Abstract
The installation of shrimp and salt ponds has contributed to the devastation of several mangroves worldwide. In semiarid regions, where few mangroves are able to cope with the harsh environment, these forests are more vulnerable to human impacts. In Brazil, several aquaculture and salt ponds have been established over mangroves and salt flats (‘apicuns’) i.e. sandier natural areas contiguous to mangroves. These areas, often hypersaline, can be colonized by mangroves if tidal coverage increases, and should, therefore, be protected to ensure mangrove conservation against predicted sea level rise. To restore a mangrove stand in an abandoned saltworks (former ‘apicum’ area) in northeast Brazil, hydrological restoration measures were applied, which involved digging channels to restore estuarine water flux to planted and water-borne propagules. In two years, mangroves developed rapidly in the intervention area, after decades of slow monospecific Avicennia germinans recovery. Survival of planted Rhizophora mangle propagules was high, and naturally established Avicennia germinans and Laguncularia racemosa reached far higher densities and heights in channels with respect to the condition before channeling. These results provide valuated insight into mangrove expansion over salt flats by increasing tidal coverage from ongoing rising sea levels. Associated with ecological changes after mangrove rehabilitation and driven by the return of plant and faunal key groups, ecological interactions like facilitation, herbivory and bioturbation increased in the restored area. Our results shed light on the processes related to forest recovery of degraded littoral areas, and contribute to improving the restoration and management of mangrove forests in semiarid coasts.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mangroves are highly productive biological communities and one of the most highly affected coastal ecosystem by human activities (Diegues 1999; Lugo 2002; Lacerda et al. 2021), despite their important social and ecological roles (Barbier et al. 1997; Alongi 2002; Manson et al. 2005; McLeod and Salm 2006). Although mangroves cover around 137,000 km2 of tropical and subtropical coasts (Worthington and Spalding 2018; Friess et al. 2019), at least 35% of these forests have been destroyed in the past decades due to human settlements, wood extraction, shrimp culture, and salt production (Valiela et al. 2001). In semiarid environments, where few mangrove tree species are able to cope with the harsh environmental conditions, these forests are more vulnerable to changes caused by both the environment and humans. The mangroves in arid-semiarid coasts have been underrepresented in global maps and conservation measures, although recent conservative accounts estimate an extension of over 6,000 km2 of these forests (Adame et al. 2021). Despite their low productivity and slow growth due to the high saline environment and low nutrient input, these mangroves are one of the few woody ecosystems that grow in hot-arid climates, and they can stock considerable amounts of carbon belowground (Adame et al. 2018).
Climatic conditions (high temperatures and evaporation rates) in northeast Brazil favor the installation of saltworks in estuaries and coasts (Reis-Neto et al. 2013). Salt or aquaculture ponds are constructed in the same littoral zone where mangroves occur, so the mangrove forest is usually cleared for this purpose. After mangrove clearing, shrimp and salt production replaced one another in several regions, impairing the return of natural hydrological and soil features in occupied lands and leading to critical levels of mangrove degradation in the last decades (Ferreira and Lacerda 2016; Lacerda et al. 2019). Shrimp culture is still the main threat to mangroves around the world. In northeast Brazil, however, several abandoned saltworks remain and the environmental alterations they caused still impair mangrove recovery. Several saltworks were established over salt flats or ‘salinas’, also called ‘apicuns’ in Brazil. ‘Apicuns’ (or ‘apicum’ in the singular) are sandy areas adjacent to fully canopied mangroves that act as buffer areas for forest persistence (Schaeffer-Novelli 2000; Schmidt et al. 2013). The use of these mangrove-associated wetlands for salt or shrimp production, livestock breeding or human occupation has altered their natural estuary dynamics and severely contributed to their degradation. Despite this problem, gaps in legislation have not compelled deactivated enterprises to restore degraded mangrove or ‘apicum’ areas, or remove dams or structures to restore tidal flushing and hydrological connectivity. Impairing tidal flushing prevents the arrival of mangrove propagules for ecosystem recuperation, thus delaying recovery by decades (Reis-Neto et al. 2013).
In the Pacoti River (state of Ceará, Brazil), a saltworks was installed over an ‘apicum’ area bordered by mangroves at the end of the 1960s, and abandoned around the beginning of the 1980s. Since the abandonment of the saltworks activities in this area, only trees of black mangrove Avicennia germinans (and very few of white mangrove Laguncularia racemosa) have been able to colonize around 30% of the total converted area. Avicennia germinans is a common mangrove on Brazilian coasts (Cintrón and Schaeffer-Novelli 1983; Kjerfve and Lacerda 1993), occurring alone or together with red mangrove Rhizophora spp. and/or L. racemosa. Black mangrove is able to establish in higher and dryer portions of littoral habitat and can cope better than other mangroves with saline soil and limited tidal flooding (Soares et al. 2008; Costa et al. 2014; Adame et al. 2021); however, their recovery in the saltwork was too slow. Therefore, we hypothesized that the limited mangrove recovery of the disused saltworks area is caused by hydrological limitations and associated low propagule recruitment.
In this paper, we aim to test the ability of native mangrove species to develop in an abandoned saltworks through hydrological management and propagule planting. We focused our study and based the success of the experiment on whether the system restarted some functionalities through i) the development of unplanted propagules, both from external or internal origin; ii) an increase in sediment organic matter content; and iii) the presence of key faunal groups, which are indicatives of their ability to self perpetuate (Wyant et al. 1995; Jansen 1997; Choi 2004; Lewis 2005). Specifically, Brachyuran crabs are a key group of ecosystem engineers, and fiddler crabs play a significant role in the composition (grain size, chemistry) and topography of mangrove sediments due to their burrowing (Kristensen 2008). Therefore, we hypothesize that their abundance and detritivorous habits can be related to changes in the sediment features of the restored area after management.
Since the persistence of such abandoned saltworks promotes their occupation and conversion, it is paramount to increase the speed of mangrove recovery (Ferreira and Lacerda 2016) and, subsequently, mitigate increasing human impacts on the Pacoti River estuary. Moreover, this knowledge is important for replication by mangrove restorers, especially in terms of mangrove areas converted to saltworks and promoting the return of mangrove ecosystems and their functionality in semi-arid environments.
Material and Methods
Study Area
The regional climate (Fig. 1) is tropical with an average annual rainfall of below 875 mm (Barreto et al. 2012), mainly in the wet season from February to June. Although drought in northeast Brazil is seasonal, the region is undergoing a period of decreasing average rain volumes since the 1990s that has intensified in the past decade (Marengo et al. 2017). Temperature ranges from 22 to 34 °C and can reach 40 °C in the dry season from July to February. Tides are semidiurnal with maximum tidal amplitude of about 3.2 m and a minimum of -0.1 m, which is considered a mesotidal regime (Borges et al. 2017). In around March and September (Fig. 3A), equinox high tides increase the input of marine water in the estuaries, reaching high levels in coastal areas and allowing the penetration of more water inside the studied saltworks. These tides can start in the previous month and extend their influence after their occurrence, mainly in the second semester, when trade winds can increase water input in the estuary from July–August to November. Higher rain levels occur in the first half of each year, in the rainy season, with higher levels in general between February and April (Fig. 3B).
