Abstract
Background
Patients with hepatocellular carcinoma (HCC) beyond the traditional criteria (advanced HCC) are typically offered palliation, which is associated with a 3-year survival rate lower than 30%. This study aimed to describe the outcomes for a subset of patients with advanced HCC who satisfied the Extended Toronto Criteria (ETC) and were listed for liver transplantation (LT).
Methods
All patients listed in the Toronto liver transplantation program with HCC beyond both the Milan and University of California, San Francisco criteria were included in this study. Data were extracted from the prospectively collected electronic database. All radiologic images were reviewed by two independent radiologists. The primary end point was patient survival.
Results
Between January 1999 and August 2014, 96 patients with advanced HCC were listed for LT, and 62 (65%) of these patients received bridging therapy while on the waiting list. Bridging therapy led to a significant reduction in tumor progression (p = 0.02) and tumor burden (p < 0.001). The majority of those listed underwent LT (n = 69, 72%). Both tumor progression on waiting list (hazard ratio [HR] 4.973; range1.599–15.464; p = 0.006) and peak alpha-fetoprotein (AFP) at 400 ng/ml or higher (HR, 4.604; range 1.660–12.768; p = 0.003) were independently associated with waiting list dropout. Post-LT HCC recurrence occurred in 35% of the patients (n = 24). Among those with HCC recurrence, survival was significantly better for those who received curative treatment (p = 0.004). The overall actuarial survival rates from the listing were 76% at 1 year, 56% at 3 years, and 47% at 5 years, and the corresponding rates from LT were 93, 71, and 66%.
Conclusion
Liver transplantation provides significantly better survival rates than palliation for patients with selected advanced HCC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Besides being a potentially curative treatment for early hepatocellular carcinoma (HCC),1,2 liver transplantation (LT) has the added advantage of removing the underlying cirrhosis and thereby minimizing the future risk of de novo HCC. Since the introduction of the Milan criteria,3 a post-LT recurrence rate lower than 15% and a 5-year survival rate higher than 70% have become the acceptable norm.3,4 A number of expanded criteria have since been proposed and shown to achieve comparable outcomes. The most widely recognized are the University of California, San Francisco (UCSF) criteria,5,6 which are associated with a recurrence rate of approximately 10% and a 5-year survival rate of 75% to 80%. Generally, LT is not recommended for patients with advanced HCC1,2,7 due to previous undesirable experience with unrestricted access.8
In most Western centers, palliative therapy remains the only treatment option for patients with advanced HCC. Locoregional therapies such as transarterial chemoembolization (TACE), stereotactic body radiation (SBRT), and systemic therapy (sorafenib) are used for palliation, resulting in a 3-year survival rate lower than 30% at best.9,10
Improvements in understanding HCC biology and its role in predicting outcomes has rekindled interest in LT for advanced HCC.11 Encouragement from the improved outcomes with LT for HCC and dissatisfaction with the poor outcomes with palliative therapy has led to reexamination of the role of LT in advanced HCC.
The Extended Toronto Criteria (ETC) place no restriction on the number HCC lesions or tumor size. Patients with any number and any size of HCC lesions are eligible for LT provided no evidence exists for vascular invasion or extrahepatic disease, no cancer-related constitutional symptoms are observed, and a targeted biopsy of the largest lesion does not show poor differentiation. Findings show that the LT outcomes for patients beyond Milan but within the ETC were inferior compared with those within Milan, but were still acceptable.12 However, no studies have specifically focused on the outcomes for patients with advanced HCC from the time of listing as described in this report. This study aimed to investigate and report on the outcomes of patients listed for LT with advanced HCC (within ETC) who would have received palliative treatment in most Western centers.
Materials and Methods
Study Population
The analysis included patients listed in the Toronto liver transplant program for advanced HCC, patients listed for nonadvanced HCC who had progressed to advanced HCC while on the waiting list, and patients listed for non-HCC indications who had developed advanced HCC while on the waiting list. Advanced HCC was defined using pre-transplantation imaging demonstrating tumors beyond both the Milan and UCSF criteria but within the ETC.
