Abstract
This study aimed to identify the prevalence, clinical and radiographic characteristics, and risk factors for cognitive dysfunction in patients with Neuromyelitis optica spectrum disorder (NMOSD). Eighty-three participants who were diagnosed with NMOSD were recruited. Cognitive function was assessed using the Montreal Cognitive Assessment (MoCA) and Frontal Assessment Battery (FAB). The mean age of the patients was 47.78 ± 13.14 years, with an average of 12.05 ± 4.62 years of formal education. The majority (54%) exhibited cognitive impairment, defined by a MoCA score < 25 (mean: 22.96 ± 3.82). Disease severity (evaluated by the Expanded Disability Status Scale) and lower formal education levels were associated with cognitive impairment (p = 0.011 and < 0.001, respectively). The annualized relapse rate, disease duration, and AQP4 antibody status were not associated with cognitive impairment. Interestingly, informant-reported cognitive decline was associated with poorer cognitive performance (p = 0.027). Radiographic findings of lesion location and severity were associated with MoCA-assessed cognitive performance, particularly for lesions in the right parietal lobes (p = 0.023). Hippocampal atrophy was negatively correlated with FAB scores. In conclusion, approximately half of the Thai patients with NMOSD exhibited cognitive impairment, which was associated with age, formal education level, disease severity, relative perception, and specific radiological findings. Further studies incorporating comprehensive neuropsychological tests and subjective cognitive complaints are warranted.
Similar content being viewed by others
Introduction
Neuromyelitis optica spectrum disorder (NMOSD) is an autoimmune demyelinating disease that leads to inflammation and destruction of white matter in the central nervous system1. NMOSD affects all age groups and predominates in females2,3. In Thailand, the disease is more prevalent than multiple sclerosis, another autoimmune demyelinating disease, resulting in greater morbidity and impaired quality of life2,4.
According to studies3,5,6,7,8,9,10,11, the reported prevalence of cognitive decline in patients with NMOSD is 30–70%, a range that varies with the study methodology and cognitive assessment tests employed. The primary deficits are slowed information processing, episodic memory decline, executive function impairment, and attention deficits3,6,7,9,12,13. Higher educational levels are protective against cognitive decline3,9,10. Physical factors have demonstrated both correlations and noncorrelations with cognitive levels3,8,9,10,13,14. Older adults tend to exhibit greater attention and verbal memory impairments than younger individuals3,8,15. Furthermore, radiological characteristics observed in brain magnetic resonance imaging (MRI) correlate with the degree and types of cognitive decline in patients with NMOSD3,7.
Various assessments have been used to evaluate cognitive function in patients with NMOSD. However, in resource-constrained settings, more accessible and less comprehensive tools are prioritized. The Montreal Cognitive Assessment (MoCA) is widely used for quickly assessing global cognitive function16. Studies have shown significantly lower MoCA scores in patients with NMOSD than in healthy controls6,17. The validated Thai version of the MoCA uses a cutoff score of less than 25 to indicate cognitive impairment18. The Frontal Assessment Battery (FAB), consisting of six subtests, is a simple tool for assessing frontal lobe dysfunction and can be administered by any practitioner19. No studies have yet evaluated FAB in patients with NMOSD.
This research aimed to study the prevalence and characteristics of cognitive impairment in patients with NMOSD at Siriraj Hospital using accessible tools, including the MoCA and FAB, and their correlation with radiological features. Additionally, we examined factors related to cognitive impairment, such as baseline patient characteristics, subjective cognitive complaints from patients and informants, and other NMOSD-related factors.
Methods
Participants
This cross-sectional study received approval from the Institutional Review Board, Faculty of Medicine Siriraj Hospital (Si 576/2019), in full compliance with international guidelines for human research protection, including the Declaration of Helsinki and the International Conference on Harmonization in Good Clinical Practice. Written informed consent was obtained from each participant. Eligible participants were recruited from Siriraj Hospital between August 2019 and August 2020. The inclusion criterion for patients was a diagnosis of NMOSD according to the 2015 International Panel for Neuromyelitis Optica Diagnosis criteria1, with the last attack occurring more than 3 months before enrollment. Patients were excluded if they did not voluntarily participate, had a dementia diagnosis from other causes, had any active primary psychiatric disorder other than depression, or had physical limitations for cognitive testing based on the Expanded Disability Status Scale (EDSS).