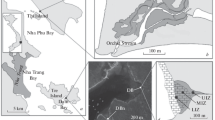
Satellite image showing the lower part of the Pacoti estuary and the mangrove forests along its margins; dune formations can be seen in lighter colors. The lower right corner shows an enlarged view of the study area. The image also shows mangroves colonizing the area from the borders toward the inner part of the area. Black lines and white numbers indicate channels constructed for water inlet to the area at the restoration stage: three channels in the west border (1 to 3) and two channels in the east border (4 and 5). Point with the asterisk indicates the control area
Estuarine littoral areas of the Pacoti River are currently being subjected to great variability due to the influence of climate, rising sea levels, and human intervention such as dam construction upstream and conversion to shrimp ponds and saltworks (Lacerda and Marins 2002; Lacerda et al. 2007). Research was conducted at an abandoned saltworks on the margins of the Pacoti River, inside a State ‘Permanent Protection’ Conservation Unit located in the metropolitan region of the city of Fortaleza, state of Ceará (northeast Brazil) (3°49′37–3°50′00 S/38°25′09–38°25′24 W) (Fig. 1). Aerial images show that at the end of the 1950s, the study area was a salt flat (‘apicum’) with a mangrove forest located in the northeastern half. The selected saltworks was installed in the 1960s and occupied the salt flat and part of the mangrove stand. This and other saltworks operated along the river course until the early 1980s, when the ponds were abandoned. Cyanophyta and Diatomacea algae blooms are periodic in the abandoned saltworks associated with changes in hydrological factors (tides, rains), and cover the soil with filamentous green mat.
Currently, the selected area is surrounded by mangrove forests (Fig. 1) although remains of dykes or barriers constructed to enclose salt ponds restricted tidal water intrusion into the saltworks for decades by higher spring and equinox tide events. A restricted tidal influence enters through the northwestern sector of the experimental area; however, their limited coverage has not counteracted the hypersalinization and desiccation of the soil, thus restricting mangrove development to Avicennia germinans. Some individuals of Laguncularia racemosa had already colonized and sparsely established in the marginal areas of the saltworks in recent years, but no individuals of Rhizophora mangle were established inside the saltworks, which remains restricted to river and creek coasts. Since local mangrove genera are monospecific, species will henceforth only be referred by genus.
Restoration and Management
Hydrological Restoration
Field monitoring and tests were conducted in 2016 to understand the hydrology and other physical and biological characteristics of the selected areas. Soil salinity in the areas reached 91 ppt in previous survey, when observations of tagged seedlings showed that the natural development of Avicennia in the saltworks progressed through periods of seedling mortality in dry season and pulses of seedling growth in wet seasons and/or equinox tides. Although slow, the growth of Avicennia seedlings occurs over the influence of tides and rains (Moncunill 2006) and by the ecological facilitation of nurse herbaceous plants such as Batis maritima, Sesuvium portulacastrum, and Eleocharis sp. In contrast, the absence of natural Rhizophora and Laguncularia propagules was associated with the inability to cope with hard environmental conditions such as high salinity, temperature and hydrological limitations (Elster 2000), thus revealing that these mangroves need measures of hydrological restoration to develop and recover.
In 2017, five channels were dug perpendicular to the river course (0.5–1 m wide/0.3–0.5 m deep) towards the central part of the saltworks to promote mangrove development in the experimental areas and allow estuarine water influx and circulation (Fig. 1, Fig. 2A). Channels allow input of tidal water flux and propagules from mangrove forests adjacent to the saltworks area. Three (3) channels were excavated from a densely vegetated mangrove creek that borders the western side of the saltworks (Fig. 2B, C) and two (2) channels were excavated directly from the main river course in the eastern limit (Fig. 2D, E). The three channels on the west side (channels 1–3) were constructed relatively close to one another to detect changes in mangrove development in a spatial microscale. The channels constructed on the east side, on the river coast (channels 4 and 5), were at a 150 m distance from each other. Channel 5, which already existed, crosses a patch of Avicennia and forms a creek between developed Rhizophora trees in their final portion that opens to the river. This channel has a high concentration of Avicennia roots that prevented soil collection and hindered fiddler crab burrowing, making the ecological characteristics of this channel different from those of the other channels. Red mangrove was planted at this location to determine whether Black mangrove prevents the development of Rhizophora. A flat bare area on the northern side of the saltworks (Fig. 1) with similar soil and level to the area chosen for restoration was selected as a control. Given the similarity of channel 4 to the western channels, this channel was joined to the others to compare changes in the restored area with the control area. The rain and tide levels throughout the experiment period appear in Fig. 3.
Abandoned saltwork in the Pacoti River mangrove, Ceará, Brazil. A. Central part of the saltworks; the image was taken at the start of mangrove establishment in west channels. B, C. Channels dug to bring water into the western limit of the area. D, E. Channels dug to bring water directly from the main river course into the eastern side. The channel in image E was the only preexistent channel; it runs along a patch of Avicennia (Image) and continues through a creek between Rhizophora until it reaches the river
Mangrove Plantings
For planting, we selected Rhizophora mangle, a species commonly found in the estuary, due to its rapid growth, low predation and high primary production, which allows the early return of forest and key faunal groups such as Brachyuran crabs (Ferreira et al. 2015, 2019a, b), and for its ability in retain mangrove propagules between their roots. Healthy 973 propagules were planted at 30 cm distances inside the channels in the wet season, in late March 2017, using the methodology of Ferreira et al (2007). Growth and number of planted Rhizophora were measured annually in dry season (8, 20 and 32 months after planting). Since Laguncularia is an opportunist species able to colonize impacted areas, it was allowed to colonize the area freely, together with Avicennia. Data on density and growth of naturally established seedlings of Laguncularia and Avicennia in the first 2 years could not be collected, due to the continuous process of loss and replacement of these small seedlings, and also by the difficulty to measure them when covered by algae mats or during flooding periods of several days. Therefore, the measurements were focused in the Rhizophora seedlings’ growth, since, due to their rapid development and easy measurability, is the most suitable species to indicate restoration success.
Counting and Surveying of Carcinofauna
Populations of Minuca rapax and Leptuca leptodactyla in the edge of the channels and in the bare control area were surveyed by counting their burrows between 2017 and 2018, to compare the changes in their abundance and eventual relationships with soil changes and mangrove development in the channel area. Burrows were counted at randomly positioned square plots in the control area. In the channel area, counting was not performed inside the channels because water coverage leads the crabs to remain in the burrows covered by mud. Instead, they were counted by positioning plots randomly along the edge of the channels. Permanent faunal surveys were performed to follow the ecological succession in the recovering forest.
Soil Surveying
Sampled Areas
Prior to channel construction (wet season, 2017), sediment was also collected and analyzed, and further sediment samplings from the border of the channels were performed in the next dry season (also in 2017, 8th month), and every six months, namely in the wet season of 2018 (14th month) and in the dry season of 2018 (20th month). Soil samples in the control area were collected in the same periods as those of the channel area. The biotic variable abundance of fiddler crabs, was measured together with the soil abiotic variables.
