Abstract
Alternative pre-mRNA splicing is a critical mechanism that generates multiple mRNA from a single gene, thereby increasing the diversity of the proteome. Recent research has highlighted the significance of specific splicing isoforms in cellular processes, particularly in regulating cell numbers. In this review, we examine the current understanding of the role of alternative splicing in controlling cancer cell growth and discuss specific splicing factors and isoforms and their molecular mechanisms in cancer progression. These isoforms have been found to intricately control signaling pathways crucial for cell cycle progression, proliferation, and apoptosis. Furthermore, studies have elucidated the characteristics and functional importance of splicing factors that influence cell numbers. Abnormal expression of oncogenic splicing isoforms and splicing factors, as well as disruptions in splicing caused by genetic mutations, have been implicated in the development and progression of tumors. Collectively, these findings provide valuable insights into the complex interplay between alternative splicing and cell proliferation, thereby suggesting the potential of alternative splicing as a therapeutic target for cancer.
Similar content being viewed by others
Introduction
Most human pre-mRNAs contain two regions: one is the exon, which codes for the mRNA and produces the specific protein, and the other is the intron, which does not code for the protein sequence and which is removed from the pre-mRNA by a sophisticated biological process called RNA splicing. Although the intronic sequences are not pivotal for protein translation, many eukaryotic genes in yeasts to vertebrates possess them. Interestingly, the higher the evolutionary level of the species, the longer and more complicated the intronic sequence [1, 2].
The complexity of intron sequences promotes alternative splicing, in which exons are selectively included in the mRNA or the introns are not excluded. If alternative splicing events alter the combinations of exons that possess protein-coding sequences (CDS), the resulting alteration in protein products may affect their subcellular localization, secretory activity, stability, enzymatic activity, post-translational modification (PTM), or protein interactions [3,4,5]. Even if alternative splicing changes only the untranslated region (UTR) in the mRNA, the translation efficiency or mRNA stability could change between splicing-generated mRNA variants [6, 7]. Since 95% of multi-exon human genes undergo alternative splicing, systemic changes in alternative splicing can regulate most cellular processes including cell growth and survival [8]. Although a wide range of alternative splicing events may occur by chance because of misprocessed RNA splicing, many alternative splicing events are highly conserved among vertebrates, and they play a crucial role in determining cell fates [9]. These events are mostly finely regulated by the expression and activation of splicing factors according to tissue type, developmental stage, and signal transduction [10, 11]. However, genetic mutations in the splicing factor or cis-regulatory RNA element, as well as abnormal expression of the splicing factor, can induce aberrant alternative splicing, which can promote the expression of undesired protein isoforms instead of the appropriate protein isoforms, leading to the progression of human diseases, including cancer [12].
Comprehensive analyses of alternative splicing from patients with cancer have identified the global alteration of splicing pools in cancer tissues [13]. Additionally, recent studies have demonstrated that alteration of alternative splicing in specific genes is potentially associated with oncogenic properties including cell growth and survival [14]. Appropriate cell proliferation monitored through checkpoints is essential for growth, regeneration, and maintenance of tissue functions. Cells can arrest the cell cycle when they sense DNA damage or undergo apoptosis because of excessive DNA damage with unsuccessful DNA repairs [15, 16]. They can exit the cell cycle by differentiating into specific cell types or entering a quiescent stage. However, cancer cells display uncontrolled cell cycle progression and resistance to cell death [17, 18]. These cancer cells exhibit transcriptomic abnormalities that promote cancer cell transformation and proliferation. Aberrant alternative splicing may play a critical role in these transcriptome abnormalities by affecting expression of specific cancer-associated isoforms.
In this review, we describe the current understanding of the role of alternative splicing in controlling cell division and survival by discussing specific splicing factors and isoforms and their molecular mechanisms for regulating cell proliferation. Finally, we provide a novel insight that the proteome diversity generated by alternative splicing is a key to resolving the complexity of cell proliferation mechanisms and to understanding the detailed mechanism underlying human diseases related to cell proliferation.
Regulatory mechanism of alternative splicing
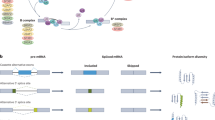
Splicing occurs through a sequential catalytic process involving the spliceosome complexes U1, U2, U4, U5, and U6, in the canonical splicing process [19]. The spliceosome complexes comprise small nuclear ribonucleoproteins (snRNPs) consisting of small nuclear RNA (snRNA) and numerous proteins [20]. During the transcription of multi-exon genes, splicing occurs simultaneously to remove introns from the transcripts via recruitment of spliceosome complexes. The primary criterion for defining introns and exons within the pre-mRNA is the presence of intronic consensus sequences located at the 5′ and 3′ splice sites (ss). These sequences are recognized by the snRNA of the spliceosome via base-pairing with the target pre-mRNA [21, 22] (Fig. 1A). The U1 snRNP binds to the GU motif at the intron boundary of the 5′ss. SF1 and U2AF2 bind to the branch point site (BPS) and polypyrimidine tract (PPT), respectively, and U2AF1 binds to the AG motif at the intron boundary of the 3′ss. Subsequently, SF1 protein is released; U2 snRNP binds to the BPS, and it is stabilized by SF3B1. Next, the U4/U6•U5 tri-snRNP complex is recruited, following which U1 and U4 snRNPs are released to form a catalytically activated spliceosome. Finally, the RNA undergoes conformational rearrangement and is catalyzed in two transesterification steps: one involves 5′ss cleavage to form a lariat intron and the other involves catalysis of exon ligation. Consequently, mRNA is generated, and snRNPs are recycled for additional splicing processes.
Overview of splicing mechanisms and alternative splicing types. (A) Simplified schematic illustration of pre-mRNA splicing. Evolutionarily conserved 5′ splice sites (5′ss; GU) and 3′ splice sites (3′ss; AG) are located at the 5′ and 3′ ends of introns, respectively. Within the upstream intron of the 3′ss, a branchpoint sequence (BPS) and a polypyrimidine tract (PPT) are present. The 5′ss is recognized by the U1 snRNP, the PPT by the U2AF2, and the 3′ss by the U2AF1. The U2 snRNP recognizes the BPS. It then undergoes two successive transesterification steps to generate splicing products (spliced exon and lariat intron). (B) Major types of alternative splicing events. Cells generate different mRNA transcripts by exon skipping of alternative exons, the selection of alternative 5 or 3′ss, retention of introns, selection of the mutually exclusive exons, and selection of alternative promoters or polyadenylation. Exons are represented by colored boxes, introns by horizontal lines, and distinct alternative splicing events by dotted lines
Although splicing occurs according to splice site sequences, several human introns frequently have weak intronic consensus sequences [2, 23]. Moreover, numerous splicing factors are expressed, they bind to intronic or exonic splicing regulatory sequences, and these trans-acting splicing factors contribute to or interfere with the recruitment of spliceosome to the splice site, depending on the specific site to which they bind [24]. Consequently, splicing factors affect the splice site selection of spliceosome, particularly showing greater effects on splice site selection in introns containing weak consensus sequences [25]. Therefore, although the transcripts are expressed from one gene, depending on the expression and activation of splicing factors, mRNA can be expressed as different combinations of exons, producing alternative splicing variants. In particular, in higher mammals, alternative splicing plays a pivotal role in providing complexity to cellular system regulation by diversifying the transcriptome and fine-tuning gene functions [23].
Most alternative splicing events can be classified into seven types according to the pattern of splice site changes (Fig. 1B). Exon skipping is the most predominant event of alternative splicing in humans. Specific exons that are included or excluded in the mRNA are called cassette exons. Alternative 5′ss or 3′ss change the length of the exon. Some introns can be retained in the mRNA. Intron sequences mainly encode nonfunctional protein sequences and involve the termination codon, potentially leading to a reduction of protein function. Mutually exclusive exons represent an alternative splicing pattern in which only one exon is included between consecutive alternative exons. Lastly, alternative promoters or alternative polyadenylation generate different splicing variants.
Comprehensive bioinformatics analyses have revealed that alternative splicing globally and dynamically occurs in most human genes as various patterns of exon selection [9]. To explore the detailed function of genes for further understanding of complex cellular mechanisms, such as cell proliferation and survival, the study of alternative splicing patterns of transcripts and their differential functions of protein isoforms is indispensable, especially in understanding human disease.
Alternative splicing of pathway components related to cancer cell growth
Cell cycle pathway
The mitotic cell cycle is a process in which duplicated chromosomes are separated into two cells to obtain two genetically identical daughter cells (Fig. 2A). Chromosomes are replicated through two stages, interphase and M phase. The cell cycle is controlled by complex mechanisms such as cell cycle checkpoints that monitor the appropriate cell size, complete chromosome replication, and correct cell segregation for timely and accurate cell division [26]. The main mechanism of action of the cell cycle checkpoint is to regulate the activity of cyclin-dependent kinases (CDKs), a family of serine/threonine protein kinases. Cyclin/CDK complexes are formed and activated at various stages of the cell cycle, and they control cell cycle progression through phosphorylation of downstream targets such as the tumor suppressor retinoblastoma (Rb) [27].
Key signaling pathways that affect cancer cell growth and alternative splicing events that regulate these pathways. Alternative splicing is involved in the regulation of significant signaling transduction pathways that affect cancer cell growth. The five major signal transduction pathways that affect cancer cell growth include the cell cycle, MYC, NRF2, PI3K/Akt, and Ras/Raf pathways. Through alternative splicing, genes within these pathways can generate diverse protein isoforms with distinct functions and expression levels. Through alternative splicing, these genes can dynamically modulate the signaling landscape, contributing to the regulation of cancer cell growth. Genes regulating these pathways through alternative splicing are shown in gray boxes, and genes with abnormal splicing due to genetic mutations are shown in pink boxes. Representative alternative splicing events among these genes are depicted. The main domains of these genes are marked with colored boxes, and the domain regions changed according to alternative splicing and the type of alternative splicing are indicated
In keratinocytes, increased eIF2α phosphorylation by UVB enhanced translation of cyclin dependent kinase inhibitor 1 A (CDKN1A, also called p21) transcript variant 4 through a mechanism mediated in part by upstream open reading frames (ORFs) situated in the 5′-leader of CDKN1A mRNA [28]. eIF2α phosphorylation protects cells from stress, such as UVB irradiation, by regulating cell cycle control and determining cell fate through a specific splicing variant of CDKN1A that promotes G1 arrest and subsequent DNA repair. In addition, a mutation that destroys the splice site of CDKN1A has been found to induce the skipping event of exon 2 and, interestingly, cause transcript fusion between CDKN1A and RAB44, which are different genes [29].
Cyclin dependent kinase inhibitor 2 A (CDKN2A), located on human chromosome 9, encodes the p16INK4A and p14ARF proteins. A splice site mutation in CDKN2A, found in a family with melanomas, neurofibromas, and multiple dysplastic nevi, was reported to cause the skipping of exon 2, which encodes more than 50% of the p16INK4A and p14ARF proteins [30]. In addition, splice site variation of CDKN2A was found by sequencing of 167 melanoma-prone families [31]. The AGgt-to-AGtt and AGgt-to-ATgt mutations in CDKN2A disrupted the normal splicing of exon 2. The AGgt-to-CGgt mutation of exon 1 disrupted splicing and affected the function of p16INK4A, confirming that the splice site mutation plays an important role in melanoma susceptibility.
Cyclin E1 encoded by CCNE1 controls the G1/S transition. In the Ccne1 splicing variant IN3 (ORF shifted from exon 2 to exon 3 due to alternative 5′ss selection), exon 4 skipping and exon 5 skipping are associated with retarded proliferation in murine hepatocellular carcinoma [32]. Isoforms with partial loss of exons 3 and 8 and complete loss of exons 4, 5, 6, and 7 are localized in the cytoplasm due to the lack of a nuclear localization signal. This Ccne1 isoform is predominantly expressed in G0 in hepatocytes and forms inactive complexes to sequester CDK2 in the cytoplasm, delaying cell cycle re-entry of hepatocytes after G1 arrest.
Human cyclin D1 (CCND1) splicing occurs at the exon 4/intron 4 boundary. When splicing occurs at this site, the cyclin D1a protein is produced; when splicing fails, the cyclin D1b protein, which contains a part of intron 4 instead of exon 5 and terminates prematurely, is produced. Cyclin D1b splicing is increased by G/A870 polymorphism and SRC-associated in mitosis of 68 kDa (SAM68) [33, 34]. Cyclin D1b is upregulated in various carcinomas, including prostate cancer, and exhibits a higher carcinogenic potential than cyclin D1a because of its stronger nuclear localization [35,36,37]. The cyclin D2 (Ccnd2) splice variant found in the mouse heart contains exon 1 and the longer exon 2, which generate 20 novel residues in the C-terminal [38]. This isoform aggregates in the endoplasmic reticulum (ER), Golgi, and lysosomal compartments and functions as a negative cell cycle regulator by aggregating and sequestering several cell cycle proteins. The 17 kDa truncated cyclin D2 isoform localizes to the cytoplasm and interacts with CDK4; however, the complex is unable to phosphorylate the target pRb [39].
The long isoform of CDK2 with 48 residues inserted owing to partial insertion of intron 5 has approximately half the specific activity compared to the normal form of CDK2 when forming a complex with cyclin A [40]. Poly(C)-binding proteins (PCBPs) bind to C-rich PPT in intron 4 and promote inclusion of CDK2 exon 5 [41]. Exon 5 exclusion of CDK2 dramatically reduces CDK2 protein expression, affecting cell cycle kinetics.
E2F transcription factor 1 (E2F1) is a transcription factor that regulates the S phase transition from G0/G1. Intron 5 retention and exon 6 skipping variants were identified in the central nervous system of rats, and these were terminated early, resulting in C-terminal truncation [42]. E2F transcription factor 3 (E2F3) produces isoforms called E2F3a and E2F3b owing to an alternative promoter [43, 44]. E2F3a is tightly regulated by cell growth and is expressed only at the G1/S boundary, whereas E2F3b is detected throughout the cell cycle and expressed in both quiescent and proliferating cells. Additionally, in quiescent cells, the E2F3b protein binds to Rb, resulting in a predominant E2F-Rb complex [45].
In RB1, the G1/S checkpoint regulator, 27% of coding mutations disrupted splicing [46]. Of the RB1 exon splicing mutations, 58% were blocked primarily at the A complex converting to the B complex, and 33% at the B complex. Mutation-induced RB1 exon loss or intronic sequence exonization induced premature termination, resulting in the restriction of RB1 expression [47, 48]. Inhibition of RB1 expression induced a high expression of p16INK4A. In addition, the RB1 splicing mutation is associated with prognosis and low penetrance in patients with non-small cell lung cancer (NSCLC) or retinoblastoma [47, 49].