Demographic data and laboratory assessment
We collected primary demographic data and clinical information from participants and informants: age, sex, handedness, formal education level, occupation, comorbidities, and subjective cognitive complaints (such as episodic memory loss or slow thought-processing speed). Regarding the clinical characteristics of patients with NMOSD, we recorded age at onset, disease duration, presentation of each attack, aquaporin-4 (AQP4) antibody status, which was determined by cell-based assay, number of relapses, and previous and current medications. The EDSS scores were obtained20. Brain MRIs from each participant were evaluated for T2 lesions, their locations, and gadolinium-enhanced lesions.
Cognitive assessment
Eligible participants and their informants were questioned about any perceived cognitive decline in the former. All participants underwent cognitive assessment using the Thai version of the MoCA18 and the FAB19. One investigator determined the scores for all seven MoCA subcategories (Visuospatial/Executive, Naming, Attention, Language, Abstraction, Delayed Recall, and Orientation), the total MoCA score, and the time required to complete the test. In the letter fluency task of the MoCA Language subcategory and in the Lexical Fluency subcategory of the FAB, the number of words starting with the Thai letters “Koh” and “Soh,” respectively, was recorded. The Memory Index Score21, which is derived from the Delayed Recall subtest in the MoCA, was calculated (score range: 0–15). A MoCA cutoff score of less than 25 was used to define the cognitively impaired group16,22.
MRI characteristics
The MRI Brain scans at remission of eligible participants were reviewed. The deep white matter lesions were assessed using the Fazekas scale23. The Fazekas scale assigns scores of 1 for punctate lesions, 2 for mild confluence lesions, and 3 for large confluence lesions. The Fazekas scale was also applied to evaluate white matter lesions in each lobe of the brain. The degree of medial temporal lobe atrophy was evaluated using the medial temporal lobe atrophy scale24,25, which employs scores ranging from 0 to 4. This assessment evaluates the choroidal fissure width, temporal horn, and hippocampal formation height on a five-point scale (0–4). Higher scores indicate greater hippocampal atrophy. Additionally, global brain shrinkage was assessed using the global cortical atrophy scale, with scores ranging from 0 (no atrophy) to 3 (severe atrophy)26.
Statistical analysis
Data analyses were performed using Stata Statistical Software, release 18 (StataCorp LLC, College Station, TX, USA). The data distribution was tested using the Kolmogorov–Smirnov test and Levene’s test for equality of variances. Baseline characteristics and clinical data were analyzed using descriptive statistics. The results are presented as frequencies, percentages, means with standard deviations (SDs), or medians with interquartile ranges (IQRs). Comparisons between patients with cognitive impairment (MoCA score < 25) and those with preserved cognition (MoCA score ≥ 25) were analyzed using unpaired t-tests, chi-square tests, or Fisher’s exact tests. A subsequent partial correlation analysis was conducted for participant characteristics, accounting for age, sex and years of formal education. Correlations between continuous data on clinical and radiological characteristics and cognitive test scores, including each subsection, are presented using Spearman’s correlation coefficients. A p value less than 0.05 indicated statistical significance.
Results
Demographic data
Eighty-three patients with NMOSD participated in this study (Table 1). The cohort was predominantly female (82.9%), with a mean age of 47.8 ± 13.1 years and an average formal education level of 12.1 ± 4.6 years. During the study, 75.9% of patients were employed. Fifty-five participants (66.3%) complained of memory decline, and 47 (56.6%) reported slow brain-processing speed. Informant complaints about cognitive problems, primarily from relatives, were noted in 25 participants (30.1%). Only six participants (7.2%) had a diagnosis of depression.
Clinical characteristics of Patients with NMOSD
Serum AQP4 IgG was detected in 74 (89.2%) participants. The average age at onset was 38.4 ± 13.5 years, with a mean disease duration of 9.2 ± 7.2 years. The median annualized relapse rate was 0.44 (IQR 0.27–0.83). The most common presentations were myelitis (68.7%) and optic neuritis (62.7%). The median duration since the last relapse was 42 months (IQR 15–74).
NMOSD severity, according to the EDSS score, was mild (EDSS < 3) in approximately half of the participants (54%). Residual cerebral lesion symptoms, such as decreased mental acuity or fatigue, were detected in only 1.2% of patients during the remission period. Participants used various medications that may affect cognition, including gamma-aminobutyric acid modulating agents (46, 55.4%), anticholinergic drugs (60, 72.3%), and muscle relaxants (71, 85.5%). Vitamin D levels were within the standard limits in almost all patients; most patients received vitamin D supplements regardless of their levels.