Sediment Analyses
Soil penetrability (i.e. substrate softness) was measured by vertically releasing a steel centimeter measuring rod of 45 cm in length, 1.3 cm in diameter, and 370 g in weight, from a height of 1.2 m (Ferreira et al. 2013). The average penetrating depth of three releases was calculated for each point. Then, sediment was sampled with a core (∅ 10 cm), and packaged in plastic bags for physicochemical analysis. Three random replicates from each channel were collected and further mixed as one sample for each channel, and a similar procedure (three replicates) was performed in the control area. Samples were taken to the laboratory, where they were dried for organic matter and particle size analyses. Salinity was measured from percolated water after core extraction with an optical refractometer. When this was not possible, measurements were taken from laboratory centrifuged samples. To measure sediment particle size, samples previously dried at 60 °C were weighed in triplicates of 100 g each and washed in a 0.062 mm mesh sieve to separate the sand from the silt–clay fraction. The sand retained in the sieve was removed, dried at 60 °C, and weighed. The weight was deducted from the initial 100 g and the silt–clay percentages were calculated (Colares and Melo 2013). To determine sediment organic matter content, the method of weight loss on ignition, described by Schulte and Hopkins (1996), was used (Colares and Melo 2013). When necessary, a 100 g subsample of the sampled sediment was centrifuged at 10,000 g for 20 min to remove interstitial water and to measure salinity.
Statistical Procedure
A Spearman’s correlation was performed to explore the correlation between abiotic (salinity, penetrability, fine sediment content, organic matter content) and biotic (fiddler crab abundance) variables before the first experiment setting.
To explore the relationship among the populations of fiddler crabs and behavior of abiotic variables after channel construction in the restored area, the response variable fiddler crab abundance and the explanatory variables (fix effect – all standardized by z-scores according to Marquardt 1980) salinity, organic matter, silt + clay, sand, penetrability, sample period (wet’17, dry ‘17, wet ‘18, and dry’18), channel (considering ‘without’ in wet’17, and ‘with’ in the remaining seasons), and season (wet, dry) were related using generalized linear mixed models (GLMM). The channels (1 to 4) analyzed throughout the study periods (months 1, 8, 14, and 20) were introduced in the model as random effect. For the GLMM, the variable fiddler crab abundance, which is a discrete quantitative variable, was modeled from Poisson type error distribution and the ‘log’ link function. Before model formulation, exploratory analyses examined the collinearity of the variance inflation factor (VIF), hence removing the more collinear fix effect variables (sand, season, and channel) (Zuur et al. 2010). Several models were developed and reduced to the lower value of the Akaike information criterion (AIC) and Bayesian information criterion (BIC). The model analyses were conducted in function ‘glmer’, from the lme4 package (Bates et al. 2018), and (z-score) explanatory variables were standardized with the ‘decostand’ function (Oksanen et al. 2019) in R software (R Development Core Team 2018). The variances of model residuals were considered homogeneous. The distribution of residuals was Poisson-type, verified with the ‘descdist’ function of the fitdistrplus package (Delignette-Muller and Dutang 2015).
Next, to determine whether management procedures reached the criteria for restoration success, counteracting stressing conditions and promoting organic matter retention and the presence of key faunal groups, the relationship of the variables salinity, organic matter content, silt + clay percentage, sand percentage, penetrability and fiddler crab abundance, as well as time in months (response explanatory variables) was established through simple linear regression (LM) for each explanatory variable. The models were performed for the individual channels (1–4) and control area. The time variable was converted to ‘month’, starting at 1 and ending in 20, during the 2-year sampling period. The assumptions required for the LM were verified (temporal autocorrelation, normality, and homoscedasticity of the residuals). The regression model was developed using the ‘lm’ function, the temporal autocorrelation was verified with the ‘dwtest’ function, of the lmtest package (Zeileis and Horthorn 2002), and normality and homoscedasticity were verified using residuals plots from the regression model. Since channel 5 have no fiddler crabs populations, we did not include it in the models exploring relationship among biotic and abiotic variables.
It is important to note that the size and patched structure of the saltworks posed practical limitations concerning channels location and delimitation of control and management areas. Conversely, however, it allowed to observe microscale changes in restoration processes that can be replicated at larger scales. The monitoring of abiotic/biotic soil variables and plant growth/number are partially decoupled, since soil variables surveys were performed around every six months after planting (wet and dry seasons: 0, 8, 14 and 20 months), but planted seedlings measurements were made annually in dry seasons until the third year (8, 20 and 32 months). This impaired identification of more accurate cause-effect relationships between soil features and seedlings development.
Results
Propagule Development
After planting, the survival of Rhizophora seedlings was high. In one month, propagules were growing and sprouting. Considering all the channels, in the dry season (8th month) [Fig. 4, Table 3 (Appendix)], around 91% of propagules were alive, with several pairs of leaves, and growing. Survival was high in the western channels (channels 1–3) and in the eastern channel 4, ranging from 93.51% (channel 4) to 98.8% (channel 3), and at around 70% in channel 5. Near the end of the first year, Laguncularia seedlings established in channels 1 to 3, colonizing from the beginning towards the middle portion of the channels, and grew relatively fast compared to natural seedlings observed elsewhere in the river. Avicennia seedlings established all along the channels, but growth was low. Few individuals of Laguncularia and Avicennia colonized channel 4.
Growth of planted and naturally established propagules in zero time (planting), and 8, 20 and 32 months in channels 1 to 5 (1–3 west channels, 4–5 east channels). In red Rhizophora, green Avicennia, and yellow Laguncularia. Top figure, the height of seedlings by channel in 8, 20 and 32 months (x-axis); the Laguncularia trees in channel 5 appear as an average size 2,5 m bar. Down figure, the number of planted (Rhizophora) and naturally established Avicennia and Laguncularia seedlings in the different channels. The Laguncularia trees in channel 5 was not included, since were nor measured
Around the end of the wet season of 2018 (approximately 15 months from experiment onset), Rhizophora individuals in the western channels were infected by an insect that mostly attacked terminal buds, thus promoting apical bifurcation and decreasing growth averages. Towards the end of the second year (2018, 20 months), an average of 51.47% (± 32.43) of the planted propagules were alive, mainly in channels 3 and 4 (81.5% and 90%, respectively) [Fig. 4, Table 3 (Appendix], with an average height close to 60 cm. Survival was intermediate in channels 5 and 2 (56.5% and 62%, respectively). In channel 5, 54% of the planted propagules developed, 23.7% withered or were uprooted by water flow, and 22.3% disappeared or were partially seized, among which around 70% were cut close to their base, damage likely caused by Ucides cordatus crabs. Seedlings of Avicennia also established in this channel between the adult conspecific trees, together with a few individuals of Laguncularia.
Further surveying performed in the 3rd year (at the end of 2019, 32 months) [Fig. 4, Table 3 (Appendix)] revealed low plant survival in some western channels (1 and 2, 0% and 15.7%, respectively) and relatively high survival in channel 3 and channel 4 (68.45% and 78.7%, respectively), while in channel 5 survival was intermediate (52.69%). Naturally established seedlings and saplings of Avicennia and Laguncularia that colonized the western channels surpassed in height and number the planted red mangroves. In 32 months, 290 saplings of Laguncularia and 463 of Avicennia were established. Differential levels of plant survival in channels 1 to 4 were observed in 3 years, and in the channel with the highest survival of Rhizophora and Laguncularia, the survival of Avicennia was lower (channel 3) and vice-versa (channel 1). All the planted Rhizophora in channel 1 died in the 3rd year [Fig. 4, Table 3 (Appendix)] but developed prop roots in all the other channels. All Laguncularia individuals in channel 5 and several in the western channels surpassed 2 m in height, but they were not measured by operative limitations.
Soil Abiotic Variables and Crab Populations
Table 1 shows abiotic and biotic parameters for both the channels and the control area before (wet season in the first year) and after hydrological restoration.
