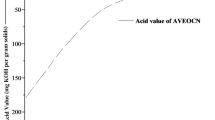
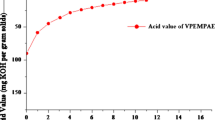
Abstract
Diels-Alder adduct of gum rosin and maleic anhydride (MPA) were treated with trimethylolpropane to obtain trimethylolpropane modified maleopimaric acid adduct (TMPA). This adduct was then epoxidized using epichlorohydrin and KOH as catalyst to synthesize an epoxy resin (TMPAE), which was further esterified using methacrylic acid in the presence of triphenyl phosphine as catalyst and hydroquinone as inhibitor to produce bio-based multifunctional vinyl ester resin (VTMPAE). This article also reports the synthesis of lignin model compounds i.e. methacrylated eugenol (ME) and methacrylated guaiacol (MG) as bio-based reactive monomers for vinyl ester resins. The chemical structures of all synthesized products were analyzed using FTIR, 1HNMR and 13C NMR spectroscopic techniques. The thermal and mechanical properties of the samples were evaluated using Differential Scanning Calorimetry (DSC), Thermogravimetric Analyzer (TGA) and Universal Testing Machine (UTM), respectively. Chemical and corrosion resistance of the above cured VTMPAE samples coated on steel panels were also evaluated as a function of % weight loss by immersing the VTMPAE samples in 1 M HCl, 1 M NaOH, and 1 M NaCl solutions for 90 days. The morphological changes that appear upon such an exposure were also studied with the help of Scanning electron microscopy (SEM).
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
J. R. Brown and Z. Mathys, Compos. Part A 28, 675 (1997).
S. A. Lane, T. E. Even, T. W. Austill, and P. A. Johnson, US Patent 6 187 442 (2001).
A. P. Mouritz and Z. Mathys, Compos. Struct. 47, 643 (1999).
J. L. Love, US Patent 5?961 825 (2001).
S. Zhang, L. Ye, and Y. W. Mai, Appl. Compos. Mater. 7, 125 (2000).
S. Jaswal and B. Gaur, Rev. Chem. Eng. 30, 567 (2014).
S. Jaswal and B. Gaur, Chem. Eng. Trans. 32, 1591 (2013).
G. F. Chen, Prog. Org. Coat. 20, 139 (1992).
X. Liu, W. Xin, and J. Zhang, Green Chem. 11, 1018 (2009).
J. J. La Scala, J. M. Sands, J. A. Orlicki, et al., Polymer 45, 7729 (2004).
J. J. La Scala, J. A. Orlicki, J. M. Sands, and G. R. Palmese, in Proceedings of Composites 2004, Convention and Trade Show, American Composites Manufacturers Association, Tampa, Fl. USA, 2004.
J. J. La Scala, J. A. Orlicki, C. Winston, et al., Polymer 46, 2908 (2005).
D. Grob, Recommended Methods for the Analysis of Alkyd Resins (International Union of Pure and Applied Chemistry, Verlag Butterworth and Co. Ltd., London, 1973).
D. W. Knoll, D. H. Nelson, and P. W. Keheres, in Proceedings of 134th Am. Chem. Soc. Meet, Chicago, Division of Paint, Plastics and Printing Ink Chemistry, 1958, Paper No. 5, p. 20.
J. F. Stanzione, J. M. Sadler, J. J. La Scala, and R. P. Wool, Chem. Sus. Chem. 7, 1291 (2012).
B. Gaur and J. S. P. Rai, Polymer 33, 4210 (1992).
B. Gaur and J. S. P. Rai, Eur. Pol. J. 29, 1149 (1993).
M. Malik, V. Choudhary, and I. K. Varma, J. Appl. Pol. Sci. 82, 416 (2001).
R. Bhatnagar and I. K. Varma, J. Therm. Anal. Calorim. 35, 1241 (1989).
O.C. Zaske, “Unsaturated polyester and vinyl ester resins,” in Handbook of Thermoset Plastics, Ed. by S. H. Goodman (William Andrew Inc., New Jersey, 1998), pp. 122–168.
B. Gawdzik and T. Matynia, J. Appl. Pol. Sci. 81, 2062 (2001).
S. Aggarwal, R. Singhal, and J. S. P. Rai, J. Macromol. Sci. Pure Appl. Chem. 36, 741 (1999).
M. Sultania, J. S. P. Rai, and D. Srivastava, Eur. Pol. J. 46, 2019 (2010).
J. P. J. Higgins and K. E. Weale, J. Pol. Sci., Part A-1 6, 3007 (1968).
F. Heatley, P. A. Lovell, and J. Mcdonald, Eur. Pol. J. 29, 255 (1993).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Jaswal, S., Gaur, B. Structure-property correlation study of bio-based multifunctional vinyl ester resin in presence of methacrylated lignin model compounds. Polym. Sci. Ser. B 57, 417–433 (2015). https://doi.org/10.1134/S1560090415050048
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1560090415050048