Demographic and clinical data were retrospectively extracted from the prospectively collected electronic transplant database. The study was approved by the Research Ethics Board (15–9989–CE).
Assessment of Change in Tumor Burden
All the patients underwent radiologic imaging every 3 months irrespective of bridging therapy and were independently reviewed by radiologists (S.G.B. and B.S.) blinded to the outcome. Tumor burden was assessed according to the modified Response Evaluation Criteria in Solid Tumors (mRECIST).13 At baseline, HCC lesions were divided into target and non-target lesions based on their longest diameter. Up to five lesions (instead of two lesions as specified by mRECIST), each larger than 1 cm in at least one dimension, were identified as target lesions. In the event of more than five lesions each larger than 1 cm, the largest five lesions were identified as target lesions. All other lesions were identified as non-target lesions.
Change in both target and non-target lesions was considered in the assessment of the overall change in tumor burden. In the event of bridging therapy, only the maximum length of the viable tumor was measured. The overall change in tumor burden was reported according to mRECIST criteria as complete response, partial response, stable disease, or progressive disease (Supplementary Table 1).
Bridging Therapy
Patients without decompensation who were expected to have more than 6 months wait time underwent bridging therapy. In general, percutaneous ablation was recommended for lesions up to 3 cm in size. The use of TACE was recommended for non-ablatable lesions with satisfactory liver function, and SBRT was considered for lesions not amenable to ablation and TACE. Surgical resection was not used as a bridging therapy, but patients with prior resection were included.
The utility and technology of bridging therapy methods evolved during the study period. In brief, these methods include relative obsolescence of percutaneous ethanol injection, use of drug-eluting bead TACE rather than conventional TACE, routine practice of superselective TACE to maximize the impact on tumor and minimize the liver injury, and routine use of SBRT as a bridge to LT (since 2006) for patients not eligible to undergo ablation and TACE.14
Waiting List Dropout
Patients who experienced death or tumor progression beyond the ETC while awaiting transplant were considered dropouts. Those who remained within ETC despite tumor progression were not delisted.
End Points
The primary end point of the study was patient survival, defined from the time of listing and transplantation (for those who underwent LT) to dropout or death. Survivors were censored at the time of their last clinic visit.
Data Analysis and Statistics
Statistical analyses were performed using either GraphPad prism 5 (GraphPad; San Diego, CA, USA) or SPSS for Windows v20 (SPSS, Chicago, IL, USA). For analysis purposes, peak alpha-fetoprotein (AFP) was reclassified as a binary variable of less than 400 or as 400 ng/ml or more, as previously reported.15 Overall change in tumor burden was reclassified as progressive disease or nonprogressive disease (including complete response, partial response, and stable disease). Univariate analysis with the Mann–Whitney U test or one-way analysis of variance (ANOVA) followed by the multivariable logistic regression model was used to determine independent associations with waiting list dropout. Survival and disease recurrence estimates were calculated using univariate Cox proportional hazard models and the multivariable Cox proportional hazard model.
Results
Waiting List Characteristics
Demographics
Between January 1999 and August 2014, 96 listed patients fulfilled the inclusion criteria (Table 1). The median age at listing was 59 years (interquartile range [IQR] 52–63 years), and the majority (n = 83, 86%) were men. Chronic hepatitis C (n = 41, 43%) was the most common etiology of liver disease, followed by chronic hepatitis B, alcohol-related liver disease, and non–alcohol-related steatohepatitis.
Tumor Burden
At baseline, the median total number of HCC lesions (target and non-target lesions) was 5 (IQR 3–8), the median diameter of the largest lesion was 4.4 cm (IQR 2.6–6.3 cm), and the median total diameter of the target lesions was 8.9 cm (IQR 6.4–12.7 cm). Immediately before LT or dropout, the median total number of HCC lesions was 6 (IQR 2–11), the median diameter of the largest lesion was 3 cm (IQR 1.2–5.8 cm), and the median total diameter of the target lesions was 6.3 cm (IQR 1.2–11.4 cm). More than half of the patients (n = 55, 57%) had progressive disease with or without bridging therapy.
