SD, and the significant differences are indicated as *p < 0.05, **p < 0.01, and ***p < 0.001
Nordalbergin reveals protective effects against LPS-induced tissue damages in mice subjected to LPS challenge
The murine endotoxemia model has been widely adopted as a method of studying sepsis and septic diseases. In our research, intraperitoneal (i.p.) injection was performed to acquire LPS-challenged mice. To testify the protective effects of nordalbergin against LPS-induced tissue damage, mice were first intraperitoneally injected with nordalbergin for 1 h and then with LPS for 5 h. Afterward, murine tissues (lung, liver, and kidney) were harvested and stained by an H&E staining kit. Simultaneously, blood was collected, and the levels of aspartate transaminase (AST), alanine transaminase (ALT), and blood urea nitrogen (BUN) in the serum were measured by Mic Fujifilm Dri-Chem NX-500 chemistry analyzer.
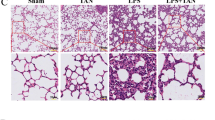
As shown in Fig. 6A, the administration of LPS triggered an increase in the infiltration of inflammatory cells, causing lung, liver, and kidney injury obviously. By contrast, a decrease in inflammatory cell infiltration can notably be observed in cells administered by nordalbergin. In addition, nordalbergin reduced levels of AST, ALT and BUN in mice serum (Fig. 6B-D). Collectively, our experimental results further verified the protective effects of nordalbergin against septic-induced tissue damage by murine endotoxemia model.
Effects of nordalbergin on serum inflammatory markers and tissue damage (lung, liver, and kidney) in mice. Mice were intraperitoneally injected with nordalbergin (10 or 20 mg/kg) or DMSO (control group), followed by the intraperitoneal injection of 50 mg/kg LPS. Tissues of the lung, liver, and kidney were harvested after 5 h of LPS injection. (A) H&E staining (200x magnification). Infiltration and damaged sites (neutrophils) were indicated specifically by red arrows. Indicative values of (B) AST, (C) ALT, and (D) BUN in mice serum were measured by Fujifilm DriI-Chem NX-500 chemistry analyzer. The analyzed results were performed from three independent experiments. Values are presented as means ± SD (n = 3), and the significant differences are indicated as *p < 0.05, **p < 0.01, and ***p < 0.001
Nordalbergin decreases the secretion of ROS by LPS/ATP- and LPS/nigericin-induced J774A.1 cells
The role of ROS in NLRP3 activation has recently been well-acknowledged. Intracellular (NOX-derived) ROS triggers activation of NLRP3 inflammasome in macrophages upon stimulation with DAMPs, PAMPs, ATP as well as pore-forming toxins [32]. In addition, ROS act as secondary messengers in the activation of NF-κB and MAPK pathways [33]. Taken together, the secretion of ROS can be used as a prominent indicator to investigate the anti-inflammatory and antioxidant effects of potential therapeutic agents. To investigate ROS production, ATP and nigericin were applied as mediators to activate NLRP3 inflammasome. J774A.1 cells were pre-treated with various concentrations (10 and 20 µM) of nordalbergin for 1 h and then primed with LPS (1 µg/mL) for 24 h. Before collecting the living cells, samples were treated with either ATP (5 mM) or nigericin (10 µM) for 30 min. H2DCFDA (5 µM) staining was performed to stain the cells. Afterward, the levels of LPS/ATP- and LPS/nigericin-induced ROS in J774A.1 macrophages were measured by flow cytometry. The experimental results demonstrated that nordalbergin significantly inhibited ROS production triggered by NLRP3 inflammasome activation (Fig. 7). By reducing ROS levels, nordalbergin likely leads to attenuate NF-κB and MAPK pathways and decrease NLRP3 inflammasome activation, providing a comprehensive view of how nordalbergin modulates LPS-induced inflammation.
