Abstract
Macrobenthos organisms are vital ecological components of intertidal wetlands. This work studied effects of non-native mangrove rehabilitation (Kandelia obovata) and salt marsh invasion (Spartina alterniflora) on macrobenthos communities in Ximen Island (Zhejiang, China). The macrobenthos communities were compared between rehabilitated K. obovata forests of five different stand ages (2-, 8-, 11-, 16-, and 60-year-old) and adjacent S. alterniflora marsh and non-vegetated mudflat. Compared to the mudflat, S. alterniflora increased the abundance, Shannon–Wiener diversity index (H’), and Margalef's richness index (d’) of macrobenthos. Gastropoda abundance was high in the S. alterniflora marsh, whereas, malacostraca biomass was low. Within mangrove forests, macrobenthos abundance was positively correlated with the stand age of mangroves (Spearman Correlation, P < 0.05), but the corresponding H’, d’ and Pielou's evenness index (J’) showed negative correlations. Gastropoda dominated over macrobenthos in the younger stands (2–11 years in age), whereas, phascolosomatidea and bivalvia were more abundant in the older stands (16 and 60 years in age). Noticeably, the highest biomass for malacostraca was recorded from the 11-year-old stand. Macrobenthos communities differed among habitats, with the divergence in community structure between the S. alterniflora marsh and mangrove stands increasing with mangrove stand age, to be significantly different for stands planted over 16 years ago. Since the 16-year-old stand exhibited similar macrobenthos features to the 60-year-old stand, we speculated that, in the case of macrobenthos, a rehabilitated K. obovata forest can reach its maturity level at a time period of about 15 years in Ximen Island.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Mangrove ecosystems are widely considered to be among the most productive and biodiverse ecosystems on the planet, providing a wide range of benefits to humans (Ellison et al. 2020). However, human population growth and coastal development have induced a substantial loss of mangroves globally (Romañach et al. 2018), impacting on coastal ecosystems and human livelihoods (Duke et al. 2007). Growing awareness of the importance of mangroves has led to increased mangrove conservation and restoration projects around the world, including China (Chu et al. 2015; Ellison et al. 2020; Romañach et al. 2018). Recent assessments based on remote sensing, indicates that mangroves in China have recovered from 18,702 ha in 2000 to 22,419 ha in 2015 (Jia et al. 2018).
Spartina alterniflora, a cordgrass native to North America, is among the most successful invasive plants in China (An et al. 2007). This aggressive invader has spread to all mangrove locations in coastal China (Zuo et al. 2012), and covers vast areas of previously bare mudflats around mangroves as well as gaps within mangrove forests (Zhang et al. 2012). The expansion of the invasive S. alterniflora threatens native mangrove habitats (Chen et al. 2014), including preventing the establishment of mangrove seedlings, especially in areas under strong human disturbance, which facilitate S. alterniflora’ growth (Zhang et al. 2012). Additional negative consequences of the expansion of the invasive S. alterniflora have been reported, including reduction of local biodiversity, competitive exclusion of native intertidal plants, modifications of coastal landscapes, and impacts on mudflat aquaculture (Mao et al. 2019; Wan et al. 2009). To address these issues, mangrove plantation has been proved efficient in preventing the spread of S. alterniflora while restoring mangrove stands (Chen et al. 2014; Feng et al. 2014; Zhou et al. 2015).
Macrobenthos play key roles in regulating ecological processes in coastal ecosystems. For example, they mediate the turnover and exportation of organic matter, facilitate the flow of energy and materials recycling, and act as links in coastal food webs (Lee 2008). Changes in coastal vegetation may cause changes in the associated macrobenthos assemblages (Fondo and Martens 1998; Neira et al. 2005). Hence, mangrove restoration to control S. alterniflora invasions also need recover biodiversity (e.g. abundance, diversity, richness, evenness) and community structure of macrobenthos to recover all its functional properties. Thus, assessment of macrobenthos community help assess the effectiveness of restoration efforts (Ashton et al. 2003; Koo et al. 2011), and can be applied to assess the outcome of mangrove restoration to control S. alterniflora invasion in Chinese coastal areas.
Yueqing Bay, Zhejiang Province, China, has experienced severe S. alterniflora invasions since 1990s. Ximen Island, the biggest island in Yueqing Bay, is the northern boundary of mangrove distribution in China. In fact, no natural mangroves were found in Zhejiang Province. The mangrove species Kandelia obovata, a cold-resistant species with broad environmental tolerance, was introduced to Ximen Island in 1957 from Fujian Province. Successive K. obovata plantations have since been conducted in the island, but most of them were subsequently destroyed by human activities. Several rehabilitation projects of K. obovata forests were conducted since 2000 to compensate for the destroyed mangroves and to mitigate S. alterniflora invasion, resulting in an estimated K. obovata area of ≈ 30 ha in Ximen Island at present.