Bridging Therapy
Nearly two thirds of the patients (n = 62, 65%) received bridging therapy. The majority (n = 49, 79%) received bridging therapy by a single method. The median number of bridging therapy sessions of any method was 2 (IQR 1–2).
The most common method was TACE (n = 47, 76%), used either alone (n = 38, 61%) or in combination with other methods (n = 9, 15%). Radiofrequency ablation was used for 18 patients (29%), whereas percutaneous ethanol injection therapy and SBRT, either alone or in combination, were used for four patients (6%).
A comparison between the patients who received bridging therapy and those who did not is summarized in Table 2. The distribution for etiology of liver disease differed significantly (p = 0.02) between the two groups. Hepatitis B was common among the patients who received bridging therapy, whereas hepatitis C and alcohol-related liver disease were more common among those who did not. Liver disease severity (sodium-MELD score) was less (p < 0.001) in patients who received bridging therapy. Both peak AFP and tumor burden (total number of HCC lesions, diameter of the largest target lesion, and total diameter of all the target lesions) at listing were similar between the two groups. As expected, bridging therapy led to a significant reduction in tumor burden. Receiving bridging therapy per se did not have an impact on the waiting list outcome. However, tumor progression on the waiting list was evident in a significantly greater proportion of patients who did not receive bridging therapy (n = 25, 74% vs. n = 30, 48%; p = 0.02).
Waiting List Outcome: Transplantation Versus Dropout
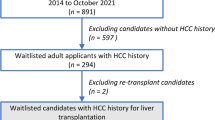
Nearly three fourths of the patients (n = 69, 72%) underwent LT (Fig. 1). The majority received a deceased donor graft (n = 57, 83%). All but one patient dropped out due to tumor progression beyond the ETC—extrahepatic spread (n = 10), macrovascular invasion (n = 9), tumor progression with development of cancer-related symptoms (n = 4), or tumor rupture (n = 3).
In the univariate analysis (Table 3), the tumor burden at listing did not differ between the groups. Female sex (p = 0.03) and peak AFP of 400 ng/ml or higher (p = 0.0007), tumor burden at LT or dropout, and tumor progression on the waiting list (p = 0.03) were associated with dropout.
Multivariate analysis was performed using sex, time on the waiting list, peak AFP higher than 400 ng/ml, and tumor progression on the waiting list as the explanatory variables. Other significant variables in the univariate analysis were not included because they were either measured concurrently with the outcome (tumor burden at LT/dropout) or were reflected in a statistically more powerful way using categorical variables (change in tumor burden). In the multivariate analysis, both tumor progression on the waiting list (hazard ratio [HR] 4.973; range 1.599–15.464; p = 0.006) and peak AFP of 400 ng/ml or higher (HR 4.604; range 1.660–12.768; p = 0.003) were independently associated with dropout.
Post-LT HCC Recurrence
More than a third of the patients who had transplantation (n = 24, 35%) experienced recurrence after a median time of 17 months (IQR 8–25 months) (Fig. 1). The cumulative risk of recurrence was 13% at 1 year, 34% at 3 years, and 41% at 5 years. In the univariate analysis, no tested variables were associated with recurrence (Supplementary Table 2).
Lungs (n = 9) were the most common site of HCC recurrence followed by bone (n = 8), liver (n = 5), and lymph nodes (n = 5), whereas adrenal glands and abdominal wall were infrequent sites of recurrence. All patients with recurrence were considered for curative treatment (surgery or ablation). Seven patients (29%) underwent curative treatment (surgery/ablation), whereas eight patients (33%) underwent noncurative treatment (radiotherapy/palliative surgery/sorafenib treatment), and the remaining patients (n = 9, 38%) received best supportive care.