Effects of nordalbergin on the secretion of reactive oxygen species (ROS) by LPS/ATP- and LPS/nigericin-induced J774A.1 cells. Cells were pre-treated with various concentrations (10 and 20 µM) of nordalbergin for 1 h and then primed with LPS (1 µg/mL) for 24 h. Samples were incubated with either ATP (5 mM) or nigericin (10 µM) for 30 min. H2DCFDA (5 µM) was added to stain the cells. (A, B) The relative fluorescence intensity for ROS generation was analyzed and is shown in histograms by flow cytometry. (C, D) Intracellular ROS levels were quantified. The analyzed results were from three independent experiments. Values are presented as means ± SD, and the significant differences are indicated as *p < 0.05, **p < 0.01, and ***p < 0.001
Discussion
Sepsis is an overwhelming systemic response to infection that frequently results in tissue damage, organ failure and even death. The excessive production of pro-inflammatory mediators like NO and PGE2, along with pro-inflammatory cytokines (such as TNF-α, IL-6, and IL-1β), is believed to be linked to the immunostimulatory process in sepsis [7, 23, 34]. Dalbergia sissoo DC., commonly known as Indian rosewood or Sheesham, is a traditional medicinal plant with a long history of use for its analgesic, anti-nociceptive, anti-inflammatory, and immuno-modulatory properties, especially for the treatment of joint inflammation and joint pain [18]. Nordalbergin, a coumarin derived from Dalbergia sissoo DC, has been shown to stimulate the differentiation of human promyelocytic leukemia HL-60 cells [19]. Recent research has indicated that nordalbergin exhibits anti-neuroinflammatory properties by significantly reducing inflammatory reactions in microglia [20]. However, its anti-inflammatory effects in the context of endotoxemia have not been explored. Therefore, this study aimed to investigate the anti-inflammatory potential of nordalbergin on LPS-stimulated macrophages using both in vitro and ex vivo methods, as well as in an LPS-induced endotoxemia mouse model. Our experimental results demonstrated that nordalbergin inhibited LPS-induced NO production by murine J774A.1 cells, PEM, and BMDM; additionally, it significantly decreased the expression of iNOS and COX-2 and suppressed the production of PGE2 by LPS-induced J774A.1 cells. On the other hand, our experimental results also showed that nordalbergin reduced LPS-induced IL-6 and TNF-α production by murine J774A.1 cells, PEM, and BMDM, indicating that nordalbergin inhibits LPS-induced inflammatory responses by macrophages. Consistent to previous study, nordalbergin also inhibited NO production, iNOS and COX-2 expressions, and IL-6 and TNF-α production by LPS-induced microglia [20]. These results indicate that nordalbergin is a critical compound in anti-inflammatory property of Dalbergia sissoo DC.
Bacterial LPS is the most common microbial mediator in sepsis and septic shock [5], which binds to TLR4 and activates its downstream signaling cascades, including MAPKs and NF-κB signaling pathway [7, 23]. Our experimental results pointed out that nordalbergin not only inhibited JNK and p38 phosphorylations but also reduced NF-κB activity by LPS-induced macrophages. A previous study indicated that inhibition of JNK with a JNK inhibitor SP600125 in a murine sepsis model exhibits protective effects against sepsis-induced acute lung injury [35]. Moreover, O’Sullivan et al. indicated that both p38 MAPK inhibitor SB-202,190 and NF-κB inhibitor SN-50 exhibit a significant survival benefit in the cecal ligation and puncture-induced sepsis animal model [10]. In the context of sepsis, the elevation of serum markers such as AST, ALT, and BUN is indicative of multi-organ dysfunction, particularly affecting the liver and kidneys [36]. For the pathophysiology of sepsis, neutrophils are the first responders to inflammatory signals in the lungs, and the infiltration of neutrophils is associated with the release of proteases, ROS, and other cytotoxic substances that can damage the alveolar-capillary barrier [37]. Similar to the lungs, inflammatory cells infiltrate the liver, releasing enzymes and oxidative species that cause hepatocellular injury and necrosis [38]. Our experimental results demonstrated that nordalbergin significantly reduced the levels of AST, ALT, and BUN, and decreased the infiltration of inflammatory cells in lung, liver, and kidney tissues in LPS-challenged mice, indicating that nordalbergin exhibits protective effects against LPS-induced tissue damages in endotoxemia. Collectively, these results suggest that nordalbergin attenuated LPS-induced inflammatory response and protected LPS-induced tissue damages through attenuating JNK, p38, and NF-κB signaling pathways.