Here, we compare the macrobenthos communities in different aged stands rehabilitated K. obovata, areas invaded by S. alterniflora and unvegetated mudflats in Ximen Island. Specifically, we (1) examined changes in the macrobenthos community with S. alterniflora invasion; (2) elucidate changes of the macrobenthos communities with stand age of rehabilitated mangrove forests; and (3) estimate the time required for a rehabilitated mangrove forest of non-native species to develop to maturity status.
2 Materials and Methods
2.1 Sampling Sites Descriptions
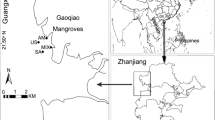
This study was conducted in Ximen Island (28°33′N, 121°18′E) (Fig. 1), Yueqing Bay, Zhejiang Province, China. Ximen Island covers a land area of 6.98 km2 and a mudflat area of 15.11 km2. This region has a humid subtropical climate. The mean (over the past 30 years) annual air temperature is 18.3 °C, the mean annual precipitation is 1595.7 mm, and the mean annual sunshine duration is 1714.6 h per year. Tides are regular semi-diurnal with a maximum range of 8.34 m and an average range of 4.54 m (http://www.yueqing.gov.cn/).
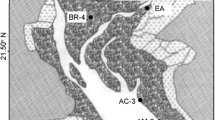
To achieve our goals seven sampling sites were selected (Fig. 1). To examine the influence of invasive S. alterniflora on intertidal macrobenthos communities, one S. alterniflora marsh (denoted as SA) and one bare mudflat were selected (denoted as MF). In addition, five rehabilitated K. obovata stands, being planted in 1957, 2001, 2006, 2009, and 2015, respectively (denoted as KO60, KO16, KO11, KO8, KO2, respectively), were selected to understand the changes in macrobenthos communities at different successional stages of the mangrove forest.
2.2 Sampling
Seasonal samplings of macrobenthos at each one of above sites were undertaken in October 2018, December 2018, March 2019 and July 2019 to represent the autumn, winter, spring and summer seasons, respectively. At each sampling time and site, triplicate sediment samples of 25 cm × 25 cm × 30 cm were randomly collected. A metal frame of the designated volume was pushed down into sediment prior to shoveling. The collected sediment samples were then sieved by a 0.5 mm mesh to retrieve the macrobenthos. All the macrobenthos specimens were preserved into 5% formalin, and the organisms were identified at the lowest taxonomic level possible under the Stemi508 (ZEISS), were counted and their fresh weighted to the nearest 0.0001 g on a precision electronic laboratory balance.
2.3 Data Analyses
The following parameters were calculated to summarize the macrobenthos community structure for each sample: Shannon–Wiener diversity index (H’; log e based), Margalef' s richness index (d), Pielou' s evenness index (J’), the total abundance, and the total biomass. Two-way ANOVA was applied to test for contributions of seasonal and spatial changes to the variance in these parameters. Kolmogorov–Smirnov test was performed for the normality and Levene’s test for the homogeneity of variances. Because of the presence of heteroscedasticity, all parameters were analyzed by the Kruskal–Wallis test. The Mann–Whitney U test was then applied for pairwise comparisons if any significant differences were found in the Kruskal–Wallis test. The linear relationship between mangrove stand age and the parameters was tested using Spearman correlation analysis. The Excel 2019 and MATLAB 2019a software were employed for the analyses.
Multivariate analyses were performed using PRIMER 7.0. The seasonal and spatial variations of the macrobenthos community structure were evaluated using non-metric multidimensional scaling (n-MDS) ordination in combination with hierarchical cluster analysis (group-average linking) (Anderson et al. 2016). The Bray–Curtis dissimilarity matrix was constructed based on the square root transformed abundance data. To reduce the impacts of rare species, species that contributed less than 1% of the total abundance were eliminated, and then species that accounted for more than 3% abundance of each sample were included again. Two-way analysis of similarities (ANOSIM) was employed to determine the statistical significance of differences among seasons and sites (Anderson et al. 2016).
The ABC curve (Abundance Biomass Comparison curve) was implemented to detect the stability of macrobenthos communities. When the biomass curve is above the abundance curve, the macrobenthos community can be considered undisturbed exhibiting high stability; when the two curves intersect or overlap with each other, the macrobenthos community is considered mediately disturbed exhibiting low stability; when the biomass curve is below the abundance curve, the macrobenthos community is considered heavily disturbed (Warwick 2008). This analysis was performed using the R software of the R package “ABC analysis”.
3 Results
3.1 Species Composition and Biodiversity
Over the four seasons considered, a total of 62 macrobenthos species were identified (Table S1). Gastropoda (40.32%, 25 species), polychaeta (20.97%, 13 species), and malacostraca (19.35%, 12 species) dominated the community species richness, followed by bivalvia (9.68%, 6 species), actinopterygii (6.45%, 4 species), phascolosomatidea (1.61%, 1 species), and nemertea (1.61%, 1 species).
