Abstract
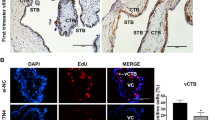
Objective: High-mobility group A1 (HMGA1) protein is known to express in trophoblast; however, the role of migration has not been reported to date. In this study, we investigated the role of HMGA1 on the pathogenesis of preeclampsia using immortalized human trophoblast cell (HTR-8/SVneo). Materials and Methods: We investigated HMGA1 expression in cytotrophoblasts derived from our preeclampsia model mouse, the CD40L mouse, using immunofluorescence. Wound healing and transwell migration assays were also performed using HTR-8/SVneo (extravillous trophoblast) cells transfected with DNA or siRNA of HMGA1. The effect of extranuclear translocation of HMGA1 on the migration of extravillous trophoblastic cells was evaluated using deoxycholic acid (DCA). Results: HMGA1 was expressed exclusively in the nuclei of trophoblasts derived from control mice; cytoplasmic expression was observed only in CD40L mice with preeclampsia. Furthermore, overexpression of HMGA1 in the nuclei of HTR-8/SVneo cells stimulated cell proliferation and migration. Translocation of nuclear HMGA1 to cytoplasm treated with DCA reduced cell migration. Conclusions: Collectively, these findings demonstrate that proper subcellular localization of HMGA1 is important for its function in trophoblast cells, and suggest that aberrant cytoplasmic expression of HMGA1 contributes to the pathogenesis of preeclampsia through impairment of trophoblast migration.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Pijnenborg R, Dixon G, Robertson WB, Brosens I. Trophoblastic invasion of human decidua from 8 to 18 weeks of pregnancy. Placenta. 1980;1(1):3–19.
Pijnenborg R, Bland JM, Robertson WB, Brosens I. Uteroplacental arterial changes related to interstitial trophoblast migration in early human pregnancy. Placenta. 1983;4(4):397–413.
Khong TY, De Wolf F, Robertson WB, Brosens I. Inadequate maternal vascular response to placentation in pregnancies com-plicated by preeclampsia and by small for gestational age infants. Br J Obstet Gynecol. 1986;93(10):1049–1059.
Johnson KR, Lehn DA, Elton TS, Barr PJ, Reeves R. Complete murine cDNA sequence, genomic structure, and tissue expression of the high mobility group protein HMG-I(Y). J Biol Chem. 1988; 263(34):18338–18342.
Reeves R, Nissen MS. The A.T-DNA-binding domain of mam-malian high mobility group I chromosomal proteins. A novel peptide motif for recognizing DNA structure. J Biol Chem. 1990;265(15):8573–8582.
Nagpal S, Ghosn C, DiSepio D, et al. Retinoid-dependent recruitment of a histone H1 displacement activity by retinoic acid receptor. J Biol Chem. 1999;274(32):22563–22568.
Chiappetta G, Aventaggiato V, Visconti R, et al. High level expression of the HMGI(Y) gene during embryonic development. Oncogene. 1996;13(11):2439–2446.
Fusco A, Fedele M. Roles of HMGA proteins in cancer. Nat Rev Cancer. 2007;7(12):899–910.
Bamberger AM, Makrigiannakis A, Ro¨ser K, et al. Expression of the high-mobility group protein HMGI (Y) in human trophoblast: potential role in trophoblast invasion of maternal tissue. Virchows Arch. 2003;443(5):649–654.
Matsubara K, Matsubara Y, Mori M, et al. Immune activation during implantation phase causes preeclampsia-like symptoms through the CD40-CD40 ligand pathway in pregnant mice. Hypertens Res. 2016;39(6):407–414.
Chaiwarapongsa T, Chaemsaithong P, Yeo L, Romero R. Pre-eclampsia part 1: current understanding of its pathophysiology. Nat Rev Nephrol. 2014;10(8):466–480.
Andersson U, Erlandsson-Harris H. HMGB1 is a potent trigger of arthritis. J Intern Med. 2004;255(3):344–350.
Wang H, Yang H, Tracey KJ. Extracellular role of HMGB1 in inflammation and sepsis. J Intern Med. 2004;255(3):320–331.
de Souza AW, Westra J, Limburg PC, Bijl M, Kallenberg CG. HMGB1 in vascular disease: Its role in vascular inflammation and atherosclerosis. Autoimmun Rev. 2012;11(12):909–917.
Lotze MT, Tracey KJ. High-mobility group box 1 protein (HMGB1) nuclear weapon in the immune arsenal. Nat Rev Immu-nol. 2005;5(4):331–342.
van Beijnum JR, Buurman WA, Griffioen AW. Convergence and amplification of toll-like receptor (TLR) and receptor for advanced glycation end products (RAGE) signaling pathways via high mobility group B1 (HMGB1). Angiogenesis. 2008;11(1):91–99.
Bartram HP, Scheppach W, Schmid H, et al. Proliferation of human colonic mucosa as an intermediate biomarker of carcino-genesis: effects of butyrate, deoxycholate, calcium, ammonia, and pH. Cancer Res. 1993;53(14):3283–3288.
Fujii K, Luo Y, Sasahira T, Denda A, Ohmori H, Kuniyasu H. Co-treatment with deoxycholic acid and azoxymethane accelerates section of HMGB1 in IEC6 intestinal epithelial cells. Cell Prolif. 2009;42(5):701–709.
Naruse K, Lash GE, Innes BA, et al. Localization of matrix metal-loproteinase (MMP)-2, MMP-9 and tissue inhibitors for MMPs (TIMPs) in uterine natural killer cells in early human pregnancy. Hum Reprod. 2009;24(3):553–561.
Bussemakers MJ, Van de Ven WJ, Debruyne FM, Schalken J. Identification of high mobility group protein I (Y) as potential progression marker for prostate cancer by differential hybridiza-tion analysis. Cancer Res. 1991;51(2):606–611.
Tamimi Y, van der Poel HG, Denyn MM, et al. Increased expression of high mobility group protein I (Y) in high grade prostate cancer determined by in situ hybridization. Cancer Res. 1993; 53(22):5512–5516.
Chen Q, Yin YX, Wei J, et al. Increased expression of high mobility group box 1 (HMGB1) in the cytoplasm of placental syncytiotrophoblast from preeclamptic placentae. Cytokine. 2016;85:30–36.
Lash GE, Otun HA, Innes BA, et al. Regulation of extravillous trophoblast invasion by uterine natural killer cells is dependent on gestational age. Hum Reprod. 2010;25(5):1137–1145.
Godbole G, Suman P, Gupta SK, Modi D. Decidualized endome-trial stromal cell derived factors promote trophoblast invasion. Fertil Steril. 2011;95(4):1278–1283.
Renaud SJ, Postovit LM, Macdonald-Goodfellow SK, McDonald GT, Caldwell JD, Graham CH. Activated macrophages inhibit human cytotrophoblast invasiveness in vitro. Biol Reprod. 2005; 73(2):237–243.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Uchikura, Y., Matsubara, K., Muto, Y. et al. Extranuclear Translocation of High-Mobility Group A1 Reduces the Invasion of Extravillous Trophoblasts Involved in the Pathogenesis of Preeclampsia: New Aspect of High-Mobility Group A1. Reprod. Sci. 24, 1630–1638 (2017). https://doi.org/10.1177/1933719117697254
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719117697254