The tumor suppressor p53 induces cell cycle arrest at multiple stages, including G/S and G2/M checkpoints, in situations such as DNA damage. The use of an alternative 3′ss located in tumor protein P53 (TP53) intron 6 results in the p53Ψ isoform lacking major parts of the DNA-binding domain, nuclear localization sequence, and tetramerization domain [50]. The p53Ψ isoform is not capable of DNA binding and transactivation, but it induces mesenchymal-like characteristics and improves motility and invasion of normal and malignant cells. TP53, a gene encoding the tumor suppressor protein p53, called “guardian of the genome”, has been reported to have splice site mutations in patients with various cancers, including colorectal cancer [51, 52]. Exon 4–intron 4 junctions mutations identified in pediatric adrenocortical tumors induce erroneous splicing, resulting in protein instability, altered intracellular localization, and loss of function [53]. Exon 6-truncating mutants, similar to the p53Ψ isoform, lack transcriptional activities and the ability to respond to DNA damage, as well as pro-tumorigenic functions that promote cancer cell proliferation, survival, and metastasis [54].
MYC pathway
The MYC oncogene family consists of three members: MYC, MYCN, and MYCL, which encode c-Myc, N-Myc, and L-Myc, respectively (Fig. 2B). Myc is a basic helix-loop-helix and leucine zipper (bHLH-LZ) transcription factor that regulates approximately 15% of the total transcriptome [55, 56]. Thus, Myc proteins mediate various biological processes including cell growth and proliferation, apoptosis, differentiation, cell cycle, and metabolism [57]. Myc promotes transcription by forming a heterodimer with Myc-associated factor X (MAX) through the bHLH-LZ domain required for DNA-protein interaction. Unlike Myc, which forms a heterodimer only with MAX, MAX forms a homodimer or binds to the MXD, MGA, and MNT proteins [58]. This process induces an antagonistic effect on the Myc family by sequestering MAX from Myc.
MAX with exon 2 inclusion was considerably more effective at binding homodimeric DNA than exon 2 skipping of MAX [59]. In addition, this isoform induced a decrease in Myc expression, slowed growth, and accelerated apoptosis during growth factor deprivation. In another MAX isoform, the basic region, helix 1, and the loop of the helix-loop-helix region were deleted [60]. Since this MAX isoform cannot bind to E-box Myc site DNA because the basic region does not exist, it appears to function as a dominant negative regulator. The MAX protein with a C-terminus truncated by intron 4 retention was shown to retain its ability to bind to the CACGTG motif in complex with c-Myc; however, it was located in the cytoplasm [61]. In addition to the wild type of MAX, hypoxia induced intron 4 retention and alternative splicing including cassette exon located within intron 4 [62]. The isoforms produced by intron 4 retention are highly destabilized by 36 isoform-specific amino acids, which destabilize heterologous proteins. The variant containing the cassette exon located within intron 4 is degraded by nonsense-mediated mRNA decay (NMD), and both isoforms play a role in downregulating the wild-type MAX isoform.
MAX dimerization protein 3 (MXD3) has a variant that uses exon 6 as the last exon and a variant that uses exon 7 instead of exon 6 as the last exon [63]. These two splice variants are most likely a result of alternative polyadenylation. According to an analysis of The Cancer Genome Atlas (TCGA) data, exon 7 mRNA was expressed at higher levels in normal cells than in cancer cells, whereas exon 6 mRNA was expressed at higher levels in cancer cells. The exon 7 inclusion isoform containing a considerably longer 3′UTR has a greater reduction in protein expression compared to the exon 6 inclusion isoform. In addition, the exon 7 inclusion isoform undergoes phosphorylation, and it is localized throughout the nucleus; in contrast, the exon 6 inclusion isoform does not appear to be phosphorylated, and is mainly confined to the nuclear foci.
NRF2 pathway
The NRF2 pathway regulates transcriptional responses of genes important for oxidative and electrophilic stress responses (Fig. 2C) [64,65,66]. Kelch-like ECH-associated protein 1 (Keap1), a substrate adaptor for a cullin-3 (Cul3)-based ubiquitin ligase, inhibits the transcriptional activity of nuclear factor-erythroid factor 2-related factor 2 (Nrf2) by promoting ubiquitination and proteasomal degradation of the transcription factor Nrf2 under basal conditions [67,68,69,70]. Thiols of Keap1 cysteines are modified by oxidants, electrophiles, and Nrf2 activators, and Nrf2 is dissociated from Keap1 [65, 71]. The dissociated Nrf2 translocates to the nucleus without ubiquitination and proteasome degradation, forming a heterodimer with small musculoaponeurotic fibrosarcoma oncogene homologue (sMAF) [72, 73]. The Nrf2/sMaf heterodimer binds to the antioxidant response element (ARE) and induces the transcription of a battery of antioxidant and detoxification genes [74,75,76].
Exon 2 or exon 2 + 3 skipping of NFE2 like BZIP transcription factor 2 (NFE2L2), the gene coding for Nrf2, was found in some NSCLCs and patients with head and neck squamous carcinoma (HNSC) [77]. These abnormal transcript variants lack the Keap1 interaction domain, resulting in loss of interaction with Keap1. Thus, Nrf2 is stabilized, resulting in an Nrf2 transcriptional response and Nrf2 pathway dependence. NFE2L2 can be spliced using a second alternative promoter (P2) that exists downstream. Because these P2 transcripts start translation using a different AUG than those that use the upstream promoter (P1), they miss the part of exon 1 that encodes the 16 amino acids and thus have a shorter N-terminus. The ΔN-Nrf2, a protein of the NFE2L2-P2 transcript, is more stable and abundant in cells in the absence of stress owing to its impaired binding to Keap1 [78]. Tert-butylhydroquinone (tBHQ) induces electrophilic stress, and the full-length protein isoform of the NFE2L1-P1 transcript was strongly detected in the nucleus, whereas ΔN-Nrf2 translocation from the cytoplasm to the nucleus was not observed. Further studies are needed to determine the function of ΔN-Nrf2 in the cytoplasm, in addition to its transcriptional activity, and whether this function is related to carcinogenesis.
Keap1 splice variants were found in human highly-metastatic hepatoma (MHCC97H) cells and other cell lines. This Keap1 splice variant (Keap1ΔC) lacks exons 4 and 5, resulting in the loss of 180 amino acid residues [79]. Since the missing amino acid residues are located in most of the double glycine-repeat (DGR) domain and the adjacent C-terminal region essential for interaction with Nrf2, these Keap1 isoforms retain no or little ability to inhibit Nrf2. Therefore, the isoform in which exons 4 and 5 are skipped acts as a dominant-negative competitor of intact Keap1 because of its antagonist effect on Keap1-mediated turnover of Nrf2.
PI3K/Akt pathway
The PI3K/Akt pathway is involved in several cellular physiological processes such as cell cycle, growth, proliferation, survival, and autophagy (Fig. 2D). PI3K activation is stimulated by various oncogenes and growth factor receptors, resulting in the conversion of phosphatidylinositol (3,4)-bisphosphate (PIP2) to phosphatidylinositol (3,4,5)-trisphosphate (PIP3) [80]. PIP3 serves as a plasma membrane docking site for the recruitment and activation of several pleckstrin homology (PH) domain-containing proteins such as AKT, SGK, and PDK1 [81]. Phosphorylation of Akt Thr308 by PDK1 at the membrane leads to partial activation of Akt, and Ser473 phosphorylation by a complex involving mTOR/Rictor (TORC2) results in full activation of this enzyme. Activated Akt phosphorylates downstream effectors, including GSK3β, FoxO, MDM2, and BAD, in the cytoplasm and nucleus, triggering various biological processes such as cell cycle, apoptosis, and glucose metabolism. Thr308 and Ser473 of Akt are dephosphorylated by PP2A and PHLPP1/2, respectively, and PTEN dephosphorylates PIP3 to inhibit Akt activity [82,83,84]. Akt causes TSC2 phosphorylation and inactivation and induces RHEB to activate the mTOR/Raptor (TORC1) complex, which affects mRNA translation, nucleotide and lipid synthesis, cell growth, and autophagy.
PI3K is a heterodimeric enzyme composed of a p110 catalytic subunit and a regulatory subunit. The phosphoinositide-3-kinase regulatory subunit 1 (PIK3R1) encodes the PI3K regulatory subunits p85α, p55α, and p50α, with the same C-terminal but different N-terminals as a result of alternative splicing. The p85α protein with the longest N-terminal region can form a homodimer through SH3 domain-PR1 (proline-rich motif 1) and BH-BH domain interactions [85,86,87]. Additionally, p85α binds to PTEN through the N-terminal SH3-BH region, which is absent in p55α and p50α, and enhances enzymatic activity. Through this process, p85α plays a role in maintaining the balance of PI3K signaling by regulating not only p110-PI3K but also PTEN-PI3K. Stat3 directly binds to the p55α and p50α promoters in vivo to induce their expression, and the overexpression of p55α or p50α reduced the level of activated Akt [88]. PIK3R1 splice acceptor and donor sites mutations in exon 11 induce exon 11 exclusion, resulting in a protein lacking a portion (Δ434_475) of the iSH2 domain [89, 90]. The protein thus produced has been shown to interfere with the regulation of p110δ, resulting in increased p110δ signaling and triggering activated phosphoinositide 3-kinase δ syndrome (APDS) type 2, which causes primary antibody deficiency, senescent T cells, developmental abnormalities within the T lymphocyte compartment, and immune dysregulation [91,92,93,94,95].
The isoform generated by partial insertion of intron 8 of PTEN lacks part of the C2 domain, C-tail, and PDZ-binding domains [96]. Consequently, it induces reduced cell migration, adhesion, and enhanced apoptosis, which, similar to the full-length isoform, acts as a tumor suppressor. Additionally, studies have reported variants containing segments (3a, 3b, 3c; 5a, 5b, 5c) of different sizes of intron 3 or intron 5 and variants in which a part of exon 5 or all of exon 6 were excluded [97, 98]. The PTEN variant was regulated by p53 and differentially expressed in heritable breast cancer syndrome, sporadic breast cancers, and Cowden syndrome compared with the normal tissues. The PTEN 5a isoform functions similar to full-length PTEN by reducing cyclin D1 promoter activity and Akt phosphorylation; in contrast, PTEN 5b and 5c appear to exert the opposite function by decreasing cyclin D1 promoter activity. Mutations in the splice junction or intron region of the PTEN gene have been identified in patients with Cowden syndrome and Bannayan Zonana syndrome, an autosomal dominant genetic disorder characterized by hamartomas [99, 100]. These mutations caused either out-of-frame skipping of an entire exon or activation of cryptic splice sites, resulting in partial intronic sequence inclusion, premature transcript termination, or polyadenylation within exon 8. Samples with splicing changes due to these mutations exhibited lower PTEN protein expression and higher Akt phosphorylation compared with samples without splicing changes even with mutations; however, p-ERK1/2 did not increase. These findings suggest that PTEN variants may contribute to the pathogenesis of various diseases and may serve as diagnostic markers.
Ras/Raf pathway
Consisting of the Ras-Raf-MEK-ERK signaling cascade, the mitogen-activated protein kinase (MAPK) pathway is an essential cellular network for inter- and intra-cell communication that transmits, amplifies, and integrates signals from various stimuli, such as growth factors, hormones, tumor-promoting substances, and differentiation factors (Fig. 2E). This pathway activates transcription factors and regulates genes that control cell proliferation, differentiation, angiogenesis, inflammation, development, apoptosis, integrin signaling, and migration. Ras small GTPase, the first component of MAPK signaling, contains three genes, HRAS, NRAS, and KRAS, that encode four RAS proteins (HRas, NRas, KRas4A, and KRas4B) [101]. The Raf family comprises three genes (ARAF, BRAF, and RAF1), and the MEK family, five genes (MEK1, MEK2, MEK3, MEK4, and MEK5). When the ligand binds to the tyrosine kinase receptor, the guanine exchange factor is recruited and activated, leading to the exchange of GDP with GTP in Ras. Activated Ras induces Raf activation to form Ras homo- or heterodimer. Active RAF dimers recruit MEK and subsequently activate ERK. Activated ERK1/2 alters gene expression in cells by phosphorylating multiple substrates and regulating various transcription factors. Conversely, when Ras becomes inactive GDP-bound state by GTPase activating protein, downstream signaling is turned off.
HRAS exon 2 has a relatively weak 3′ss; therefore, inclusion and exclusion are regulated by the balance of positive and negative splicing regulatory factors such as SRSF2 and hnRNPF/H [102]. Therefore, exon 2 mutation identified in attenuated patients with Costello syndrome have been shown to simultaneously interfere with exonic splicing enhancer function and exonic splicing silencer generation, causing exon 2 skipping. These splicing abnormality abrogated the production of HRAS protein and inhibited cancer cell proliferation. Since HRAS exon 2 inclusion may also affect the onset of Costello syndrome and carcinogenic potential, splice switching oligonucleotides that induce exon 2 exclusion can serve as a suitable strategy for therapy.
KRAS generates K-Ras4A and K-Ras4B according to the use of the alternative fourth exon. Exon 4 encodes HVRs, a membrane binding and targeting motif, and K-Ras4A and 4B are differentially expressed in mouse development and adult tissues [103, 104]. K-Ras4A is palmitoylated, whereas K-Ras4B lacks a palmitoylation site [103,104,105]. Thus, in addition to the CAAX motif, K-Ras4A has a dual membrane-targeting motif, as a site of palmitoylation and bipartite polybasic region exists at its C-terminus. Therefore, K-Ras4A plays an important role in K-Ras-induced tumors, and the mechanism by which K-Ras4A accesses the plasma membrane differs from that of K-Ras4B [106]. Additionally, unlike K-Ras4B, K-Ras4A does not bind to the cytosolic chaperone δ-subunit of cGMP phosphodiesterase type 6 (PDE6δ); therefore, a significant difference has been observed in their subcellular trafficking. K-Ras4A has been shown to respond to hypoxia and K-Ras4B to ER stress; KRAS4A splicing is controlled by the DCAF15/RBM39 pathway [107]. Cells with a low KRAS4A/KRAS4B ratio have shown higher sensitivity to cancer treatment drugs and an association with high KRAS signaling and a poor patient outcome, suggesting that they could serve as biomarkers of sensitivity to existing cancer treatments [107, 108].
Five splicing isoforms have been reported for NRAS: isoform 1; isoform 2 containing exon 3b; isoform 3 lacking exon 3; and isoform 4 lacking exon 3 and 4 simultaneously; and isoform 5, in which the first 17 codons of exon 2 and 3 codons at the end of exon 5 are fused [109]. Isoforms 1, 2, and 4 are located only in the cytoplasm, whereas isoforms 3 and 5 are located both in the nucleus and in the cytoplasm. Each isoform has different binding affinities for downstream targets, differentially regulating the RAS signaling pathway. In addition, the different protein expression levels of each isoform are different; isoform 5 has shown almost 1000-fold lower expression than isoform 1. However, forced expression of isoform 5, which consists of 20 amino acids, has been reported to increase cell proliferation and transformation by activation of the NRAS target. The splicing isoform of A-Raf, called DA-Raf1, contains a Ras-binding domain, and it can bind to both Ras and M-Ras; however, it lacks a kinase domain and acts as a dominant-negative antagonist by interfering with the ERK pathway [110, 111].