According to results of the Akaike information criterion (AIC) and Bayesian information criterion (BIC), model 5 with salinity, silt + clay, and period as variables reached a low value (AIC = 87.9 and BIC = 91.8) (Table 4, Appendix). The survey period obtained a higher coefficient, followed by silt + clay and salinity. After the channels were dug, the survey period (i.e. time) (two dry seasons and two wet seasons), fine sediment content (silt + clay), and salinity were significantly correlated with fiddler crab abundance. Salinity and fiddler crab abundance were positively related among them and to dry periods. No significant differences were observed among closely located channels 1 to 3 in terms of biotic and abiotic sediment variables. Spearman’s test showed correlations of 0.52 to 0.63 between organic matter and fine sediment, salinity and fiddler crab abundance (Fig. 6).
Comparing the LM models for the restored and control area (Fig. 5), it was observed that organic matter content, penetrability, and fiddler crab density increased with time in the restored area, as shown in the relationship between month and OM (ANOVA; F1,14 = 8.47; p = 0.01), Penet (ANOVA; F1,14 = 3.47; p = 0.08), and fiddler crab density (ANOVA; F1,14 = 5.65; p = 0.03), according to Table 2. There was no relationship between month and the explanatory variables for the control area. Salinity showed great amplitude in time (seasonal), with no homoscedasticity, which hindered modeling.
Changes in organic matter (OM) content, sand, silt + clay (SC), Penetrability and fiddler crab (Uca) density (y axes) in the experimental rehabilitated areas (channels) (top graphs, red line) and control area (bottom graphs, blue line) in the Pacoti saltworks. The time scale (x axis) are the 24 months (2 years) of monitoring, with the variables measured in 4 moments: before channel digging (wet season, month 1), and then around every six months in following dry season 2017, wet season 2018 and dry season 2018
Minuca rapax and Leptuca leptodactyla populations increased in the channel areas in two years, with less fluctuation in number than in the control area (Table 1, Fig. 5). Small individuals of another fiddler crab, Leptuca cumulanta (a species that lives close to mangrove trees), were established in the initial portion of the west channels. After the first semester, burrows and individuals of Callinectes danae and C. exasperatus became more common in and around the channels, but only when flooded. Populations of Sesarma rectum also increased at the borders of the east channels.
Discussion
Mangrove Recovery in the Pacoti Saltworks
The Pacoti saltworks was installed in an area with a mangrove forest and a salt flat (‘apicum’) and later deactivated. ‘Apicuns’ have restricted tidal flooding and high evaporation that generally lead to hypersaline soil maintained by further geochemical processes such as sulfidization (Albuquerque et al. 2014). After deactivation, the saltworks remained as a near-enclosed system due to the permanence of dams that surround the salt ponds. Since topography and hydroperiod are important drivers of mangrove development, mainly in the seedling and juvenile stages, hydrological restoration was recognized as a primary management measure to promote mangrove recovery in the degraded sites (McKee 1995; Lewis 2005; Flores-Verdugo et al. 2007, 2015; Pérez-Ceballos et al. 2017).
Hydrological management created a more frequently flooded area in and around the channels (Fig. 7A, B) and enabled the survival of more than 90% of the planted seedlings of Rhizophora in the first semester (Fig. 8C, D, E, F, G). In one year, the development of Rhizophora was still high (Fig. 9A), despite the infestation by a lepidopteran insect that decreased growth levels (Fig. 7D), and in the 20th month (second year), 51.47% of Rhizophora saplings were still alive, several of which with prop roots (Fig. 9D, E, F, H). The Rhizophora propagules planted in the east channel 4 were able to develop in a dry and herbaceous environment due to the close input of estuarine water, while the propagules planted in the previously existent east channel 5 among Avicennia trees were the most developed, showing that preexistent Avicennia trees did not impair the development of red mangrove and can be used together in restoration projects. As expected, Rhizophora seedlings contributed in retaining other propagules in the channels, thus allowing their rooting.
A. The Pacoti saltworks before the construction of channels and planting (y. 2016); B. Developed mangrove in the channel constructed in the same point after management (y. 2020). C. Avicennia growing outward and into patches of Sesuvium portulacastrum and Batis maritima. D. Damage in an apical bud caused by a lepidopteran infestation; in the lower right corner, a detailed image of the damage in the apical part of the sapling
Experimental area in the Pacoti saltworks (west channels) in the initial year of management (2017). In the bottom of all images are visible the predominant Avicennia forests in the saltworks. A, B. Soil previous to channel construction, showing Cyanophyta biofilms (A). C, D. Planted Rhizophora seedlings in channels at time 0 (March). E, F. Same seedlings in the 4th month (July). G. Rhizophora seedlings in the 7th month (October). H. Naturally established Laguncularia colonizing a channel
Experimental area in the Pacoti saltworks (west channels) in the first (2017) and second (2018) year of management. A. Seedlings of planted Rhizophora (1 year). B. Naturally established Laguncularia (1 year). C. Seedlings of naturally established Laguncularia (17th month, August 2018). D. Planted Rhizophora (17th month). E, F. Planted Rhizophora (20.th month, November 2018). G. Saplings of naturally established Laguncularia (2 years). H. planted Rhizophora (2 years)
Although mangrove regeneration is poor in dry regions with limited tidal flux (Toledo et al. 2001), the conditions generated by the west channels greatly improved the establishment of Avicennia and Laguncularia seedlings in and around the channels. After the management measures, in three years, several Avicennia trees reached the size of trees established more than a decade before. After channel digging, seedlings of Laguncularia colonized the channels gradually from their openings to their end in the center of the saltworks (Figs. 8H, 9B, C, G), and many propagules produced by these plants are growing in the channels. Moreover, the seedlings grew and developed far more canopy than Rhizophora (Figs. 9G, 10E), contributing to primary production through the release of leaves. One channel exhibited the highest density of Avicennia but the lowest colonization of Laguncularia and survival of planted Rhizophora, which all died. Since none of the abiotic variables showed significant difference among the channels to explain such low survival in Rhizophora seedlings, we hypothesize a synergic effect of lepidopteran infection and a frequent covering of seedlings by algal mat fragments (Kodikara et al. 2017), which can include potential toxin releasing Cianophyta), considering these factors were more intense in this channel. Since no significant differences were observed among channels 1 to 3 in terms of biotic and abiotic sediment variables, the variability in mangrove development among them may be related to other factors not measured, such as seed properties (viable period, dispersal, buoyancy, size, susceptibility to burial by sediment), micro-topography (influenced by burrowing crabs), facilitation/competition among mangroves, and other ecological processes (Rabinowitz 1978; Vogt et al. 2018; Ferreira et al. 2019a, b).
A. Two propagules of Rhizophora truncated close to the ground, between Avicennia pneumatophores. B. A collapsed burrow of a Portunid crab Callinectes spp. between fiddler crab burrows close to planted seedlings. C. Grapsoid Sesarma rectum at the border of east channel 4. D, E. Bioturbation of soil by fiddler crabs burrowing at channel sides. F. Fiddler crab Minuca rapax (Ocypodoidea), feeding in algae biofilm (greenish-dark soil at the center of the image). G. A close-up of M. rapax. H. Grapsoid Goniopsis cruentata feeding in a Rhizophora propagule
Abiotic and Biotic Changes Associated with Restoration
A decrease in soil saline levels caused by water flux from the channels in two years was expected, but analyses could not detect such a trend, probably because of the extreme seasonal variations of this parameter and higher resilience of saline conditions due to the historical use of the area. The model built to explore the relationship between the populations of fiddler crabs and behavior of abiotic variables in the restored area revealed a positive correlation between salinity and fiddler crab abundance and between fiddler crabs and fine sediment content after channel digging. The positive correlation between salinity and dry seasons was expected, while the correlation of salinity with fiddler crab abundance can be due to organic carbon food flocculation promoted by high soil salinity (Kida et al. 2017). The increase in fine sediment fractions (silt and clay) is probably responsible for the increase in fiddler crab abundance, since fine sediments can contain more organic matter to serve as food for these detritivores.
