Abstract
Sharks play a key role in the functioning of marine ecosystems and maintenance of trophic web balance, including life cycles of parasites co-occurring in their habitats. We investigated the structure of parasite communities of three sympatric shark species (Etmopterus spinax, Galeus melastomus, and Scyliorhinus canicula) and explored both the influence of host features in shaping the communities and their role as biological indicators of environment stability in the Gulf of Naples (central Mediterranean Sea), a geographical area characterized by strong anthropic pressure. Parasites found were all trophic transmitted helminths with a complex life cycle, except Lernaeopoda galei, that is a ecto-parasite copepod. Communities were all similarly impoverished with 4–5 component species and low values of species richness and diversity. Higher abundance of cestode larvae of the genus Grillotia was found in G. melastomus, although their dominance in all host species suggests that the three sharks have a similar role as intermediate/paratenic hosts in local food webs. Similarly, high abundance of Grillotia larvae could also suggest the occurrence of high abundance of largest top predators in the area. Host morphological (fork length in S. canicula and G. melastomus and body condition index in G. melastomus) and physiological (sex and gonadosomatic and hepatosomatic indices in S. canicula) variables were differently correlated to parasite community structures depending by host species. Potential reasons for the present impoverished parasite communities are discussed.
Similar content being viewed by others
Introduction
Parasites are ubiquitous inhabitants of most ecosystems, occurring in all food webs at all trophic levels1,2,3,4. In the marine realm, the majority of them are trophic transmitted and exhibit a complex life cycle, requiring a number of intermediate/paratenic and definitive invertebrate and vertebrate hosts to reach their adult stage1,2,3. As these hosts include obligate zooplanktonic and benthic taxa, the study of parasite communities often allows to obtain information on potential preys and predator–prey interactions more efficiently (quickly and cheaper) than proper studies of food habits and trophic position of their hosts, especially in deep marine habitats2,3,4,5. Moreover, parasites may also provide information about health status and/or deterioration of ecosystems. Perturbations in ecosystem structure and function affect food web topology, and thus also impact upon parasite transmission, modifying communities in composition and abundances2,4,6,7,8,9,10,11. Finally, parasites are even able to impair behaviour, physiology, and morphology of their host individuals from species to population, community, and ecosystem level12,13. Therefore, the incorporation of parasite communities in the study of marine food webs is essential to understand predator–prey interactions3,12.
Sharks are apex predators and act as key elements with high structural importance within the ecosystem, serving as important links between diverse components of the marine trophic webs14. Studies showed that the loss of apex predators such as sharks caused inevitably changes in the intermediate levels of food webs15,16, with potential repercussion on trophic interactions and transmission dynamics of parasites with complex life cycles. As such, parasite communities of sharks could be used as useful indicators of biodiversity, food web structure, and environmental stress2,5,13,17. Among sharks living in the north-eastern Atlantic and Mediterranean Sea, Scyliorhinus canicula (Linnaeus, 1758), Galeus melastomus Rafinesque 1810, and Etmopterus spinax (Linnaeus, 1758) are carnivorous generalist occupying the mid-high marine trophic levels, thus playing not only an important role as predators, but also serving as prey for largest taxa18,19,20,21,22. Given the key position in trophic food webs, they also host a wide variety of heteroxenous parasites; despite of that, comparative analyses of their parasites communities only exist from the Balearic Sea (north-western Mediterranean Sea)18,19,20.
Using these studies as an excellent starting point, we performed a comparative work in the Gulf of Naples (central Mediterranean Sea), a geographical area where these shark species are commonly encountered and that is generally characterized by strong anthropic pressure23,24,25. In particular, this study aimed to: (i) investigate the structure of parasite communities in the three species when living in sympatry; (ii) explore the influence of host features (physiological and morphological) in shaping the composition and abundance of parasite communities; and (iii) discuss the use of parasite communities as biological indicators of environment stability.