ERK1 exon 4 encodes a phosphorylation site for ERK1 kinase. SMNDC1, which is highly expressed in patients with pancreatic and ovarian cancers and associated with poor patient outcomes, has been reported to induce ERK1 exon 4 inclusion [112]. Forced exclusion of exon 4 using antisense oligonucleotides has been shown to significantly reduce oncogenic ERK1, survival, and pro-apoptotic signaling and suppress target gene expression and tumor cell growth.
Splicing factors that affect cancer progression
RBM4
RNA binding motif protein 4 (RBM4), an RNA-binding protein that shuttles between the nucleus and cytoplasm, serves as a splicing factor that modulates alternative splicing by binding to the RNA recognition motifs (RRMs) GTAACG or CGGCGG, functioning as a general splicing inhibitor (Fig. 3A) [113, 114]. Following mRNA-sequencing analysis in RBM4-expressing H157 cells, alternative splicing events regulated by RBM4 were investigated using gene ontology. This analysis revealed that RBM4 target genes were associated with crucial cellular processes including cell proliferation, cell cycle regulation, apoptosis, migration, and tumorigenesis [115]. RBM4 demonstrated the ability to inhibit proliferation and migration in various cancer cell lines and effectively hindered cancer progression in tumor xenograft models [115,116,117]. Moreover, RBM4 influenced the utilization of the 5′ss of Bcl-x, an apoptosis regulator, inducing a shift from the anti-apoptotic isoform (Bcl-xL) to the pro-apoptotic isoform (Bcl-xS), thereby promoting apoptosis and impeding tumor progression [118, 119]. In addition, RBM4 downregulated the protein level of serine/arginine-rich splicing factor 1 (SRSF1), a splicing factor that functions as a proto-oncogene, and inhibited mTOR activation [120,121,122]. In gastric cancer cell lines, RBM4 has been shown to impede cancer progression by suppressing the expression of MAPK-dependent signaling pathway proteins [117]. Another study provided evidence that RBM4 exerted control over cell proliferation and mediated inflammatory responses by regulating the alternative splicing of transcription factors and co-activators [116]. Consistent with its role as a tumor suppressor, RBM4 expression has been found to be decreased in patients with NSCLC, breast cancer, pancreatic cancer, and gastric cancer [123]. In addition, patients with lung, breast, ovarian, and gastric cancers presenting higher expression of RBM4 exhibited higher survival rates, suggesting that RBM4 could serve be a target for human cancer treatment.
Key splicing factors that modulate cancer progression. Splicing factors play a crucial role in influencing cancer progression by modulating the process of alternative splicing. Several key splicing factors have been reported to significantly affect cancer progression. These factors include RBM4, NOVA, SF3B1, SF3B4, SRSF3 and PTB. Their specific roles involve regulating alternative splicing events in target gene pre-mRNAs that are associated with cancer progression. The intricate involvement of these splicing factors in alternative splicing contributes to the dynamic control of cellular processes related to cancer progression. Intracellular processes that are enhanced (red up arrows) or attenuated (blue down arrows) by these splicing factors are indicated, and the target genes involved are indicated
NOVA1
Neuro-oncological ventral antigen 1 (NOVA1) is a well-known brain-specific splicing factor that plays a crucial role in alternative splicing regulation in neurons by binding to the YCAY sequence on target pre-mRNAs (Fig. 3B) [124,125,126,127,128,129,130]. In the context of gastric cancer, increased levels of miR-146b-5p in the surrounding tissues following gastrectomy have been associated with tumor recurrence and poor survival rates. Notably, miR-146b-5p inhibits NOVA1 expression in immune cells and stromal spindle cells within the remnant microenvironment [131]. Decreased NOVA1 levels in T cells within gastric cancer tissues are correlated with a decrease in FOXP3-positive regulatory T cells and are indicative of poor patient prognosis. These findings suggest that NOVA1 may serve as a potential biomarker for predicting the prognosis of gastric cancer patients and the presence of occult residual disease in remnant tissues post-gastrectomy.
Further studies by the same group found that attenuation of NOVA1 expression in T cells, tumor cells, and stromal spindle cells reduced FOXP3 + Treg density. In contrast, the infiltration of CD68 + macrophages and CD163 + M2 macrophages was increased, and NOVA1 expression was found to be associated with changes in immune cell composition [132]. Frequent NOVA1 inhibition was observed in the gastric cancer microenvironment, and decreased NOVA1 expression in tumor cells was strongly associated with tumor progression and poor prognosis. However, in contrast to these findings, NOVA1 mRNA has been reported to be significantly elevated in gastric cancer compared to that in non-tumor tissues, and high NOVA1 expression is associated with poorer prognosis [133]. In gastric cancer, miR-339 expression was downregulated, and mRNA and protein levels of NOVA1 were suppressed in miR-339-overexpressing cells. Overexpression of miR-339 inhibited gastric cancer cell proliferation, cell cycle progression, and invasion, and overexpression of NOVA1 impaired the inhibitory effect of miR-339 on gastric cancer cells. Another negative regulator of NOVA1 is miR-181b-5p, and its overexpression has been shown to suppress proliferation, migration, and invasion while promoting apoptosis in astrocytoma cells [134]. Similarly, downregulation of NOVA1 resulted in reduced cell growth, inhibited migration and invasion, and promoted apoptosis in U87 cells. Notably, patients with astrocytoma presenting high levels of NOVA1 expression exhibited worse survival outcomes compared with those with low NOVA1 expression levels.
Intratumoral Nova1 has been reported to be strongly correlated with hepatocellular carcinoma (HCC) poor survival and increased early recurrence [135]. HCC cell lines overexpressing Nova1 promoted cell proliferation, invasion, and migration. Nova1 plays a crucial role in promoting the inclusion of exon 9 in the inhibitory neurotransmitter receptor subunits GABAARγ2 [136,137,138]. Moreover, it interacts with the GABAARγ2 protein and demonstrates an inverse relationship with the expression levels of GABAARγ2 and GABA. Additionally, the upregulation of Nova1 expression promoted the growth of subcutaneous HCC in nude mice, indicating a potential oncogenic function for Nova1. Compared with normal lung tissue, NSCLC tissue showed significantly elevated NOVA1 expression, which correlated with indicators of poor differentiation, TNM stage, T stage, and lymph node metastasis [139]. Additionally, patients with NSCLC exhibiting high NOVA1 expression experienced shorter survival compared to those with low expression. NOVA1 facilitated the proliferative and invasive capacities of NSCLC cells by regulating the activation of the Wnt/β-catenin signaling pathway. Moreover, NOVA1 expression promoted the inclusion of exons 7 and 8 of human telomerase reverse transcriptase (hTERT), generating enzymatically active telomerase and influencing telomere length [140]. Notably, NOVA1 knockdown significantly reduced tumor growth in a xenograft model.
Additionally, NOVA1 has been associated with an unfavorable prognosis in patients with colorectal cancer (CRC) [141]. It upregulates MMP-2, MMP-7, and MMP-9 by regulating JAK2/STAT3 signaling through binding to and stabilizing IL6 mRNA. Consequently, NOVA1 has been identified as a novel regulator that influences the proliferation and metastasis of CRC cells. NOVA1 has been reported to exhibit a tumor-suppressive effect depending on the specific environment; however, studies conducted till date collectively indicate that NOVA1 appears to have a dominant tumor-promoting effect affecting proliferation, invasion, migration, and telomerase activity.
SF3B1
The SF3B complex regulates splicing of pre-mRNA by binding to the U2 snRNP and recognizing BPS [142]. The SF3B complex consists of seven proteins (SF3B1–7) with a molecular weight of 10 ~ 155 kDa. SF3B components, including SF3B1, have been implicated in cancer and various genetic disorders. Splicing factor 3B subunit 1 (SF3B1) is the largest protein within the SF3B complex, and it contains a HEAT [Huntingtin, elongation factor 3, subunit A of protein phosphatase 2 A, phosphatidylinositol 3-kinase (PI3K) target of rapamycin 1] domain consisting of 22 tandem repeats (Fig. 3C) [143]. The HEAT domain serves as the central region for RNA and protein binding in the SF3B complex. In human diseases, most SF3B1 mutations are localized in the HEAT 4–12 region. SF3B1 mutation has been detected in 30% of patients with myelodysplastic syndrome (MDS) and in 80% of patients presenting MDS subtype with ringed sideroblasts (RARS) [144,145,146,147]. Additionally, mutations have been detected in 20% of patients with MDS/myeloproliferative neoplasms and 15% of patients with chronic lymphocytic leukemia (CLL) [146, 148,149,150]. Furthermore, mutations in SF3B1 have been reported at low frequencies in patients with acute myeloid leukemia, breast cancer, prolactinomas, uveal melanoma, leptomeningeal melanoma, blue nevus-like cutaneous melanoma, pancreatic ductal adenocarcinoma, and prostate cancer [151,152,153,154,155,156,157,158,159].
Although some reports have suggested no significant effect, SF3B1 mutations in patients with MDS have been associated with a favorable prognosis and long survival rates [146, 160,161,162]. However, in most diseases except MDS, but especially cancer, mutations in SF3B1 have been associated with poor prognosis and survival [163]. Furthermore, SF3B1 is overexpressed in glioblastoma, hepatocellular carcinoma, prostate cancer, and endometrial cancer [164,165,166,167]. Similar to its mutations, SF3B1 overexpression has been directly correlated with adverse patient prognosis, lower survival rates, and drug resistance. Dysregulation caused by SF3B1 mutations or changes in expression modulates oncogenic splicing variants such as BCL2L1-xL, KLF6-SV1, AR-v7 (androgen receptor variant 7), and In1-ghrelin [163,164,165,166, 168, 169]. The blockade or silencing of SF3B1 has been demonstrated to regulate the expression levels of essential components involved in mRNA homeostasis, including spliceosome, splicing factors, exon-junction complex (EJC) and SMG-1Upf1–eRF1–eRF3 (SURF) components, and NMD factors [164, 166]. Furthermore, it has been shown to modulate the AKT/mTOR/ß-catenin, JNK, PDK1, GSK3b, ERK, and AMPK signaling pathways [164,165,166]. Additionally, SF3B1 inhibition has been found to suppress proliferation, migration, apoptosis, and the formation of tumor spheres and colonies, as well as angiogenesis [164,165,166].
Capitalizing on the oncogenic properties of SF3B1 and its potential for suppressing tumor growth through blockade or silencing, several drugs have been developed to target SF3B1. These drugs include pladienolide B, spliceostatins, herboxidiene, sudemycins, and H3B-8800 [170,171,172,173,174]. Notably, H3B-800 entered phase I clinical trials in 2016, further affirming SF3B1 as a promising biomarker and a target for pharmacological treatment [175, 176].
SF3B4
Splicing factor 3b subunit 4 (SF3B4) is a major subunit of the SF3B complex, consisting of two N-terminal RRMs and a C-terminal proline-rich (PR) domain (Fig. 3D) [177]. In addition to its role in pre-mRNA splicing, SF3B4 is implicated in cell signaling, transcription, and translation processes [178].
According to a TCGA analysis, only SF3B4 has shown high expression in the SF3B complex in cervical squamous cell carcinoma and endocervical adenocarcinoma [179]. SF3B4 has been reported to enhance the proliferation and invasion of cervical cancer cells, thereby promoting their malignant behavior. RNA-seq analysis conducted in SF3B4-knockdown HeLa cells revealed that differentially expressed genes (DEGs) were enriched in cellular processes such as regulation of cell proliferation, transcription, apoptotic process, and cell adhesion. Among the downstream targets of SF3B4, the gene exhibiting the most significant change in mRNA expression was sperm-associated antigen 5 (SPAG5). SF3B4 knockdown reduced SPAG5 expression by inducing the retention of SPAG5 intron 21, subsequently causing premature termination of the transcript. SPAG5 is a mitotic spindle-binding protein involved in regulating mitosis [180]. It has been reported to promote the proliferation and progression of not only cervical cancer but also hepatocellular cancer and breast cancer by modulating the cell cycle [179, 181,182,183,184]. Notably, the presence of SPAG5 intron retention transcripts correlated with extended survival times in patients. Collectively, these findings underscore the oncogenic role of SF3B4 in cervical cancer by virtue of its regulatory influence on SPAG5 splicing.
SF3B4 has been reported to be up-regulated in hepatocellular carcinoma [185, 186]. High SF3B4 expression has been found to be associated with intrahepatic metastasis and poor prognosis [187]. Overexpression of SF3B4 has been shown to trigger the SF3b complex, which induced exon skipping of the tumor suppressor Kruppel-like factor 4 (KLF4), resulting in non-functional KLF4 transcripts [188]. Consequently, cyclin-dependent kinase inhibitor 1B (CDKN1B; p27Kip1) became transcriptionally inactive, disrupting cell cycle regulation, while the activation of the Snail family transcriptional repressor 2 (SNAI2) gene promoted epithelial–mesenchymal transition (EMT). These events contributed to the malignant transformation and proliferation of liver cells. Furthermore, SF3B4 outperformed the existing diagnostic markers for hepatocellular carcinoma, namely glypican 3 (GPC3), glutamine synthetase (GS), and heat-shock protein 70 (HSP70) and demonstrated its potential as a reliable diagnostic marker for early-stage hepatocellular carcinoma.
Mutations in SF3B4 mostly lead to reduced expression due to frameshifts, which cause acrofacial dysostosis, Nager syndrome, and Rodriguez syndrome [189, 190]. The SF3B4 mutation affects the regulation of gene expression and abnormal splicing of crucial genes involved in skeletal development within growth plate chondrocytes [191, 192]. Consequently, this disruption ultimately manifests as defects in craniofacial and limb development, which are observed in acrofacial dysostosis.
SF3B4 has exhibited significant upregulation in ovarian cancer and a correlation with unfavorable patient prognosis [193]. The expression of SF3B4 has been found to be negatively regulated by miR-509-3p. RAD52, involved in DNA damage repair, assumes an oncogenic role in various tumors [194,195,196]. Loss of SF3B4 has been reported to reduce RAD52 expression by inducing retention of intron 8 of RAD52 and generating premature termination codons. SF3B4 has been shown to act as an oncogene by modulating the alternative splicing of RAD52, thereby facilitating the proliferation, migration, and invasion of ovarian cancer cells. Additionally, SF3B4 has been reported to function as an oncogene in esophageal squamous cell carcinoma (ESCC) [197]. Conversely, in pancreatic cancer, the protein level of SF3B4 has shown a reduction compared with that in adjacent symptomatic tissue, and this diminished expression of SF3B4 has been found to facilitate the proliferation and migration of pancreatic cancer cells, indicating an inhibitory role for SF3B4 in pancreatic cancer [198]. These findings suggest that SF3B4 may have different biological functions, depending on the tumor type.