In two years, the digging of channels promoted a significant increase in organic matter content, in sediment softness (penetrability), and in fiddler crab density in comparison to the unmanaged area. Evidence suggests that the rapid growth of mangroves in restored area can be attributed to input and retention of plant matter and detritus brought by the channels, mainly on the western side. Increased sediment softness is likely the consequence of the bioturbation of fiddler crabs (Botto and Iribarne 2000; Gutiérrez and Jones 2006) (Figs. 9C, 10D, E, F). Since these crabs are important regulators of sediment organic matter content (Kristensen 2008), results suggest that the two variables, fiddler crab density and organic matter content, are linked by the higher amount of fine sediment (Ferreira et al. 2019b). Notably, the periodical growth of algal mats in the saltworks, which fiddler crabs feed on, could have also influenced crab patchiness and density.
In essence, hydrological restoration allowed the rapid development of mangroves, with higher diversity and growth of trees than in the decades prior to our management measures. Conversely, the control area have no established mangroves to this day. It was not possible to clearly separate the effect of herbivore infestation and sediment biotic/abiotic factors on the survival/mortality of planted seedlings. Nevertheless, the results confirm the reach of restoration criteria, shown by the positive effects of management over mangrove recovery and growth, increased organic matter, and the increase of key Brachyuran populations that are important for mangrove functionality (Dahdouh-Guebas and Cannicci 2021), even in the arid realm of the saltworks. Moreover, the management applied here showed that these areas of ‘apicuns’ can rapidly recover, initially with Laguncularia and Avicennia, when tides and water-borne propagules reach the areas. Furthermore, the management confirmed the effect of Rhizophora seedlings in retaining propagules and increasing their recruitment in the channels. In natural conditions, mangrove roots and stems facilitate stranding and, hence, the rapid rooting of propagules (Toledo et al. 2001; Vogt et al. 2018). Our experiment also shows that salt flats are part of mangrove ecosystems and offer a suitable environment for mangrove refuge and perpetuation in the presence of rising sea levels, thus revealing pathways of mangrove resilience to climate change.
The Role of Biota before and after Restoration
The conversion of the studied area from ‘apicum’ and mangrove ecosystem to a saltworks, its subsequent deactivation, and the ongoing mangrove recovery altered the dynamics and ecological features of the ecosystem. The environmental conditions created in the saltworks after it was abandoned are not only influenced by climatic and other abiotic factors, but also by increasing biotic interactions between the flora and fauna, such as facilitation and herbivory, visible even in the short time of the experiment.
After the saltworks was deactivated, the settlement and development of Avicennia propagules were facilitated by patches of herbaceous plants, such as Batis maritima, Sesuvium portulacastrum, and Eleocharis sp. (Fig. 7C). This facilitation proceeds through sediment desalination, decreasing temperature, and propagule retention (Milbrandt and Tinsley 2006; McKee et al. 2007; Peterson and Bell 2012; Donnelly and Walters 2014; Teutli-Hernández et al. 2019). The seedlings that grow outside the nurse plant patches (Fig. 7C) exhibited densodependence when they are clumped, suggesting competition between these seedlings; furthermore, competition between herbaceous patches and mangrove saplings has been reported (Bracho et al. unpublished). The roots of planted Rhizophora also facilitated the development of other mangroves.
Algae are extremely conspicuous in the studied saltworks, even after channel digging. They grow as extended mats or biofilms of variable thickness (millimeters) in the soil in flooding periods (Fig. 8A). Cyanophyta algae are common in active or inactive saltworks environments, and Lyngbya spp. and Coleofasciculus spp. were identified in the saltworks. Bacillariophyta (Diatoms), such as Melosira spp. and Entomoneis spp., in fewer numbers, were also found. In dry periods, these films dry out and form leather-like crusts over soil, after which they decay and disappear in a few weeks. Since tidal flux in the saltworks before management was irregular, the life-death cycles of these algae were similar. Furthermore, these algae are associated with hypersaline soil conditions (Oren 2015; Vogt et al. 2018). Since some can release toxic compounds, they can cause the underdevelopment of mangroves in the bare central areas, where these algae develop more frequently, and the mortality of planted seedlings.
Brachyuran crabs are ‘ecosystem engineers’ of mangroves and play significant roles in the topography and biogeochemistry of the sediment and in plant diversity, structure, and biomass through herbivory and bioturbation (Ferreira et al. 2019b). The two fiddler crab species observed in the present study, namely Minuca rapax (Fig. 10F, G )and Leptuca leptodactyla, inhabit sandier and more sun exposed substrates than other fiddler crab species (Bezerra et al. 2006). Moreover, they are able to tolerate the high thermohaline stress posed by the saltworks environment by digging burrows (Ferreira et al. 2019b). After the restoration measures, populations of M. rapax and L. leptodactyla increased and remained dense around the channels (Figs. 9C, 10D,E), while maintained seasonal fluctuations in the control area. Crab bioturbation, in turn, can promote an increase in the diversity of sediment meiofauna (Citadin et al. 2016). M. rapax feeds on microalgal mats, which probably constitute a food source that influences the population dynamics (density and patchiness) of these detritivore crabs in the bare areas of the saltworks.
Sesarma rectum crab populations along the borders of the east channels increased significantly (Fig. 10C) and, together with Goniopsis cruentata, can increase the consumption of Avicennia and Laguncularia propagules, thus influencing their recruitment (Ferreira et al. 2013). Also, burrows of swimming crabs Callinectes danae and C. exasperatus became more common when the experimental area was flooded, revealing a diversification in the food chain since these crabs are fish and mollusc predators and increased sediment bioturbation (Fig. 10B). Leptuca cumulanta, a fiddler crab species that lives close to mangrove trees, established in the initial portion of the western channels. Marks in leaves of Laguncularia can indicate the presence of the arboreal herbivorous crab Aratus pisonii. Herbivory on Rhizophora propagules was significant in channel 5, where more than 22% of experimentally planted propagules were consumed. Characteristics of the propagule cuts close to the soil surface (Fig. 10A) suggest that they were made by the large mangrove crab Ucides cordatus, while Goniopsis cruentata, another locally abundant propagule consumer, cannot manage with firmly standing Rhizophora propagules (Ferreira et al. 2007, 2013) (Fig. 10H).
The lepidopteran infestation caused great apical bud damage in planted Rhizophora seedlings in the western channels (Fig. 7D), thus decreasing their vertical growth. This uncommon event partly explains the differential growth of seedlings among the channels. Close to 48% of red mangrove seedlings died in 20 months by insect infestation and likely also by thermo-haline stress. The main effect of the parasite on saplings was observed in apical buds and resulted in the falling of dried stipules and leaves; moreover, this effect of the parasite promotes pre-apical bifurcation of the stem. Unfortunately, it was not possible to identify the Lepidopteran, and we could not determine whether it inhabits the mangrove or if it is an introduced species. The damage was very similar to the one described for the moth Ecdytolopha sp. in the Caribbean (Onuf et al. 1977; Feller and Chamberlain 2007). Significant herbivory on Rhizophora mangroves is rare due to their high tannin content; however, the young age of the saplings and the stressful environment conditions likely decreased their defenses, thus allowing the massive infestation. Since similar infestations were previously observed causing massive defoliation in northeast Brazil, the present findings suggest that their effects can be more deleterious in stressful environments and on small trees, and thus can have a significant effect in young plantations. Increase in temperature due to global warming can influence herbivore insects assemblages, and these defoliation events could intensify.