The Kruskal–Wallis tests revealed significant spatial differences for every parameter (H’, d, J’, the total abundance, and the total biomass) among sites (P < 0.05), while no significant seasonal difference was found (P > 0.05). Hence, the data from different seasons were pooled together to examine the spatial patterns. Both H’ (average values ranged from 1.09 to 1.98 among seven sites) and d (ranged from 0.88 to 1.95) showed significant lower values in the 16-year-old mangrove stand and significant higher values in the 2-year-old stand (both P < 0.05) (Fig. 2 a and b). J’ (ranged from 0.57 to 0.86) in the 16-year-old stand was significantly lower than those in the other sites (P < 0.05) except the 60-year-old stand (Fig. 2c).
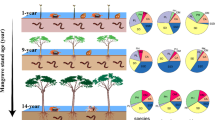
The pairwise Mann–Whitney U test showed that the abundance (average values ranged from 261.33 to 1032.00 ind∙m−2 among seven sites) in the mudflat was significantly lower than those in the other sites (P < 0.05), and the highest value of abundance occurred in the 60-year-old mangrove stand (Fig. 3a). Gastropods generally dominated the macrobenthos communities in the mudflat, S. alterniflora marsh, and the three younger mangrove stands (2-, 8-, 11-year-old stands), with high percentage of three species Optediceros breviculum, Assiminea lutea, and Mainwaringia leithii in most of these sites. The macrobenthos communities in the two older mangrove stands (16-, 60-year-old stands) were represented by Phascolosoma arcuatum belonging to phascolosomatidea and Xenostrobus atratus belonging to bivalve (Fig. 3a, seasons Table 1). The average biomass (ranged from 98.07 to 255.22 g·m−2) showed significant higher values in 11-, 16-, 60-year-old mangrove stands than the other four sites (P < 0.05) (Fig. 3b). Malacostraca species Tubuca arcuata accounted for high percentage of biomass in every site, and the highest biomass of this species was found in the 11-year-old mangrove stand. High dominance of other two carbs Scylla serrata and Metaplax longipes were found in the mudflat. And, P. arcuatum also dominated in the biomass of 16-, 60-year-old mangrove stands (Fig. 3b, seasons Table 1).
Their seasonal mean abundance (or biomass) and relative percentage to the total abundance (or biomass) are shown.
Within the five mangrove sites, significant positive correlations were found (Spearman Correlation, P < 0.05) between the total abundance and the total biomass and stand age, but significant negative correlations (P < 0.05) were found between H’, d and J’ and stand age (Fig. 4).
3.2 Macrobenthos Community Structure
The n-MDS analysis found significant spatial differences in the macrobenthos community structure among different sites (Fig. 5a). Three distinct groupings can be classified based on sites, as a group of sampling sites composed of samples collected from the 16-, 60-year-old mangrove stands, a group constituted by samples collected from the 2-, 8-, 11-year-old mangrove stands and the S. alterniflora marsh, and a separate group representing the community sampled in the mudflat. However, no obvious seasonal variation in the macrobenthos community structure was found (Fig. 5b). Result of the cluster analysis for spatial differences (Fig. 5c) was consistent with the n-MDS analysis. Two-way ANOSIM revealed that the macrobenthos community structure changed significantly among sites (Global R = 0.545, P = 0.001) and seasons (Global R = 0.198, P = 0.001), showing that spatial variations were noticeably stronger than seasonal variations (Table S2a). Pairwise tests found significant differences between all combinations of sites and seasons (P < 0.05), except between the sites of S. alterniflora marsh-, 11-year-old mangrove stand- and 2-year-old mangrove stand (P > 0.05), and the seasons of Spring-, Autumn- and Winter (P > 0.05) (Table S2b).
3.3 Abundance Biomass Comparison (ABC) Curve
The ABC curve suggested moderate disturbance within all sites in certain seasons, apart from the 2-year-old mangrove stand and S. alterniflora marsh that were undisturbed during all seasons (Fig. S1). In spring, only the mudflat was moderately disturbed; in summer, moderate disturbances were observed in 11-, 16-, 60-year-old mangrove stands; in autumn and winter, 16-, 60- year-old mangrove stands had moderately disturbed macrobenthos communities (Fig. S1).
4 Discussion
4.1 Changes of Macrobenthos Communities with S. alterniflora Invasion
Our results showed that the macrobenthos community was significantly altered by alien S. alterniflora. Specifically, compared to the mudflat, the abundance of macrobenthos was significantly higher in the Spartina marsh, where much higher gastropods abundance was observed. By contrast, malacostraca showed a higher biomass in the mudflat than the S. alterniflora marsh.