Cognitive assessment results
The mean MoCA score was 22.96 ± 3.82, and the mean time to complete the MoCA was 10.4 ± 2.57 min. On average, Patients with NMOSD could recall only 1.96 items in the 5-item Delayed Recall subcategory. The mean Memory Index Score derived from the MoCA was 8.9 ± 3.7 out of 15, indicating moderate improvement of recall memory with a cue.
The mean FAB score was 15.27 ± 2.47, suggesting only mild frontal lobe dysfunction in the patients with NMOSD. The two affected subcategories were Lexical Fluency and Similarities.
Table 1 presents the details of the patient characteristics and cognitive test results.
Correlation between subjective cognitive complaints and cognitive assessments
The groups that had informant complaints had lower adjusted MoCA scores (mean = 21.56, SD = 0.81) than those without complaints (mean = 23.57, SD = 0.47; t[81] = − 2.25, p = 0.03). Analysis of individual MoCA components revealed that informant complaints were associated with decreased scores for the Delayed Recall (t[81] = − 2.61, p = 0.01) and Orientation (t[81] = − 2.39, p = 0.02) subcategories, as well as with a lower Memory Index Score (t[81] = − 2.52, p = 0.01). No correlation was found between participant or informant complaints and FAB scores. Supplementary Table S1 includes additional analyses between complaints and cognitive assessments.
Radiological characteristics of participants
Complete brain or spinal cord MRI data were available for 76 participants, with a median duration of 15 months before study enrollment. T2 hyperintense lesions were present in 93.9% of participants, and 92.2% of those lesions were compatible with an NMOSD diagnosis. Most T2 lesions were located in the spinal cord (65.9%), cerebral hemisphere (61.4%), and optic nerve (48.8%). Lesions associated with cognitive pathways were found in the caudate (3, 3.95%), globus pallidus (8, 10.53%), and thalamus (13, 17.11%). Gadolinium-enhanced lesions were observed in 24.7% of the participants.
Correlations between radiological findings and cognitive assessments
The MoCA score (r = − 0.285, p = 0.023) and the Delayed Recall subscore of the MoCA (r = − 0.268, p = 0.033) were negatively correlated with subcortical white matter lesions in the right parietal lobe. The Memory Index Score was also associated with right parietal lobe lesions (r = − .313, p = 0.012). The letter fluency for the Thai letter “Koh” in the MoCA was associated with lesions in both parietal lobes (left: r = − 0.356, p = 0.004; right: r = − 0.285, p = 0.020). In contrast, the letter fluency for the Thai letter “Soh” in the FAB was not associated with any lesion. The time to complete the MoCA was positively correlated with lesions in the left frontal lobe (r = 0.289, p = 0.021) and both parietal lobes (left: r = 0.362, p = 0.004; right: r = 0.406, p = 0.001). However, the FAB scores did not correlate with subcortical white matter lesions.
Left hippocampal atrophy was associated with the time to complete the MoCA (r = 0.322, p = 0.018). Both left and right hippocampal atrophy were significantly associated with the total FAB score (left: r = − 0.288, p = 0.038; right: r = − 0.363, p = 0.007) and the Go No-Go subscore of the FAB (left: r = − 0.312, p = 0.025; right: r = − 0.393, p = 0.050). Left hippocampal atrophy was also associated with the FAB Similarity subscore (r = − 0.327, p = 0.016).
Global cortical atrophy was positively correlated with the time to complete the MoCA (r = 0.328, p = 0.018). Cerebral microbleeds were associated with the Visuospatial/Executive subscore of the MoCA (r = − 0.284, p = 0.044) and the Similarities subscore of the FAB (r = − 0.355, p = 0.008). No discernible connection was found between cognitive performance and periventricular lesions.
Table 2 presents the correlations between the radiographic findings and the cognitive test results.
Comparison between participants with or without cognitive impairments
Patients with cognitive impairment had significantly lower formal education levels than those with preserved cognition (9.9 vs 14.6 years, p < 0.001). No significant differences were found between participants with impaired cognition (MoCA < 25) or preserved cognition (MoCA ≥ 25) in age, sex, vascular comorbidities, subjective cognitive complaints in either the memory or nonmemory domain, AQP4 antibody status, age at NMOSD onset, or disease duration (all p > 0.05).