Perspectives of Mangrove Restoration in Semiarid Coasts
In terms of minimizing the severe environmental and social effects of global warming, mangrove reforestation is known to promote atmospheric carbon fixing and other benefits (Ferreira et al. 2019a; Hoegh-Guldberg et al. 2019). Although semiarid mangroves are undervalued in global maps due to their low height and low carbon stock aboveground, they contribute to carbon fixation and reduce warming in this realm (Adame et al. 2018; Dittmann et al. 2019). Since mangroves in northeast Brazil represent only 10% of the mangroves nationwide and they are pressured by the semiarid climate, degraded stands must be rehabilitated and restored as soon as possible since other factors, such as growing urbanization and livestock breeding (cattle, goats, pigs), can strongly hinder the return of these forests and their essential ecosystem goods and services (Alongi 2002; Lugo 2002; Manson et al. 2005; McLeod and Salm 2006; Ferreira and Lacerda 2016).
We showed that mangrove restoration can recuperate forest areas converted from saltworks and shrimp ponds in semiarid coasts. According to the specific conditions of each location, hydrological rebuilding can be an effective measure to accelerate mangrove recovery, either by the most suitable tree species for drought conditions or by a wider range of mangrove species present in the regional realm. It could also be applied at a higher scale to recuperate larger areas. Furthermore, our results indicate that the current fast sea-level rise (Strauss et al. 2021; Ward and Lacerda 2021) could have the same effect of our hydrological restoration, allowing mangroves to establish in new ‘apicuns’ and sedimentary grounds, thus supporting that the conservation of salt flats will provide refuge for mangroves to avoid increased flood of intertidal areas. The self-recovery of degraded areas can be a low-cost solution for mangrove restoration (Ferreira et al. 2015), although recuperation is slow (Reis-Neto et al. 2013). In stands in dry environments, additional management measures may be necessary to restore the mangrove ecosystem, like hydrological management.
Finally, this study reveals the urgent need for a legal framework that forces deactivating shrimp and salt enterprises to fully remove barriers and enclosures that prevent estuarine water flux in these converted/degraded areas (Ferreira and Lacerda 2016). Until the expected institutional restoration policies are not established, the partial removal of barriers can help increase the rates of forest self-recovery in several regions, contributing in increase rates of carbon sequestration.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Adame MF, Brown CJ, Bejarano M, Herrera-Silveira JA, Ezcurra P, Kauffman JB, Birdsey R (2018) The undervalued contribution of mangrove protection in Mexico to carbon emission targets. Conservation Letters 11:e12445. https://doi.org/10.1111/conl.12445
Adame MF, Reef R, Santini NS, Najera E, Turschwell MP, Hayes MA, Masque P, Lovelock CE (2021) Mangroves in arid regions: ecology, threats, and opportunities. Estuarine Coastal and Shelf Science 248:106796. https://doi.org/10.1016/j.ecss.2020.106796
Albuquerque AGBM, Ferreira TO, Cabral RL, Nóbrega GN, Romero RE, Meireles AJA, Otero XL (2014) Hypersaline tidal flats (apicum ecosystems): the weak link in the tropical wetlands chain. Environmental Review 22:1–11. https://doi.org/10.1139/er-2013-0026
Alongi DM (2002) Present state and future of the world’s mangrove forests. Environmental Conservation 29:331–349. https://doi.org/10.1017/S0376892902000231
Barbier EB, Acreman MC, Knowler D (1997) Economic valuation of wetlands: A guide for policy makers and planners. Ramsar Convention Bureau, Gland, Switzerland
Barreto HBF, Santos WO, da Cruz CM (2012) Analysis of the distribution of mean annual rainfall in the state of Ceará. Revista Verde 7:122–128
Bates D, Maechler M, Bolker B, Walker S, Christensen RHB, Singmann H, Dai B, Scheipl F, Grothendieck G, Green P (2018) Package ‘lme4’. Version, 1: 17. https://CRAN.R-project.org/package=lme4
Bezerra LEA, Dias CB, Santana GX, Matthews-Cascon H (2006) Spatial distribution of fiddler crabs (genus Uca) in a tropical mangrove of northeast Brazil. Scientia Marina 70:759–766. https://doi.org/10.3989/SCIMAR.2006.70N4759
Borges R, Ferreira AC, Lacerda LD (2017) Systematic planning and ecosystem-based management as strategies to reconcile mangrove conservation with resource use. Frontiers in Marine Science 4:353. https://doi.org/10.3389/fmars.2017.00353
Botto F, Iribarne O (2000) Contrasting effects of two burrowing crabs (Chasmagnathus granulata and Uca uruguayensis) on sediment composition and transport in estuarine environments. Estuarine, Coastal and Shelf Science 51:141–151. https://doi.org/10.1006/ecss.2000.0642
Choi YD (2004) Theories for ecological restoration in changing environment: Toward ‘futuristic’ restoration. Ecological Restoration 19:75–81. https://doi.org/10.1111/j.1440-1703.2003.00594_19_1.x
Cintrón C, Schaeffer-Novelli Y (1983) Introducción a la ecología del manglar. Oficina Regional de Ciencia y Tecnologia de la UNESCO para AL y el Caribe (ROSTLAC), Montevideo
Citadin M, Costa TM, Netto SA (2016) The response of meiofauna and microphytobenthos to engineering effects of fiddler crabs on a subtropical intertidal sandflat. Austral Ecology 41:572–579. https://doi.org/10.1111/aec.12346
Colares GB, Melo VMM (2013) Relating microbial community structure and environmental variables in mangrove sediments inside Rhizophora mangle L. habitats. Applied Soil Ecology 64:171–177. https://doi.org/10.1016/j.apsoil.2012.12.004
Costa DFS, Rocha RM, Cestaro LA (2014) Phytoecological analysis and zonation of mangrove of the hypersaline estuary. Mercator 13:119–126. https://doi.org/10.4215/RM2014.1301.0009
Dahdouh-Guebas F, Cannicci S (2021) Mangrove restoration under shifted baselines and future uncertainty. Frontiers in Marine Science 8:799543. https://doi.org/10.3389/fmars.2021.799543
Delignette-Muller ML, Dutang Ch (2015) fitdistrplus: An R Package for Fitting Distributions. Journal of Statistical Software 64:1–34. http://www.jstatsoft.org/v64/i04/
Diegues AC (1999) Human populations and coastal wetlands: conservation and management in Brazil. Ocean and Coastal Management 42:187–210
Dittmann S, Mosley L, Clanahan M, Quinn J, Crooks S, Emmer I, Thomson S, Seaman R (2019) Proof of concept for tidal re-connection as a blue carbon offset project. Goyder Institute for Water Research Technical Report Series No. 19/29
Donnelly M, Walters L (2014) Trapping of Rhizophora mangle propagules by coexisting early successional species. Estuaries and Coasts 37:1562–1571. https://doi.org/10.1007/s12237-014-9789-2
