Summary
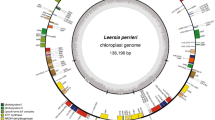
The entire chloroplast genome of the monocot rice (Oryza sativa) has been sequenced and comprises 134525 bp. Predicted genes have been identified along with open reading frames (ORFs) conserved between rice and the previously sequenced chloroplast genomes, a dicot, tobacco (Nicotiana tabacum), and a liverwort (Marchantia polymorpha). The same complement of 30 tRNA and 4 rRNA genes has been conserved between rice and tobacco. Most ORFs extensively conserved betweenN. tabacum andM. polymorpha are also conserved intact in rice. However, several such ORFs are entirely absent in rice, or present only in severely truncated form. Structural changes are also apparent in the genome relative to tobacco. The inverted repeats, characteristic of chloroplast genome structure, have expanded outward to include several genes present only once per genome in tobacco and liverwort and the large single copy region has undergone a series of inversions which predate the divergence of the cereals. A chimeric tRNA pseudogene overlaps an apparent endpoint of the largest inversion, and a model invoking illegitimate recombination between tRNA genes is proposed which accounts simultaneously for the origin of this pseudogene, the large inversion and the creation of repeated sequences near the inversion endpoints.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- PSII:
-
photosystem II
- PSI:
-
photosystem I
- RuBisCo:
-
ribulose 1,5-bisphosphate carboxylase
- IRA and IRB :
-
denote the inverted repeat regions distal and proximal tondhF respectively
References
deHeij HT, Lustig H, Moeskops D-JM, Bovenberg WA, Bisanz C, Groot GSP (1983) Chloroplast DNAs ofSpinacia, Petunia andSpirodela have a similar gene organization. Curr Genet 7:1–6
Gupta KC, Patwardhan S (1988) ACG, the initiator codon for a Sendai virus protein. J. Biol Chem 263:8553–8556
Hirai A, Ishibashi T, Morikami A, Iwatsuki N, Shinozaki K, Sugiura M (1985) Rice chloroplast DNA: a physical map and the location of the genes for the large subunit of ribulose 1,5-bisphosphate carboxylase and the 32 KD photosystem II reaction center protein. Theor Appl Genet 70:117–122
Howe CJ (1985) The endpoints of an inversion in wheat chloroplast DNA are associated with short repeated sequences containing homology toatt. Curr Genet 10:139–145
Kohchi T, Ogura Y, Umesono K, Yamada Y, Komano Y, Ozeki H, Ohyama K (1988) Ordered processing and splicing in a polycistronic transcript in liverwort chloroplasts. Curr Genet 14:147–154
Kozak M (1983) Comparison of initiation of protein synthesis in procaryotes, eucaryotes, and organelles. Microbiol Rev 47:1–45
McLaughlin WE, Larrinua IM (1988) The sequence of the maize plastid encodedrpl23 locus. Nucleic Acids Res 16:8183
Meng BY, Tanaka M, Wakasugi T, Ohme M, Shinozaki K, Sugiura M (1988) Cotranscription of the genes for ribosomal protein CS14: determination of the transcriptional initiation site by in vitro capping. Curr Genet 14:395–400
Moon E, Kao T-H, Wu R (1988) Rice mitochondrial genome contains a rearranged chloroplast gene cluster. Mol Gen Genet 213:247–253
Murata N, Miyao M, Hayashida N, Hidaka T, Sugiura M (1988) Identification of a new gene in the chloroplast genome encoding a low-molecular-mass polypeptide of photosystem II complex. FEBS Lett 235:283–288
Ogihara Y, Terachi T, Sasakuma T (1988) Intramolecular recombination of chloroplast genome mediated by short direct-repeat sequences in wheat species. Proc Natl Acad Sci USA 85:8573–8577
Ohyama K, Fukuzawa H, Kohchi T, Shirai H, Sano T, Sano S, Umesono K, Shiki Y, Takeuchi M, Chang Z, Aota S-I, Inokuchi H, Ozeki H (1986) Chloroplast gene organization deduced from complete sequence of liverwortMarchantia polymorpha chloroplast DNA. Nature 322:572–574
Ohyama K, Fukuzawa H, Kohchi T, Sano T, Sano S, Shirai H, Umesono K, Shiki Y, Takeuchi M, Chang Z, Aota S-I, Inokuchi H, Ozeki H (1988) Structure and organization ofMarchantia polymorpha chloroplast genome. I. Cloning and gene identification. J Mol Biol 203:281–298
Palmer JD (1985) Comparative organization of chloroplast genomes. Annu Rev Genet 19:325–354
Palmer JD, Stein DB (1986) Conservation of chloroplast genome structure among vascular plants. Curr Genet 10:823–833
