Abstract
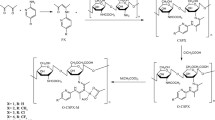
Antifungal activity of synthetic metal complexes of quaternized N-(propyl) chitosan derivatives with Сu(II) against yeastlike (Saccharomyces cereviseae, Rodothorula rubra, and Candida albicans) and mycelial fungi (Fusarium oxysporum, Alternaria alternata, Cladosporium herbarum) was studied. In vitro application (at 250‒500 μg/mL) of the metal complex of quaternized N-(propyl) derivative of low-molecular chitosan with 53% substitution and 1.3% copper ions proved efficient against F. оxysporum, one of ten most common fungal plant pathogens. Water-soluble quaternized N-(propyl) chitosan derivatives with 40−58% degree of substitution were synthesized using glycidyltrimethylammonium chloride under optimally adjusted conditions. Metal complexes of the chitosan derivative with 53% degree of substitution with Сu(II) ions were obtained by dialysis. The quantity of copper ions in the metal complexes was determined by atomic emission spectrometry. The structure of chitosan derivatives was confirmed by spectral analysis (IR, 1H NMR).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Badawy, M.E.I. and Rabea, E.I., Synthesis and antifungal property of N-(aryl) and quaternary N-(aryl) chitosan derivatives against Botrytis cinerea, Cellulose, 2014, vol. 21, no. 4, pp. 3121–3137.
Badawy, M.E.I., Structure and antimicrobial activity relationship of quaternary N-alkyl chitosan derivatives against some plant pathogens, J. Appl. Polym. Sci., 2010, vol. 117, no. 2, pp. 960–969.
Bratskaya, S.Yu. and Pestov, A.V., Khelatiruyushchie proizvodnye khitozana (Chelating Chitosan Derivatives), Vladivostok: Dal’nauka, 2016.
Cho, J., Grant, J., Piquette-Miller, M., and Allen, C., Synthesis and physicochemical and dynamic mechanical properties of a water-soluble chitosan derivative as a biomaterial, Biomacromol, 2006, vol. 7, no. 10, pp. 2845–2855.
de Oliveira Pedro, R., Takaki, M., Gorayeb, T.C., Bianchi, V.L., Thomeo, J.C., Tiera, M.J., and de Oliveira Tiera, V.A., Synthesis, characterization and antifungal activity of quaternary derivatives of chitosan on Aspergillus flavus, Microbiol. Res., 2013, vol. 168, no. 1, pp. 50–55.
Guo, Z., Xing, R., Liu, S., Zhong, Z., Ji, X., Wang, L., and Li, P., The influence of the cationic of quaternized chitosan on antifungal activity, Int. J. Food Microbiol., 2007, vol. 118, no. 2, pp. 214–217.
Il’ina, A.V., Kulikov, S.N., Chalenko, G.I., Gerasimova, N.G., and Varlamov, V.P., Obtaining and study of monosaccharide derivatives of low-molecularweight chitosan, Appl. Biochem. Microbiol., 2008, vol. 45, no. 5, pp. 551–558.
Kulikov, S.N., Khairullin, R.Z., Tyurin, Yu.A., Lisovskaya, S.A., Glushko, N.I., Levov, A.N., and Varlamov, V.P., Fungicidal activity of chitosan against mycelial fungi, Prakt. Med., 2010, no. 1 (40), pp. 119–120.
Khaldeeva, E.V., Glushko, N.I., Lisovskaya, S.A., Parshakov, V.R., and Saifieva, O.V., Allergenic fungi in presentday dwellings, Prakt. Med., 2011, vol. 51, no. 3, pp. 122–124.
Kulikov, S.N., Lisovskaya, S.A., Bezrodnykh, E.A., Zelenikhin, P.V., Shakirova, D.R., and Tikhonov, V.E., Antifungal activity of oligochitosans (short chain chitosans) against some Candida species and clinical isolates of C. albicans: molecular weight–activity relationship, Eur. J. Med. Chem., 2014, vol. 74, pp. 169–178.
Kulikov, S.N., Nemtsev, S.V., Varlamov, V.P., Alimova, F.K., and Zakharova, N.G., Biological preparations with different mechanisms of action for protecting potato against fungal diseases, Appl. Biochem. Microbiol., 2006, vol. 42, no. 1, pp. 77–83.