SRSF3
The serine/arginine-rich splicing factor (SRSF) protein family comprises RNA-binding proteins that regulate various RNA biological processing such as mRNA transport and polyadenylation, as well as constitutive and alternative splicing [199,200,201,202,203,204,205,206]. Currently, 12 members (SRSF1–12) of the SRSF family have been identified in humans, with SRSF3 being the smallest member (Fig. 3E). SRSF3 has been observed to exhibit high expression levels in a wide range of tumors, including breast cancer, cervical cancer, colorectal cancer, gastric cancer, glioblastoma, head and neck squamous cell carcinoma, hepatocellular carcinoma, non-small cell lung cancer, oral squamous cell carcinoma, ovarian cancer, and retinoblastoma [203, 207,208,209,210,211,212,213,214,215,216,217,218,219,220,221,222,223,224,225].
SRSF3, a potential exonic splicing enhancer that is upregulated in glioblastoma, binds to the CA(G/C/A)CC(C/A) motif and alters more than 1,000 alternative splicing events [215]. Particularly, the knockout of SRSF3 leads to the exclusion of exon 7 in the ETS variant 1 (ETV1) gene and the replacement of the terminal exon 9 in the nudE neurodevelopment protein 1 (NDE1) gene with a mutually exclusive exon 9′. The ETV1 isoform with exon 7 inclusion and the NDE1 isoform with terminal exon 9 have been confirmed to be important for mitosis and cell proliferation of tumor cells, significantly increasing oncogenic activity. In addition, SRSF3 has been shown to inhibit PDCD4 protein expression by participating in alternative splicing, cytoplasmic export and translation of PDCD4, a tumor suppressor gene involved in antiproliferation, apoptosis, and antimetastasis [203, 226, 227].
MDM4 is an oncogene that suppresses the p53 tumor suppressor [228, 229]. SRSF3 is necessary for the inclusion of exon 6 (exon 7 in mice) in the human MDM4 gene [230]. When this exon is excluded, it produces an unstable transcript containing a premature termination codon, which is subject to NMD. Consequently, this splicing event affects MDM4 protein levels. The in vitro and in vivo induction of MDM4 exon 6 skipping using an antisense oligonucleotide have been reported to inhibit MDM4 protein abundance and melanoma growth and to increase sensitivity to MAPK-targeting therapeutics.
TAR DNA-binding protein (TDP43) is overexpressed in triple-negative breast cancer (TNBC) and is a major regulator of unique alternative splicing in TNBC [209]. SRSF3 interacts with these TDP43 to control specific splicing events, including that of PAR3 and NUMB. SRSF3/TDP43 knockdown has been shown to inhibit cell proliferation by inducing NUMB exon 12 exclusion and inhibition of cell migration and invasion by inducing PAR3 exon 12 inclusion. SRSF3 knockdown has been shown to induce G1 arrest and apoptosis by promoting downregulation of G1/S transition-related genes, BCL2 protein reduction, and homeodomain-interacting protein kinase-2 (HIPK2) exon 8 exclusion [214].
The oncogene Erb-B2 receptor tyrosine kinase 2 (ERBB2) is overexpressed in 20–30% of invasive breast cancer and is associated with poor prognosis [231]. In breast cancer cells, SRSF3, along with hnRNP H1, has been identified as a regulator responsible for controlling the production of distinct splice variants of ERBB2 with different functionalities [232]. In particular, the knockdown of SRSF3 converted the oncogenic variant (exon 16 skipping) to a cell proliferation suppressive variant (premature stop codon generated by intron 15 inclusion). Additionally, SRSF3 binds to exon 18 of the interleukin enhancer-binding factor 3 (ILF3), leading to the production of an ILF3 isoform that facilitates cell growth [233]. Furthermore, SRSF3 has been demonstrated to be strongly related to the PI3K-AKT signaling pathway [201]. Cumulative evidence from various studies supports the role of SRSF3 in tumorigenesis, proliferation, and anti-apoptosis. Consequently, targeting the downregulation of SRSF3 holds potential as a therapeutic strategy for anticancer treatment.
PTB
Polypyrimidine tract binding protein (PTB) shuttles between the nucleus and cytoplasm, and it is involved in various mRNA metabolic pathways, such as polyadenylation, mRNA stability, and initiation of translation, as well as regulation of pre-mRNA splicing (Fig. 3F) [234, 235]. PTB has a high affinity for binding to CU rich sequences including UCUU and CUCUCU [236, 237].
PTB is found to be overexpressed in epithelial ovarian tumors, glioma, and various cancer cell lines [238,239,240]. Knockdown of PTB has been shown to decrease cell proliferation, anchorage-independent growth, and invasiveness in these cell lines. Specifically, PTBP1 knockdown enhances the inclusion of exon 3 in reticulon 4 (RTN4) [241]. The presence of exon 3 in RTN4 isoforms is associated with reduced cell proliferation, suggesting that PTB-induced cell proliferation in glioma cells is partly mediated by RTN4 splicing. Another study reported that in glioblastoma, PTBP1 recognizes an alternative 5′ss within ubiquitin specific peptidase 5 (USP5) exon 15, resulting in the generation of USP5 isoform 2 with a shorter exon length [242]. Enforced expression of USP5 isoform 1 through antisense targeting in glioblastoma cell lines has been demonstrated to inhibit cell growth and migration. These findings demonstrate that both the regulation of RTN4 splicing by PTBP1 and the splicing of USP5 play important roles in gliomagenesis.
Keloid is a fibrotic skin disease characterized by excessive accumulation of extracellular matrix due to the proliferation of dermal fibroblasts [243, 244]. Its pathological features are similar to that of tumors, including tissue invasion and recurrence. PTB is overexpressed in keloid tissues and fibroblasts, and as in glioblastoma, alternative splicing changes in RTN4 and USP5 by PTB have been observed [245]. Furthermore, suppressing PTB has been shown to decrease the expression of fibronectin 1 (FN1) in transplanted keloid tissues and TGF-β1-treated keloid fibroblasts. FN1 is an important protein involved in cell adhesion, migration, and differentiation and is closely associated with cancer and fibrosis. Additionally, inhibition of PTB led to a reduction in excessive deposition of collagen type III alpha 1 chain (COL3A1), demonstrating that PTB siRNA promoted regression of keloid tissue in vivo by regulating both dermal cell proliferation and extracellular matrix accumulation.
PTB induces the exclusion of α-exon in fibroblast growth factor receptor 1 (FGFR1), which is associated with proliferation, and regulates the mutually exclusive splicing of exons IIIb and IIIc in fibroblast growth factor receptor 2 (FGFR2) [246, 247]. Moreover, PTB inhibits the inclusion of caspase 2 (CASP2) exon 9 and FAS exon 6, which are involved in apoptosis [248,249,250]. The binding of PTBP1 to pyruvate kinase M1/2 (PKM) intron 8 leads to the skipping of exon 9 in PKM, resulting in the generation of a PKM2 isoform that promotes the Warburg effect [251, 252]. This increased expression of PKM2 confers drug resistance in pancreatic ductal adenocarcinoma, suggesting that PKM2 and PTBP1 could serve as potential therapeutic targets to enhance the response to chemotherapy [253].
Conclusions and future perspectives
After the development of RNA sequencing technology, comprehensive analyses of alternative splicing shed light on the remarkable diversity of mRNA variants in human genes. Recent studies have identified the functional importance of specific splicing isoforms and splicing factors in cancer progression, associated with regulation of cell numbers. Collectively, published and upcoming unpublished data uncover the effects of alternative splicing and provide the key to understanding the complexity of regulatory systems that determine cell fate. However, our understanding of the systemic regulation of alternative splicing by combinatorial activation or inhibition by hundreds of splicing factors and their fine-tuning control of the proteome is only at the initial stage. Moreover, short-lead bulk RNA sequencing, which has been the most common method for global splicing analysis, presents issues in precision when detecting alternative splicing patterns, especially for the small-size exons or transcripts with low expression levels. However, with the continuous development of alternative splicing analysis methods, such as long-read sequencing, and the application of bioinformatics techniques, such as deep learning, we expect that detailed cellular mechanisms can be profiled by analyzing not only gene expression levels but also alternative splicing patterns [254, 255].
In this review, we highlight the regulation of cancer cell proliferation through alternative splicing. Most studies have focused only on the expression level and PTMs of cell-number-regulating proteins. However, this approach is insufficient to fully explain the different functional outcomes of these genes depending on cancer types and stages, or individual patients with cancer. Recent studies have revealed that alternative splicing-mediated isoforms exhibit differential cellular functions in promoting cell growth. Therefore, alterations in alternative splicing within cancer cells can contribute to cell proliferation. Consequently, analyzing alternative splicing patterns in genes that regulate cell number may provide clues to identify the causes of abnormal cell growth and survival of cancer cells. Ultimately, exploration of global alternative splicing regulation with the role of individual splicing isoforms will suggest novel therapeutic splicing modulators or diagnostic markers for pathological splicing phenotypes and will provide a strategy to precisely predict therapeutic outcomes for precision medicine by revealing detailed information about tumor tissues.
Data Availability
Not applicable.
Abbreviations
- CDS:
-
Coding sequence
- PTM:
-
Post-translational modification
- UTR:
-
Untranslated region
- snRNPs:
-
Small nuclear ribonucleoproteins
- snRNA:
-
Small nuclear RNA
- ss:
-
Splice sites
- BPS:
-
Branch point site
- PPT:
-
Polypyrimidine track
- CDK:
-
Cyclin-dependent kinase
- Rb:
-
Retinoblastoma
- CDKN1A:
-
Cyclin dependent kinase inhibitor 1 A
- CDKN2A:
-
Cyclin dependent kinase inhibitor 2 A
- CCND1:
-
Cyclin D1
- SAM68:
-
SRC-associated in mitosis of 68 kDa
- Ccnd2:
-
Cyclin D2
- ER:
-
Endoplasmic reticulum
- PCBP:
-
Poly(C)-binding protein
- E2F1:
-
E2F transcription factor 1
- E2F3:
-
E2F transcription factor 3
- NSCLC:
-
Non-small cell lung cancer
- TP53:
-
Tumor protein P53
- bHLH-LZ:
-
Basic helix-loop-helix and leucine zipper
- MAX:
-
Myc-associated factor X
- MXD3:
-
MAX dimerization protein 3
- TCGA:
-
The Cancer Genome Atlas
- Keap1:
-
Kelch-like ECH-associated protein 1
- Cul3:
-
Cullin-3
- Nrf2:
-
Nuclear factor-erythroid factor 2-related factor 2
- sMAF:
-
Small musculoaponeurotic fibrosarcoma oncogene homologue
- ARE:
-
Antioxidant response element
- HNSC:
-
Head and neck squamous carcinoma
- P:
-
Promoter
- tBHQ:
-
Tert-butylhydroquinone
- DGR domain:
-
Double glycine-repeat domain
- PIP2:
-
Phosphatidylinositol (3,4)-bisphosphate
- PIP3:
-
Phosphatidylinositol (3,4,5)-trisphosphate
- PH domain:
-
Pleckstrin homology domain
- PIK3R1:
-
Phosphoinositide-3-kinase regulatory subunit 1
- PR:
-
Proline-rich motif
- APDS:
-
Activated phosphoinositide 3-kinase δ syndrome
- MAPK:
-
Mitogen-activated protein kinase
- PDE6δ:
-
δ-subunit of cGMP phosphodiesterase type 6
- SRSF:
-
Serine/arginine-rich splicing factor
- NOVA1:
-
Neuro-oncological ventral antigen 1
- HCC:
-
Hepatocellular carcinoma
- hTERT:
-
Human telomerase reverse transcriptase
- CRC:
-
Colorectal cancer
- SF3B1:
-
Splicing factor 3B subunit 1
- PI3K:
-
Phosphatidylinositol 3-kinase
- MDS:
-
Myelodysplastic syndrome
- RARS:
-
Ringed sideroblasts
- CLL:
-
Chronic lymphocytic leukemia
- AR-v7:
-
androgen receptor variant 7
- EJC:
-
Exon-junction complex
- SURF:
-
SMG-1Upf1–eRF1–eRF3
- NMD:
-
Nonsense-mediated mRNA decay
- RRM:
-
RNA recognition motif
- SF3B4:
-
Splicing factor 3b subunit 4
- DEGs:
-
Differentially expressed genes
- SPAG5:
-
Sperm-associated antigen 5
- KLF4:
-
Kruppel-like factor 4
- CDKN1B:
-
Cyclin-dependent kinase inhibitor 1B
- SNAI2:
-
Snail family transcriptional repressor 2
- EMT:
-
Epithelial–mesenchymal transition
- GPC3:
-
Glypican 3
- GS:
-
Glutamine synthetase
- HSP70:
-
Heat-shock protein 70
- ESCC:
-
Esophageal squamous cell carcinoma
- ETV1:
-
ETS variant 1
- NDE1:
-
nudE neurodevelopment protein 1
- TDP43:
-
TAR DNA-binding protein
- TNBC:
-
Triple-negative breast cancer
- HIPK2:
-
Homeodomain-interacting protein kinase-2
- ERBB2:
-
Erb-B2 receptor tyrosine kinase 2
- ILF3:
-
Interleukin enhancer-binding factor 3
- PTB:
-
Polypyrimidine tract binding protein
- RTN4:
-
Reticulon 4
- USP5:
-
ubiquitin specific peptidase 5
- FN1:
-
Fibronectin 1
- COL3A1:
-
Collagen type III alpha 1 chain
- FGFR:
-
Fibroblast growth factor receptor
- CASP2:
-
Caspase 2
- PKM:
-
Pyruvate kinase M1/2
References
McCoy MJ, Fire AZ. Intron and gene size expansion during nervous system evolution. BMC Genomics. 2020;21(1):360.
Irimia M, Roy SW. Evolutionary convergence on highly-conserved 3 ' Intron Structures in Intron-Poor Eukaryotes and Insights into the ancestral eukaryotic genome. PLOS Genet. 2008;4(8):e1000148.
Choi S, Cho N, Kim KK. Non-canonical splice junction processing increases the diversity of RBFOX2 splicing isoforms. Int J Biochem Cell Biol. 2022;144:106172.
Choi S, Lee HS, Cho N, Kim I, Cheon S, Park C, et al. RBFOX2-regulated TEAD1 alternative splicing plays a pivotal role in Hippo-YAP signaling. Nucleic Acids Res. 2022;50(15):8658–73.