Materials and methods
Collection and shark examination
Individuals of three sympatric benthic shark species were collected from the Gulf of Naples. Sampling included 102 S. canicula, 91 G. melastomus, and 39 E. spinax. All specimens were obtained between July and August 2020 in the trawling area between Ischia and Capri Islands (~ 40.575816 N, 13.966513 E), at ~ 400–600 m depth22. Specimens were frozen until dissections were performed. They constituted the bycatch of scientific and commercial trawling operations (red shrimps’ and pink shrimps’ fishery activities) held with a commercial fishing vessel equipped with bottom trawl nets (mouth of 3 × 4 m in height and width, respectively; 40 mm mesh size), towed at ~ 2–2.5 kn on muddy bottoms22,26. Samplings were approved by the ethics institutional review board of Italian Ministry of Agricultural, Food and Forestry Policies and performed in accordance with the permit n. 0008453 (issued May 15, 2020) by the Italian Ministry of Agricultural, Food and Forestry Policies, the guide for the care and use of animals by the Italian Ministry of Health, and the ARRIVE guidelines.
After thawing, the sharks were weighed to the nearest 0.1 g and measured (fork length-FL) to nearest 0.1 cm; sex was determined by gonadal examination. A macroscopic gonadal maturity score (GMS) was recorded to investigate the phase of the reproductive cycle (1 = immature, 2 = maturing, 3 = mature, 4 = resting/regressing) as described in Follesa and Carbonara27.
Body condition index (BCI, whole weight/fork length3) was calculated as described by Le Cren28. The gonadosomatic (GSI, gonad weight/host eviscerated weight × 100) and hepatosomatic indices (HSI, liver weight/host eviscerated weight × 100) were calculated as suggested by Mouine et al.29.
Parasitological study
The skin, gills, mouth cavity, digestive tract, liver, heart, gonads, visceral cavity, and mesenteries of each individual were examined under a dissecting microscope for parasites10,30. The musculature of each specimen was cut in thin slices (about 0.5 cm in thickness) and examined under a dissecting microscope for trypanorhynch larvae22. For each organ/tissue, ecto- and endo-parasites were collected, counted, washed in physiological saline solution, and preserved in 70% ethanol or frozen (− 20 °C) for morphological and genetic analyses, respectively.
For identification, crustaceans were clarified in 20% potassium hydroxide, and cestodes and trematodes were stained with Mayer’s acid carmine and mounted in Canada balsam or clarified in Amman’s lactophenol. Parasites were studied by light microscope and identified according to the available morphological identification keys31,32,33,34,35,36.
Larval forms of Anisakis Dujardin, 1845 spp. and trypanorhynchs were identified or confirmed at the species level using a molecular approach. Genomic DNA from ∼2 mg of middle portion of each larva was extracted using Quick-gDNA Miniprep Kit (ZYMO RESEARCH) following the standard manufacturer-recommended protocol.
For the identification of Anisakis larvae, the ITS (Internal Transcribed Spacer) region of rDNA including first internal transcribed spacer (ITS-1), the 5.8S gene, the second transcribed spacer (ITS-2), and ∼70 nucleotides of the 28S gene, was amplified using the primers NC5 (forward; 5′-GTAGGTGAACCTGCGGAAGGATCATT-3′) and NC2 (reverse; 5′-TTAGTTTCTTTTCCTCCGCT-3′)37. Polymerase chain reactions (PCRs) were carried out in a 15 µL volume containing 0.3 µL of each primer 10 mµ, 2.5 µL of MgCl2 25 mM (Promega), 15 µL of 5× buffer (Promega), 0.3 µL of DMSO 0.3 mM, 0.3 µL of dNTPs 10 mM (Promega), 0.3 µL (5 U/μL) of Go-Taq Polymerase (5 U/μl) (Promega), and 2 µL of total DNA. PCR temperature conditions were the following: 94 °C for 5 min (initial denaturation), followed by 30 cycles at 94 °C for 30 s (denaturation), 55 °C for 30 s (annealing), 72 °C for 30 s (extension), followed by post-amplification at 72 °C for 5 min.