Palmer JD, Thompson WF (1982) Chloroplast DNA rearrangements are more frequent when a large inverted repeat sequence is lost. Cell 29:537–550
Palmer JD, Jansen RK, Michaels HJ, Chase MW, Manhart JR (1988) Chloroplast DNA variation and plant phylogeny. Ann Missouri Bot Garden 75:1180–1218
Posno M, van Noort M, Debise R, Groot GSP (1984) Isolation, characterization, phosphorylation and site of synthesis ofSpinacia chloroplast ribosomal proteins. Curr Genet 8:147–154
Quigley F, Weil JH (1985) organization and sequence of five tRNA genes and of an unidentified reading frame in the wheat chloroplast genome: evidence for gene rearrangements during the evolution of chloroplast genomes. Curr Genet 9:495–503
Rodermel S, Orlin P, Bogorad L (1987) The transcription termination region between two convergently-transcribed photoregulated operons in the maize plastid chromosome containsrps14,trnR (UCC) and a putativetrnfM pseudogene. Nucleic Acids Res 15:5493
Sanger F, Nicklen S, Coulson AR (1977) DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA 74:5463–5467
Schmidt GW, Mishkind ML (1986) The transport of proteins into chloroplasts. Annu Rev Biochem 55:879–912
Shinozaki K, Ohme M, Tanaka M, Wakasugi T, Hayashida N, Matsubayashi T, Zaita N, Chunwongse J, Obokata J, Yamaguchi-Shinozaki K, Ohto C, Torazawa K, Meng BY, Kusuda J, Takaiwa F, Kato A, Tohdoh N, Shimada H, Sugiura M (1986) The complete nucleotide sequence of the tobacco chloroplast genome: its gene organization and expression. EMBO J 5:2043–2049
Stein DB, Palmer JD, Thompson WF (1986) Structural evolution and flip-flop recombination of chloroplast DNA in the fern genusOsmunda. Curr Genet 10:835–841
Stern DB, Gruissem W (1987) Control of plastid gene expression: 3′ inverted repeats act as mRNA processing and stabilizing elements, but do not terminate transcription. Cell 51:1145–1157
Stewart WN (1983) Paleobotany and the evolution of plants. Cambridge University Press, Cambridge, UK
Sugiura M, Shinozaki K, Zaita N, Kusuda M, Kumano M (1986) Clone bank of the tobacco (Nicotiana tabacum) chloroplast genome as a set of overlapping restriction endonuclease fragments: mapping of eleven ribosomal protein genes. Plant Sci 44:211–216
Tanaka M, Wakasugi T, Sugita M, Shinozaki K, Sugiura M (1986) Genes for the eight ribosomal proteins are clustered on the chloroplast genome of tobacco (Nicotiana tabacum): similarity to the S10 andspc operons ofEscherichia coli. Proc Natl Acad Sci USA 83:6030–6034
Thach RE, Sundararajan TA, Dewey DF, Brown JC, Doty P (1966) Translation of synthetic messenger RNA. Cold Spring Harbor Symp Quant Biol 31:85–97
Wakasugi T, Ohme M, Shinozaki K, Sugiura M (1986) Structures of tobacco chloroplast genes for tRNAIle (CAU), tRNALeu (CAA), tRNACys (GCA), tRNASer (UGA) and tRNAThr (GGU): a compilation of tRNA genes from tobacco chloroplasts. Plant Mol Biol 7:385–392
Wolfe KH, Sharp PM (1988) Identification of functional open reading frames in chloroplast genomes. Gene 66:215–222
Zaita N, Torazawa K, Shinozaki K, Sugiura M (1987)Trans splicting in vivo: joining of transcripts from the ‘divided’ gene for ribosomal protein S12 in the chloroplasts of tobacco. FEBS Lett 210:153–156
Zurawski G, Clegg MT (1987) Evolution of higher-plant chloroplast DNA-encoded genes: implications for structure-function and phylogenetic studies. Annu Rev Plant Physiol 38:391–418
Zurawski G, Bottomley W, Whitfield PR (1984) Junctions of the large single copy region and the inverted repeats inSpinacia oleracea andNicotiana debneyi chloroplast DNA: sequence of the genes for tRNAHis and the ribosomal proteins S19 and L2. Nucleic Acids Res 12:6547–6558
Author information
Authors and Affiliations
Additional information
Communicated by R.G. Herrmann
Rights and permissions
About this article
Cite this article
Hiratsuka, J., Shimada, H., Whittier, R. et al. The complete sequence of the rice (Oryza sativa) chloroplast genome: Intermolecular recombination between distinct tRNA genes accounts for a major plastid DNA inversion during the evolution of the cereals. Molec. Gen. Genet. 217, 185–194 (1989). https://doi.org/10.1007/BF02464880
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02464880