Cho N, Joo J, Choi S, Kang BG, Lee AJ, Youn SY, et al. A novel splicing variant of DJ-1 in Parkinson’s disease induces mitochondrial dysfunction. Heliyon. 2023;9(3):e14039.
Lee S, Jung H, Choi S, Cho N, Kim EM, Kim KK. Intron retention decreases METTL3 expression by inhibiting mRNA export to the cytoplasm. BMB Rep. 2023;56(9):514–9.
Chan JJ, Zhang B, Chew XH, Salhi A, Kwok ZH, Lim CY, et al. Pan-cancer pervasive upregulation of 3’ UTR splicing drives tumourigenesis. Nat Cell Biol. 2022;24(6):928–39.
Pan Q, Shai O, Lee LJ, Frey JB, Blencowe BJ. Deep surveying of alternative splicing complexity in the human transcriptome by high-throughput sequencing. Nat Genet. 2008;40(12):1413–5.
Mazin PV, Khaitovich P, Cardoso-Moreira M, Kaessmann H. Alternative splicing during mammalian organ development. Nat Genet. 2021;53(6):925–34.
Choi S, Cho N, Kim KK. The implications of alternative pre-mRNA splicing in cell signal transduction. Exp Mol Med. 2023;55(4):755–66.
Fu XD, Ares M. Context-dependent control of alternative splicing by RNA-binding proteins. Nat Rev Genet. 2014;15(10):689–701.
Daguenet E, Dujardin G, Valcárcel J. The pathogenicity of splicing defects: mechanistic insights into pre-mRNA processing inform novel therapeutic approaches. EMBO Rep. 2015;16(12):1640–55.
Kahles A, Lehmann KV, Toussaint NC, Hüser M, Stark SG, Sachsenberg T, et al. Comprehensive Analysis of Alternative Splicing Across Tumors from 8,705 patients. Cancer Cell. 2018;34(2):211–24.
Bradley RK, Anczuków O. RNA splicing dysregulation and the hallmarks of cancer. Nat Rev Cancer. 2023;23(3):135–55.
Scully R, Panday A, Elango R, Willis NA. DNA double-strand break repair-pathway choice in somatic mammalian cells. Nat Rev Mol Cell Biol. 2019;20(11):698–714.
Orth JD, Loewer A, Lahav G, Mitchison TJ. Prolonged mitotic arrest triggers partial activation of apoptosis, resulting in DNA damage and p53 induction. Mol Biol Cell. 2012;23(4):567–76.
Matthews HK, Bertoli C, de Bruin RAM. Cell cycle control in cancer. Nat Rev Mol Cell Biol. 2022;23(1):74–88.
Hanahan D, Weinberg RA. Hallmarks of Cancer: the Next Generation. Cell. 2011;144(5):646–74.
Wahl MC, Will CL, Lührmann R. The spliceosome: Design Principles of a dynamic RNP machine. Cell. 2009;136(4):701–18.
Matera AG, Wang ZF. A day in the life of the spliceosome. Nat Rev Mol Cell Biol. 2014;15(2):108–21.
Denisov SV, Bazykin GA, Sutormin R, Favorov AV, Mironov AA, Gelfand MS, et al. Weak negative and positive selection and the drift load at Splice Sites. Genome Biol Evol. 2014;6(6):1437–47.
Montemayor EJ, Curran EC, Liao HH, Andrews KL, Treba CN, Butcher SE, et al. Core structure of the U6 small nuclear ribonucleoprotein at 1.7-Å resolution. Nat Struct Mol Biol. 2014;21(6):544–51.
Barbosa-Morais NL, Irimia M, Pan Q, Xiong HY, Gueroussov S, Lee LJ, et al. The Evolutionary Landscape of Alternative Splicing in Vertebrate Species. Science. 2012;338(6114):1587–93.
Van Nostrand EL, Freese P, Pratt GA, Wang XF, Wei XT, Xiao R, et al. A large-scale binding and functional map of human RNA-binding proteins. Nature. 2020;583(7818):711–9.
Marasco LE, Kornblihtt AR. The physiology of alternative splicing. Nat Rev Mol Cell Biol. 2023;24(4):242–54.
Barnum KJ, O’Connell MJ. Cell cycle regulation by checkpoints. Methods Mol Biol. 2014;1170:29–40.
Ding L, Cao JQ, Lin W, Chen HJ, Xiong XH, Ao HS, et al. The roles of cyclin-dependent kinases in cell-cycle progression and therapeutic strategies in human breast Cancer. Int J Mol Sci. 2020;21(6):1960.
Collier AE, Spandau DF, Wek RC. Translational control of a human CDKN1A mRNA splice variant regulates the fate of UVB-irradiated human keratinocytes. Mol Biol Cell. 2018;29(1):29–41.
Sun H, Nguyen M, Mueller W, Cheng Z, Zeng H, Zhu C, et al. CDKN1A-RAB44 transcript fusion and activation in cancers. bioRxiv. 2019. https://doi.org/10.1101/111856.
Petronzelli F, Sollima D, Coppola G, Martini-Neri ME, Neri G, Genuardi M. CDKN2A germline splicing mutation affecting both P16(ink4) and P14(arf) RNA processing in a melanoma/neurofibroma kindred. Genes Chromosom Cancer. 2001;31(4):398–401.
Loo JCY, Liu L, Hao AH, Gao LZ, Agatep R, Shennan M, et al. Germline splicing mutations of CDKN2A predispose to melanoma. Oncogene. 2003;22(41):6387–94.
Zschemisch NH, Liedtke C, Dierssen U, Nevzorova YA, Wüstefeld T, Borlak J, et al. Expression of a cyclin E1 isoform in mice is correlated with the quiescent cell cycle status of hepatocytes in vivo. Hepatology. 2006;44(1):164–73.
Knudsen KE, Diehl JA, Haiman CA, Knudsen ES. Cyclin D1: polymorphism, aberrant splicing and cancer risk. Oncogene. 2006;25(11):1620–8.
Paronetto MP, Cappellari M, Busà R, Pedrotti S, Vitali R, Comstock C, et al. Alternative splicing of the cyclin D1 Proto-Oncogene is regulated by the RNA-Binding protein Sam68. Cancer Res. 2010;70(1):229–39.
Comstock CES, Augello MA, Benito RP, Karch J, Tran TH, Utama FE, et al. Cyclin D1 splice variants: polymorphism, risk, and isoform-specific regulation in prostate Cancer. Clin Cancer Res. 2009;15(17):5338–49.
Solomon DA, Wang Y, Fox SR, Lambeck TC, Giesting S, Lan Z, et al. Cyclin D1 splice variants - Differential effects on localization, RB phosphorylation, and cellular transformation. J Biol Chem. 2003;278(32):30339–47.
Lu FM, Gladden AB, Diehl JA. An alternatively spliced cyclin D1 isoform, cyclin D1b, is a nuclear oncogene. Cancer Res. 2003;63(21):7056–61.
Sun Q, Zhang FX, Wafa K, Baptist T, Pasumarthi KBS. A splice variant of cyclin D2 regulates cardiomyocyte cell cycle through a novel protein aggregation pathway. J Cell Sci. 2009;122(10):1563–73.
Denicourt C, Legault P, McNabb FA, Rassart E. Human and mouse cyclin D2 splice variants: transforming activity and subcellular localization. Oncogene. 2008;27(9):1253–62.
Ellenrieder C, Bartosch B, Lee GYC, Murphy M, Sweeney C, Hergersberg M, et al. The long form of CDK2 arises via alternative splicing and forms an active protein kinase with cyclins a and E. DNA Cell Biol. 2001;20(7):413–23.
Ji XJ, Humenik J, Yang D, Liebhaber SA. PolyC-binding proteins enhance expression of the CDK2 cell cycle regulatory protein via alternative splicing. Nucleic Acids Res. 2018;46(4):2030–44.
Jackson DP, Ting JHH, Pozniak PD, Meurice C, Schleidt SS, Dao A, et al. Identification and characterization of two novel alternatively spliced E2F1 transcripts in the rat CNS. Mol Cell Neurosci. 2018;92:1–11.
Cates HM, Heller EA, Lardner CK, Purushothaman I, Peña CJ, Walker DM, et al. Transcription factor E2F3a in Nucleus Accumbens affects Cocaine Action via transcription and alternative splicing. Biol Psychiat. 2018;84(3):167–79.
He YW, Armanious MK, Thomas MJ, Cress WD. Identification of E2F-3B, an alternative form of E2F-3 lacking a conserved N-terminal region. Oncogene. 2000;19(30):3422–33.
Leone G, Nuckolls F, Ishida S, Adams M, Sears R, Jakoi L, et al. Identification of a novel E2F3 product suggests a mechanism for determining specificity of repression by rb proteins. Mol Cell Biol. 2000;20(10):3626–32.
Cygan KJ, Soemedi R, Rhine CL, Profeta A, Murphy EL, Murray MF, et al. Defective splicing of the RB1 transcript is the dominant cause of retinoblastomas. Hum Genet. 2017;136(9):1303–12.
Bhateja P, Chiu M, Wildey G, Lipka MB, Fu PF, Yang MCL, et al. Retinoblastoma mutation predicts poor outcomes in advanced non small cell lung cancer. Cancer Med. 2019;8(4):1459–66.
Dehainault C, Michaux D, Pages-Berhouet S, Caux-Moncoutier V, Doz F, Desjardins L, et al. A deep intronic mutation in the RB1 gene leads to intronic sequence exonisation. Eur J Hum Genet. 2007;15(4):473–7.
Valverde JR, Alonso J, Palacios I, Pestana A. RBI gene mutation up-date, a meta-analysis based on 932 reported mutations available in a searchable database. BMC Genet. 2005;6.
Senturk S, Yao Z, Camioloa M, Stilesb B, Rathod T, Walsh AM, et al. p53Ψ is a transcriptionally inactive p53 isoform able to reprogram cells toward a metastatic-like state. Proc Natl Acad Sci U S A. 2014;111(32):E3287–96.
Miller M, Shirole N, Tian R, Pal D, Sordella R. The evolution of TP53 mutations: from loss-of-function to separation-of-function mutants. J Cancer Biol Res. 2016;4(4):1091.
Smeby J, Sveen A, Eilertsen IA, Danielsen SA, Hoff AM, Eide PW, et al. Transcriptional and functional consequences of TP53 splice mutations in colorectal cancer. Oncogenesis. 2019;8:35.
Pinto EM, Maxwell KN, Halalsheh H, Phillips A, Powers J, MacFarland S, et al. Clinical and functional significance of TP53 exon 4-Intron 4 Splice Junction Variants. Mol Cancer Res. 2022;20(2):207–16.
Shirole NH, Pal D, Kastenhuber ER, Senturk S, Boroda J, Pisterzi P, et al. TP53 exon-6 truncating mutations produce separation of function isoforms with pro-tumorigenic functions. eLife. 2016;5:e17929.
Dang CV, O’Donnell KA, Zeller KI, Nguyen T, Osthus RC, Li F. The c-Myc target gene network. Semin Cancer Biol. 2006;16(4):253–64.
Fernandez PC, Frank SR, Wang LQ, Schroeder M, Liu SX, Greene J, et al. Genomic targets of the human c-Myc protein. Genes Dev. 2003;17(9):1115–29.
Meyer N, Penn LZ. Reflecting on 25 years with MYC. Nat Rev Cancer. 2008;8(12):976–90.
Conacci-Sorrell M, McFerrin L, Eisenman RN. An overview of MYC and its Interactome. Cold Spring Harb Perspect Med. 2014;4(1):a014357.
Zhang H, Fan SJ, Prochownik EV. Distinct roles for MAX protein isoforms in proliferation and apoptosis. J Biol Chem. 1997;272(28):17416–24.
Arsura M, Deshpande A, Hann SR, Sonenshein GE. Variant Max protein, derived by alternative splicing, associates with C-Myc in-vivo and inhibits transactivation. Mol Cell Biol. 1995;15(12):6702–9.
Mäkelä TP, Koskinen PJ, Vastrik I, Alitalo K. Alternative forms of Max as Enhancers or Suppressors of myc-ras Cotransformation. Science. 1992;256(5055):373–7.
Kemmerer K, Weigand JE. Hypoxia reduces MAX expression in endothelial cells by unproductive splicing. FEBS Lett. 2014;588(24):4784–90.
Ngo T, Corrales A, Bourne T, Elmojahid S, Lam KS, Díaz E. Alternative splicing of MXD3 and its regulation of MXD3 levels in Glioblastoma. Front Mol Biosci. 2019;6:5.
Saha S, Buttari B, Panieri E, Profumo E, Saso L. An overview of Nrf2 Signaling Pathway and its role in inflammation. Molecules. 2020;25(22):5474.
Baird L, Yamamoto M. The Molecular Mechanisms regulating the KEAP1-NRF2 pathway. Mol Cell Biol. 2020;40(13):e00099–20.
Iso T, Suzuki T, Baird L, Yamamoto M. Absolute amounts and Status of the Nrf2-Keap1-Cul3 complex within cells. Mol Cell Biol. 2016;36(24):3100–12.
Furukawa M, Xiong Y. BTB protein keap1 targets antioxidant transcription factor nrf2 for ubiquitination by the cullin 3-Roc1 ligase. Mol Cell Biol. 2005;25(1):162–71.
Zhang DD, Lo SC, Cross JV, Templeton DJ, Hannink M. Keap1 is a redox-regulated substrate adaptor protein for a Cul3-dependent ubiquitin ligase complex. Mol Cell Biol. 2004;24(24):10941–53.
Kobayashi A, Kang MI, Okawa H, Ohtsuji M, Zenke Y, Chiba T, et al. Oxidative stress sensor Keap1 functions as an adaptor for Cul3-based E3 ligase to regulate for proteasomal degradation of Nrf2. Mol Cell Biol. 2004;24(16):7130–9.
Cullinan SB, Gordan JD, Jin JO, Harper JW, Diehl JA. The Keap1-BTB protein is an adaptor that bridges Nrf2 to a Cul3-based E3 ligase: oxidative stress sensing by a Cul3-Keap1 ligase. Mol Cell Biol. 2004;24(19):8477–86.
Hayes JD, McMahon M, Chowdhry S, Dinkova-Kostova AT. Cancer chemoprevention mechanisms mediated through the Keap1-Nrf2 pathway. Antioxid Redox Signal. 2010;13(11):1713–48.
Itoh K, Chiba T, Takahashi S, Ishii T, Igarashi K, Katoh Y, et al. An Nrf2 small maf heterodimer mediates the induction of phase II detoxifying enzyme genes through antioxidant response elements. Biochem Biophys Res Commun. 1997;236(2):313–22.
Zhou JM, Zheng QX, Chen Z. The Nrf2 pathway in Liver Diseases. Front Cell Dev Biol. 2022;10:826204.