For the molecular confirmation of trypanorhynch larvae, the partial large subunit (lsrDNA, 28S) was amplified using the primers ZX-1 (forward; 5′-ACCCGCTGAATTTAAGCATAT-3′) and 1500R (reverse; 5′-GCTATCCTGAGGGAAACTTCG-3′)38,39. PCRs were carried out in a 25 μl volume containing 0.6 μl of each primer 10 µM, 2 μl of MgCl2 25 mM (Promega), 5 μl of 5× buffer (Promega), 0.6 μl of dNTPs 10 mM (Promega), 0.2 μl of Go-Taq Polymerase (5 U/μl) (Promega), and 2 μl of total DNA. PCR temperature conditions were the following: 94 °C for 3 min (initial denaturation), followed by 35 cycles at 94 °C for 30 s (denaturation), 53 °C for 30 s (annealing), 72 °C for 2 min (extension), followed by post-amplification at 72 °C for 7 min.
The successful PCR products were purified using Agencourt AMPure XP (Beckman Coulter), following the standard manufacturer-recommended protocol. Clean PCR products were Sanger sequenced from both strands through an Automated Capillary Electrophoresis Sequencer 3730 DNA Analyzer (Applied Biosystems) using the BigDye® Terminator v3.1 Cycle Sequencing Kit (Life Technologies). The obtained contiguous sequences were assembled and edited using MEGAX v1140. Sequence identity was checked using BLASTn41.
Descriptors of parasite community
A component community comprises all parasite species recovered from a sample of a particular host species, while infracommunity refers to the parasites assemblage in one host individual. Prevalence was defined as the number of hosts infected with one or more individuals of a parasite species; parasite species with prevalence higher than 10% in any of the host species will be subsequently referred to as “common”. Abundance was measured as the number of individuals of a particular parasite species in/on a single host regardless of whether or not the host was infected. Mean abundance was measured as the number of individuals of a particular parasite in a sample of a particular host species divided by the total number of hosts of that species examined (including both infected and uninfected hosts)42.
The total mean abundance, species richness, Berger-Parker dominance index, and the Brillouin index of diversity were used as overall descriptors of infracommunities for each host species examined18,19,20,42. Total mean abundance was measured as the mean number of individuals of all parasite species, while species richness as the number of parasite species harboured by each shark specimen42. Descriptors of community were compared between hosts using the Mann–Whitney U- test.
Parasite species by host specificity were classified as ‘‘specialists’’, defined narrowly as having the bulk of reproducing adults found only in a single host species or having been reported from a single host species, and ‘‘generalists’’, when reported from a variety of related host species10,43.
Multivariate distance matrix regression
Multivariate distance matrix regression (MDMR44,45) was used to identify those predictors (X) able to explain the multivariate outcome (Y) given by the observed distribution of parasites within each shark species. MDMR is a two-step procedure that first computes the pairwise (dis)similarity between samples’ scores along all variables comprising Y. Then, pairwise distances are arranged into a symmetric distance matrix (D) and the association between X and D is measured by decomposing the sums of squares of D into a portion attributable to regression onto X and a portion due to residual. Interestingly, MDMR allows the use of any (dis)similarity metric to quantify the distance between samples (e.g., Euclidean, Bray–Curtis, Manhattan), making MDMR a flexible and robust alternative to methods such as multivariate multiple regression (MMR) and multivariate analysis of variance (MANOVA). Here, morphological (FL and BCI) and physiological (sex, GMS, GSI and HSI) variables10 were used as predictors for the parasite’s community. Ecological similarity between individuals of each shark species was measured with the Bray–Curtis index, by using the whole infracommunity data and by removing the contribution of extremely rare species (i.e., those parasites present with a single individual in a single specimen).