Spartina marsh is usually considered as a more suitable habitat for gastropods when compared to bare mudflat, because the presence of vegetation can attenuate the harsh living conditions for gastropods in intertidal environments via increasing habitat complexity and heterogeneity (Hedge and Kriwoken 2000), serving as structural refuge against wave energy and predation (Lana and Guiss 1991), offering shade and hence reduced heat and dehydration stress (Sueiro et al. 2012), and providing food directly or through the epiphytic algae (Lana and Guiss 1991; Tang and Kristensen 2010). Moreover, tidal level is another key factor in limiting the distribution of gastropods (Ma et al. 2020; Tang and Yu 2007). In the present study, the mudflat is located in the lower intertidal than the S. alterniflora marsh, the lengthy periods of inundation could be detrimental to the survivorship of gastropods (Bao et al. 2008). In the S. alterniflora marsh, the gastropods assemblage was mainly consisted of two deposit feeders A. lutea (Assimineidae) and M. leithii (Littorinidae). The small-scale distribution pattern of gastropods may be determined by their digestive enzyme activity level (Liu et al. 2014). Species with greater capacity to digest saltmarsh-derived carbohydrates may obtain a higher dominance in salt marsh (Liu et al. 2014).
The aboveground vegetations in the S. alterniflora marsh significantly reduced the biomass of a burrowing crab M. longipes. This result was similar with a previous observation by Meng (2017), who found that M. longipes was more abundant in open mudflats than Spartina marshes as its better endurance to light penetration and high temperature. This species had no dietary dependency on S. alterniflora leaves, and that the presence of vegetations could not decrease the predation risk for them (Meng 2017). Interestingly, a mud crab S. serrata was found only in the mudflat habitat. This large-size species is a roaming opportunistic carnivore with strong mobility, which may be impaired by dense vegetation.
4.2 Changes of Macrobenthos Communities with Stand Age of Rehabilitated Mangrove Forests
Macrobenthos abundance, biomass and community composition differed among habitats, with the divergence in community structure between the S. alterniflora marsh and mangrove stands increasing with mangrove stand age, to be significantly different for stands planted over 16 years ago. The older stands (16-, 60-year-old stands) possessed a quite similar community structure, but it was significantly different from that of the three younger stands (2-, 8-, 11-year-old stands). The abundance and biomass of macrobenthos were positively correlated with stand age, whereas, the H’, d and J’ showed negative correlations. Stand age significantly affected biotic parameters of the macrobenthos communities of rehabilitated mangrove forests.
For the two older stands, the macrobenthos assemblages characterized by the distinct dominance of P. arcuatum (i.e. the sole species belonging to phascolosomatidea), which resulted in the reduced macrobenthos diversity in the habitats. This finding was consistent with result reported for the Jiulongjiang Estuary by Chen et al. (2007), who found that P. arcuatum was the dominant species in the 43-year-old rehabilitated K. obovata forest, while it was not yet dominated 4-, 7- and 19-year-old forests. Soil organic matter (SOM) contents, which are food sources of P. arcuatum (Chen et al. 2007), have been demonstrated to increase with the development of mangrove forests (Salmo et al. 2013), because mature mangrove forests are very efficient contribute higher organic loads and are more efficient at retaining organic-rich sediment particles (Li et al. 2017). In addition, microbial respiration rates supporting by excess organic matters also lead to oxygen depletion and the accumulation of sulfide from anaerobic metabolisms (Magni et al. 2015). P. arcuatum is particularly resistant to such environments, as this species has special ability to detoxify sulfide in anaerobic conditions (Ip et al. 1997).
X. atratus (belongs to bivalvia), another species particularly well represented in the older stands, is a common fouling animal in Chinese mangrove ecosystems. Seawater salinity (Lin et al. 2006) and distance from land (Ning and Lin 2004) are the main factors driving their distribution pattern. Fouling species can exert adverse impacts on mangroves and can even be led to mangrove mortality (Li et al. 2009), suggesting that control of the abundance of this species maybe required to maintain healthy mangrove stands.
Changes in macrobenthos assemblages with mangrove development are also related to the vegetation characteristics (e.g. canopy closure, tree height, diameter at breast height, etc.) (Li et al. 2017; Tang et al. 2012). Benthic environments shaded by mangrove canopies become cooler and wetter, promoting epifauna (Kon et al. 2010). However, heavy shade may lead to reduction in biomass of microphytobenthos, which are highly productive and are an essential source of food for mangrove epifauna (Kon et al. 2010). The development of the mangrove forest results in denser canopy in older stands with concomitant stronger shading effects. The mangrove forest at the late stage of succession is characterized by high shading, high salinity, and massive litter accumulation, which have been shown to reduce microphytobenthos growth (Chen et al. 2015). On the contrary, the more open canopied of younger stands lead to higher availability of microphytobenthos, which can support a more diverse epifaunal community. In the present study, restored mangrove stands of 2 to 11 years in age were characterized by high epifaunal gastropods abundance, basically Assimineidae spp. (e.g. O. breviculum and A. lutea) and Littorinidae spp. (e.g. M. leithii), which were depleted in the communities in 16- and 60-year-old mangrove stands. Similar results were reported by Macintosh et al. (2002) in Thailand, who found that Littorinidae, Assimineidae and Cerithidea were more representative snails in younger stands, whereas Neritidae and Ellobiidae were more abundant in mature forests.