Elster C (2000) Reasons for reforestation success and failure with three mangrove species in Colombia. Forest Ecology and Management 131:201–214. https://doi.org/10.1016/S0378-1127(99)00214-5
Feller IC, Chamberlain A (2007) Herbivore responses to nutrient enrichment and landscape heterogeneity in a mangrove ecosystem. Oecologia 153:607–616. https://doi.org/10.1007/s00442-007-0760-9
Ferreira AC, Lacerda LD (2016) Degradation and conservation of Brazilian mangroves, status and perspectives. Ocean and Coastal Management 125:38–46. https://doi.org/10.1016/j.ocecoaman.2016.03.011
Ferreira AC, Ganade G, Freire FAM, Attayde JL (2013) Propagule predation in a Neotropical mangrove: the role of the Grapsid crab Goniopsis cruentata. Hydrobiologia 707:135–146. https://doi.org/10.1007/s10750-012-1416-2
Ferreira AC, Ganade G, Attayde JL (2015) Restoration versus natural regeneration in a neotropical mangrove: effects on plant biomass and crab communities. Ocean and Coastal Management 110:38–45. https://doi.org/10.1016/j.ocecoaman.2015.03.006
Ferreira AC, Bezerra LEA, Mathews-Cascon H (2019a) Aboveground stock in a restored Neotropical mangrove: Influence of management and brachyuran crab assemblage. Wetlands Ecology and Management 27:223–242. https://doi.org/10.1007/s11273-019-09654-7
Ferreira AC, Alencar CERD, Bezerra LEA (2019b) Interrelationships among ecological factors of brachyuran crabs, trees and soil in mangrove community assemblage in Northeast Brazil. Community Ecology 20:277–290. https://doi.org/10.1556/168.2019.20.3.8
Ferreira AC, Pimenta HCD, Silva LDR, Souza AS (2007) Gestão ambiental de áreas degradadas: um estudo de caso nas nascentes e manguezais do rio Jaguaribe em Natal-RN. In: Assoc. Bras. Eng. Sanitária Ambient. (ABES) (ed) Trabalhos Tecnicos do XXIV Congresso Brasileiro de Engenharia Sanitária e Ambiental, 24. ABES, São Paulo, pp 1–11
Flores-Verdugo F, Moreno-Casasola P, Agraz-Hernández CM, López-Rosas H, Benítez-Pardo D, Travieso-Bello AC (2007) La topografía y el hidroperíodo: dos factores que condicionan la restauración de los humedales costeros. Boletín De La Sociedad Botánica De México 80:33–47
Flores-Verdugo F, Zebadua-Penagos F, Flores-de-Santiago F (2015) Assessing the influence of artificially constructed channels in the growth of afforested black mangrove (Avicennia germinans) within an arid coastal region. Journal of Environmental Management 160:113–120. https://doi.org/10.1016/j.jenvman.2015.06.024
Friess DA, Rogers K, Lovelock CE, Krauss KW, Hamilton SE, Lee SY, Lucas R, Primavera J, Rajkaran A, Shi S (2019) The state of the world’s mangrove forests: past, present, and future. Annual Review of Environmental Resources 44:89–115. https://doi.org/10.1146/annurev-environ-101718-033302
Gutiérrez JL, Jones CG (2006) Physical ecosystem engineers as agents of biogeochemical heterogeneity. Bioscience 56:227–236. https://doi.org/10.1641/0006-3568(2006)056[0227:PEEAAO]2.0.CO;2
Hoegh-Guldberg O, Caldeira K, Chopin T, Gaines S, Haugan P, Hemer M, Howard J, Konar M, Krause-Jensen D, Lindstad E, Lovelock CE, Michelin M, Nielsen FG, Northrop E, Parker R, Roy J, Smith T, Some S, Tyedmers P (2019). The Ocean as a Solution to Climate Change: Five Opportunities for Action. World Resources Institute, Washington, DC. http://www.oceanpanel.org/climate
Jansen A (1997) Terrestrial invertebrate community structure as an indicator of the success of a tropical rainforest restoration project. Restoration Ecology 2:115–124. https://doi.org/10.1046/j.1526-100X.1997.09714.x
Kida M, Tomotsune M, Limura Y, Kinjo K, Ohtsuka T, Fujitake N (2017) High salinity leads to accumulation of soil organic carbon in mangrove soil. Chemosphere 177:51–55. https://doi.org/10.1016/j.chemosphere.2017.02.074
Kjerfve B, Lacerda LD (1993) Mangroves of Brazil. In: Lacerda LD (ed) Conservation and Sustainable Utilization of Mangrove Forests in Latin America and Africa Regions, Part I: Latin America. ITTO/ISME, Okinawa, pp 245–272
Kodikara KAS, Mukherjee N, Jayatissa LP, Dahdouh-Guebas F, Koedam N (2017) Have mangrove restoration projects worked? An in-depth study in Sri Lanka. Restoration Ecology 25:705–716. https://doi.org/10.1111/rec.12492
Kristensen E (2008) Mangrove crabs as ecosystem engineers; with emphasis on sediment processes. Journal of Sea Research 59:30–43. https://doi.org/10.1016/j.seares.2007.05.004
Lacerda LD, Marins RV (2002) River damming and changes in mangrove distribution. Glomis /ISME Electronic Journal 2:1–4
Lacerda LD, Menezes MOT, Molisani MM (2007) Changes in mangrove extension at the Pacoti River estuary, CE, NE Brazil due to regional environmental changes between 1958 and 2004. Biota Neotropica 7:67–72. https://doi.org/10.1590/S1676-06032007000300007
Lacerda LD, Borges R, Ferreira AC (2019) Neotropical mangroves: Conservation and sustainable use in a scenario of global climate change. Aquatic Conservation: Marine and Freshwater Ecosystems 29:1347–1364
Lacerda LD, Ward RD, Godoy MDP, Meireles AJA, Borges R, Ferreira AC (2021) 20-years cumulative impact from shrimp farming on mangroves of Northeast Brazil. Frontiers in Forest and Global Change 4:653096. https://doi.org/10.3389/ffgc.2021.653096
Lewis RR III (2005) Ecological engineering for successful management and restoration of mangrove forests. Ecological Engineering 24:403–418. https://doi.org/10.1016/j.ecoleng.2004.10.003
Lugo AE (2002) Conserving Latin American and Caribbean mangroves: issues and challenges. Madera y Bosques 8:5–25. https://doi.org/10.21829/myb.2002.801289
Manson FJ, Loneragan NR, Skilleter GA, Phinn SR (2005) An evaluation of the evidence for linkages between mangroves and fisheries: a synthesis of the literature and identification of research directions. In: Gibson RN, Atkinson RJA, Gordon JDM (eds) Oceanography and Marine Ecology. CRC Press, pp 485–515
Marengo JA, Alves LM, Alvala RCS, Cunha AP, Brito S, Moraes OLL (2017) Climatic characteristics of the 2010–2016 drought in the semiarid Northeast Brazil region. Annals of the Academia Brasileira De Ciências 90(2):1–13. https://doi.org/10.1590/0001-3765201720170206
Marquardt DW (1980) Comment: You should standardize the predictor variables in your regression models. Journal of the American Statistic Association 75:87–91. https://doi.org/10.1080/01621459.1980.10477430
McKee KL (1995) Seedling recruitment patterns in a Belizean mangrove forest: effects of establishment ability and physico-chemical factors. Oecologia 101:448–460. https://doi.org/10.1007/BF00329423
McKee K, Rooth JE, Feller IC (2007) Mangrove recruitment after forest disturbance is facilitated by herbaceous species in the Caribbean. Ecological Applications 17:1678–1693. https://doi.org/10.1890/06-1614.1
