Abstract
The rapid decline in the area occupied by mangrove forests has convinced the Vietnamese government to launch a program for conserving and restoring the mangrove ecosystems throughout the country. This program included an experimental planting of Rhizophora apiculata seedlings in Dam Bay in Khanh Hoa province. In the course of this work, the state of the macrobenthos was monitored in new mangrove plantations, and its quantitative and qualitative characteristics were compared with those in natural mangrove associations of this area. The bulk of the material was collected in three intertidal zones in the area of mangrove plantations in Dam Bay. In addition, natural mangrove associations were investigated: one in Dam Bay, and the other (the most mature and largest association of the area) in the neighboring Nha Phu Bay. The natural mangrove association in Dam Bay was represented by a narrow belt of Rhizophora apiculata. The intertidal zone in the area of mangrove plantations is also represented by a narrow belt of Rhizophora apiculata. Until 2004, the middle and lower zones were formed by the mudflat. In 2004, mangrove seedlings were planted in the middle intertidal zone below the mature trees of the upper zone. As the seedlings grew, they began to slow down tidal currents, and nutrients began to linger in the ecosystem. The developing crown of young trees began to give litter and shade, favorable for the development of microalgae. In addition, young trees formed a hard substrate. As a result, there was an increase in the species diversity of the macrobenthic eurybiotic species and an increase in the abundance of opportunistic species. Young trees in the middle intertidal zone and formed a single mangrove forest of a large area together with adult trees of the upper zone, which caused the appearance of specialized species of gastropods and bivalve mollusks, as well as crabs. During the monitoring period, the composition and structure of the macrobenthos of mangrove plantations were gradually approaching to those in the mature natural mangrove association in Nha Phu. However, significant differences still remain between these associations after ten years of observing the growth of mangrove seedlings. The macrobenthos of Nha Phu is represented by a large number of specialized mangrove species of gastropod mollusks, and it also has a completely different structure of the taxocene of bivalve mollusks. This is likely explained by the high floristic diversity in Nha Phu and the maturity of this mangrove association. As a result of qualitative and quantitative collection of the macrobenthos, 139 species of invertebrates were found in the three mangrove associations studied. The key groups of the macrobenthos are gastropods, bivalve mollusks, and crabs. The data obtained on the composition and diversity of the macrobenthos fit into the general pattern of the mangrove fauna of the Indo-West Pacific and Southeast Asia.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
Mangrove forests grow on the border of the sea and land on the coasts of the tropical and equatorial zones. Mangroves of Southeast Asia are of the greatest interest because they cover large areas and have a higher diversity of flora and fauna than other regions (Ellison et al., 1999). In recent decades, mangrove areas have been declining everywhere, and Vietnam is no exception (Walters et al., 2000). Earlier, Vietnamese mangroves occupied about 4000 km2, but herbicides and napalm used during the war (1962–1971) destroyed about 1050 km2 (Hong and San, 1993). Population and economic growth after the Vietnam War also led to a significant reduction in the area occupied by mangrove forests. The development of shrimp and crab farms, the construction of salt ponds, agriculture, coal mining, and oil pollution of coastal waters lead to the degradation of associations, which turned into wastelands in some places (Hong and San, 1993). The majority of the surviving mangroves are located in the Mekong Delta, and even these are predominantly secondary forests and plantations (Spalding et al., 1997).
However, it has long been known how important the role of mangrove forests is for the functioning of other ecosystems in coastal waters and for the economies of coastal countries. Mangroves serve as a habitat for a diverse fauna of invertebrates, fish, reptiles, and birds and support the productivity of other coastal communities, providing food and a place for the growth of larvae and juveniles of many species, including commercial fish and invertebrates (Kathiresan and Bingham, 2001; Twilley et al., 1992). They stabilize the coastline, filter fresh runoff, and significantly reduce damage from typhoons and tsunamis (Mazda et al., 2002, 2006). Mangroves can be an ecotourism destination (Ellison, 2008). In addition, traditional forms of mangrove use are widespread along the entire coast of Vietnam: as a building material, as a fuel, as a place for obtaining food, etc. (Hong and San, 1993).
In this regard, the need to preserve and restore mangrove forests has been recognized at the state level. The Vietnamese government has established several national parks and protected areas, as well as artificial mangrove plantations (Field 1998; Hong and San, 1993).
At present, a large amount of information has been collected and many works and reviews have been published on individual areas of study of mangroves.
Mangrove trees form a habitat for a diverse invertebrate fauna. The key invertebrate groups in the mangrove ecosystem are mollusks and crabs, which have a high diversity and population density and a significant biomass. In addition, the functioning of the ecosystem largely depends on the vital activity of these groups (Berry, 1963; Kathiresan and Bingham, 2001; Lee, 1998, 1999; Macnae, 1968).
The invertebrate species living here have different relations to the mangrove ecosystem. There are both specialized species found only in mangroves and eurybiotic species, i.e., species adapted to a wide range of conditions and inhabiting different types of ecosystems. Specialized species of gastropods include the majority of representatives of the family Potamididae, the genus Littoraria (Littorinidae), and some representatives of the families Neritidae, Cerithiidae, Assimineidae, etc. (Frey, 2010; Houbrick, 1985, 1992; Reid, 1985, 1986, 2014; Reid et al., 2008, 2010). Among bivalve mollusks, these are representatives of the genus Geloina, some representatives of the family Lucinidae, and others (Morton, 1976; Kathiresan and Bingham, 2001; Glover and Taylor, 2016). For crabs, there is no detailed information on specialized mangrove species, but it is known that a high diversity and abundance of representatives of the superfamily Grapsoidea is characteristic of mature and undisturbed mangroves, while a high abundance of species of the superfamily Ocypodoidea is inherent in young and disturbed mangrove ecosystems (Macintosh et al., 2002).