We used the square root transformed abundances to retain quantitative information while reducing the influence of dominant species (i.e., Grillotia)46. MDMR analyses were performed by using the package MDMR47 in R48, which provides analytical p-values for test statistic and uses a pseudo jack-knife procedure to quantify the conditional effects (δ) of each predictor in X (i.e., morphological and physiological traits of sharks) on each outcome variable in Y (i.e., the abundance of each parasite species). δ is a relative measure of effect size, with larger values indicating larger effects and vice versa (negative estimates are interpreted as having virtually no effect).
Results
Host and parasite data
Biological data (including sex, weight, BCI, GMS, GSI, and HIS) of the three shark species examined for parasites from the Gulf of Naples are reported in Table 1. Basic parameters of infection for each parasite taxon are presented in Table 2.
A total of 15 larvae of Anisakis spp. (Rhabditida: Anisakidae) were found and all molecularly identified at species level according to the obtained sequences (850 base pairs—bp) at the ITS region of the rDNA. One and 14 larvae were respectively assigned to the species Anisakis pegreffii Campana-Rouget & Biocca, 1955 and Anisakis physeteris (Baylis, 1923), showing 100% identity with the sequences of A. pegreffii and A. physeteris previously deposited in GenBank (accession numbers: MF422221–MF422222 and MF668924–MF668926).
The morphological identification of trypanorhynchs was confirmed on a subsample of 70 larvae, according to the obtained sequences (~ 1600 bp) at the 28S gene of the rDNA. A total of 60, 5, and 5 larvae were respectively assigned to Grillotia Guiart, 1927 sp., Heterosphyriocephalus tergestinus (Pintner, 1913) Dallarés, Carrassón & Schaeffner, 2016 and Sphyriocephalus viridis (Wagener, 1854) Pintner, 1913 showing ~ 100% identity with the sequences of Grillotia sp., H. tergestinus and S. viridis previously deposited in GenBank (accession numbers: MW838236, KX570647, and FJ572940, respectively).
Representative sequences here obtained were deposited in GenBank with the accession numbers OM279537 (A. pegreffii), OM279534–36 (A. physeteris), ON427560–61 (Grillotia sp.), ON427564-65 (H. tergestinus), and ON427562–63 (S. viridis).
Overall prevalence of infection for parasites was 82%, 95.1%, and 100% respectively in E. spinax, S. canicula, and G. melastomus. A total of 20,186 individual parasites belonging to nine taxa (four in E. spinax and five each in G. melastomus and in S. canicula) were identified in the three host species. Only larval forms of a cestode of the genus Grillotia (Trypanorhyncha: Lacistorhynchidae) and of the nematode A. physeteris were found in all host species; larval forms of the cestode S. viridis (Trypanorhyncha: Sphyriocephalidae) were found in two hosts (namely E. spinax and G. melastomus). In all sharks, the most prevalent and abundant taxon was a Grillotia species. An ectoparasite copepod, Lernaeopoda galei Krøyer, 1837 (Siphonostomatoida: Lernaeopodidae) was only found from the peri-genital skin of S. canicula. All endo-parasites were obtained from the lumen of the gastrointestinal tract, except the larvae of A. physeteris, encysted into the wall of stomach and intestine.