Kulikov, S.N., Tyurin, Yu.A., Fassakhov, R.S., and Varlamov, V.P., Antibacterial and antifungal activity of chitosan: mechanisms of action and role of the structure, Zh. Mikrobiol. Epidemiol. Immunobiol., 2009, no. 5, pp. 91–98.
Lisovskaya, S., Khaldeeva, E., Salmakova, A., and Kulikov, S.N., Evaluation of the influence of antifungal drugs on growth of Candida albicans in the biofilm, BioNanoScience, 2016, vol. 6, no. 4, p. 588–590.
Lun’kov, A.P., Shagdarova, B.Ts., and Il’ina, A.V., Synthesis of quaternized chitosan derivatives and investigation of their properties, Izv. Ufim. Nauch. Tsentr RAN, 2016, no. 3 (1), pp. 56–58.
Mari, A., Schneider, P., Wally, V., Breitenbach, M, and Simon-Nobbe, B., Sensitization to fungi: epidemiology, comparative skin tests, and IgE reactivity of fungal extracts, Clin. Exp. Allergy, 2003, vol. 33, no. 10, pp. 1429–1438.
Mekahlia, S. and Bouzid, B., Chitosan-copper (II) complex as antibacterial agent: synthesis, characterization and coordinating bond-activity correlation study, Phys. Procedia, 2009, vol. 2, no. 3, pp. 1045–1053.
Qin, C., Xiao, Q., Li, H., Fang, M., Liu, Y., Chen, X., and Li, Q. Calorimetric studies of the action of chitosan-N-2-hydroxypropyl trimethyl ammonium chloride on the growth of microorganisms, Int. J. Biol. Macromol., 2004, vol. 34, no. 1–2, pp. 121–126.
Rahman, M.H., Hjeljord, L.G., Aam, B.B., Sørlie, M., and Tronsmo, A., Antifungal effect of chito-oligosaccharides with different degrees of polymerization, Eur. J. Plant. Pathol., 2015, vol. 141, no. 1, pp. 147–158.
Rahman, M.H., Shovan, L.R., Hjeljord, L.G., Aam, B.B., Eijsink, V.G.H., and Tronsmo, M.S.A., Inhibition of fungal plant pathogens by synergistic action of chito-oligosaccharides and commercially available fungicides, PLoS One, 2014, vol. 9, no. 4, e93192.
Schmuhl, R., Krieg, H.M., and Keizer, K., Adsorption of Cu(II) and Cr(VI) ions by chitosan: kinetics and equilibrium studies, Water SA, 2001, vol. 27, no. 1, pp. 1–7.
Seyfarth, F., Schliemann, S., Elsner, P., and Hipler, U.-C., Antifungal effect of high-and low-molecular-weight chitosan hydrochloride, carboxymethyl chitosan, chitosan oligosaccharide and N-acetyl-d-glucosamine against Candida albicans, Candida krusei and Candida glabrata, Int. J. Pharmaceut., 2008, vol. 353, no. 1–2, pp. 139–148.
Shagdarova, B.Ts., Il’ina, A.V., and Varlamov, V.P., Antibacterial activity of alkylated and acylated derivatives of low-molecular weight chitosan, Appl. Biochem. Microbiol., 2016, vol. 52, no. 2, pp. 222–225.
Varma, A.J., Deshpande, S.V., and Kennedy, J.F., Metal complexation by chitosan and its derivatives: a review, Carbohydr. Polym., 2004, vol. 55, no. 1, pp. 77–93.
Xiao, B., Wan, Y., Wang, X., Zha, Q., Liu, H., Qiu, Z., and Zhang, S., Synthesis and characterization of N-(2-hydroxy)propyl-3-trimethyl ammonium chitosan chloride for potential application in gene delivery, Colloids Surf. B: Biointerfaces, 2012, vol. 91, pp. 168–174.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.V. Il’ina, B.Ts. Shagdarova, A.P. Lun’kov, S.N. Kulikov, V.P. Varlamov, 2017, published in Mikrobiologiya, 2017, Vol. 86, No. 5, pp. 586–592.
Rights and permissions
About this article
Cite this article
Il’ina, A.V., Shagdarova, B.T., Lun’kov, A.P. et al. In vitro antifungal activity of metal complexes of a quaternized chitosan derivative with copper ions. Microbiology 86, 590–595 (2017). https://doi.org/10.1134/S0026261717050101
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026261717050101