Nguyen T, Sherratt PJ, Pickett CB. Regulatory mechanisms controlling gene expression mediated by the antioxidant response element. Annu Rev Pharmacol Toxicol. 2003;43:233–60.
Yamamoto T, Kyo M, Kamiya T, Tanaka T, Engel JD, Motohashi H, et al. Predictive base substitution rules that determine the binding and transcriptional specificity of maf recognition elements. Genes Cells. 2006;11(6):575–91.
Li W, Yu S, Liu T, Kim JH, Blank V, Li H, et al. Heterodimerization with small maf proteins enhances nuclear retention of Nrf2 via masking the NESzip motif. Biochim Biophys Acta. 2008;1783(10):1847–56.
Goldstein LD, Lee J, Gnad F, Klijn C, Schaub A, Reeder J, et al. Recurrent loss of NFE2L2 exon 2 is a mechanism for Nrf2 pathway activation in human cancers. Cell Rep. 2016;16(10):2605–17.
Mikac S, Dziadosz A, Padariya M, Kalathiya U, Fahraeus R, Marek-Trzonkowska N, et al. Keap1-resistant ∆N-Nrf2 isoform does not translocate to the nucleus upon electrophilic stress. bioRxiv. 2022. https://doi.org/10.1101/2022.06.10.495609.
Qiu L, Wang M, Zhu YP, Xiang YC, Zhang YG. A Naturally-Occurring Dominant-Negative inhibitor of Keap1 competitively against its negative regulation of Nrf2. Int J Mol Sci. 2018;19(8):2150.
Jiang NN, Dai QJ, Su XR, Fu JJ, Feng XC, Peng J. Role of PI3K/AKT pathway in cancer: the framework of malignant behavior. Mol Biol Rep. 2020;47(6):4587–629.
Manning BD, Cantley LC. AKT/PKB signaling: navigating downstream. Cell. 2007;129(7):1261–74.
Stambolic V, Suzuki A, de la Pompa JL, Brothers GM, Mirtsos C, Sasaki T, et al. Negative regulation of PKB/Akt-dependent cell survival by the tumor suppressor PTEN. Cell. 1998;95(1):29–39.
Brognard J, Sierecki E, Gao TY, Newton AC. PHLPP and a second isoform, PHLPP2, differentially attenuate the amplitude of akt signaling by regulating distinct akt isoforms. Mol Cell. 2007;25(6):917–31.
Andjelković M, Jakubowicz T, Cron P, Ming XF, Han JW, Hemmings BA. Activation and phosphorylation of a pleckstrin homology domain containing protein kinase (RAC-PK/PKB) promoted by serum and protein phosphatase inhibitors. Proc Natl Acad Sci U S A. 1996;93(12):5699–704.
LoPiccolo J, Kim SJ, Shi Y, Wu B, Wu HY, Chait BT, et al. Assembly and Molecular Architecture of the phosphoinositide 3-Kinase p85α Homodimer. J Biol Chem. 2015;290(51):30390–405.
Harpur AG, Layton MJ, Das P, Bottomley MJ, Panayotou G, Driscoll PC, et al. Intermolecular interactions of the p85alpha regulatory subunit of phosphatidylinositol 3-kinase. J Biol Chem. 1999;274(18):12323–32.
Cheung LWT, Walkiewicz KW, Besong TMD, Guo HF, Hawke DH, Arold ST, et al. Regulation of the PI3K pathway through a p85α monomer-homodimer equilibrium. eLife. 2015;4:e06866.
Abell K, Bilancio A, Clarkson RWE, Tiffen PG, Altaparmakov AI, Burdon TG, et al. Stat3-induced apoptosis requires a molecular switch in PI(3)K subunit composition. Nat Cell Biol. 2005;7(4):392–8.
Lucas CL, Zhang Y, Venida A, Wang Y, Hughes J, McElwee J, et al. Heterozygous splice mutation in PIK3R1 causes human immunodeficiency with lymphoproliferation due to dominant activation of PI3K. J Exp Med. 2014;211(13):2537–47.
Deau MC, Heurtier L, Frange P, Suarez F, Bole-Feysot C, Nitschke P, et al. A human immunodeficiency caused by mutations in the PIK3R1 gene. J Clin Invest. 2014;124(9):3923–8.
Abolhassani H, Aghamohammadi A, Fang MY, Rezaei N, Jiang CY, Liu X, et al. Clinical implications of systematic phenotyping and exome sequencing in patients with primary antibody deficiency. Genet Med. 2019;21(1):243–51.
Hauck F, Magg T, Krolo A, Bilic I, Hirschmugl T, Laass M, et al. Variant PIK3R1 Hypermorphic Mutation and clinical phenotypes in a family with short statures, mild immunodeficiency and lymphoma. Klin Padiatr. 2017;229(3):113–7.
Olbrich P, Lorenz M, Daball PC, Lucena JM, Rensing-Ehl A, Sanchez B, et al. Activated PI3Kδ syndrome type 2: two patients, a novel mutation, and review of the literature. Pediatr Allergy Immunol. 2016;27(6):640–4.
Lougaris V, Faletra F, Lanzi G, Vozzi D, Marcuzzi A, Valencic E, et al. Altered germinal center reaction and abnormal B cell peripheral maturation in PI3KR1-mutated patients presenting with HIGM-like phenotype. Clin Immunol. 2015;159(1):33–6.
Elkaim E, Neven B, Bruneau J, Mitsui-Sekinaka K, Stanislas A, Heurtier L, et al. Clinical and immunologic phenotype associated with activated phosphoinositide 3-kinase delta syndrome 2: a cohort study. J Allergy Clin Immunol. 2016;138(1):210–8.
Breuksch I, Welter J, Bauer HK, Enklaar T, Frees S, Thüroff JW, et al. In renal cell carcinoma the PTEN splice variant PTEN-∆ shows similar function as the tumor suppressor PTEN itself. Cell Commun Signal. 2018;16:35.
Sharrard RM, Maitland NJ. Alternative splicing of the human PTEN/MMAC1/TEP1 gene. Biochim Biophys Acta. 2000;1494(3):282–5.
Agrawal S, Eng C. Differential expression of novel naturally occurring splice variants of PTEN and their functional consequences in Cowden syndrome and sporadic breast cancer. Hum Mol Genet. 2006;15(5):777–87.
Chen HJ, Romigh T, Sesock K, Eng C. Characterization of cryptic splicing in germline PTEN intronic variants in Cowden syndrome. Hum Mutat. 2017;38(10):1372–7.
Celebi JT, Wanner M, Ping XL, Zhang H, Peacocke M. Association of splicing defects in PTEN leading to exon skipping or partial intron retention in Cowden syndrome. Hum Genet. 2000;107(3):234–8.
Yang SF, Liu GH. Targeting the Ras/Raf/MEK/ERK pathway in hepatocellular carcinoma. Oncol Lett. 2017;13(3):1041–7.
Hartung AM, Swensen J, Uriz IE, Lapin M, Kristjansdottir K, Petersen USS, et al. The splicing efficiency of activating HRAS mutations can determine Costello Syndrome phenotype and frequency in Cancer. PLOS Genet. 2016;12(5):e1006039.
Pells S, Divjak M, Romanowski P, Impey H, Hawkins NJ, Clarke AR, et al. Developmentally-regulated expression of murine K-ras isoforms. Oncogene. 1997;15(15):1781–6.
Wang Y, You M, Wang Y. Alternative splicing of the K-ras gene in mouse tissues and cell lines. Exp Lung Res. 2001;27(3):255–67.
Laude AJ, Prior IA. Palmitoylation and localisation of RAS isoforms are modulated by the hypervariable linker domain. J Cell Sci. 2008;121(4):421–7.
Tsai FD, Lopes MS, Zhou M, Court H, Ponce O, Fiordalisi JJ, et al. K-Ras4A splice variant is widely expressed in cancer and uses a hybrid membrane-targeting motif. Proc Natl Acad Sci U S A. 2015;112(3):779–84.
Chen WC, To MD, Westcott PMK, Delrosario R, Kim IJ, Philips M, et al. Targeting KRAS4A splicing through the RBM39/DCAF15 pathway inhibits cancer stem cells. Nat Commun. 2021;12(1):4288.
Eilertsen IA, Sveen A, Strømme JM, Skotheim RI, Nesbakken A, Lothe RA. Alternative splicing expands the prognostic impact of KRAS in microsatellite stable primary colorectal cancer. Int J Cancer. 2019;144(4):841–7.
Eisfeld AK, Schwind S, Hoag KW, Walker CJ, Liyanarachchi S, Patel R, et al. NRAS isoforms differentially affect downstream pathways, cell growth, and cell transformation. Proc Natl Acad Sci U S A. 2014;111(11):4179–84.
Endo T. Dominant-negative antagonists of the Ras-ERK pathway: DA-Raf and its related proteins generated by alternative splicing of Raf. Exp Cell Res. 2020;387(2).
Yokoyama T, Takano K, Yoshida A, Katada F, Sun P, Takenawa T, et al. DA-Raf1, a competent intrinsic dominant-negative antagonist of the Ras-ERK pathway, is required for myogenic differentiation. J Cell Biol. 2007;177(5):781–93.
Zhang Y, Siraj MA, Chakraborty P, Tseng R, Ku L-T, Das S, et al. Activation of ERK by altered RNA splicing in cancer. bioRxiv. 2022. https://doi.org/10.1101/2022.08.31.505957.
Uniacke J, Holterman CE, Lachance G, Franovic A, Jacob MD, Fabian MR, et al. An oxygen-regulated switch in the protein synthesis machinery. Nature. 2012;486(7401):126–9.
Wang Y, Ma M, Xiao XS, Wang ZF. Intronic splicing enhancers, cognate splicing factors and context-dependent regulation rules. Nat Struct Mol Biol. 2012;19(10):1044–52.
Wang Y, Chen D, Qian HL, Tsai YHS, Shao SJ, Liu QT, et al. The splicing factor RBM4 controls apoptosis, proliferation, and Migration to suppress Tumor Progression. Cancer Cell. 2014;26(3):374–89.
Wang WY, Quan WL, Yang F, Wei YX, Chen JJ, Yu H, et al. RBM4 modulates the proliferation and expression of inflammatory factors via the alternative splicing of regulatory factors in HeLa cells. Mol Genet Genomics. 2020;295(1):95–106.
Yong HM, Zhao W, Zhou XY, Liu ZY, Tang Q, Shi HC, et al. RNA-Binding motif 4 (RBM4) suppresses Tumor Growth and Metastasis in Human gastric Cancer. Med Sci Monit. 2019;25:4025–34.
Rbm4-Regulated Alternative Splicing Suppresses Tumorigenesis. Cancer Discov. 2014;4(11):1253.
Adams JM, Cory S. The Bcl-2 apoptotic switch in cancer development and therapy. Oncogene. 2007;26(9):1324–37.
Anczuków O, Rosenberg AZ, Akerman M, Das S, Zhan LX, Karni R, et al. The splicing factor SRSF1 regulates apoptosis and proliferation to promote mammary epithelial cell transformation. Nat Struct Mol Biol. 2012;19(2):220–8.
Karni R, Hippo Y, Lowe SW, Krainer AR. The splicing-factor oncoprotein SF2/ASF activates mTORC1. Proc Natl Acad Sci U S A. 2008;105(40):15323–7.
Karni R, de Stanchina E, Lowe SW, Sinha R, Mu D, Krainer AR. The gene encoding the splicing factor SF2/ASF is a proto-oncogene. Nat Struct Mol Biol. 2007;14(3):185–93.
Yong HM, Zhu HJ, Zhang S, Zhao W, Wang W, Chen C, et al. Prognostic value of decreased expression of RBM4 in human gastric cancer. Sci Rep. 2016;6:28222.
Lewis HA, Musunuru K, Jensen KB, Edo C, Chen H, Darnell RB, et al. Sequence-specific RNA binding by a Nova KH domain: implications for paraneoplastic disease and the fragile X syndrome. Cell. 2000;100(3):323–32.
Jensen KB, Dredge BK, Stefani G, Zhong R, Buckanovich RJ, Okano HJ, et al. Nova-1 regulates neuron-specific alternative splicing and is essential for neuronal viability. Neuron. 2000;25(2):359–71.
Buckanovich RJ, Darnell RB. The neuronal RNA binding protein Nova-1 recognizes specific RNA targets in vitro and in vivo. Mol Cell Biol. 1997;17(6):3194–201.
Raj B, Blencowe BJ. Alternative splicing in the mammalian nervous system: recent insights into mechanisms and functional roles. Neuron. 2015;87(1):14–27.
Darnell RB. RNA protein Interaction in neurons. Annu Rev Neurosci. 2013;36:243–70.
Buckanovich RJ, Yang YYL, Darnell RB. The onconeural antigen Nova-1 is a neuron-specific RNA-Binding protein, the activity of which is inhibited by paraneoplastic antibodies. J Neurosci. 1996;16(3):1114–22.
Buckanovich RJ, Posner JB, Darnell RB. Nova, the Paraneoplastic Ri Antigen, is homologous to an rna-binding protein and is specifically expressed in the developing Motor System. Neuron. 1993;11(4):657–72.
Yoon SO, Kim EK, Lee M, Jung WY, Lee H, Kang Y, et al. NOVA1 inhibition by miR-146b-5p in the remnant tissue microenvironment defines occult residual disease after gastric cancer removal. Oncotarget. 2016;7(3):2475–95.
Kim E, Yoon SO, Jung WY, Lee H, Kang Y, Jang YJ, et al. Implications of NOVA1 suppression within the microenvironment of gastric cancer: association with immune cell dysregulation. Gastric Cancer. 2017;20(3):438–47.
Shen B, Zhang Y, Yu SR, Yuan Y, Zhong YJ, Lu JW, et al. MicroRNA-339, an epigenetic modulating target is involved in human gastric carcinogenesis through targeting NOVA1. FEBS Lett. 2015;589(20):3205–11.
Zhi F, Wang Q, Deng DN, Shao NY, Wang R, Xue L, et al. MiR-181b-5p downregulates NOVA1 to suppress Proliferation, Migration and Invasion and promote apoptosis in Astrocytoma. PLoS ONE. 2014;9(10):e109124.
Zhang YA, Zhu JM, Yin J, Tang WQ, Guo YM, Shen XZ, et al. High expression of neuro-oncological ventral Antigen 1 correlates with poor prognosis in Hepatocellular Carcinoma. PLoS ONE. 2014;9(3):e90955.
Dredge BK, Stefani G, Engelhard CC, Darnell RB. Nova autoregulation reveals dual functions in neuronal splicing. EMBO J. 2005;24(8):1608–20.
Dredge BK, Darnell RB. Nova regulates GABA(A) receptor gamma2 alternative splicing via a distal downstream UCAU-rich intronic splicing enhancer. Mol Cell Biol. 2003;23(13):4687–700.