Regulation of the activating-form inflammasome is a key mechanism of inflammation that results in the secretion of IL-1β [39]. The activation of NLRP3 inflammasome has been shown to participate in polymicrobial sepsis as well as in LPS-induced acute lung injury (ALI) in mice [40, 41]. Previous studies have revealed that multi-organ dysfunction in septic mice could be greatly reduced through inhibition of NLRP3 inflammasome [42, 43]. Our experimental results demonstrated that nordalbergin effectively reduces the activation of NLRP3 inflammasome through decreasing the cleavage of IL-1β and caspase-1 and assembly of the NLRP3 inflammasome as well as inhibits the secretion of IL-1β by LPS-induced macrophages. In addition, ROS have been demonstrated to interact with NLRP3 inflammasomes and are involved in the inflammation of sepsis progression [17]. Our experimental results showed that nordalbergin effectively suppressed the secretion of ROS in a dose-dependent manner, revealing an anti-oxidative effect of nordalbergin and consequent blockage of NLRP3 inflammasome activity in LPS/ATP- and LPS/nigericin-induced macrophages. A potent and selective NLRP3 inhibitor, MCC950, acts as the blocker of the assembly and activation of the NLRP3 inflammasome, and exhibits the therapeutic benefits in reducing IL-1β production and improving survival in septic models [44, 45]. Our experimental results demonstrated that nordalbergin also has the efficacy in inhibiting NLRP3 inflammasome assembly and activation as well as attenuating IL-1β production, indicating that nordalbergin could be an effective compound for treating sepsis.
Conclusion
Our experimental results firstly demonstrated that nordalbergin exhibits anti-inflammatory effects in LPS-induced macrophages by blocking NF-κB/MAPK signaling pathways. Moreover, nordalbergin effectively reduces the activation of NLRP3 inflammasome by decreasing the expression of NLRP3 assembly-related proteins and relieving ROS secretion. Collectively, these results suggest that nordalbergin possesses the potential to act as a therapeutic drug agent against LPS-induced tissue damage and attenuate NLRP3 inflammasome-related diseases.
Data availability
Data will be made available on request.
References
Huang M, Cai S, Su J. The pathogenesis of sepsis and potential therapeutic targets. Int J Mol Sci. 2019;20:5376. https://doi.org/10.1002/mco2.418.
Qi X, Yu Y, Sun R, Huang J, Liu L, Yang Y, et al. Identification and characterization of neutrophil heterogeneity in sepsis. Crit Care. 2021;25:50. https://doi.org/10.1186/s13054-021-03481-0.
Rudd KE, Johnson SC, Agesa KM, Shackelford KA, Tsoi D, Kievlan DR, et al. Global, regional, and national sepsis incidence and mortality, 1990–2017: analysis for the global burden of Disease Study. Lancet. 2020;395:200–11. https://doi.org/10.1016/S0140-6736(19)32989-7.
Cheng Y, Marion TN, Cao X, Wang W, Cao Y. Park 7: a novel therapeutic target for macrophages in sepsis-induced immunosuppression. Front Immunol. 2018;9:2632. https://doi.org/10.3389/fimmu.2018.02632.
Gabarin RS, Li M, Zimmel PA, Marshall JC, Li Y, Zhang H. Intracellular and extracellular lipopolysaccharide signaling in sepsis: avenues for novel therapeutic strategies. J Innate Immun. 2021;13:323–32. https://doi.org/10.1159/000515740.
Wang Z, Chen W, Li Y, Zhang S, Lou H, Lu X, et al. Reduning injection and its effective constituent luteoloside protect against sepsis partly via inhibition of HMGB1/TLR4/NF-kappaB/MAPKs signaling pathways. J Ethnopharmacol. 2021;270:113783. https://doi.org/10.1016/j.jep.2021.113783.
Hung YL, Wang SC, Suzuki K, Fang SH, Chen CS, Cheng WC, et al. Bavachin attenuates LPS-induced inflammatory response and inhibits the activation of NLRP3 inflammasome in macrophages. Phytomedicine. 2019;59:152785. https://doi.org/10.1016/j.phymed.2018.12.008.
Zhang W, Chen H, Xu Z, Zhang X, Tan X, He N, et al. Liensinine pretreatment reduces inflammation, oxidative stress, apoptosis, and autophagy to alleviate sepsis acute kidney injury. Int Immunopharmacol. 2023;122:110563. https://doi.org/10.1016/j.intimp.2023.110563.