The predominant group of parasites with respect to species diversity was the Cestoda (four species), followed by Trematoda and Nematoda (two species each), and Copepoda (one species). Regarding the parasite taxa, the local parasite fauna showed almost similar representation of adult parasite stages [four species: one copepod L. galei, two tremadodes Diphterostomum betencourti (Monticelli, 1893) Odhner, 1911 (Plagiorchiidae: Zoogonidae) and Otodistomum veliporum (Creplin, 1837) Stafford, 1904 (Plagiorchiidae: Azygiidae), and one cestode Ditrachybothridium macrocephalum Rees, 1959 (Diphyllidea: Echinobothriidae)] and larval parasite stages [five species: two nematodes A. pegreffii and A. physeteris and three cestodes H. tergestinus (Trypanorhyncha: Sphyriocephalidae), Grillotia sp., and S. viridis]; however, larval forms dominated numerically the parasite communities of all shark species. In particular, all 216 parasite specimens found in E. spinax were larval forms of endo-parasites. Out of the 16,644 parasite specimens found in G. melastomus, 76 (0.5%) were adult forms and 16,568 (99.5%) were larval forms. Out of the 3326 parasite specimens found in S. canicula, 106 (3.2%) were adult forms and 3220 (96.8%) were larval forms. Out of nine parasite taxa identified, only two (L. galei and D. betencourti) were considered to be specialist species.
Descriptors of parasite community
In all shark species, the species richness ranged from one to three, with the maximum number of parasite species observed in a single individual of E. spinax, in three individuals of G. melastomus, and in four individuals of S. canicula. Most individuals of all shark species were infected with a single species of parasite: 66.6% of E. spinax, 75.5% of S. canicula, and 77% of G. melastomus, respectively.
Descriptors of parasite infracommunities for all shark species are listed in Table 3. Only five parasites were considered common, with larval stages of Grillotia found in all hosts, S. viridis found only in G. melastomus, A. physeteris found only in E. spinax, and L. galei and D. betencourti found only in S. canicula. Scyliorhinus canicula and E. spinax showed the highest average level of infracommunity diversity indices (Table 3), although their differences were not significant (Mann–Whitney U-test, U < 1858.3 and p > 0.06 in all cases). Conversely, G. melastomus showed lower diversity values on average, being significantly different from S. canicula (Mann–Whitney U-test, U > 2,837.5 and p < 0.001 in all cases) but not when compared with E. spinax (Mann–Whitney U-test, U < 1418.5 and p > 0.7 in all cases).
Predictors of infracommunity similarity
Morphological and physiological traits were able to significantly predict the compositional similarity of parasite infracommunity in all three shark species, with the only exception of E. spinax (Table 4), also showing a negligible contribution of extremely rare species in MDMR model outcomes (Table 4). When considering each predictor, our results showed that both morphological traits (FL and BCI) were significantly (albeit low) correlated with the infracommunity structure of G. melastomus, while BCI did not correlate in S. canicula (Table 4). Overall, we did not find a significant effect of sex in G. melastomus and E. spinax, while males in S. canicula showed a similar pattern of parasitic community structure (Table 4). When considering the remaining physiological traits, the MDMR model showed a significant effect of GSI and HSI only for S. canicula, while GMS did not show any significant effect (Table 4).
Relationship between predictors and parasite abundance
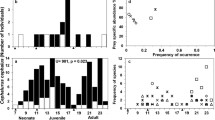
Overall, a large portion of the MDMR model outcome in each shark species was due mainly to the predictors’ effects on the abundance of Grillotia sp., which was much more pronounced on G. melastomus and S. canicula (Fig. 1). In this latter species, neither morphological nor physiological predictors had effects on the abundances of L. galei and A. physeteris, while they had a very limited effect on D. betencourti (Fig. 1). Morphological predictors had a very low association with Grillotia sp. in S. canicula, whose abundances seemed to be associated more with physiological traits such as HSI and sex (Fig. 1). Interestingly, in G. melastomus predictors had a significant influence on the abundances of all parasites, although such effect was much more pronounced for Grillotia sp. with FL being the most correlated predictors (Fig. 1). In E. spinax, the physiological predictor GMS mostly contributed to the abundances of all parasites (Fig. 1).