Macrobenthos biomass peaked at intermediate stand ages, with the highest biomass found at the 11-year-old mangrove stand, dominated by the burrowing crab T. arcuate, which is known to occupy semi-open habitats with moderate light penetration (Macintosh et al. 2002). Mangrove canopies start to close at about 10 years after they were planted (Salmo et al. 2013) and, therefore, the 11-year-old mangrove plantation could provide more suitable plant cover to T. arcuate than younger ones. Furthermore, burrowing crabs are strongly responsive to mangrove roots. Crab burrows in K. obovata forests may suffer less from the tide, since the buttresses and aerial roots of K. obovata trees consolidate the soil and protect the burrows (Li et al. 2015). However, Wang et al. (2014) proposed that intermediate levels of mangrove root density may encourage crabs to build more complicated borrows, whereas these activities may be hindered when root density is too high or too low. Thus, the peak biomass and dominance of T. arcuate in 11-year-old mangrove stands could be attributed to the moderate density of both the canopy and root system.
4.3 Disturbances in Macrobenthos Communities
Warwick (2008) argued that the ABC curve is a tool to detect anthropogenic perturbation effects on assemblages of organisms. Accordingly, in the present study, the corresponding ABC curves revealed that moderate disturbances are present in all sites except the S. alterniflora stand and the 2-year-old mangrove plantation. The existence of disturbances in these sites could due to aquaculture activities in nearby mudflats. Yang et al. (2007) found that remnant feeds and metabolites produced by fish cultures caused environmental degradation and severely disturbed the macrobenthos communities in Yueqing Bay. Moreover, the ABC curves suggested that in mangrove stands older than 2 years, the disturbances might also possibly be related to the capture of economic species, such as peanut worms, octopus, and mudskippers. When the field work of this study was conducted, we encountered many local fishermen collecting those species in mangrove stands during the course of the year. Mangrove forests can act as traps to accumulate marine garbage (Martin et al. 2019), which can enter mangrove forests through tidal flushing and human disposal. In our study, it was observed that several kinds of household waste (such as plastic products, glasses, food residues and discarded clothing) were found in almost all mangrove sites. Dissanayake and Chandrasekara (2014) suggested that, among all anthropogenic activities, domestic waste is the biggest threat to mangrove ecosystems, particularly non-degradable waste like polythene and plastics that can affect the behavior and health status of macrobenthos. The degree of macrobenthos community disturbance depends on the cumulative effects of many disturbance-contributing factors. Thus, further studies are necessary to determine how macrobenthos respond to cumulative effects at the individual, population and community levels.
4.4 A Case of Mangrove Rehabilitation of Non-native Species in China
Mangrove rehabilitation should target at optimizing ecosystem services alongside biodiversity rather than merely at recovering vegetation (Bosire et al. 2008; Brown et al. 2013). The results of the present study, as well as those of many previous studies (Chen et al. 2015; Li et al. 2017; Macintosh et al. 2002; Morrisey et al. 2003), have shown that different mangrove ages not only sustain significantly different environmental characteristics but also different macrobenthos assemblages, indicating that the ecosystem services and functions are not equivalent in different mangrove ages. Planting new stands to compensate for losses of mangroves, therefore, may only yield the deserved ecological outcomes after a lag time of stand development (Morrisey et al. 2003), which can be informed by the abundant experience on mangrove rehabilitation projects conducted in China. For example, in native K. obovata restored forests in the Jiulongjiang Estuary, macrobenthos communities and vegetation require about 20 years to reach maturation (Chen et al. 2007); in the Zhangjiang Estuary, 10-year-old restored K. obovata site is still considered to be developing toward stabilization, because of its lower macrobenthos biomass than the mature stand (Feng et al. 2014). In our study, the 16-year-old stand exhibited similar community composition and dominant species of macrobenthos to the mature stand, but younger stands had largely different macrobenthos assemblages. Therefore, it can be reasonably assumed that in Ximen Island a rehabilitated non-native K. obovata forest can reach its maturity level, when considering the development of macrobenthos, at an age of about 15 years.