The median survival period from the time of LT (60 months; IQR 37–70 vs. 27 months; IQR 14–32 months; p = 0.004) and the median survival period from the time of recurrence (32 months; IQR 25–36 months vs. 7 months; IQR 5–13 months; p = 0.004) were significantly better for those who received curative treatment than for the others (Fig. 2).
Kaplan-Meier survival estimates of patients who developed hepatocellular carcinoma recurrence following transplantation from the time of liver transplantation (a), and from the time of diagnosis of recurrence (b). The blue line represents patients who underwent curative treatment and the yellow line represents patients who underwent non-curative treatment
Survival From the Time of Listing and LT
The median follow-up period from the time of listing (all 96 patients) was 34 months (IQR 13–68 months). The actuarial patient survival rate was 76% at 1 year, 56% at 3 years, and 47% at 5 years from listing.
The median follow-up period from the time of LT (69 patients) was 39 months (IQR 24–80) months. The actuarial survival rate was 93% at 1 year, 71% at 3 years, and 66% at 5 years from LT. Survival from the time of LT was not associated with any of the tested variables (Supplementary Table 3).
The number of living donor LTs has remained relatively unchanged despite the gradual increase in the number of deceased donor LTs for advanced HCC. Overall survival from the time of listing (48 vs. 50 months; p = 0.86) and from the time of transplantation (39 vs. 45 months; p = 0.93) did not differ between deceased donor and living donor LTs.
Discussion
This study evaluated the outcomes of patients listed for LT with advanced HCC who would have received palliative treatment at most Western centers. One fourth of the listed patients dropped out. A peak AFP of 400 ng/ml or higher and tumor progression on the waiting list independently predicted dropout. The 5-year post-LT survival rate was 66%.
The patient survival in this series was lower than for those within the Milan criteria,12 but much better than the reported outcomes with palliation, whether taken from the time of transplantation (71% at 3 years; 66% at 5 years) or from the time of listing (56% at 3 years; 47% at 5 years). For selected patients with non-transplantable HCC (i.e., beyond the Milan criteria), TACE barely accomplished a 3-year survival rate of 30% even in experienced centers.9,10 The use of TACE with external beam radiotherapy16 or sorafenib17,18 also failed to demonstrate a satisfactory patient survival. Suboptimal survival (3-year survival of ~20%) also has been reported with yttrium-90 selective internal radiation therapy for patients with intermediate-stage HCC deemed unsuitable for LT.19–21 Thus, in the absence of comparable treatment options, it is not unreasonable to consider transplantation as a preferred alternative. On the other hand, it is necessary to be aware that the costs associated with LT is significantly higher than the costs associated with palliation.22 The estimated average total health care cost of LT for HCC is approximately $211,286 (95% confidence interval [CI] $193,919–$228,654). The cost of palliative TACE is approximately $39,294 (95% CI $20,455–$58,133), and the cost of sorafenib therapy is approximately $45,802 (95% CI $32,143–$59,461) (personal communication, Hla-Hla Thein). However, despite the increased costs, the quality-adjusted life year is likely better with LT than with palliation.