In addition to the three key macrobenthos groups, representatives of other taxa inhabit mangrove associations. Jellyfish (Tsingalia, 2014), burrowing anemones (Mitra and Pattanayak, 2013), annelids (Metcalfe and Glasby, 2008), sipunculids (Adrianov and Maiorova, 2012; Gopal and Chauhan, 2006; Macnae, 1968), hermit crabs (Gherardi and Vannini, 1993; Kathiresan and Bingham, 2001; Macnae, 1968), praying mantises (Ng, 2013), shrimps (Kathiresan and Bingham, 2001; Macnae, 1968; Yu et al., 1997), brachiopods (Mitra and Pattanayak, 2013a; Printrakoon and Kamlung-ek, 2013), acorn worms (Dittmann, 2002; Kumar, 2000), etc., live in mangroves of various regions of the Indo-West Pacific.
There are relatively few publications dealing with various aspects of the mangrove ecosystems in Vietnam. In addition, a significant part of them are written in Vietnamese. The fauna of Vietnamese mangroves is very poorly studied. Hong and San (1993) provide data from studies on crabs, polychaetes, and mollusks; Chertoprud et al. (2012) analyze the diversity and distribution of crabs found in mangroves in the vicinity of Nha Trang. Taxonomic studies on shrimp from Vietnamese mangroves have been published (Anker et al., 2006; Dworschak et al., 2006; Marin, 2008, 2014). In addition, there are a number of studies on the meiobenthos (Mokievsky et al., 2011; Tchesunov et al., 2010; Xuan et al., 2007).
The diversity and structure of invertebrate fauna is altered by disturbance, exploitation, management, or restoration of mangroves. Thus, these parameters can reflect the status of the mangrove association and can be used as an indicator of changes both in natural mangrove forests and in plantations (Macintosh et al., 2002).
In 2005, an experiment on planting Rhizophora apiculata Blume seedlings was started in Dam Bay (Tre Island, Nha Trang Bay). The aim of the work was to trace the qualitative and quantitative changes in the macrobenthos in plantations over time, as well as to compare the macrobenthos of plantations and natural mangrove associations. We carried out a ten-year monitoring of the macrobenthos fauna in three intertidal zones in the area of mangrove plantations of Dam Bay; the macrobenthos of these horizons correlated with the state of the nearest natural mangrove associations. Some data on gastropod mollusks have already been published as part of this large-scale project. The paper by (Zvonareva et al., 2016) provides a complete list of gastropods. In addition, some data on the quantitative characteristics of gastropods up to 2012 was published (Zvonareva et al., 2015).
This study covers all key macrobenthos groups of natural and artificial mangroves in central Vietnam, which are not often seen by researchers. For the first time, all stages of the formation of a community of macrobenthos invertebrates in mangrove plantations from the moment of planting of seedlings to their ten-year age have been recorded, which has no analogues.
MATERIAL AND METHODS
The coast of central Vietnam in Nha Trang and Nha Phu bays (Fig. 1) is often subject to storms, so mangroves are found here only in well-protected bays and lagoons (Hong and San, 1993). In Nha Trang and Nha Phu bays mangroves are scarce and often in poor condition due to anthropogenic influence. At the moment, they are present around the mariculture ponds at the mouth of the Be River within the boundaries of the city of Nha Trang and in the inner part of Nha Phu Bay. The largest natural mangrove association without mariculture is located on the northeastern coast of Nha Phu Bay. In addition, mangroves, represented by narrow belts of vegetation, are present on the islands of Nha Phu Bay and in the interior of Dam Bay.
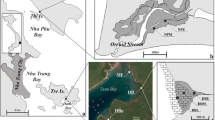
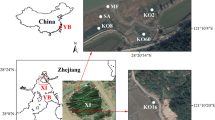
Map of areas surveyed (according to Zvonareva et al., 2015): (a) map of the study area showing its location on a map of Vietnam; (b) scheme of mangrove association in Nha Phu Bay; (c) satellite image of the natural mangrove association and mangrove plantings in Dam Bay (2014); (d) scheme of mangrove plantings in Dam Bay, indicating the location of the stations studied.
Before the mangrove seedlings were planted, the upper intertidal zone in a small depression on the coastline of the eastern coast of Dam Bay (Tre Island, Nha Trang Bay, Fig. 1) was occupied by a narrow zone of Rhizophora apiculata mangroves with a small admixture of Avicennia marina (Forsk.) Vierh. (coordinates: 12°13.116′ N, 109°18.230′ E). In 2004, seedlings of Rhizophora apiculata were planted in the sandy-silty mid intertidal zone (12°12.097′ N, 109°18.213′ E). In 2007, seedlings were planted in the lower intertidal zone (12°12.086′ N, 109°18.166′ E).