There was no significant difference in disease severity between patients with cognitive impairment and those with preserved cognition (p = 0.093). However, when stratifying patients with NMOSD based on EDSS into mild, moderate, and severe groups, there were significant differences in severity between groups of patients with cognitive impairment and preserved cognitive (p = 0.011). Table 3 presents detailed comparisons between participants with and without cognitive impairment.
Factors associated with cognitive performance
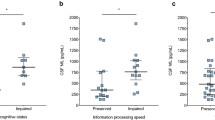
The study revealed significant associations between several cognitive measures (MoCA score, Memory Index Score, time to complete the MoCA, and FAB score) and various demographic and clinical factors. These factors were age, formal-educational level, age at onset, and EDSS score (disease severity). All associations were significant at p < 0.05 (Fig. 1 and Table 4). However, no significant correlations were observed after adjusting for age, sex, and years of formal education (p > 0.05) (Supplementary Table S2).
Association between severity of disease and Montreal Cognitive Assessment score. Abbreviations: EDSS, Expanded Disability Status Scale; MoCA, Montreal Cognitive Assessment. (A) Analysis of variance between patients with mild (EDSS < 3), moderate (EDSS 3–6), and severe (EDSS ≥ 6) disease severity, demonstrating a significant difference in MoCA scores. (B) Scatterplot showing negative correlations between EDSS and MoCA scores.
Discussion
In our study, 54% of the Thai patients with NMOSD at Siriraj Hospital experienced cognitive impairment, according to the MoCA. The mean MoCA score was 22.96, which is below the cutoff point for the Thai population16,18,22. This finding is similar to that of a previous study on Asian Patients with NMOSD by Zhang et al.8, which included the MoCA. Their investigation revealed a lower MoCA score in Patients with NMOSD than in healthy controls, with a mean score 24.42.
Previous studies on cognitive impairment in patients with NMOSD have reported a prevalence of 30–70%, which is consistent with our findings3,5,6,7,8,9. A meta-analysis reported significantly worse cognitive function in patients with NMOSD than in healthy subjects, particularly in attention, language, short-term memory, information-processing speed, and executive function6,12. Our results align with these findings, as indicated by the MoCA subscores in each domain. The cognitive tests in our study were not specifically designed to evaluate information processing speed. Consequently, we endeavored to gauge impairments in brain processing speed via the MoCA testing time as well as the number of words generated in MoCA’s Language subtest and FAB’s Lexical Fluency component.
The factors affecting cognition in the patients with NMOSD in our study were formal education level and disease severity, as defined by the EDSS score. Our comparison of EDSS scores between groups revealed significantly better scores in the cognitively preserved group. Additionally, we found significant correlations between MoCA scores and age, age at disease onset, years of formal education, disease severity according to the EDSS score, FAB score, and time to complete the MoCA. However, after adjusting for age and formal educational level, there was no significant correlation between MoCA scores and disease severity. This finding may be explained by the influence of age and years of formal education on cognitive impairment, consistent with previous studies8,10,13,14.
An interesting finding was the significance of complaints from patients’ relatives. Our study showed that the MoCA scores of patients were significantly lower when their relatives reported the patient’s cognitive problems, regardless of whether the patients themselves reported cognitive issues. In contrast, when only the patients reported experiencing cognitive difficulties, without their relatives reporting any problems, the MoCA scores did not differ significantly from those of patients whose relatives did not report any cognitive concerns. No previous NMOSD study has mentioned this finding, but studies in other diseases, including Alzheimer’s disease, have shown correlations between informant perception and objective cognitive decline27,28,29,30,31,32.
Our analysis of radiological findings revealed correlations between the severity and location of brain lesions observed on MRI scans and the affected brain areas. Specifically, lesions in the right parietal lobe were linked to lower MoCA scores and subscores (particularly for Delayed Recall) as well as Memory Index Scores. This finding was likely due to the association of the parietal cortex with episodic memory33,34. Similarly, the association between impairment in letter fluency for the Thai letter “Koh” and bilateral parietal lobe lesions aligns with previous research showing parietal lobe activation during letter fluency tasks in PET scans35,36,37. Interestingly, we noted that lesions in the left frontal and left parietal lobes were associated with the time to complete the MoCA but not with the MoCA score. This finding suggests that such lesions have a subtle effect on brain processing speed that is not directly reflected in the MoCA score.