Cao M, Wang G, Xie J. Immune dysregulation in sepsis: experiences, lessons and perspectives. Cell Death Discov. 2023;9:465. https://doi.org/10.1038/s41420-023-01766-7.
O’Sullivan AW, Wang JH, Redmond HP. NF-kappaB and p38 MAPK inhibition improve survival in endotoxin shock and in a cecal ligation and puncture model of sepsis in combination with antibiotic therapy. J Surg Res. 2009;152:46–53. https://doi.org/10.1016/j.jss.2008.04.030.
Wang W, Mao S, Yu H, Wu H, Shan X, Zhang X, et al. Pinellia pedatisecta lectin exerts a proinflammatory activity correlated with ROS-MAPKs/NF-kappaB pathways and the NLRP3 inflammasome in RAW264.7 cells accompanied by cell pyroptosis. Int Immunopharmacol. 2019;66:1–12. https://doi.org/10.1016/j.intimp.2018.11.002.
Ge Q, Chen X, Zhao Y, Mu H, Zhang J. Modulatory mechanisms of NLRP3: potential roles in inflammasome activation. Life Sci. 2021;267:118918. https://doi.org/10.1016/j.lfs.2020.118918.
Danielski LG, Giustina AD, Bonfante S, Barichello T, Petronilho F. The NLRP3 inflammasome and its role in sepsis development. Inflammation. 2020;43:24–31. https://doi.org/10.1007/s10753-019-01124-9.
Liu Y, Zhang Y, You G, Zheng D, He Z, Guo W, et al. Tangeretin attenuates acute lung injury in septic mice by inhibiting ROS-mediated NLRP3 inflammasome activation via regulating PLK1/AMPK/DRP1 signaling axis. Inflamm Res. 2024;73:47–63. https://doi.org/10.1007/s00011-023-01819-8.
Jin L, Batra S, Jeyaseelan S. Deletion of nlrp3 augments survival during polymicrobial sepsis by decreasing autophagy and enhancing phagocytosis. J Immunol. 2017;198:1253–62. https://doi.org/10.4049/jimmunol.1601745.
Lee S, Nakahira K, Dalli J, Siempos II, Norris PC, Colas RA, et al. NLRP3 inflammasome deficiency protects against microbial sepsis via increased lipoxin B(4) synthesis. Am J Respir Crit Care Med. 2017;196:713–26. https://doi.org/10.1164/rccm.201604-0892OC.
Zhao S, Chen F, Yin Q, Wang D, Han W, Zhang Y. Reactive oxygen species interact with NLRP3 inflammasomes and are involved in the inflammation of sepsis: from mechanism to treatment of progression. Front Physiol. 2020;11:571810. https://doi.org/10.3389/fphys.2020.571810.
Kothari P, Tripathi AK, Girme A, Rai D, Singh R, Sinha S, et al. Caviunin glycoside (CAFG) from Dalbergia sissoo attenuates osteoarthritis by modulating chondrogenic and matrix regulating proteins. J Ethnopharmacol. 2022;282:114315. https://doi.org/10.1016/j.jep.2021.114315.
Kawaii S, Tomono Y, Katase E, Ogawa K, Yano M. Effect of coumarins on HL-60 cell differentiation. Anticancer Res. 2000;20:2505–12.
Lo J, Wu HE, Liu CC, Chang KC, Lee PY, Liu PL, et al. Nordalbergin exerts anti-neuroinflammatory effects by attenuating MAPK signaling pathway, NLRP3 inflammasome activation and ROS production in LPS-stimulated BV2 microglia. Int J Mol Sci. 2023;24:7300. https://doi.org/10.3390/ijms24087300.
Jakobs C, Bartok E, Kubarenko A, Bauernfeind F, Hornung V. Immunoblotting for active caspase-1. Methods Mol Biol. 2013. https://doi.org/10.1007/978-1-62703-523-1_9. 1040:103 – 15.
Yazal T, Lee PY, Chen PR, Chen IC, Liu PL, Chen YR, et al. Kurarinone exerts anti-inflammatory effect via reducing ROS production, suppressing NLRP3 inflammasome, and protecting against LPS-induced sepsis. Biomed Pharmacother. 2023;167:115619. https://doi.org/10.1016/j.biopha.2023.115619.