Heatmap of the effect size showing the pairwise effect of morphological (FL and BCI) and physiological (age, sex, GMS, GSI, and HSI) predictors (conditional on the rest of the predictors) on each individual parasites considering the whole infracommunity (a) and by removing extremely rare parasite species (b, i.e., those parasites present with a single individual in a single specimen). Omnibus (blue cells) refers to the effect size of the entire design matrix on each outcome, while numbers in green cells the pairwise effect size (i.e., the effect of each predictor on each outcome variable, conditional on the rest of the predictors). Sharks in the figure have been redrawn from Compagno80,81 and downloaded from https://fishbase.org under a Creative Commons License—CC BY-NC 3.
Discussion
With the sole exception of L. galei, an ecto-parasite copepod found at least in 14 shark and two skate taxa (see https://shark-references.com/species/parasite-hosts-list/L), all the species found were trophic transmitted helminths with a complex life cycle. The present results point out on the evident trophic links between the diet of the host and the parasite occurrence and infection trends5,49. In agreement with that, studies on the feeding ecology of E. spinax, G. melastomus, and S. canicula showed that they are carnivorous generalist preying on a great variety of deep-sea invertebrates and fishes, with differences mainly related to host biological features (i.e., ontogenetic changes and gender) and abiotic factors (i.e., sampling season and geographical area). However, while the diet of E. spinax and G. melastomus mainly consists of decapod crustaceans, cephalopods, and fishes50,51,52, the diet of S. canicula consists of more diverse benthic preys, with polychaetes being prevalent in some geographic areas53,54,55.
Among the common helminth species found, a three and four or more host life-cycle has been respectively proposed for trypanorhynch cestodes of the genera Grillotia and Sphyriocephalus Pintner, 1913, with copepods and molluscs acting as first and second intermediate hosts, predatory fishes as third intermediate or paratenic hosts and elasmobranch species as definitive hosts35. The finding of larval stages only of both species suggests that the investigated sharks could acquire the infection by ingestion of both cephalopods and fishes, serving as intermediate/paratenic hosts for these parasite species. Moreover, the different values of abundance of Grillotia specimens among the three host species could suggest different amount of intermediate hosts in their diet. For instance, among the shark species studied from the north-western Mediterranean, a Grillotia infection was linked to the consumption of mycthophid fishes only in G. melastomus, as they were the dominant prey in this host species18; however, at the present no evidence exists that mycthophid fishes may play a role in the transmission of the infection.
Regarding the anisakid nematodes here detected, sharks are considered accidental or dead-end hosts of Anisakis species. Cetacean species of the families Physeteridae Gray, 1821 and Kogiidae Miller, 1923 represent the main definitive hosts for A. physeteris (see56,57). Larval stages of A. physeteris have been found sporadically in fish species, with low infection rates56,58,59, while a recent study suggests the importance of the deep-sea cephalopod Histioteuthis bonnellii (Férussac, 1834) as intermediate host in the life cycle of this parasite60. The finding of all nematode larvae of the species A. physeteris, except a single specimen molecularly identified as A. pegreffii (a common parasite of Delphinidae Gray, 1821), strongly supports that the benthonic deep habitat may represent an important food webs system for larvae of A. physeteris as previously suggested56,60.
Diphterostomum betencourti was the only common parasite found as adult in this study. According to Bray and Moore32, this trematode is known in S. canicula and S. stellaris from the Atlantic coast of Belgium, England, France, and Spain, and thus it represents a new geographical record for the Mediterranean Sea. Life cycle for species of the genus Diphterostomum Stossich, 1904 has been elucidated only for D. brusinae (Stossich, 1888) Stossich, 1904 and D. flavum Gilardoni, Etchegoin, Cribb, Pina, Rodrigues, Diez & Cremonte, 2020, two species infecting the teleost fishes. Gastropods serve as first intermediate hosts, while gastropods, bivalves, and other sedentary invertebrates—the crinoid Antedon mediterranea (Lamarck, 1816) or the polychaete Kinbergonuphis dorsalis (Ehlers, 1897)—as second intermediate hosts61,62. Saldanha et al.53 reported that polychaetes were the main prey found in the gut of S. canicula in the deep sea off southern Portugal, although polychaetes were uncommon preys in similar studies from other geographical areas51,55. It has been suggested that the variation in the prey importance could be related to the availability of different benthic assemblages among geographical areas or depths50,51,55. Unfortunately, no studies on the feeding ecology of S. canicula are available from the Gulf of Naples. The occurrence of D. betencourti as common species only in S. canicula suggests that polychaetes and/or sedentary invertebrates could represent a discrete part of the diet of this shark species in the study area, supporting the view that the availability of different benthic assemblages might influence the structure of trophic helminth community of the host63,64.