As clinical practice moves toward personalized medicine, it becomes more crucial that all treatment options are presented to the patients and their families. Transplantation seems to be a better choice for selected patients with advanced HCC, but it is limited by the scarce resource of donated organs. Deciding when it is acceptable to allocate deceased donor grafts to patients with advanced HCC will depend on the regional waiting list and organ availability. Expansion of HCC transplantation criteria can be justified only if it upholds fair organ allocation. Introduction of direct antiviral agents against hepatitic C is expected to increase the availability of grafts.23 Thus, expansion of HCC criteria may become feasible.24 However, this may never materialize due to the impending steatohepatitis epidemic.25 Another alternative would be living donor LT. This does not affect the candidacy of others, and the outcomes are comparable to deceased donor LT.26,27; therefore, would be a preferred option for selected patients with advanced HCC. The equivalence of HCC recurrence and patient survival between deceased donor and living donor LT in the current analysis further supports this notion. However, living donor LT must be undertaken in centers with extensive experience due to safety concerns of donors.28
The waiting list dropout in this study was almost 30%. Higher dropout rates also have been reported previously for patients with HCC beyond the traditional criteria.29,30 Both AFP and tumor progression (tumor biologic factors) but not tumor burden (size and numbers) were independently associated with dropout. The predictive role of tumor biology over tumor burden also has been shown in previous studies. In a recent study from the USCF group, an AFP level of 500 ng/ml or higher and rapid tumor progression predicted dropout even for patients with very early stage HCC (single tumor <2 cm).31 In another study, which included patients with HCC within and beyond Milan, the response to therapy was the only predictor of dropout, irrespective of tumor staging.32 Likewise, a high AFP level, but not total tumor volume, was associated with waiting list dropout.33
Posttransplantation HCC recurrence also was found to be higher than in patients within traditional criteria. However, the 5-year post-LT survival rate was 66%. The aggressive treatment of recurrence likely played a role in achieving such acceptable survival rates. The benefits of such aggressive treatment also has been demonstrated previously.34,35 The high recurrence rate may raise concerns about offering LT to this group of patients. However, it is important to recognize that patient survival, not recurrence, is the most clinically relevant and decisive outcome measure of transplantation.36
Against expectations, time on the waiting list did not have an impact on post-LT survival. This contradicts the recent suggestion of implementing a period of observation to identify unfavorable tumor behavior before listing/transplantation.37–39 One likely explanation for this discrepancy is the unaccounted period between the diagnosis of HCC (by the hepatologist/gastroenterologist/family physician) and listing, resulting in a bias toward the null. A substantial number of patients progress beyond the ETC during this period and are turned down before they are formally assessed for LT.
This study had strengths and limitations. The use of radiologic parameters rather than explant parameters in the analysis made the findings reflect prospective decision making in day-to-day practice. On the other hand, being a single center, retrospective study with a relatively limited number of patients should be considered a limitation of this study. Some comparisons may have been affected by the relatively small cohort. In addition, during the 15-year study period, there were significant advancements in bridging therapy and a shift toward a more aggressive management of HCC for patients on the waiting list, which had the potential for an impact on the results. Independent validation of this study’s results is therefore needed.
Efforts to expand the traditional HCC allocation criteria are already underway.24,40 One such example would be the introduction of the “up-to-seven” criteria by Mazzaferro (who initially proposed the Milan criteria) and colleagues.41 Similarly, the emphasis on tumor biology rather than tumor burden in selecting patients for LT is gaining momentum.11,24,40,42 Therefore, it is not unreasonable to speculate that LT will emerge as an acceptable treatment choice for selected patients with advanced HCC in the near future. Certain transplantation programs in Turkey and India seem to follow an unrestricted tumor burden policy, similar to that of the ETC.43 It is crucial that these programs publish their long-term outcome results, which may further encourage other transplantation programs to expand their allocation policy. In addition, the future of LT for advanced HCC relies on the availability of donor organs, and living donor LT likely will play a crucial role in this. Where available, the option of living donor LT should be discussed and offered to all patients with advanced HCC.
In conclusion, LT offers a 5-year survival rate of 66% for selected patients with advanced HCC and is a preferable alternative to palliation. Living donor LT may be the best option for this cohort because it will not have a negative impact others on the waiting list.
References
Bruix J, Sherman M, American Association for the Study of Liver D. Management of hepatocellular carcinoma: an update. Hepatology. 2011;53:1020–2.
European Association for the Study of the L, European Organisation for R, Treatment of C. EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol. 2012;56:908–43.
Mazzaferro V, Regalia E, Doci R, et al. Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis. N Engl J Med. 1996;334:693–9.
Mazzaferro V, Bhoori S, Sposito C, et al. Milan criteria in liver transplantation for hepatocellular carcinoma: an evidence-based analysis of 15 years of experience. Liver Transpl. 2011;17(Suppl 2):S44–57.
Yao FY, Ferrell L, Bass NM, et al. Liver transplantation for hepatocellular carcinoma: expansion of the tumor size limits does not adversely impact survival. Hepatology. 2001;33:1394–403.