The material was collected not only from mangrove plantations, but also from two natural associations. There is a natural mangrove association in a small bay to the south of the plantings in Dam Bay (coordinates: 12°11.858′ N; 109°17.949′ E, Fig. 1), the type and size of which correspond to the upper intertidal zone in the area of plantation before planting of seedlings on the mid and lower intertidal zones. In addition, a large natural association was investigated in Nha Phu Bay near Orchid Stream Park (NP station, coordinates: 12°24.362′ N; 109°14.407′ E, Fig. 1). It differs from the mangroves in Dam Bay by the structure of the vegetation. Trees here grow along the stream, which is somewhat freshened by runoff from the mountains (up to 18‰ in the upper reaches). Mangrove trees are represented by seven species with dominance of the Rhizophoreacea family in the lower reaches and Xylocarpus granatum and Excoecaria agallocha in the upper reaches. This association is subject to anthropogenic influence as it is located next to a recreational park.
Quantitative samples have been collected in the planting area since 2005. We used various sampling gear: bottom sampler, tubes, and frames of different sizes, and a frame with an area of 0.11 m2 was used in most cases. The number of samples at the station also varied from year to year; four samples were usually collected. The soil was removed from the frame to a depth of 15–20 cm, then the animals were separated from the soil by elutriation in a bucket. The remaining coarse fraction of the precipitate was washed through a sieve with a mesh size of 3 mm. Starting in 2014, soil from a 0.11 m2 frame was immediately placed on a sieve, and only soil from a small 0.01 m2 frame was elutriated. Animals were picked from the samples after sorting at the laboratory in Nha Trang. Worm-like animals were fixed with 4% formalin, and the rest, with 75% alcohol. For each quantitative sample, the abundance and biomass were then calculated.
In addition to quantitative samples, qualitative samples were also collected from tree trunks and roots, and large and rare animals were also collected (hand picking). Table 1 shows a list of stations and sample areas in Dam Bay and Nha Phu bay.
In this work, it was decided not to include data on the phylum Annelida, since this is a very difficult group for species identification, determination is labor-intensive, and their processing was not included in the tasks of this work.
All collections are stored in the Laboratory of Morphology and Ecology of Marine Invertebrates, Severtsov Institute of Ecology and Evolution, Russian Academy of Sciences.
RESULTS
In the mangrove associations studied by us, 139 species of macroinvertebrates were found (excluding Annelida, the species identification of which was virtually impossible). Gastropods and bivalve mollusks and crabs were the most diverse, while other groups were represented by 1–2 species.
Figure 3 shows the species diversity of macrobenthos groups at the stations studied.
Species diversity of macrobenthos invertebrates (except for annelids) in three intertidal zones of mangrove plantations and in two natural mangrove associations found over the entire study period. UIZ, upper intertidal zone in the area of mangrove plantings; MIZ, mid intertidal zone; LIZ, lower intertidal zone; DBn, natural mangrove association at Dam Bay; NP, natural mangrove association in Nha Phu Bay.
Gastropods
Gastropods are the most diverse group represented by 64 species from 24 families. The most diverse families were Neritidae (eight species), Cerithiidae (seven species), Potamididae (six species), Littorinidae (six species), Onchidiidae (six species), and Ellobiidae (five species).
Gastropods were the most diverse group at all stations, except for the lower intertidal zone. Forty-five species were found in the mangrove plantations, with the upper and mid intertidal zones having 27 and 28 species, respectively, and fewer in the lower intertidal zone (22 species). Thirty-five species were found in the natural association in Nha Phu, and 27 species were found in the natural association in Dam Bay.
Of all the species found, 17 can be called specialized species. The most diverse among them is the natural association in Nha Phu (16 species, see Fig. 2). In the plantings, the lower intertidal zone was the poorest (only three specialized species), and the upper and middle ones had 10 and 11 species, respectively. It should be noted that in the last survey two specialized mangrove species were found in the mid intertidal zone of plantings: Terebralia sulcata (Born 1778) and Cassidula nucleus (Gmelin 1791), which had not previously been found in plantings, but were present in all collections from the natural mangrove association in Nha Phu. Eight specialized mangrove species are found in the natural mangrove association at Dam Bay.
Figure 3 shows that all mangrove associations are dominated by eurybiontic species, which are characteristic mainly of the epifauna of the soft intertidal bottom. However, a significant proportion of species in the natural mangrove association in Dam Bay are eurybiotic species characteristic of the rocky intertidal bottoms, for example, Pictocolumbella ocellata (Link 1807), Planaxis sulcatus (Born 1780), etc. The majority of the mollusks found in the lower intertidal zone are species typical for the subtidal, for example, Lataxiena blosvillei (Deshayes 1832) and representatives of the families Rannelidae, Cypraeidae, etc.
The species diversity in mangrove plantations has changed over time. Figure 4 illustrates the growth of diversity in three zones. Figure 3 shows that this growth occurs both at the expense of specialized species and at the expense of eurybiotic species.
In terms of the quantitative characteristics, gastropods dominate among other groups of invertebrates at all stations, except for the lower intertidal zone. The values of abundance and biomass in the upper and mid intertidal zones have a pronounced tendency to increase (Fig. 5). The abundance and biomass in the natural mangrove association at Dam Bay were noticeably lower than those of the upper and mid intertidal zones during the late observation period. The lower zone was extremely poor in gastropods throughout the entire observation period.
The abundance and biomass of gastropods in some surveys were very high. Thus, the maximum density of gastropods was recorded in 2015 in the mid intertidal zone of plantings: 2698 ± 1127 ind./m2, and the maximum biomass was recorded in the fall of 2011: 447 ± 318 g/m2.