Zhang YA, Liu HN, Zhu JM, Zhang DY, Shen XZ, Liu TT. RNA binding protein Nova1 promotes tumor growth in vivo and its potential mechanism as an oncogene may due to its interaction with GABA(A) receptor-gamma 2. J Biomed Sci. 2016;23:71.
Qu LY, Tian YL, Wang F, Li ZX. NOVA1 promotes NSCLC proliferation and invasion by activating Wnt/beta-catenin signaling. BMC Cancer. 2022;22(1):1091.
Ludlow AT, Wong MS, Robin JD, Batten K, Yuan L, Lai TP, et al. NOVA1 regulates hTERT splicing and cell growth in non-small cell lung cancer. Nat Commun. 2018;9:3112.
Hong YG, Xu GS, Yu GY, Zhou JD, Liu QZ, Ni JS, et al. The RNA binding protein neuro-oncological ventral antigen 1 (NOVA1) regulates IL-6 mRNA stability to enhance JAK2-STAT3 signaling in CRC. Surg Oncol. 2019;31:67–74.
Sun CF. The SF3b complex: splicing and beyond. Cell Mol Life Sci. 2020;77(18):3583–95.
Cretu C, Schmitzová J, Ponce-Salvatierra A, Dybkov O, De Laurentiis EI, Sharma K, et al. Molecular Architecture of SF3b and Structural Consequences of its Cancer-related mutations. Mol Cell. 2016;64(2):307–19.
Yoshida K, Sanada M, Shiraishi Y, Nowak D, Nagata Y, Yamamoto R, et al. Frequent pathway mutations of splicing machinery in myelodysplasia. Nature. 2011;478(7367):64–9.
Song JM, Hussaini M, Qin DH, Zhang XH, Shao HP, Zhang L, et al. Comparison of SF381/DNMT3A comutations with DNMT3A or SF3B1 mutation alone in Myelodysplastic Syndrome and Clonal Cytopenia of undetermined significance. Am J Clin Pathol. 2020;154(1):48–56.
Cazzola M, Rossi M, Malcovati L, Grp AIRC. Biologic and clinical significance of somatic mutations of SF3B1 in myeloid and lymphoid neoplasms. Blood. 2013;121(2):260–9.
Jenkins JL, Kielkopf CL. Splicing factor mutations in Myelodysplasias: insights from Spliceosome Structures. Trends Genet. 2017;33(5):336–48.
Foy A, McMullin MF. Somatic SF3B1 mutations in myelodysplastic syndrome with ring sideroblasts and chronic lymphocytic leukaemia. J Clin Pathol. 2019;72(11):778–82.
Wan YZ, Wu CJ. SF3B1 mutations in chronic lymphocytic leukemia. Blood. 2013;121(23):4627–34.
Tang AD, Soulette CM, van Baren MJ, Hart K, Hrabeta-Robinson E, Wu CJ, et al. Full-length transcript characterization of SF3B1 mutation in chronic lymphocytic leukemia reveals downregulation of retained introns. Nat Commun. 2020;11(1):1438.
Yu JF, Li YM, Li T, Li YF, Xing HZ, Sun H, et al. Gene mutational analysis by NGS and its clinical significance in patients with myelodysplastic syndrome and acute myeloid leukemia. Exp Hematol Oncol. 2020;9(1):2.
Yu JF, Li YM, Zhang DF, Wan DM, Jiang ZX. Clinical implications of recurrent gene mutations in acute myeloid leukemia. Exp Hematol Oncol. 2020;9(1):4.
Banerji S, Cibulskis K, Rangel-Escareno C, Brown KK, Carter SL, Frederick AM, et al. Sequence analysis of mutations and translocations across breast cancer subtypes. Nature. 2012;486(7403):405–9.
Ellis MJ, Ding L, Shen D, Luo JQ, Suman VJ, Wallis JW, et al. Whole-genome analysis informs breast cancer response to aromatase inhibition. Nature. 2012;486(7403):353–60.
Li CZ, Xie WY, Rosenblum JS, Zhou JY, Guo J, Miao YZ, et al. Somatic SF3B1 hotspot mutation in prolactinomas. Nat Commun. 2020;11(1):2506.
Küsters-Vandevelde HVN, Creytens D, van Engen-van Grunsven ACH, Jeunink M, Winnepenninckx V, Groenen PJTA, et al. SF3B1 and EIF1AX mutations occur in primary leptomeningeal melanocytic neoplasms; yet another similarity to uveal melanomas. Acta Neuropathol Commun. 2016;4:5.
Kong Y, Krauthammer M, Halaban R. Rare SF3B1 R625 mutations in cutaneous melanoma. Melanoma Res. 2014;24(4):332–4.
Biankin AV, Waddell N, Kassahn KS, Gingras MC, Muthuswamy LB, Johns AL, et al. Pancreatic cancer genomes reveal aberrations in axon guidance pathway genes. Nature. 2012;491(7424):399–405.
Armenia J, Wankowicz SAM, Liu D, Gao JJ, Kundra R, Reznik E, et al. The long tail of oncogenic drivers in prostate cancer. Nat Genet. 2019;51(7):645–51.
Damm F, Kosmider O, Gelsi-Boyer V, Renneville A, Carbuccia N, Hidalgo-Curtis N, et al. Mutations affecting mRNA splicing define distinct clinical phenotypes and correlate with patient outcome in myelodysplastic syndromes. Blood. 2012;119(14):3211–8.
Patnaik MM, Lasho TL, Hodnefield JM, Knudson RA, Ketterling RP, Al-Kali A, et al. SF3B1 mutations are prevalent in myelodysplastic syndromes with Ring Sideroblasts but do not hold independent Prognostic Value. Blood. 2012;119(2):569–72.
Bejar R, Stevenson KE, Caughey BA, Abdel-Wahab O, Steensma DP, Galili N, et al. Validation of a Prognostic Model and the impact of mutations in patients with Lower-Risk Myelodysplastic Syndromes. J Clin Oncol. 2012;30(27):3376–82.
Zhou ZX, Gong Q, Wang Y, Li MK, Wang L, Ding HF, et al. The biological function and clinical significance of SF3B1 mutations in cancer. Biomark Res. 2020;8(1):38.
Fuentes-Fayos AC, Perez-Gomez JM, G-Garcia ME, Jimenez-Vacas JM, Blanco-Acevedo C, Sanchez-Sanchez R, et al. SF3B1 inhibition disrupts malignancy and prolongs survival in glioblastoma patients through BCL2L1 splicing and mTOR/beta-catenin pathways imbalances. J Exp Clin Cancer Res. 2022;41(1):39.
López-Cánovas JL, del Rio-Moreno M, García-Fernandez H, Jiménez-Vacas JM, Moreno-Montilla MT, Sánchez-Frias ME, et al. Splicing factor SF3B1 is overexpressed and implicated in the aggressiveness and survival of hepatocellular carcinoma. Cancer Lett. 2021;496:72–83.
Jiménez-Vacas JM, Herrero-Aguayo V, Gómez-Gómez E, León-González AJ, Sáez-Martínez P, Alors-Pérez E, et al. Spliceosome component SF3B1 as novel prognostic biomarker and therapeutic target for prostate cancer. Transl Res. 2019;212:89–103.
Popli P, Richters MM, Chadchan SB, Kim TH, Tycksen E, Griffith O, et al. Splicing factor SF3B1 promotes endometrial cancer progression via regulating KSR2 RNA maturation. Cell Death Dis. 2020;11(10):842.
Boise LH, González-García M, Postema CE, Ding LY, Lindsten T, Turka LA, et al. Bcl-X, a bcl-2-Related gene that functions as a Dominant Regulator of apoptotic cell-death. Cell. 1993;74(4):597–608.
Trecesson SD, Souaze F, Basseville A, Bernard AC, Pecot J, Lopez J, et al. BCL-X-L directly modulates RAS signalling to favour cancer cell stemness. Nat Commun. 2017;8:1123.
Kotake Y, Sagane K, Owa T, Mimori-Kiyosue Y, Shimizu H, Uesugi M, et al. Splicing factor SF3b as a target of the antitumor natural product pladienolide. Nat Chem Biol. 2007;3(9):570–5.
Kaida D, Motoyoshi H, Tashiro E, Nojima T, Hagiwara M, Ishigami K, et al. Spliceostatin A targets SF3b and inhibits both splicing and nuclear retention of pre-mRNA. Nat Chem Biol. 2007;3(9):576–83.
Hasegawa M, Miura T, Kuzuya K, Inoue A, Ki SW, Horinouchi S, et al. Identification of SAP155 as the target of GEX1A (Herboxidiene), an Antitumor Natural product. ACS Chem Biol. 2011;6(3):229–33.
Xargay-Torrent S, López-Guerra M, Rosich L, Montraveta A, Roldán J, Rodríguez V, et al. The splicing modulator sudemycin induces a specific antitumor response and cooperates with ibrutinib in chronic lymphocytic leukemia. Oncotarget. 2015;6(26):22734–49.
Seiler M, Yoshimi A, Darman R, Chan B, Keaney G, Thomas M, et al. H3B-8800, an orally available small-molecule splicing modulator, induces lethality in spliceosome-mutant cancers. Nat Med. 2018;24(4):497–504.
Steensma DP, Wermke M, Klimek VM, Greenberg PL, Font P, Komrokji RS, et al. Results of a clinical trial of H3B-8800, a Splicing Modulator, in patients with myelodysplastic syndromes (MDS), Acute Myeloid Leukemia (AML) or chronic myelomonocytic leukemia (CMML). Blood. 2019;134:673.
Steensma DP, Wermke M, Klimek VM, Greenberg PL, Font P, Komrokji RS, et al. Phase I first-in-human dose escalation study of the oral SF3B1 modulator H3B-8800 in myeloid neoplasms. Leukemia. 2021;35(12):3542–50.
Champion-Arnaud P, Reed R. The Prespliceosome Components Sap-49 and Sap-145 interact in a Complex implicated in tethering U2-Snrnp to the Branch Site. Genes Dev. 1994;8(16):1974–83.
Xiong F, Li S. SF3b4: a versatile player in eukaryotic cells. Front Cell Dev Biol. 2020;8:14.
Li YW, Diao YC, Wang ZX, Wang SR, Peng JL, Kong BH. The splicing factor SF3B4 drives proliferation and invasion in cervical cancer by regulating SPAG5. Cell Death Discov. 2022;8(1):326.
He J, Green AR, Li Y, Chan SYT, Liu DX. SPAG5: an emerging Oncogene. Trends Cancer. 2020;6(7):543–7.
Yang YF, Zhang MF, Tian QH, Fu J, Yang X, Zhang CZY, et al. SPAG5 interacts with CEP55 and exerts oncogenic activities via PI3K/AKT pathway in hepatocellular carcinoma. Mol Cancer. 2018;17:117.
Liu HL, Hu JW, Wei R, Zhou LF, Pan H, Zhu HC, et al. SPAG5 promotes hepatocellular carcinoma progression by downregulating SCARA5 through modifying beta-catenin degradation. J Exp Clin Cancer Res. 2018;37:229.
Li M, Li AQ, Zhou SL, Lv H, Yang WT. SPAG5 upregulation contributes to enhanced c-MYC transcriptional activity via interaction with c-MYC binding protein in triple-negative breast cancer. J Hematol Oncol. 2019;12:14.
Canu V, Donzelli S, Sacconi A, Lo Sardo F, Pulito C, Bossel N, et al. Aberrant transcriptional and post-transcriptional regulation of SPAG5, a YAP-TAZ-TEAD downstream effector, fuels breast cancer cell proliferation. Cell Death Differ. 2021;28(5):1493–511.
Wong YH, Wu CC, Lin CL, Chen TS, Chang TH, Chen BS. Applying NGS data to find Evolutionary Network biomarkers from the early and late stages of Hepatocellular Carcinoma. BioMed Res Int. 2015;2015:2015.
Xu WJ, Huang HX, Yu L, Cao LH. Meta-analysis of gene expression profiles indicates genes in spliceosome pathway are up-regulated in hepatocellular carcinoma (HCC). Med Oncol. 2015;32(4):96.
Iguchi T, Komatsu H, Masuda T, Nambara S, Kidogami S, Ogawa Y, et al. Increased Copy Number of the Gene Encoding SF3B4 indicates poor prognosis in Hepatocellular Carcinoma. Anticancer Res. 2016;36(5):2139–44.
Shen Q, Eun JW, Lee K, Kim HS, Yang HD, Kim SY, et al. Barrier to autointegration factor 1, procollagen-lysine, 2-oxoglutarate 5-dioxygenase 3, and splicing factor 3b subunit 4 as early-stage cancer decision markers and drivers of hepatocellular carcinoma. Hepatology. 2018;67(4):1360–77.
Bernier FP, Caluseriu O, Ng S, Schwartzentruber J, Buckingham KJ, Innes AM, et al. Haploinsufficiency of SF3B4, a component of the Pre-mRNA Spliceosomal Complex, causes Nager Syndrome. Am J Hum Genet. 2012;90(5):925–33.
McPherson E, Zaleski C, Ye Z, Lin S. Rodriguez syndrome with SF3B4 mutation: a severe form of Nager syndrome? Am J Med Genet A. 2014;164(7):1841–5.
Devotta A, Juraver-Geslin H, Gonzalez JA, Hong CS, Saint-Jeannet JP. Sf3b4-depleted Xenopus embryos: a model to study the pathogenesis of craniofacial defects in Nager syndrome. Dev Biol. 2016;415(2):371–82.
Marques F, Tenney J, Duran I, Martin J, Nevarez L, Pogue R, et al. Altered mRNA splicing, Chondrocyte Gene expression and abnormal skeletal development due to SF3B4 mutations in Rodriguez Acrofacial Dysostosis. PLOS Genet. 2016;12(9):e1006307.
Diao YC, Li YW, Wang ZX, Wang SR, Li P, Kong BH. SF3B4 promotes ovarian cancer progression by regulating alternative splicing of RAD52. Cell Death Dis. 2022;13(2):179.
Li P, Xu YZ, Zhang QL, Li Y, Jia WX, Wang X, et al. Evaluating the role of RAD52 and its interactors as novel potential molecular targets for hepatocellular carcinoma. Cancer Cell Int. 2019;19(1):279.
Lieberman R, Xiong DH, James M, Han YH, Amos CI, Wang L, et al. Functional characterization of RAD52 as a lung cancer susceptibility gene in the 12p13.33 locus. Mol Carcinog. 2016;55(5):953–63.
Toma M, Sullivan-Reed K, Śliwiński T, Skorski T. RAD52 as a potential target for synthetic lethality-based Anticancer Therapies. Cancers. 2019;11(10):1561.