Yao FY, Xiao L, Bass NM, et al. Liver transplantation for hepatocellular carcinoma: validation of the UCSF-expanded criteria based on preoperative imaging. Am J Transplant. 2007;7:2587–96.
Bruix J, Reig M, Sherman M. Evidence-based diagnosis, staging, and treatment of patients with hepatocellular carcinoma. Gastroenterology. 2016;150:835–53.
Iwatsuki S, Gordon RD, Shaw BW Jr, Starzl TE. Role of liver transplantation in cancer therapy. Ann Surg. 1985;202:401–7.
Llovet JM, Real MI, Montana X, et al. Arterial embolisation or chemoembolisation versus symptomatic treatment in patients with unresectable hepatocellular carcinoma: a randomised controlled trial. Lancet. 2002;359:1734–9.
Lo CM, Ngan H, Tso WK, et al. Randomized controlled trial of transarterial lipiodol chemoembolization for unresectable hepatocellular carcinoma. Hepatology. 2002;35:1164–71.
Cillo U, Giuliani T, Polacco M, et al. Prediction of hepatocellular carcinoma biological behavior in patient selection for liver transplantation. World J Gastroenterol. 2016;22:232–52.
Sapisochin G, Goldaracena N, Laurence JM, et al. The extended Toronto criteria for liver transplantation in patients with hepatocellular carcinoma: a prospective validation study. Hepatology. 2016;64:2077–88.
Lencioni R, Llovet JM. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin Liver Dis. 2010;30:52–60.
Sandroussi C, Dawson LA, Lee M, et al. Radiotherapy as a bridge to liver transplantation for hepatocellular carcinoma. Transpl Int. 2010;23:299–306.
Toso C, Meeberg G, Hernandez-Alejandro R, et al. Total tumor volume and alpha-fetoprotein for selection of transplant candidates with hepatocellular carcinoma: a prospective validation. Hepatology. 2015;62:158–65.
Yang P, Zeng ZC, Wang BL, et al. The degree of Lipiodol accumulation can be an indicator of successful treatment for unresectable hepatocellular carcinoma (HCC) patients—in the case of transcatheter arterial chemoembolization (TACE) and external beam radiotherapy (EBRT). J Cancer. 2016;7:1413–20.
Zhao Y, Li H, Bai W, et al. Early sorafenib-related adverse events predict therapy response of TACE plus sorafenib: a multicenter clinical study of 606 HCC patients. Int J Cancer. 2016;139:928–37.
Lencioni R, Llovet JM, Han G, et al. Sorafenib or placebo plus TACE with doxorubicin-eluting beads for intermediate stage HCC: the SPACE trial. J Hepatol. 2016;64:1090–8.
Lance C, McLennan G, Obuchowski N, et al. Comparative analysis of the safety and efficacy of transcatheter arterial chemoembolization and yttrium-90 radioembolization in patients with unresectable hepatocellular carcinoma. J Vasc Interv Radiol. 2011;22:1697–705.
Moreno-Luna LE, Yang JD, Sanchez W, et al. Efficacy and safety of transarterial radioembolization versus chemoembolization in patients with hepatocellular carcinoma. Cardiovasc Intervent Radiol. 2013;36:714–23.
El Fouly A, Ertle J, El Dorry A, et al. In intermediate-stage hepatocellular carcinoma: radioembolization with yttrium-90 or chemoembolization? Liver Int. 2015;35:627–35.
Thein HH, Isaranuwatchai W, Campitelli MA, et al. Health care costs associated with hepatocellular carcinoma: a population-based study. Hepatology. 2013;58:1375–84.
Belli LS, Berenguer M, Cortesi PA, et al. Delisting of liver transplant candidates with chronic hepatitis C after viral eradication: a European study. J Hepatol. 2016;65:524–31.
Mazzaferro V. Squaring the circle of selection and allocation in liver transplantation for HCC: an adaptive approach. Hepatology. 2016;63:1707–17.