At all stations, the dominant species were eurybiotic and opportunistic species Battilaria sp., Clithon oualaniense (Lesson 1831), and Pirenella cingulata (Gmelin 1791). In some surveys at the late period of monitoring, their total contribution to abundance (Fig. 6a) and biomass exceeded 90%. An increase in the abundance and biomass in the upper and mid intertidal zones also occurs exclusively due to these three species, which are capable of forming very dense aggregations (Fig. 6b).
Dominant species of gastropods in plantings and natural mangrove associations: (a) the percentage of the abundance of dominant species of gastropods in the upper two intertidal zones in the area of mangrove plantations in Dam Bay (the lower zone is not shown, since the abundance of gastropods is negligible here) and in two natural mangrove associations at the time of the last survey (2014 for Nha Phu, 2015 for DBn); (b) aggregation of gastropods Battilaria sp., Clithon oualaniense, and Pyrenella cingulata in the mid intertidal zone.
Bivalve Mollusks
In the course of this study, 43 species of bivalve mollusks belonging to 18 families were found. In terms of species diversity, this group follows gastropods at all stations, except for the lower intertidal zone, where the diversity of bivalve mollusks exceeds the diversity of gastropods, and Nha Phu, where bivalve mollusks are extremely scarce.
The lower intertidal zone is the most species-diverse among the mangrove associations studied (26 species). Slightly fewer species were found in the mid intertidal zone (21 species), and the upper intertidal zone (15 species) was the poorest in mangrove plantations. The species diversity in the natural mangrove association in Dam Bay roughly corresponded to that of the upper zone (14 species). The natural mangrove association in Nha Phu has the lowest species diversity with only three species of bivalve mollusks found here.
Only two specialized species, Geloina erosa (Lightfoot 1786) and Pegophysema philippiana (Reeve 1850), were found in the associations studied. Both were found in the natural mangrove association in Nha Phu and in the mangrove plantations of Dam Bay, and only P. philippiana was found in the natural mangrove association in Dam Bay. The rest of the bivalve mollusks found during the study are eurybiotic species typical for the infauna of soft s intertidal and subtidal bottoms (for example, representatives of the families Tellinidae and Cardiidae). In addition to infauna, representatives of the epifauna are found on the roots and trunks of mangrove trees, for example, Saccostrea cuccullata (Born 1778) and representatives of the family Mytilitdae. The proportion of subtidal species in the lower intertidal zone is higher than at other stations. In addition, large species, for example, representatives of the families Veneridae and Pinnidae, are also found at this station in qualitative samples.
At the initial period of monitoring (2005–2006), the diversity of bivalve mollusks did not exceed 1–2 species. In subsequent years, their diversity reached eight species in the upper zone, nine species in the middle zone, and 12 species in the lower intertidal zone (Fig. 7).
The abundance and biomass of bivalve mollusks experienced strong fluctuations throughout the period of the study, with no apparent tendency to increase or decrease over time. The highest abundance was recorded in the lower zone in 2008 (153 ± 138 ind./m2). The highest biomass was recorded in 2013 in the natural mangrove association in Dam Bay (338 ± 151 ind./m2). The greatest contribution to the abundance and biomass in the upper and mid intertidal zones, as well as in the natural mangrove association in Dam Bay, is made by the eurybiotic species Gafrarium pectinatum (Linnaeus 1758).
Crabs
Eighteen crab species were found in the three mangrove associations over the entire observation period. Eleven species of crabs were found in the upper intertidal zone, 14 species were found in the middle zone, and eight species were found in the lower zone. The natural mangrove associations were the poorest in terms of species, and the diversity in Dam Bay and Nha Phu was represented by six species. The superfamily Ocypodoidea is represented in the studied associations by nine species. The superfamily Grapsoidea is represented by only two species: (1) Metopograpsus latifrons (White 1847) found at all stations and (2) a single specimen of Sesarmidae gen. sp. found in the last survey (October 2015) in the mid intertidal zone (MIZ).
The species diversity increased over time from one or two species in the first two surveys to six species in the upper, seven in the middle, and five species in the lower intertidal zone in different years of the late period of monitoring (Fig. 8).
The biomass and abundance of crabs experienced significant fluctuations with no visible trends throughout the monitoring period. In general, their values were low compared to the abundance of mollusks. In most cases, the abundance of crabs did not exceed 40 ind./m2, and the biomass did not exceed 16 g/m2.
At all stations studied, the greatest contribution to the abundance and biomass was made by representatives of the superfamily Ocypodoidea. In the lower intertidal zone, it was made by the species Macrophthalmus miloti (Crosnier 1965), while at other stations there were representatives of the genus Uca.
Other Groups of Invertebrates
Apart from the three key groups of invertebrates and annelids, few representatives of other taxonomic groups were found in the studied mangrove associations. In total, 13 species were found: the jellyfish Cassiopea sp., the burrowing sea anemone Edwarsiidae gen. sp., the chiton Acanthochitona ostreaphila (Sirenko et Saito 2017), three species of hermit crabs (Clibanarius sp., Paguroidea gen. sp., and Coenobitidae gen. sp.), two species of shrimps (Caridea), one species of Stomatopoda, two species of Sipunculida, the brachiopod Lingula anatina Lamarck 1801, and one acorn worm (Ptychoderidae gen. sp.). Shrimps and hermit crabs were encountered at all stations studied. The rest of the species listed were found only in the lower intertidal zone in mangrove plantations of Dam Bay. None of these taxonomic groups was abundant, and no trends were revealed in the dynamics of their quantitative values.