5 Conclusion
Our results demonstrated, (a) the striking dissimilarities between macrobenthos communities in the S. alterniflora marsh and in barren mudflat, and (b) changes in biodiversity and community structure of macrobenthos with planting age of K. obovata stands. The 16-year-old stand reached a similar macrobenthos community structure as that of the mature stand. Accordingly, we conclude that the macrobenthos community structure can be significantly altered by S. alterniflora invasion and K. obovata plantation; and the macrobenthos community structure of rehabilitated K. obovata forest reaches full development about 15 years after it has been planted. These insights are valuable for governing the aggressive invasion of S. alterniflora and monitoring the ecological development of mangrove rehabilitation sites in China, particularly in regions out the natural range of mangrove distribution. Considering the importance of the macrobenthos community for the management and rehabilitation of mangrove ecosystems, further studies are needed to evaluate the long-term effects of artificially planted mangroves on local habitats and macrobenthos assemblages and to explore the mechanisms of the potential ecological consequences.
References
An SQ, Gu BH, Zhou CF, Wang ZS, Deng ZF, Zhi YB, Li HL, Chen L, Yu DH, Liu YH (2007) Spartina invasion in China: implications for invasive species management and future research. Weed Res 47:183–191. https://doi.org/10.1111/j.1365-3180.2007.00559.x
Anderson M, Gorley R, Clarke K (2016) PERMANOVA+ for PRIMER: Guide to Software and Statistical Methods. PRIMER-E
Ashton E, Hogarth P, Macintosh D (2003) A comparison of brachyuran crab community structure at four mangrove locations under different management systems along the Melaka Straits-Andaman Sea Coast of Malaysia and Thailand. Estuaries 26:1461–1471. https://doi.org/10.1007/BF02803654
Bao YX, Hu ZY, Li HH, Ge BB, Cheng HY (2008) Seasonal variation and functional groups of macrobenthic communities at diked and natural tidal flat, Lingkun Island, China. Curr Zool 54:416–427. https://doi.org/10.3321/j.issn:1000-0933.2008.04.017
Bosire JO, Dahdouh GF, Walton M, Crona BI, Lewis RR, Field C, Kairo JG (2008) Functionality of restored mangroves: A review. Aquat Bot 89:251–259. https://doi.org/10.1016/j.aquabot.2008.03.010
Brown DA, Howe C, Mace GM, Knight AT (2013) Do mangrove forest restoration or rehabilitation activities return biodiversity to pre-impact levels? Environ Evid 2:1–8. https://doi.org/10.1186/2047-2382-2-20
Chen GC, Ye Y, Lu CY (2007) Changes of macrobenthic faunal community with stand age of rehabilitated Kandelia candel mangrove in Jiulongjiang Estuary, China. Ecol Eng 31:215–224. https://doi.org/10.1016/j.ecoleng.2007.07.002
Chen H, Liao BW, Liu BE, Peng CH, Zhang Y, Guan W, Zhu QA, Yang G (2014) Eradicating invasive Spartina alterniflora with alien Sonneratia apetala and its implications for invasion controls. Ecol Eng 73:367–372. https://doi.org/10.1016/j.ecoleng.2014.09.096
Chen Q, Li J, Zhang LM, Lu HF, Ren H, Jian SG (2015) Changes in the Macrobenthic Faunal Community during Succession of a Mangrove Forest at Zhanjiang, South China. J Coast Res 300:315–325. https://doi.org/10.2112/JCOASTRES-D-13-00019.1
Chu V, Russell M, Brown S, Dart P (2015) Using Shoreline Video Assessment for coastal planning and restoration in the context of climate change in Kien Giang. Vietnam Ocean Sci J 50:413–432. https://doi.org/10.1007/s12601-015-0038-9
Dissanayake N, Chandrasekara U (2014) Effects of Mangrove Zonation and the physicochemical parameters of soil on the distribution of macrobenthic fauna in kadolkele mangrove forest, a tropical mangrove forest in Sri Lanka. Adv Ecol 2014:1–13. https://doi.org/10.1155/2014/564056
Duke N, Meynecke J, Dittmann S, Ellison AM, Anger K, Berger U, Cannicci S, Diele K, Ewel KC, Field CD, Koedam N, Lee SY, Marchand C, Nordhaus I, Dahdouh-Guebas F (2007) A world without mangroves? Science 317:41b–42b. https://doi.org/10.1126/science.317.5834.41b
Ellison AM, Felson AJ, Friess DA (2020) Mangrove rehabilitation and restoration as experimental adaptive management. Front Mar Sci 7:327–355. https://doi.org/10.3389/fmars.2020.00327