Consistent with previous studies, FAB and subtests, such as the Go No-Go test, were negatively associated with medial cortical atrophy and hippocampal atrophy38. Our study showed that while global cortical atrophy is not associated with MoCA scores, it is associated with the time to complete the MoCA, indicating slower brain processing speed. Furthermore, we observed associations between cortical microbleeds and visuospatial function, as reflected in the MoCA subscores, similar to previous reports in patients with Alzheimer’s disease39.
It is worth mentioning that this study enrolled a large number of patients with NMOSD and was the first to explore cognition among patients with NMOSD in Thailand. We utilized the accessible MoCA and FAB, which can be easily applied in clinical practice. This study also included the assessment of subjective cognitive complaints reported by patients (self-reports) and their relatives (informant reports). This approach has not been previously utilized for patients with NMOSD.
There are several limitations to this study. First, we used only the MoCA and the FAB to assess cognitive function, which might not fully represent all affected cognitive domains. More studies using comprehensive neuropsychological tests are required to describe the characteristics of cognitive problems in patients with NMOSD. However, the tests we used (MoCA and FAB) are accessible and applicable to outpatient settings with limited time and resources. Second, the time to complete the MoCA may vary based on the speech speed of different examiners. To minimize this issue, we used only one examiner for the instructions. Third, only a small number of participants reported a depressed mood. Depression is a well-described factor associated with cognitive decline and has a high prevalence in patients with NMOSD, reaching 42.8–58.3%5,10,11. The surprisingly low prevalence of depression in this study might be because the screening assessment we used for depression was only the self-reported Patient Health Questionnaire-240. More assessments of mood problems are needed, and the correlation between depression and cognitive problems should be examined further.
Conclusions
Most of the Thai patients with NMOSD had cognitive impairment, which was associated with formal education level, disease severity, and the perceptions of their relatives. Correlations were found between cognition and NMOSD radiographic characteristics. Further studies using more comprehensive cognitive tests, including evaluations of mood, behavior, and subjective cognitive complaints, are needed for better future care of patients with NMOSD.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Wingerchuk, D. M. et al. International consensus diagnostic criteria for neuromyelitis optica spectrum disorders. Neurology 85, 177–189. https://doi.org/10.1212/wnl.0000000000001729 (2015).
Siritho, S., Nakashima, I., Takahashi, T., Fujihara, K. & Prayoonwiwat, N. AQP4 antibody-positive Thai cases: Clinical features and diagnostic problems. Neurology 77, 827–834. https://doi.org/10.1212/WNL.0b013e31822c61b1 (2011).
Liu, Y. et al. Structural MRI substrates of cognitive impairment in neuromyelitis optica. Neurology 85, 1491–1499. https://doi.org/10.1212/wnl.0000000000002067 (2015).
Tisavipat, N. et al. The epidemiology and burden of neuromyelitis optica spectrum disorder, multiple sclerosis, and MOG antibody-associated disease in a province in Thailand: A population-based study. Mult. Scler Relat. Disord. 70, 104511. https://doi.org/10.1016/j.msard.2023.104511 (2023).
Moore, P. et al. Cognitive and psychiatric comorbidities in neuromyelitis optica. J. Neurol. Sci. 360, 4–9. https://doi.org/10.1016/j.jns.2015.11.031 (2016).
Meng, H. et al. Cognitive dysfunction in adult patients with neuromyelitis optica: A systematic review and meta-analysis. J. Neurol. 264, 1549–1558. https://doi.org/10.1007/s00415-016-8345-3 (2017).
Oertel, F. C., Schließeit, J., Brandt, A. U. & Paul, F. Cognitive impairment in neuromyelitis optica spectrum disorders: A review of clinical and neuroradiological features. Front. Neurol. 10, 608. https://doi.org/10.3389/fneur.2019.00608 (2019).
Zhang, N. et al. Cognitive impairment in Chinese neuromyelitis optica. Mult. Scler. 21, 1839–1846. https://doi.org/10.1177/1352458515576982 (2015).
Kim, S. H. et al. Cognitive impairment differs between neuromyelitis optica spectrum disorder and multiple sclerosis. Mult. Scler. 22, 1850–1858. https://doi.org/10.1177/1352458516636246 (2016).