DISCUSSION
The diversity of gastropods of these associations was discussed in detail in (Zvonareva et al., 2016). Comparison of lists of species in a number of works on Thailand, the Philippines, Malaysia, Vietnam, and others showed that the mangrove associations of Dam Bay and Nha Phu bay are characterized by a rather high diversity of gastropods.
The diversity of bivalve mollusks was assessed mainly on the basis of quantitative samples (as opposed to the assessment of gastropods and crabs), since the representatives of this group mainly belong to the infauna, are located in the sediment, and are poorly visible. The species diversity found depends to a large extent on the number of samples. A work on natural mangroves in northern Vietnam provides a list of 91 species of bivalve mollusks (Hong and San, 1993). Only three species are present in the material collected in this study, but the majority of the species from this list belong to the fauna of the subtidal. From four to 44 species of bivalve mollusks were found in various studies from other regions of Southeast Asia (Wells, 1983; Jiang and Lee, 1995; Macintosh et al., 2002; Lozouet and Plaziat, 2008). Thus, the species list of bivalve mollusks in this study is one of the longest. Perhaps this is explained by the extensive sampling effort and the fact that the samples were taken in the lower intertidal zone, where many species typical for the subtidal zone were found, for example, Vasticardium angulatum, Ruditapes philippinarum, etc.
Chertoprud et al. (2012) provides a list of the species from mangrove plantations in Dam Bay. We managed to find all the species from this list, except for Parasesarma ungulatum, since it occurs in the spray zone. In our study, the sampling was carried out only in the intertidal zone, but we found five species that were absent in the study of Chertoprud et al., including one species of the family Sesarmidae. For the fauna of Nha Phu Bay, the same study lists five more species that are absent in our samples (including P. ungulatum). In that study, the collection was carried out in another mangrove association in the innermost part of the bay, that is the reason for the difference in the species lists. Hong and San (1993) report 44 species of crabs in the mangrove forests of northern Vietnam, with only four species in common with our study. In the natural associations of Malaysia in the Sarawak region, 31 crab species were found (Ashton, 2002); 30 crab species were found in the Selangor region (Sasekumar, 1974); 25 crab species were found in natural mangrove associations, 23 species were found in eight-year plantings (ten of them belong to the family Grapsidae), 13 species were found in one-year old plantations in Thailand (seven of them belong to the family Grapsidae) (Macintosh et al., 2002). Thus, the species diversity of crabs in our study is inferior to the species diversity of crabs both in natural associations and in the plantings of other regions of Southeast Asia. The number of species of the superfamily Ocypodoidea is comparable to their number in other regions, but the families Grapsidae and Sesarmidae are very poorly represented. These differences in the taxonomic composition and species diversity may be caused by imperfect sampling techniques (crabs are very mobile and quickly hide in deep burrows, and, possibly, additional fishing methods are required, for example, crab traps, to better cover the entire variety of crabs). It is also possible that the mangrove associations of other regions differ greatly in the structure and degree of complexity from those studied in this work and therefore have more diverse fauna.
The most diverse and abundant groups in the associations studied are the same key groups as in other regions of the Indo-West Pacific (Berry, 1963; Macnae, 1968; Lee, 1998, 1999; Kathiresan and Bingham, 2001). The composition of less significant groups of the macrobenthos is also typical for the mangroves in the Indo-West Pacific (Kathiresan and Bingham, 2001). Thus, the data obtained on the composition and diversity of the macrobenthos fit into the general pattern of the mangrove fauna of the Indo-West Pacific and Southeast Asia.
Summarizing the data obtained on the dynamics of the species composition and abundance, it is possible to reconstruct the events that occurred over ten years of the existence of mangrove plantations. Before the planting of seedlings in the MIZ, mangroves were represented by a very narrow zone of Rhizophora apiculata with the inclusion of several Avicennia marina trees in the upper intertidal zone. This mangrove association had a small area and was simply organized, and nutrients were freely washed away by the tides. This can explain the low diversity of the macrobenthos and the low abundance of mollusks at the initial stage of observing mangrove plantations. In contrast to mollusks, crabs had a high abundance in the first year of observation, and these values are achieved exclusively by the Uca spp. crabs, which (according to the data published) are characteristic of immature mangroves and fragmented mangroves (Macintosh et al., 2002).
The planting of the R. apiculata monoculture in the MIZ in 2004 resulted in a number of changes both at this station and in the upper zone. In the middle zone, the seedlings took root well and grew quite quickly. Therefore, a large amount of free organic matter appeared in the middle zone directly in the form of litter, and also indirectly: the appearance of areas shaded by mangroves created favorable conditions for the development of microalgae and macrophytes. The appearance of additional organic matter in the ecosystem caused a sharp increase in the diversity of nonspecialized species of mollusks and crabs, an increase in the abundance of opportunistic species, predators and scavengers nonspecialized to mangroves. The attraction of new species was also facilitated by the appearance of a solid substrate in the form of growing trees. A similar increase in the abundance of the macrobenthos is reported in a study comparing plantings of different ages in Thailand (Macintosh et al., 2002). Young trees in the middle zone slow down tidal currents and prevent the leaching of organic matter from the upper zone. There is also an increase in the number of opportunistic and nonspecialized species. Thus, mangroves now occupy a significant area and provide a large number of microenvironments. As a result, specialized mangrove species begin to appear in the fauna. According to the published data (which is confirmed by our observations), the area of the mangrove association and the diversity of its vegetation are the main factors determining the diversity of mangrove fauna and especially specialized species (Hutchings and Saenger, 1987; Lee, 1998; Ellison et al., 1999). Sharp fluctuations in the biomass and abundance of different groups of the macrobenthos can be explained by natural variability.