Feng JX, Guo JM, Huang Q, Jiang JX, Huang GM, Yang ZM, Lin GH (2014) Changes in the community structure and diet of benthic macrofauna in invasive Spartina alterniflora wetlands following restoration with native mangroves. Wetlands 34:673–683. https://doi.org/10.1007/s13157-014-0533-2
Fondo EN, Martens EE (1998) Effects of mangrove deforestation on macrofaunal densities, Gazi Bay, Kenya. Mangroves Salt Marshes 2:75–83. https://doi.org/10.1023/A:1009982900931
Hedge P, Kriwoken LK (2000) Evidence for effects of Spartina anglica invasion on benthic macrofauna in Little Swanport estuary, Tasmania. Austral Ecol 25:150–159. https://doi.org/10.1046/j.1442-9993.2000.01016.x
Ip Y, Tan G, Kuah S, Chew S (1997) Detoxification of environmental sulfide to sulfane sulfur in the intertidal sipunculid Phascolosoma arcuatum. J Comp Physiol 167:213–220. https://doi.org/10.1007/s003600050067
Jia MM, Wang ZM, Zhang YZ, Mao DH, Wang C (2018) Monitoring loss and recovery of mangrove forests during 42 years: The achievements of mangrove conservation in China. Int J Appl Earth Obs Geoinformation 73:535–545. https://doi.org/10.1016/j.jag.2018.07.025
Kon K, Kurokura H, Tongnunui P (2010) Effects of the physical structure of mangrove vegetation on a benthic faunal community. J Exp Mar Biol Ecol 383:171–180. https://doi.org/10.1016/j.jembe.2009.11.015
Koo B, Je J, Woo H (2011) Experimental restoration of a salt marsh with some comments on ecological restoration of coastal vegetated ecosystems in Korea. Ocean Sci J 46:47–53. https://doi.org/10.1007/s12601-011-0004-0
Lana C, Guiss C (1991) Influence of Spartina alterniflora on structure and temporal variability of macrobenthic associations in a tidal flat of Paranagua Bay (southeastern Brazil). Mar Ecol Prog Ser 73:231–244
Lee SY (2008) Mangrove macrobenthos: Assemblages, services, and linkages. J Sea Res 59:16–29. https://doi.org/10.1016/j.seares.2007.05.002
Li SW, Chan BKK, Tam N (2009) Barnacle fouling impedes the gaseous exchange and food production of the mangroves Kandelia obovata, a dominant mangrove species in Hong Kong and Taiwan. Hydrobiologia 618:199–203. https://doi.org/10.1007/s10750-008-9576-9
Li W, Cui LJ, Zhang MY, Wang YF, Zhang YQ, Lei YR, Zhao XS (2015) Effect of mangrove restoration on crab burrow density in Luoyangjiang Estuary. China for Ecosyst 2:21. https://doi.org/10.1186/s40663-015-0046-3
Li YF, Du FY, Gu YG, Ning JJ, Wang LG (2017) Changes of the macrobenthic faunal community with stand age of a non-native mangrove species in Futian Mangrove National Nature Reserve, Guangdong. China Zool Stud 56:19. https://doi.org/10.6620/ZS.2017.56-19
Lin X, Lu CY, Wang Y, Ye Y (2006) Preliminary study on effects of salinity on the attachment of marine fouling fauna barnacles to planted mangrove seedlings. Mar Environ Sci S1:25–28. https://doi.org/10.3969/j.issn.1007-6336.2006.z1.006
Liu W, Tanimura A, Imai T, Kanaya G, Niiyama T (2014) Distribution of gastropods in a tidal flat in association with digestive enzyme activities. Plankton Benthos Res 9:156–167. https://doi.org/10.3800/pbr.9.156
Ma W, Wang WQ, Tang C, Chen GC, Wang M (2020) Zonation of mangrove flora and fauna in a subtropical estuarine wetland based on surface elevation. Ecol Evol 10:7404–7418. https://doi.org/10.1002/ece3.6467
Macintosh DJ, Ashton EC, Havanon S (2002) Mangrove rehabilitation and intertidal biodiversity: a study in the Ranong Mangrove Ecosystem, Thailand. Estuar Coast Shelf Sci 55:331–345. https://doi.org/10.1006/ecss.2001.0896
Magni P, Draredja B, Melouah K, Como S (2015) Patterns of seasonal variation in lagoonal macrozoobenthic assemblages (Mellah lagoon, Algeria). Mar Environ Res 109:168–176. https://doi.org/10.1016/j.marenvres.2015.07.005
Mao D, Liu M, Wang Z, Li L, Man WD, Jia MM, Zhang YZ (2019) Rapid invasion of Spartina alterniflora in the Coastal Zone of Mainland China: Spatiotemporal Patterns and Human Prevention. Sensors 19:2308. https://doi.org/10.3390/s19102308
Martin C, Hanan A, Carlos M (2019) Mangrove forests as traps for marine litter. Environ Pollut 247:499–508. https://doi.org/10.1016/j.envpol.2019.01.067
Meng HY (2017) Study on the Distribution and Habitat election of two burrowing crabs in response to Spartina alterniflora invasion in mangroves of Zhangjiang Estuary, China. Ms.D Thesis, p 156