Huang CH, Wang SC, Chen IC, Chen YT, Liu PL, Fang SH, et al. Protective effect of piplartine against LPS-induced sepsis through attenuating the MAPKs/NF-kappaB signaling pathway and NLRP3 inflammasome activation. Pharmaceuticals. 2021;14:588. https://doi.org/10.3390/ph14060588.
Sahan-Firat S, Canacankatan N, Korkmaz B, Yildirim H, Tamer L, Buharalioglu CK, et al. Increased production of nitric oxide mediates selective organ-specific effects of endotoxin on oxidative stress. Antiinflamm Antiallergy Agents Med Chem. 2012;11:161–72. https://doi.org/10.2174/187152312803305740.
Gao Y, Jiang W, Dong C, Li C, Fu X, Min L, et al. Anti-inflammatory effects of sophocarpine in LPS-induced RAW 264.7 cells via NF-kappaB and MAPKs signaling pathways. Toxicol Vitro. 2012;26:1–6. https://doi.org/10.1016/j.tiv.2011.09.019.
Perez-Sala D, Lamas S. Regulation of cyclooxygenase-2 expression by nitric oxide in cells. Antioxid Redox Signal. 2001. https://doi.org/10.1089/152308601300185197. 3:231 – 48.
Ricciotti E, FitzGerald GA. Prostaglandins and inflammation. Arterioscler Thromb Vasc Biol. 2011;31:986–1000. https://doi.org/10.1161/ATVBAHA.110.207449.
Dorrington MG, Fraser IDC. NF-kappaB Signaling in macrophages: Dynamics, Crosstalk, and Signal Integration. Front Immunol. 2019;10:705. https://doi.org/10.3389/fimmu.2019.00705.
Al-Qahtani AA, Alhamlan FS, Al-Qahtani AA. Pro-inflammatory and anti-inflammatory interleukins in infectious diseases: a comprehensive review. Trop Med Infect Dis. 2024;9:13. https://doi.org/10.3390/tropicalmed9010013.
Dinarello CA, Simon A, van der Meer JW. Treating inflammation by blocking interleukin-1 in a broad spectrum of diseases. Nat Rev Drug Discov. 2012;11:633–52. https://doi.org/10.1038/nrd3800.
Zhang C, Boini KM, Xia M, Abais JM, Li X, Liu Q, et al. Activation of nod-like receptor protein 3 inflammasomes turns on podocyte injury and glomerular sclerosis in hyperhomocysteinemia. Hypertension. 2012;60:154–62. https://doi.org/10.1161/HYPERTENSIONAHA.111.189688.
Rendra E, Riabov V, Mossel DM, Sevastyanova T, Harmsen MC, Kzhyshkowska J. Reactive oxygen species (ROS) in macrophage activation and function in diabetes. Immunobiology. 2019;224:242–53. https://doi.org/10.1016/j.imbio.2018.11.010.
Checa J, Aran JM. Reactive oxygen species: drivers of physiological and pathological processes. J Inflamm Res. 2020;13:1057–73. https://doi.org/10.2147/JIR.S275595.
Gao N, Chen J, Li Y, Ding Y, Han Z, Xu H, et al. The CYP2E1 inhibitor Q11 ameliorates LPS-induced sepsis in mice by suppressing oxidative stress and NLRP3 activation. Biochem Pharmacol. 2023;214:115638. https://doi.org/10.1016/j.bcp.2023.115638.
Lou L, Hu D, Chen S, Wang S, Xu Y, Huang Y, et al. Protective role of JNK inhibitor SP600125 in sepsis-induced acute lung injury. Int J Clin Exp Pathol. 2019;12:528–38.
Li JL, Li G, Jing XZ, Li YF, Ye QY, Jia HH, et al. Assessment of clinical sepsis-associated biomarkers in a septic mouse model. J Int Med Res. 2018;46:2410–22. https://doi.org/10.1177/0300060518764717.
Yang SC, Tsai YF, Pan YL, Hwang TL. Understanding the role of neutrophils in acute respiratory distress syndrome. Biomed J. 2021. https://doi.org/10.1016/j.bj.2020.09.001. 44:439 – 46.