Kidogami S, Iguchi T, Sato K, Yoshikawa Y, Hu Q, Nambara S, et al. SF3B4 plays an oncogenic role in esophageal squamous cell carcinoma. Anticancer Res. 2020;40(5):2941–6.
Zhou WT, Ma N, Jiang H, Rong YF, Deng YZ, Feng YY, et al. SF3B4 is decreased in pancreatic cancer and inhibits the growth and migration of cancer cells. Tumour Biol. 2017;39(3):1010428317695913.
Che YY, Fu L. Aberrant expression and regulatory network of splicing factor-SRSF3 in tumors. J Cancer. 2020;11(12):3502–11.
Zhou ZX, Gong Q, Lin ZJ, Wang Y, Li MK, Wang L, et al. Emerging roles of SRSF3 as a therapeutic target for Cancer. Front Oncol. 2020;10:577636.
Xiong J, Chen YS, Wang WP, Sun J. Biological function and molecular mechanism of SRSF3 in cancer and beyond. Oncol Lett. 2022;23(1):21.
Huang YQ, Steitz JA. Splicing factors SRp20 and 9G8 promote the nucleocytoplasmic export of mRNA. Mol Cell. 2001;7(4):899–905.
Park SK, Jeong S. SRSF3 represses the expression of PDCD4 protein by coordinated regulation of alternative splicing, export and translation. Biochem Biophys Res Commun. 2016;470(2):431–8.
Ratnadiwakara M, Archer SK, Dent CI, De Los Mozos IR, Beilharz TH, Knaupp AS, et al. SRSF3 promotes pluripotency through nanog mRNA export and coordination of the pluripotency gene expression program. eLife. 2018;7:e37419.
Lou H, Neugebauer KM, Gagel RF, Berget SM. Regulation of alternative polyadenylation by u1 snRNPs and SRp20. Mol Cell Biol. 1998;18(9):4977–85.
Maciolek NL, McNally MT. Serine/arginine-Rich proteins contribute to negative regulator of splicing element-stimulated polyadenylation in rous sarcoma virus. J Virol. 2007;81(20):11208–17.
Sotiriou C, Wirapati P, Loi S, Harris A, Fox S, Smeds J, et al. Gene expression profiling in breast cancer: understanding the molecular basis of histologic grade to improve prognosis. J Natl Cancer Inst. 2006;98(4):262–72.
Schmidt M, Böhm D, von Törne C, Steiner E, Puhl A, Pilch H, et al. The humoral immune system has a key prognostic impact in node-negative breast cancer. Cancer Res. 2008;68(13):5405–13.
Ke H, Zhao LM, Zhang HL, Feng X, Xu HB, Hao JJ, et al. Loss of TDP43 inhibits progression of triple-negative breast cancer in coordination with SRSF3. Proc Natl Acad Sci U S A. 2018;115(15):E3426–35.
Su YA, Yang J, Tao L, Nguyen H, He P. Undetectable and decreased expression of KIAA1949 (phostensin) encoded on chromosome 6p21.33 in human breast cancers revealed by Transcriptome Analysis. J Cancer. 2010;1:38–50.
Kim YJ, Kim BR, Ryu JS, Lee GO, Kim HR, Choi KH, et al. HNRNPA1, a Splicing Regulator, is an effective target protein for cervical Cancer detection comparison with conventional tumor markers. Int J Gynecol Cancer. 2017;27(2):326–31.
Torres S, García-Palmero I, Marín-Vicente C, Bartolomé RA, Calviño E, Fernández-Aceñero MJ, et al. Proteomic characterization of transcription and splicing factors Associated with a metastatic phenotype in Colorectal Cancer. J Proteome Res. 2018;17(1):252–64.
Kuranaga Y, Sugito N, Shinohara H, Tsujino T, Taniguchi K, Komura K, et al. SRSF3, a Splicer of the PKM Gene, regulates cell growth and maintenance of Cancer-Specific Energy metabolism in Colon Cancer cells. Int J Mol Sci. 2018;19(10):3012.
Kurokawa K, Akaike Y, Masuda K, Kuwano Y, Nishida K, Yamagishi N, et al. Downregulation of serine/arginine-rich splicing factor 3 induces G1 cell cycle arrest and apoptosis in colon cancer cells. Oncogene. 2014;33(11):1407–17.
Song X, Wan XC, Huang TZ, Zeng C, Sastry N, Wu BL, et al. SRSF3-Regulated RNA alternative splicing promotes Glioblastoma Tumorigenicity by affecting multiple Cellular processes. Cancer Res. 2019;79(20):5288–301.
Xu LF, Shen JX, Jia J, Jia R. Inclusion of hnRNP L alternative exon 7 is Associated with Good Prognosis and inhibited by Oncogene SRSF3 in Head and Neck squamous cell carcinoma. BioMed Res Int. 2019;2019:2019.
Wang HL, Lekbaby B, Fares N, Augustin J, Attout T, Schnuriger A, et al. Alteration of splicing factors’ expression during liver disease progression: impact on hepatocellular carcinoma outcome. Hepatol Int. 2019;13(4):454–67.
Chen H, Gao F, He M, Ding XF, Wong AM, Sze SC, et al. Long-read RNA sequencing identifies alternative splice variants in Hepatocellular Carcinoma and Tumor-Specific Isoforms. Hepatology. 2019;70(3):1011–25.
Yea S, Narla G, Zhao X, Garg R, Tal-Kremer S, Hod E, et al. Ras promotes growth by alternative splicing-mediated inactivation of the KLF6 tumor suppressor in hepatocellular carcinoma. Gastroenterology. 2008;134(5):1521–31.
Jia R, Zhang S, Liu MM, Zhang Y, Liu Y, Fan MW, et al. HnRNP L is important for the expression of oncogene SRSF3 and oncogenic potential of oral squamous cell carcinoma cells. Sci Rep. 2016;6:35976.
Liu PQ, Guo ZZ, Yin YT, Jia J, Guo JH, Jia R. Expression of SRSF3 is correlated with carcinogenesis and progression of oral squamous cell carcinoma. Int J Med Sci. 2016;13(7):533–9.
He XL, Ee PLR, Coon JS, Beck WT. Alternative splicing of the multidrug resistance protein 1/ATP binding cassette transporter subfamily gene in ovarian cancer creates functional splice variants and is associated with increased expression of the splicing factors PTB and SRp20. Clin Cancer Res. 2004;10(14):4652–60.
Iborra S, Hirschfeld M, Jaeger M, zur Hausen A, Braicu I, Sehouli J, et al. Alterations in expression pattern of splicing factors in epithelial ovarian Cancer and its clinical impact. Int J Gynecol Cancer. 2013;23(6):990–6.
Villegas VM, Gold AS, Wildner A, Ehlies F, Murray TG. Genomic landscape of retinoblastoma. Clin Exp Ophthalmol. 2014;42(1):2–3.
Kooi IE, Mol BM, Massink MPG, de Jong MC, de Graaf P, van der Valk P, et al. A Meta-analysis of Retinoblastoma Copy numbers refines the list of possible driver genes involved in Tumor Progression. PLoS ONE. 2016;11(4):e0153323.
Lankat-Buttgereit B, Göke R. The tumour suppressor Pdcd4: recent advances in the elucidation of function and regulation. Biol Cell. 2009;101(6):309–17.
Kim J, Park RY, Chen JK, Kim J, Jeong S, Ohn T. Splicing factor SRSF3 represses the translation of programmed cell death 4 mRNA by associating with the 5’-UTR region. Cell Death Differ. 2014;21(3):481–90.
Danovi D, Meulmeester E, Pasini D, Migliorini D, Capra M, Frenk R, et al. Amplification of Mdmx (or Mdm4) directly contributes to tumor formation by inhibiting p53 tumor suppressor activity. Mol Cell Biol. 2004;24(13):5835–43.
Wade M, Li YC, Wahl GM. MDM2, MDMX and p53 in oncogenesis and cancer therapy. Nat Rev Cancer. 2013;13(2):83–96.
Dewaele M, Tabaglio T, Willekens K, Bezzi M, Teo SX, Low DHP, et al. Antisense oligonucleotide-mediated MDM4 exon 6 skipping impairs tumor growth. J Clin Invest. 2016;126(1):68–84.
Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, Mcguire WL. Human-breast Cancer - correlation of Relapse and Survival with amplification of the Her-2 Neu Oncogene. Science. 1987;235(4785):177–82.
Gautrey H, Jackson C, Dittrich AL, Browell D, Lennard T, Tyson-Capper A. SRSF3 and hnRNP H1 regulate a splicing hotspot of HER2 in breast cancer cells. RNA Biol. 2015;12(10):1139–51.
Aigner A, Juhl H, Malerczyk C, Tkybusch A, Benz CC, Czubayko F. Expression of a truncated 100 kDa HER2 splice variant acts as an endogenous inhibitor of tumour cell proliferation. Oncogene. 2001;20(17):2101–11.
Romanelli MG, Diani E, Lievens PMJ. New Insights into functional roles of the polypyrimidine tract-binding protein. Int J Mol Sci. 2013;14(11):22906–32.
Sawicka K, Bushell M, Spriggs KA, Willis AE. Polypyrimidine-tract-binding protein: a multifunctional RNA-binding protein. Biochem Soc Trans. 2008;36:641–7.
Ray D, Kazan H, Chan ET, Castillo LP, Chaudhry S, Talukder S, et al. Rapid and systematic analysis of the RNA recognition specificities of RNA-binding proteins. Nat Biotechnol. 2009;27(7):667–70.
Reid DC, Chang BL, Gunderson SI, Alpert L, Thompson WA, Fairbrother WG. Next-generation SELEX identifies sequence and structural determinants of splicing factor binding in human pre-mRNA sequence. RNA. 2009;15(12):2385–97.
He X, Arslan AD, Pool MD, Ho TT, Darcy KM, Coon JS, et al. Knockdown of splicing factor SRp20 causes apoptosis in ovarian cancer cells and its expression is associated with malignancy of epithelial ovarian cancer. Oncogene. 2011;30(3):356–65.
Wang C, Norton JT, Ghosh S, Kim J, Fushimi K, Wu JY, et al. Polypyrimidine tract-binding protein (PTB) differentially affects malignancy in a cell line-dependent manner. J Biol Chem. 2008;283(29):20277–87.
He X, Pool M, Darcy KM, Lim SB, Auersperg N, Coon JS, et al. Knockdown of polypyrimidine tract-binding protein suppresses ovarian tumor cell growth and invasiveness in vitro. Oncogene. 2007;26(34):4961–8.
Cheung HC, Hai T, Zhu W, Baggerly KA, Tsavachidis S, Krahe R, et al. Splicing factors PTBP1 and PTBP2 promote proliferation and migration of glioma cell lines. Brain. 2009;132:2277–88.
Izaguirre DI, Zhu W, Hai T, Cheung HC, Krahe R, Cote GJ. PTBP1-Dependent regulation of USP5 Alternative RNA splicing plays a role in Glioblastoma Tumorigenesis. Mol Carcinog. 2012;51(11):895–906.
Qu M, Song N, Chai G, Wu XL, Liu W. Pathological niche environment transforms dermal stem cells to keloid stem cells: a hypothesis of keloid formation and development. Med Hypotheses. 2013;81(5):807–12.
Appleton I, Brown NJ, Willoughby DA. Apoptosis, necrosis, and proliferation - possible implications in etiology of keloids. Am J Pathol. 1996;149(5):1441–7.
Jiao H, Dong P, Yan L, Yang ZG, Lv XY, Li QC, et al. TGF-β1 induces polypyrimidine tract-binding protein to alter fibroblasts proliferation and fibronectin deposition in Keloid. Sci Rep. 2016;6:38033.
Jin W, Bruno IG, Xie TX, Sanger LJ, Cote GJ. Polypyrimidine tract-binding protein down-regulates fibroblast growth factor receptor 1 alpha-exon inclusion. Cancer Res. 2003;63(19):6154–7.
Carstens RP, Wagner EJ, Garcia-Blanco MA. An intronic splicing silencer causes skipping of the IIIb exon of fibroblast growth factor receptor 2 through involvement of polypyrimidine tract binding protein. Mol Cell Biol. 2000;20(19):7388–400.
Côté J, Dupuis S, Wu JY. Polypyrimidine track-binding protein binding downstream of caspase-2 alternative exon 9 represses its inclusion. J Biol Chem. 2001;276(11):8535–43.
Izquierdo JM. Hu antigen R (HuR) functions as an alternative pre-mRNA splicing regulator of Fas apoptosis-promoting receptor on exon definition. J Biol Chem. 2008;283(27):19077–84.
Izquierdo JM, Majós N, Bonnal S, Martínez C, Castelo R, Guigó R, et al. Regulation of fas alternative splicing by antagonistic effects of TIA-1 and PTB on exon definition. Mol Cell. 2005;19(4):475–84.
Christofk HR, Vander Heiden MG, Harris MH, Ramanathan A, Gerszten RE, Wei R, et al. The M2 splice isoform of pyruvate kinase is important for cancer metabolism and tumour growth. Nature. 2008;452(7184):230–43.
David CJ, Chen M, Assanah M, Canoll P, Manley JL. HnRNP proteins controlled by c-Myc deregulate pyruvate kinase mRNA splicing in cancer. Nature. 2010;463(7279):364–8.
Calabretta S, Bielli P, Passacantilli I, Pilozzi E, Fendrich V, Capurso G, et al. Modulation of PKM alternative splicing by PTBP1 promotes gemcitabine resistance in pancreatic cancer cells. Oncogene. 2016;35(16):2031–9.
Glinos DA, Garborcauskas G, Hoffman P, Ehsan N, Jiang LH, Gokden A, et al. Transcriptome variation in human tissues revealed by long-read sequencing. Nature. 2022;608(7922):353–9.
Gao DD, Morini E, Salani M, Krauson AJ, Chekuri A, Sharma N, et al. A deep learning approach to identify gene targets of a therapeutic for human splicing disorders. Nat Commun. 2021;12(1):3332.
Acknowledgements
Not applicable.
Funding
This work was supported by the National Research Foundation of Korea (RS-2023-0024733131482092640001, NRF-2022R1A2C1003870, and NRF-2021R1I1A1A01051949) and the Korea Environment Industry & Technology Institute (KEITI) grants (NO. 2022002960005) through the Environment Health Action Program, funded by the Korea Ministry of Environment (MOE).
Author information
Authors and Affiliations
Contributions
KKK conceptualized the manuscript; SC and KKK drafted the manuscript; NC prepared the figures; KKK, SC, NC, and EK contributed in the discussion and edited the manuscript. SC and NC contributed equally to this work. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Choi, S., Cho, N., Kim, EM. et al. The role of alternative pre-mRNA splicing in cancer progression. Cancer Cell Int 23, 249 (2023). https://doi.org/10.1186/s12935-023-03094-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12935-023-03094-3