Morrisey DJ, Skilleter GA, Ellis JI, Burns BR, Kemp CE, Burt K (2003) Differences in benthic fauna and sediment among mangrove (Avicennia marina var. australasica) stands of different ages in New Zealand. Estuar Coast Shelf Sci 56:581–592. https://doi.org/10.1016/S0272-7714(02)00208-1
Neira C, Levin LA, Grosholz ED (2005) Benthic macrofaunal communities of three sites in San Francisco Bay invaded by hybrid Spartina, with comparison to uninvaded habitats. Mar Ecol Prog 292:111–126. https://doi.org/10.3354/meps292111
Ning Q, Lin Y (2004) Characteristics of quantitative distribution and species composition of fouling fauna in Dongwan mangrove stands of Fangchenggang, Guangxi. J Trop Oceanogr 01:64–68. https://doi.org/10.3969/j.issn.1009-5470.2004.01.009
Romañach SS, DeAngelis DL, Koh HL, Li YH, Lee SY, Zhai L, Raja Barizan RS, Zhai L (2018) Conservation and restoration of mangroves: Global status, perspectives, and prognosis. Ocean Coast Manag 154:72–82. https://doi.org/10.1016/j.ocecoaman.2018.01.009
Salmo SG, Lovelock C, Duke NC (2013) Vegetation and soil characteristics as indicators of restoration trajectories in restored mangroves. Hydrobiologia 720:1–18. https://doi.org/10.1007/s10750-013-1617-3
Sueiro MC, Bortolus A, Schwindt E (2012) The role of the physical structure of Spartina densiflora Brong. in structuring macroinvertebrate assemblages. Aquat Ecol 46:25–36. https://doi.org/10.1007/s10452-011-9379-3
Tang M, Kristensen E (2010) Associations between macrobenthos and invasive cordgrass, Spartina anglica, in the Danish Wadden Sea. Helgol Mar Res 64:321–329. https://doi.org/10.1007/s10152-009-0187-2
Tang YJ, Yu SX (2007) Spatial zonation of macrobenthic fauna in Zhanjiang Mangrove Nature Reserve, Guangdong, China. Acta Ecol Sin 27:1703–1714. https://doi.org/10.1016/S1872-2032(07)60042-0
Tang Y, Fang Z, Zhong Y, Zhang ZW, Chen K, An D, Yang XB, Liao BW (2012) Succession of macrofauna communities in wetlands of Sonneratia apetala artificial mangroves during different ecological restoration stages. Acta Ecol Sin 32:3160–3169. https://doi.org/10.5846/stxb201104210525
Wan SW, Qin P, Liu JN, Zhou H (2009) The positive and negative effects of exotic Spartina alterniflora in China. Ecol Eng 35:444–452. https://doi.org/10.1016/j.ecoleng.2008.05.020
Wang SK, Chu TJ, Huang DQ, Li B, Wu JH (2014) Incorporation of exotic Spartina alternifiora into diet of deposit-feeding snails in the Yangtze River Estuary Salt Marsh: Stable Isotope and Fatty Acid Analyses. Ecosystems 17:567–577. https://doi.org/10.1007/s10021-013-9743-3
Warwick RM (2008) Abundance biomass comparison method. In: Jørgensen SE, Fath BD (eds) Encyclopedia of Ecology. Elsevier Science, pp 11–15
Yang JY, Gao AG, Ning XR, Zhang DS (2007) Characteristics of macrofauna and their response to aquiculture in Yueqing Bay, China. Acta Ecol Sin 27:34–40. https://doi.org/10.1016/S1872-2032(07)60008-0
Zhang Y, Huang G, Wang WQ, Chen LZ, Lin GH (2012) Interactions between mangroves and exotic Spartina in an anthropogenically disturbed estuary in southern China. Ecology 93:588–597. https://doi.org/10.1890/11-1302.1
Zhou T, Liu SC, Feng ZL, Liu G, Gan Q, Peng SL (2015) Use of exotic plants to control Spartina alterniflora invasion and promote mangrove restoration. Sci Rep 5:12980. https://doi.org/10.1038/srep12980
Zuo P, Zhao S, Liu C, Wang CH, Liang YB (2012) Distribution of Spartina spp. along China’s coast. Ecol Eng 40:160–166. https://doi.org/10.1016/j.ecoleng.2011.12.014
Acknowledgements
We are grateful to Runjie Jin, Hengwei Wang, Dan Li, Hein Zar Htwe and Nuttiga Hempattarasuwan for assistance with field work. We also thank for doctor Yibo Liao and researcher Hangjun Wang for species identification of macrobenthos.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, Q., Song, L., Agusti, S. et al. Changes of the Macrobenthos Community with Non-native Mangrove Rehabilitation (Kandelia obovata) and Salt Marsh Invasion (Spartina alterniflora) in Ximen Island, Zhejiang, China. Ocean Sci. J. 56, 395–405 (2021). https://doi.org/10.1007/s12601-021-00037-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12601-021-00037-9