The natural mangrove association in Dam Bay had much in common with the upper intertidal zone in the planting area before 2004 (small area and vegetation structure). This fact explains the low diversity of specialized mangrove fauna in the association, as well as the relatively low abundance of gastropods.
The natural mangrove association at Nha Phu was the most extensive and had a complex spatial structure and a high floristic diversity. This is probably the reason for the high species diversity of specialized gastropods. This association differs from the other stations studied by the extremely low diversity and abundance of bivalve mollusks. This may be explained by the fact that mangroves are not the most favorable environment for bivalve mollusks to live in. The majority of bivalve mollusks are filter feeders, and for many of them it is problematic to live in conditions of rare flooding in the upper intertidal zone. In natural conditions, mangroves are able to inhabit only the upper part of the intertidal zone (Morton, 1976). In addition, the soil in mature mangrove forests is known to be acidic, and only bivalve mollusks with a very thick shell are able to live in it (Morton, 1976). Thus, only specialized bivalve mollusks, which have certain adaptations to the difficult conditions, can live in mature mangroves while the rest live here at the limit of their ecological plasticity (Morton, 1976).
The fauna of the lower intertidal zone differed sharply from the upper and middle zones and from both natural mangrove associations in the area of mangrove plantings in all aspects even eight years after the planting of seedlings. The growth of seedlings is depressed here, and the fauna is more similar in composition to the fauna of the mudflats and upper subtidal than to the fauna of mangroves. This is probably caused by the influence of too frequent and prolonged flooding, as such conditions are not suitable for either Rhizophora seedlings or normal mangrove fauna.
At the time of the last survey, it can be concluded that the structure and composition of the fauna after ten years of development of the new community are still very different from that of the mature mangrove association in Nha Phu. At the same time, the fauna of the upper and middle zones of mangrove plantations is already more similar to the fauna of mature mangroves than the fauna of the natural association in Dam Bay, which is characterized by a small area, simplicity of the spatial structure, and low vegetation diversity.
REFERENCES
Adrianov, A. and Maiorova, A., Peanut worms of the phylum Sipuncula from the Nha Trang Bay (South China Sea) with a key to species, Zootaxa, 2012, vol. 3166, pp. 41–58.
Anker, A., Poddoubtchenko, D., and Marin, I.N., On the presence of the alpheid shrimp genus Bermudacaris Anker and Iliffe, 2000 (Crustacea: Decapoda: Caridea) in the Pacific Ocean, with description of a new species from Vietnam, J. Nat. Hist., 2006, vol. 40, nos. 27–28, pp. 1675–1686.
Ashton, E.C., Mangrove sesarmid crab feeding experiments in Peninsular Malaysia, J. Exp. Mar. Biol. Ecol., 2002, vol. 273, no. 1, pp. 97–119.
Berry, A.J., Faunal zonation in mangrove swamps, Bull. Natl. Mus. State Singapore, 1963, vol. 32, pp. 90–98.
Chertoprud, E.S., Spiridonov, V.A., Marin, I.N., and Mokievsky, V.O., Brachyuran crabs (Crustacea Decapoda Brachyura) of the mangrove intertidal zone of southern Vietnam, in Benthic Fauna of the Bay of Nhatrang, Southern Vietnam, 2012, vol. 2, pp. 258–295.
Dittmann, S., Benthic fauna in tropical tidal flats—a comparative perspective, Wetlands Ecol. Manage., 2002, vol. 10, pp. 189–195.
Dworschak, P.C., Marin, I., and Anker, A., A new species of Naushonia Kingsley, 1897 (Decapoda: Thalassinidea: Laomediidae) from Vietnam and the Philippines with notes on the genus Espeleonaushonia Juarrero and Martínez-Iglesias, 1997, Zootaxa, 2006, vol. 16, pp. 1–16.
Ellison, A.M., Managing mangroves with benthic biodiversity in mind: moving beyond roving banditry, J. Sea Res., 2008, vol. 59, pp. 2–15.
Ellison, A.M., Fransworth, E.J., and Merkt, R.E., Origins of mangrove ecosystems and the mangrove biodiversity anomaly, Global Ecol. Biogeogr., 1999, vol. 8, no. 2, pp. 95–115.
Field, C.D., Rehabilitation of mangrove ecosystems an overview, Mar. Pollut. Bull., 1998, vol. 37, nos. 8–12, pp. 383–392.
Frey, M.A., A revised classification of the gastropod genus Nierita, Veliger, 2010, vol. 51, no. 1, pp. 1–7.
Gherardi, F. and Vannini, M., Hermit crabs in a mangrove swamp: proximate and ultimate factors in the clustering of Clibanarius laevimanus, J. Exp. Mar. Biol. Ecol., 1993, vol. 168, no. 2, pp. 167–187.
Glover, E. and Taylor, J., Lucinidae of the Philippines: highest known diversity and ubiquity of chemosymbiotic bivalves from intertidal to bathyal depths (Mollusca: Bivalvia), in Tropical Deep-Sea Benthos, Heros, V., Strong, E., and Bouchet, P., Eds., Paris: Memoires du Museum National d’Histoire Naturelle, 2016, pp. 65–234.