McLeod E, Salm RV (2006) Managing mangroves for resilience to climate change. World Conservation Union (IUCN), Gland
Milbrandt EC, Tinsley MN (2006) The role of saltwort (Batis maritima L.) in regeneration of degraded mangrove forests. Hydrobiologia 568:369–377. https://doi.org/10.1007/s10750-006-0203-3
Moncunill DF (2006) The rainfall trend over Ceará and its implications. In: Instituto Nacional de Pesquisas Espaciais (INPE) (ed) Proceedings of 8 International Conference on Southern Hemisphere Meteorology and Oceanography (ICSHMO). Foz do Iguaçu, INPE, pp 315–323
Oksanen J, Guillaume-Blanchet F, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O'Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs R, Wagner H (2019) vegan: Community Ecology Package. R package version 2.5–6. https://CRAN.R-project.org/package=vegan
Onuf CP, Teal JM, Valiela I (1977) Interactions of nutrients, plant growth and herbivory in a mangrove ecosystem. Ecology 58:514–526. https://doi.org/10.2307/1939001
Oren A (2015) Cyanobacteria in hypersaline environments: biodiversity and physiological properties. Biodiversity and Conservation 24:781–798. https://doi.org/10.1007/s10531-015-0882-z
Pérez-Ceballos R, Echeverría-Ávila S, Zaldívar-Jiménez A, Zaldívar-Jiménez T, Herrera-Silveira J (2017) Contribution of microtopography and hydroperiod to the natural regeneration of Avicennia germinans in a restored mangrove forest. Ciencias Marinas 43:55–67. https://doi.org/10.7773/cm.v43i1.2683
Peterson JM, Bell SS (2012) Tidal events and salt-marsh structure influence black mangrove (Avicennia germinans) recruitment across an ecotone. Ecology 93:1648–1658. https://doi.org/10.1890/11-1430.1
R Development Core Team (2018) A Language and Environment for Statistical Computing. R Found Stat Comput Vienna, Austria, 2. https://www.R-project.org/
Rabinowitz D (1978) Mortality and initial propagule size in mangrove seedlings in Panamá. Journal of Ecology 66:45–51
Reis-Neto AS, Andrade AJ, Lignon MC (2013) Analyses of the mangroves’ rehabilitation process in abandoned saltwork area, in the Ceará River, Northeast Brazil. International Journal of Applied Natural Sciences 2:27–36
Schaeffer-Novelli Y (2000) Grupo de ecossistemas: Manguezal, marisma e apicum. Caribbean Ecological Research, São Paulo
Schmidt AJ, Bemvenuti CE, Diele D (2013) Sobre a definição da zona de apicum e sua importância ecológica para populações de caranguejo Uçá Ucides cordatus (Linnaeus, 1763). Boletim Técnico-Científico Do CEPENE 19:9–25
Schulte EE, Hopkins BG (1996) Estimation of sediment organic matter by weight loss-on ignition. In: Magdoff FR, Tabatabai MA, Hanlon EA (eds) Sediment Organic Matter: Analysis and Interpretation. Sediment Science Society of America, Madison, pp 21–31
Soares MLG, Chaves FO, Estrada GCD, Cavalcanti VF, Portugal AMM, Barbosa B (2008) Caracterização das florestas de mangue do complexo estuarino de Caravelas (Bahia-Brasil). Boletim Técnico-Científico Do CEPENE 16:23–41
Strauss BH, Kulp SA, Rasmussen DJ, Levermann A (2021) Unprecedented threats to cities from multi-century sea level rise. Environmental Research Letters 16:114015
Teutli-Hernández C, Herrera-Silveira JA, Comín FA, López MM (2019) Nurse species could facilitate the recruitment of mangrove seedlings after hydrological rehabilitation. Ecological Engineering 130:263–270. https://doi.org/10.1016/j.ecoleng.2017.07.030
Toledo G, Rojas A, Bashan Y (2001) Monitoring of black mangrove restoration with nursery-reared seedlings on an arid coastal lagoon. Hydrobiologia 444:101–109. https://doi.org/10.1023/A:1017552305401
Valiela I, Bowen JL, York JK (2001) Mangrove forests: one of the World’s threatened major tropical environments. Bioscience 51:807–815. https://doi.org/10.1641/0006-3568(2001)051[0807:MFOOTW]2.0.CO;2
Vogt JC, Abed RMM, Albach DC, Palinska KA (2018) Bacterial and archaeal diversity in hypersaline cyanobacterial mats along a transect in the intertidal flats of the sultanate of Oman. Environmental Microbiology 75:331–347. https://doi.org/10.1007/s00248-017-1040-9
Ward R, Lacerda LD (2021). Responses of mangrove ecosystems to sea level change. In: Friess D, Sidik F (eds) Dynamic Sedimentary Environment of Mangrove Coasts. Amsterdam, Elsevier, pp 235–253. https://doi.org/10.1016/B978-0-12-816437-2.00002-1
Worthington T, Spalding M (2018) Mangrove Restoration Potential: A global map highlighting a critical opportunity. IUCN/The Nature Conservancy, 1–19. https://www.iucn.org/sites/dev/files/content/documents/mangrove-tnc-report-final.31.10.lowspreads.pdf
Wyant JG, Meganck RA, Ham SH (1995) The need for an environmental restoration decision framework. Ecological Engineering 5:417–420. https://doi.org/10.1016/0925-8574(95)00007-0
Zeileis A, Hothorn T (2002) Diagnostic Checking in Regression Relationships. R News. 2:7–10. URL https://CRAN.R-project.org/doc/Rnews/
Zuur AF, Ieno EN, Elphick CS (2010) A protocol for data exploration to avoid common statistical problems. Methods in Ecology and Evolution 1:3–14. https://doi.org/10.1111/j.2041-210X.2009.00001.x
Acknowledgements
We would like to thank CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior/MEC, Brazil) for providing the post-doctoral grant (A.C. Ferreira). L.E.A. Bezerra would like to thank the to Conselho Nacional de Desenvolvimento Científico e Tecnológico – CNPq (Research Productivity Fellowship, 310165/2020-2). We also thank Ravena S.A. Nogueira for the laboratory analyses, Cesar V.C. Freitas and Carolina C. Bracho for their valuable help in the field, and Hortência S. Barroso for helping in algae identification. We are also grateful to anonymous reviewers who provided constructive comments to improve this manuscript.
Funding
This work was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico – CNPq/MCTI, Brazil, Grant number 310165/2020–2 (Luis Ernesto A. Bezerra). Author Alexander C. Ferreira has received research support from CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior/MEC, Brazil).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Alexander Cesar Ferreira, Luis Ernesto Arruda Bezerra, Fulvio Aurélio Moraes Freire, and Jose Vitor Machado Vaconcelos. The first draft of the manuscript was written by Alexander Cesar Ferreira and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ferreira, A.C., Freire, F.A.M., Rodrigues, J.V.M. et al. Mangrove Recovery in Semiarid Coast Shows Increase of Ecological Processes from Biotic and Abiotic Drivers in Response to Hydrological Restoration. Wetlands 42, 80 (2022). https://doi.org/10.1007/s13157-022-01603-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13157-022-01603-0