Loomba R, Sanyal AJ. The global NAFLD epidemic. Nat Rev Gastroenterol Hepatol. 2013;10:686–90.
Olthoff KM, Smith AR, Abecassis M, et al. Defining long-term outcomes with living donor liver transplantation in North America. Ann Surg. 2015;262:465–75, discussion 473–465.
Selzner M, Kashfi A, Cattral MS, et al. Live donor liver transplantation in high MELD score recipients. Ann Surg. 2010;251:153–7.
Rossler F, Sapisochin G, Song G, et al. Defining benchmarks for major liver surgery: a multicenter analysis of 5202 living liver donors. Ann Surg. 2016;264:492–500.
Bittermann T, Niu B, Hoteit MA, Goldberg D. Waitlist priority for hepatocellular carcinoma beyond milan criteria: a potentially appropriate decision without a structured approach. Am J Transplant. 2014;14:79–87.
Schmitt TM, Kumer SC, Shah N, et al. Liver transplantation for T3 lesions has higher waiting list mortality but similar survival compared to T1 and T2 lesions. Ann Hepatol. 2010;9:390–6.
Mehta N, Sarkar M, Dodge JL, et al. Intention to treat outcome of T1 hepatocellular carcinoma with the “wait and not ablate” approach until meeting T2 criteria for liver transplant listing. Liver Transpl. 2016;22:178–87.
Vitale A, D’Amico F, Frigo AC, et al. Response to therapy as a criterion for awarding priority to patients with hepatocellular carcinoma awaiting liver transplantation. Ann Surg Oncol. 2010;17:2290–302.
Merani S, Majno P, Kneteman NM, et al. The impact of waiting list alpha-fetoprotein changes on the outcome of liver transplant for hepatocellular carcinoma. J Hepatol. 2011;55:814–9.
Sapisochin G, Goldaracena N, Astete S, et al. Benefit of treating hepatocellular carcinoma recurrence after liver transplantation and analysis of prognostic factors for survival in a large Euro-American series. Ann Surg Oncol. 2015;22:2286–94.
Bodzin AS, Lunsford KE, Markovic D, et al. Predicting mortality in patients developing recurrent hepatocellular carcinoma after liver transplantation: impact of treatment modality and recurrence characteristics. Ann Surg. 2016.
Yao FY. Liver transplantation for hepatocellular carcinoma: beyond the Milan criteria. Am J Transplant. 2008;8:1982–9.
Toso C, Mentha G, Kneteman NM, Majno P. The place of downstaging for hepatocellular carcinoma. J Hepatol. 2010;52:930–6.
Roberts JP, Venook A, Kerlan R, Yao F. Hepatocellular carcinoma: ablate and wait versus rapid transplantation. Liver Transpl. 2010;16:925–9.
Pomfret EA, Washburn K, Wald C, et al. Report of a national conference on liver allocation in patients with hepatocellular carcinoma in the United States. Liver Transpl. 2010;16:262–78.
Mulligan DC. The ongoing quest to find the appropriate patients to transplant with hepatocellular carcinoma: Milan to San Francisco to Toronto and beyond. Hepatology. 2016;64:1853–55.
Mazzaferro V, Llovet JM, Miceli R, et al. Predicting survival after liver transplantation in patients with hepatocellular carcinoma beyond the Milan criteria: a retrospective, exploratory analysis. Lancet Oncol. 2009;10:35–43.
Lee HW, Suh KS. Liver transplantation for advanced hepatocellular carcinoma. Clin Mol Hepatol. 2016;22:309–18.
Moray G, Karakayali F, Yilmaz U, et al. Expanded criteria for hepatocellular carcinoma and liver transplantation. Transplant Proc. 2007;39:1171–4.
Disclosure
There are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Aravinthan, A.D., Bruni, S.G., Doyle, A.C. et al. Liver Transplantation is a Preferable Alternative to Palliative Therapy for Selected Patients with Advanced Hepatocellular Carcinoma. Ann Surg Oncol 24, 1843–1851 (2017). https://doi.org/10.1245/s10434-017-5789-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-017-5789-3