Gopal, B. and Chauhan, M., Biodiversity and its conservation in the Sundarban mangrove ecosystem, Aquat. Sci., 2006, vol. 68, no. 3, pp. 338–354.
Hong, P.N. and San, H.T., Mangroves of Vietnam, Bangkok: IUCN, 1993.
Houbrick, R.S., Genus Clypeomorus Jousseaume (Cerithiidae—Prosobranchia), Smithson. Contrib. Zool., 1985, vol. 403, pp. 1–131.
Houbrick, R.S., Monograph of the genus Cerithium Bruguiere in the Indo-Pacific (Cerithiidae—Prosobranchia), Smithson. Contrib. Zool., 1992, vol. 510, pp. 1–211.
Hutchings, P. and Seanger, P., Ecology of Mangroves, Heatwall, H., Ed., St. Lucia: University of Queensland Press, 1987.
Jiang, J.X. and Li, R.G., An ecological study on the Mollusca in mangrove areas in the estuary of the Jiulong River, Hydrobiologia, 1995, vol. 295, nos. 1–3, pp. 213–220.
Kathiresan, K. and Bingham, B.L., Biology of mangroves and mangrove ecosystems, Adv. Mar. Biol., 2001, vol. 40, pp. 81–251.
Kumar, R.S., A review of biodiversity studies of soil dwelling organisms in Indian mangroves, Zoos’ Print J., 2000, vol. 15, pp. 221–227.
Lee, S.Y., Ecological role of grapsid crabs in mangrove ecosystems: a review, Mar. Freshwater Res., 1998, vol. 49, no. 4, pp. 335–343.
Lee, S.Y., Tropical mangrove ecology: physical and biotic factors influencing ecosystem structure and function, Aust. Ecol., 1999, vol. 24, no. 4, pp. 355–366.
Lozouet, P. and Plaziat, J.-C., Mangrove Environments and Mollusks Abatan River, Bohol and Panglao Islands, Central Philippines, Hackenheim: ConchBooks, 2008.
Macintosh, D.J., Ashton, E.C., and Havanon, S., Mangrove rehabilitation and intertidal biodiversity: a study in the Ranong mangrove ecosystem, Thailand, Coast. Shelf Sci., 2002, vol. 55, no. 3, pp. 331–345.
Macnae, W., A general account of the fauna and flora of mangrove swamps and forests in the Indo-West-Pacific region, Adv. Mar. Biol., 1968, vol. 6, pp. 73–270.
Marin, I.N., Description of two new species from the genera Palaemonella Dana, 1852 and Vir Holthuis, 1952 (Crustacea: Caridea: Palaemonidae: Pontoniinae), Zool. Mededelingen, 2008, vol. 82, no. 35, pp. 375–390.
Marin, I., The first record of an association between a pontoniine shrimp (Crustacea: Decapoda: Palaemonidae: Pontoniinae) and a thalassematid spoon worm (Echiura: Thalassematidae), with the description of a new shrimp species, Zootaxa, 2014, vol. 3847, no. 4, pp. 557–566.
Mazda, Y., Magi, M., Nanao, H., Kogo, M., Miyagi, T., et al., Coastal erosion due to long-term human impact on mangrove forests, Wetlands Ecol. Manage., 2002, vol. 10, no. 1, pp. 1–9.
Mazda, Y., Magi, M., Ikeda, Y., Kurokawa, T., and Asano, T., Wave reduction in a mangrove forest dominated by Sonneratia sp., Wetlands Ecol. Manage., 2006, vol. 14, no. 4, pp. 365–378.
Metcalfe, K.N. and Glasby, C.J., Diversity of Polychaeta (Annelida) and other worm taxa in mangrove habitats of Darwin Harbour, northern Australia, J. Sea Res., 2008, vol. 59, nos. 1–2, pp. 70–82.
Mitra, S. and Pattanayak, J., Diversity and distribution of sea-anemones (Cnidaria: Actiniaria) in the estuaries and mangroves of Odisha, India, Zool. Surv. India, 2013, vol. 113, no. 3, pp. 113–118.
Mitra, S. and Pattanayak, J., Studies on Lingula anatina (Brachiopoda: Inarticulata) in Subarnarekha estuary, Odisha with special reference to habitat, Zool. Surv. India, 2013a, vol. 113, no. 3, pp. 49–53.
Mokievsky, V.O., Tchesunov, A.V., Udalov, A.A., and Nguen, D.T., Meiobenthos of a mangrove intertidal area of Nha Trang Bay, Vietnam, South China Sea: quantitative distribution and community structure with special reference to free-living nematodes, Biol. Morya, 2011, vol. 37, pp. 262–273.
Morton, B., The biology, ecology and functional aspects of the organs of feeding and digestion of the S.E. Asian mangrove bivalve, Enigmonia aenigmatica (Mollusca: Anomiacea), J. Zool., 1976, vol. 179, pp. 437–466.
Ng, Y.P., The ecology of stomatopods in Matang waters with emphasis on Miyakea nepa and Oratosquillina perpensa, PhD Thesis, KualaLumpur: University of Malaya, Faculty of Science, 2013.
Printrakoon, C. and Kamlung-ek, A., Socioeconomic study and economic value of living fossil, Alingula sp. in mangrove ecosystem in Trat Province, Thailand, Chin. J. Popul. Sci., 2013, vol. 11, no. 3, pp. 187–199.