Yan J, Li S, Li S. The role of the liver in sepsis. Int Rev Immunol. 2014;33:498–510. https://doi.org/10.3109/08830185.2014.889129.
Latz E, Xiao TS, Stutz A. Activation and regulation of the inflammasomes. Nat Rev Immunol. 2013;13:397–411. https://doi.org/10.1038/nri3452.
Fattahi F, Zetoune FS, Ward PA. Complement as a major inducer of harmful events in infectious sepsis. Shock. 2020;54:595–605. https://doi.org/10.1097/SHK.0000000000001531.
He W, Dong H, Wu C, Zhong Y, Li J. The role of NLRP3 inflammasome in sepsis: a potential therapeutic target. Int Immunopharmacol. 2023;115:109697. https://doi.org/10.1016/j.intimp.2023.109697.
Liu LL, Yan X, Xue KY, Wang XM, Li LY, Chen HY, et al. Prim-O-glucosycimifugin attenuates liver injury in septic mice by inhibiting NLRP3 inflammasome/caspase-1 signaling cascades in macrophages. Phytomedicine. 2022;106:154427. https://doi.org/10.1016/j.phymed.2022.154427.
Li YF, Sheng HD, Qian J, Wang Y. The Chinese medicine babaodan suppresses LPS-induced sepsis by inhibiting NLRP3-mediated inflammasome activation. J Ethnopharmacol. 2022;292:115205. https://doi.org/10.1016/j.jep.2022.115205.
Miao R, Huang J. MCC950 improves lipopolysaccharide–induced systemic inflammation in mice by relieving pyroptosis in blood neutrophils. Exp Ther Med. 2023;26:417. https://doi.org/10.3892/etm.2023.12117.
Li S, Guo Z, Zhang ZY. Protective effects of NLRP3 inhibitor MCC950 on sepsis-induced myocardial dysfunction. J Biol Regul Homeost Agents. 2021;35:141–50. https://doi.org/10.23812/20-662-A.
Acknowledgements
The authors would like to thank the Center for Research Resources, and Development of Kaohsiung Medical University for support in confocal microscopy, and thank the Center for Laboratory Animals in Kaohsiung Medical University for the animal care. This study was supported by grants from the National Science and Technology Council, Taiwan, R.O.C. (grant No. NSTC 111-2218-E-037-001, NSTC 111-2314-B-037-071-MY3, NSTC 112-2314-B-037-127, NSTC 112-2314-B-037-128, NSTC 112-2926-I-037-501-G, NSTC 112-2314-B-037-089, NSTC 112-2311-B-039 -001, NSTC 112-2622-E-039-001, NSTC 111-2622-E-039-004, NSTC 113-2311-B-039-001-MY3, NSTC 113-2314-B-037-017 and NSTC 112-2218-E-037-001), China Medical University (grant No. CMU111-IP-04 and CMU112-MF-25), China Medical University Hospital (grant No. DMR-112-056, DMR-113-190, and DMR-113-191), and NTHU-KMU Joint Research Project (grant No. KT113P010).
Author information
Authors and Affiliations
Contributions
Pin-Rong Chen: Conceptualization, Data curation, Writing–original draft. Chia-Yang Li: Conceptualization, Funding acquisition, Writing– review & editing. Taha Yazal: Formal analysis, Methodology. I-Chen Chen: Formal analysis. Po-Len Liu: Data curation. Yi-Ting Chen: Formal analysis. Ching-Chih Liu: Methodology. Jung Lo: Methodology. Tzu-Chieh Lin: Methodology. Ching-Tang Chang: Methodology. Hsin-En Wu: Methodology. Yuan-Ru Chen: Data curation. Wei-Chung Cheng: Conceptualization, Funding acquisition. Chien-Chih Chiu: Data curation. Chi-Shuo Chen: Data curation. Shu-Chi Wang: Conceptualization, Funding acquisition, Writing– review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Communicated by Artur Bauhofer.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, PR., Li, CY., Yazal, T. et al. Protective effects of nordalbergin against LPS-induced endotoxemia through inhibiting MAPK/NF-κB signaling pathway, NLRP3 inflammasome activation, and ROS production. Inflamm. Res. (2024). https://doi.org/10.1007/s00011-024-01922-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00011-024-01922-4