Vanotti, S. et al. Cognitive performance of neuromyelitis optica patients: Comparison with multiple sclerosis. Arq. Neuropsiquiatr. 71, 357–361. https://doi.org/10.1590/0004-282x20130038 (2013).
Hollinger, K. R. et al. Cognition, mood, and purpose in life in neuromyelitis optica spectrum disorder. J. Neurol. Sci. 362, 85–90. https://doi.org/10.1016/j.jns.2016.01.010 (2016).
He, D., Chen, X., Zhao, D. & Zhou, H. Cognitive function, depression, fatigue, and activities of daily living in patients with neuromyelitis optica after acute relapse. Int. J. Neurosci. 121, 677–683. https://doi.org/10.3109/00207454.2011.608456 (2011).
Blanc, F. et al. Cognitive functions in neuromyelitis optica. Arch. Neurol. 65, 84–88. https://doi.org/10.1001/archneurol.2007.16 (2008).
Blanc, F. et al. White matter atrophy and cognitive dysfunctions in neuromyelitis optica. PLoS One 7, e33878. https://doi.org/10.1371/journal.pone.0033878 (2012).
Saji, E. et al. Cognitive impairment and cortical degeneration in neuromyelitis optica. Ann. Neurol. 73, 65–76. https://doi.org/10.1002/ana.23721 (2013).
Nasreddine, Z. S. et al. The Montreal Cognitive Assessment, MoCA: A brief screening tool for mild cognitive impairment. J. Am. Geriatr. Soc. 53, 695–699. https://doi.org/10.1111/j.1532-5415.2005.53221.x (2005).
Vlahovic, L. et al. Prevalence, demographic, and clinical factors associated with cognitive dysfunction in patients with neuromyelitis optica spectrum disorder. Neurology 102, e207965. https://doi.org/10.1212/wnl.0000000000207965 (2024).
Tangwongchai, S. et al. The validity of thai version of the montreal cognitive assessment (MoCA-T). Vol. 3 (2009).
Dubois, B., Slachevsky, A., Litvan, I. & Pillon, B. The FAB: A frontal assessment battery at bedside. Neurology 55, 1621–1626. https://doi.org/10.1212/wnl.55.11.1621 (2000).
Kurtzke, J. F. Rating neurologic impairment in multiple sclerosis: An expanded disability status scale (EDSS). Neurology 33, 1444–1452. https://doi.org/10.1212/wnl.33.11.1444 (1983).
Kaur, A., Edland, S. D. & Peavy, G. M. The MoCA-memory index score: An efficient alternative to paragraph recall for the detection of amnestic mild cognitive impairment. Alzheimer Dis. Assoc. Disord. 32, 120–124. https://doi.org/10.1097/wad.0000000000000240 (2018).
Hemrungrojn, S. et al. Use of the montreal cognitive assessment thai version to discriminate amnestic mild cognitive impairment from Alzheimer’s disease and healthy controls: Machine learning results. Dement. Geriatr. Cogn. Disord. 50, 183–194. https://doi.org/10.1159/000517822 (2021).
Fazekas, F., Chawluk, J. B., Alavi, A., Hurtig, H. I. & Zimmerman, R. A. MR signal abnormalities at 1.5 T in Alzheimer’s dementia and normal aging. AJR Am. J. Roentgenol. 149, 351–356. https://doi.org/10.2214/ajr.149.2.351 (1987).
Wahlund, L. O., Julin, P., Johansson, S. E. & Scheltens, P. Visual rating and volumetry of the medial temporal lobe on magnetic resonance imaging in dementia: a comparative study. J. Neurol. Neurosurg. Psychiatry 69, 630–635. https://doi.org/10.1136/jnnp.69.5.630 (2000).
Scheltens, P., Launer, L. J., Barkhof, F., Weinstein, H. C. & van Gool, W. A. Visual assessment of medial temporal lobe atrophy on magnetic resonance imaging: Interobserver reliability. J. Neurol. 242, 557–560. https://doi.org/10.1007/bf00868807 (1995).
Pasquier, F. et al. Inter- and intraobserver reproducibility of cerebral atrophy assessment on MRI scans with hemispheric infarcts. Eur. Neurol. 36, 268–272. https://doi.org/10.1159/000117270 (1996).
Caselli, R. J. et al. Subjective cognitive decline: self and informant comparisons. Alzheimers Dement. 10, 93–98. https://doi.org/10.1016/j.jalz.2013.01.003 (2014).