Reid, D.G., Habitat and zonation patterns of Littoraria species (Gastropoda: Littorinidae) in Indo-Pacific mangrove forests, Biol. J. Linn. Soc., 1985, vol. 26, pp. 39–68.
Reid, D.G., The Littorinid Molluscs of Mangrove Forests in the Indo-Pacific Region: the Genus Littoraria, London: British Museum (Natural History), 1986.
Reid, D.G., The genus Cerithidea Swainson, 1840 (Gastropoda: Potamididae) in the Indo-West Pacific region, Zootaxa, 2014, vol. 3775, no. 1, pp. 1–65.
Reid, D.G., Dyal, P., Lozouet, P., Glaubrecht, M., and Williams, S.T., Mudwhelks and mangroves: the evolutionary history of an ecological association (Gastropoda: Potamididae), Mol. Phylogenet. Evol., 2008, vol. 47, no. 2, pp. 680–699.
Reid, D.G., Dyal, P., and Williams, S.T., Global diversification of mangrove fauna: a molecular phylogeny of Littoraria (Gastropoda: Littorinidae), Mol. Phylogenet. Evol., 2010, vol. 55, no. 1, pp. 185–201.
Spalding, M., Blasco, F., and Field, C., World Mangrove Atlas, Okinawa: International Society for Mangrove Ecosystems, 1997.
Tchesunov, A.V., Mokievsky, V.O., and Thanh, N.V., Three new free-living nematode species (Nematoda, Enoplida) from mangrove habitats of Nha Trang, Central Vietnam, Russ. J. Nematol., 2010, vol. 18, no. 1, pp. 155–172.
Tsingalia, M.H., Population and spatial dynamics mangrove jellyfish Cassiopeia sp. at KeNha’s Gazi Bay, Am. J. Life Sci., 2014, vol. 2, no. 6, pp. 395–399.
Twilley, R.R., Chen, R., and Hargis, T., Carbon sinks in mangroves and their implications to carbon budget of tropical coastal ecosystems, Water, Air Soil Pollut., 1992, vol. 64, pp. 265–288.
Walters, B.B., Local mangrove planting in the Philippines: are fisherfolk and fishpond owners effective restorationists?, Restor. Ecol., 2000, vol. 8, no. 3, pp. 237–246.
Wells, F., An analysis of marine invertebrate distributions in a mangrove swamp in northwestern Australia, Bull. Mar. Sci., 1983, vol. 33, no. 3, pp. 736–744.
Xuan, Q., Vanreusel, A., Thanh, N.V., and Smol, N., Biodiversity of meiofauna in the intertidal Khe Nhan mudflat, Can Gio mangrove forest, Vietnam with special emphasis on free living nematodes, Ocean Sci. J., 2007, vol. 42, no. 3, pp. 135–152.
Yu, R., Chen, G.Z., Wong, Y.S., Tam, N.F.Y., and Lan, C.Y., Benthic macrofauna of the mangrove swamp treated with municipal wastewater, Hydrobiologia, 1997, vol. 347, pp. 127–137.
Zvonareva, S.S. and Kantor, Yu.I., Checklist of gastropod molluscs in mangroves of Khanh Hoa province, Vietnam, Zootaxa, 2016, vol. 4162, no. 3, p. 401.
Zvonareva, S.S., Kantor, Yu.I., Li, X., and Britayev, T.A., Long-term monitoring of Gastropoda (Mollusca) fauna in planted mangroves in central Vietnam, Zool. Stud., 2015, vol. 54, p. 39.
ACKNOWLEDMENTS
We are grateful to the participants of the Vietnam expeditions in 2005–2011: colleagues from the Laboratory of Morphology and Ecology of Marine Invertebrates, Severtsov Institute of Ecology and Evolution, Russian Academy of Sciences (A.E. Fedosov, P.Yu. Dgebuadze, E.S. Mekhova, I.N. Marin, N.A. Zaitseva, A. Zykova, and T.A. Britaev), the staff of the Faculty of Biology, Moscow State University (E.S. Chertoprud, A.E. Zhadan, and E.V. Vortsepneva), the employees of the Shirshov Institute of Oceanology Russian Academy of Sciences (A.A. Udalov, V.A. Spiridonov, and V.O. Mokievskii), and an employee of the Zoological Institute, Russian Academy of Sciences (B.I. Sirenko) for collecting and primary and laboratory processing of the material. We would like to express our gratitude to the entire team of the Joint Russian–Vietnamese Tropical Center for providing the opportunity to carry out this study, and especially to the staff of the Laboratory of Ecology in Nha Trang (Nguyen T.H. Thanh, Vo T. Ha, Tran Q. Hoan, and Nguyen V. Quang) for direct assistance in organizing the work and collecting material.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
CONFLICT OF INTEREST
The authors declare no conflict of interest.
COMPLIANCE WITH ETHICAL STANDARDS
This article does not contain any research involving humans and animals as research objects.
Additional information
Translated by M. Shulskaya
Rights and permissions
About this article
Cite this article
Zvonareva, S.S., Kantor, Y.I., Nguyen, T.T. et al. Diversity and Long-Term Dynamics of the Macrobenthos in a Mangrove Plantation and Two Natural Associations of Khanh Hoa Province, Vietnam. Biol Bull Russ Acad Sci 47, 1115–1125 (2020). https://doi.org/10.1134/S1062359020090150
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1062359020090150