Gifford, K. A. et al. Inclusion of an informant yields strong associations between cognitive complaint and longitudinal cognitive outcomes in non-demented elders. J. Alzheimers Dis. 43, 121–132. https://doi.org/10.3233/jad-131925 (2015).
Gifford, K. A. et al. The source of cognitive complaints predicts diagnostic conversion differentially among nondemented older adults. Alzheimers Dement. 10, 319–327. https://doi.org/10.1016/j.jalz.2013.02.007 (2014).
Nosheny, R. L. et al. Online study partner-reported cognitive decline in the Brain Health Registry. Alzheimers Dement. 4, 565–574. https://doi.org/10.1016/j.trci.2018.09.008 (2018).
Nosheny, R. L. et al. Study partner-reported decline identifies cognitive decline and dementia risk. Ann. Clin. Transl. Neurol. 6, 2448–2459. https://doi.org/10.1002/acn3.50938 (2019).
Rattanabannakit, C. et al. The Cognitive change index as a measure of self and informant perception of cognitive decline: Relation to neuropsychological tests. J. Alzheimers Dis. 51, 1145–1155. https://doi.org/10.3233/JAD-150729 (2016).
Cabeza, R., Ciaramelli, E., Olson, I. R. & Moscovitch, M. The parietal cortex and episodic memory: An attentional account. Nat. Rev. Neurosci. 9, 613–625. https://doi.org/10.1038/nrn2459 (2008).
Davidson, P. S. et al. Does lateral parietal cortex support episodic memory? Evidence from focal lesion patients. Neuropsychologia 46, 1743–1755. https://doi.org/10.1016/j.neuropsychologia.2008.01.011 (2008).
Baldo, J. V., Schwartz, S., Wilkins, D. & Dronkers, N. F. Role of frontal versus temporal cortex in verbal fluency as revealed by voxel-based lesion symptom mapping. J. Int. Neuropsychol. Soc. 12, 896–900. https://doi.org/10.1017/s1355617706061078 (2006).
Gourovitch, M. L. et al. A comparison of rCBF patterns during letter and semantic fluency. Neuropsychology 14, 353–360. https://doi.org/10.1037/0894-4105.14.3.353 (2000).
Curtis, V. A. et al. Attenuated frontal activation during a verbal fluency task in patients with schizophrenia. Am. J. Psychiatry 155, 1056–1063. https://doi.org/10.1176/ajp.155.8.1056 (1998).
Nagata, T. et al. Association between executive dysfunction and hippocampal volume in Alzheimer’s disease. Int. Psychogeriatr. 23, 764–771. https://doi.org/10.1017/s1041610210002164 (2011).
Paradise, M. et al. The relationship of cerebral microbleeds to cognition and incident dementia in non-demented older individuals. Brain Imaging Behav. 13, 750–761. https://doi.org/10.1007/s11682-018-9883-3 (2019).
Kroenke, K., Spitzer, R. L. & Williams, J. B. The patient health questionnaire-2: Validity of a two-item depression screener. Med. Care 41, 1284–1292. https://doi.org/10.1097/01.Mlr.0000093487.78664.3c (2003).
Acknowledgements
The authors acknowledge Dr Saowalak Hunnangkul for the statistical data analysis.
Author information
Authors and Affiliations
Contributions
N.A.: Data analysis and interpretation; review of results; drafting of the manuscript. C.L.: Study conception and design; research operations; data collection; data analysis and interpretation; review of results; drafting of the manuscript. I.M.: Study conception and design; research operations; data collection; data analysis and interpretation; review of results; drafting of the manuscript. J.W.: Study conception and design; research operations; data collection; data analysis. S.S.: Study conception and design; research operations; data collection; data analysis; review of results; manuscript revision. J.J.: Study conception and design; research operations; data collection; data analysis. T.O.: Research operations; data collection; manuscript revision. N.P.: Study conception and design; research operations; manuscript revision. V.S.: Study conception and design; research operations; manuscript revision. C.R.: Study conception and design; research operations; data collection; data analysis and interpretation; review of results; critical revision of the manuscript for important intellectual content; final approval of the submitted manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Apiraksattayakul, N., Lerttanatum, C., Maethasith, I. et al. Cognitive performance in patients with neuromyelitis optica: clinical and imaging characteristics. Sci Rep 14, 20201 (2024). https://doi.org/10.1038/s41598-024-71176-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-71176-x
- Springer Nature Limited