The present parasite communities (at both component and infracommunity level) of the three sympatric shark species were all similarly impoverished with 4–5 component species, low species richness ranging from one to three, and low diversity values. Higher total mean abundance was found in G. melastomus, but this was due to a higher number of Grillotia larvae, which in turn also dominated the communities of other shark species, suggesting that they have a similar role in the trophic food web of the Gulf of Naples. The high abundance of infection with larvae of Grillotia in the present three sharks’ species could also suggest high abundance of largest top predators in the area22. The finding of two additional trypanorhynch larval stages—H. tergestinus (Pintner, 1913) Dallarés, Carrassón & Schaeffner, 2016 and S. viridis—in E. spinax also underlines the role of this small-sized shark species as intermediate/paratenic host for members of trypanorhynch cestodes.
Previous studies have already suggested the general idea that Mediterranean small-medium sized sharks have impoverished parasite communities with respect to conspecifics from European Atlantic waters, and that the low values of species richness and diversity coupled with high values of dominance indices are common to small sized sharks from different geographical areas65. For instance, seven and 11 taxa of parasites were found in E. spinax from Norwegian66 and north-east Atlantic waters of Spain65, as well as 10 parasite taxa were found in S. canicula from off the coasts of Great Britain67. However, qualitative and quantitative differences were also evident when we compared the present results to those of the same hosts from the Balearic Sea19,20. In particular, five taxa of parasites were found in 41 individuals of S. canicula, with no taxon matching the present taxa19; a total of 13 parasite taxa were found in 120 individuals of G. melastomus, with only two—D. macrocephalum and S. viridis—of those found in the present study19; and only two taxa of cestodes—Aporhynchus norvegicus (Olsson, 1868) Nybelin, 1918 and an unidentified tetraphyllidean—were found in 11 individuals of E. spinax20. Differences were also found among values of infracommunities19,20. In particular, total mean abundance in S. canicula from Balearic Sea was higher (ranging from 29.14 to 80.56), while Brillouin’s (ranging from 0.03 to 0.09) and Berger-Parker’s (ranging from 0.96 to 0.99) indices were lower than those found in the present study; in contrast, total mean abundance in G. melastomus was lower (ranging from 0.08 to 3) and Brillouin’s index higher (ranging from 0 to 0.11) than our results, with similar value of Berger-Parker’s index19. Finally, very similar results were found between the present individuals of E. spinax and those from the Balearic Sea except for the total mean abundance that in the present study was lower20.
Qualitative and quantitative differences in parasite community of the same fish species coming from different geographical areas and depths are expected because specific abiotic factors may be important drivers influencing the presence/absence and abundance of a parasite species in a specific habitat. For example, the nematode Proleptus obtusus Dujardin, 1845 was the most prevalent species (until 100%) in individuals of S. canicula from Balearic Sea (sampling from 53 to 68 m depth19,20), coast of England (sampling depth not stated67), and Portugal (sampling depth not stated68). However, it was found only in 19.2% of S. canicula studied from the north-eastern Aegean Sea (sampling from 40 to100 m depth69), and no individuals of this species were found in the present study. Proleptus obtusus Dujardin, 1845 is a parasite involved in the shallow marine food web, and the deep environment (400–600 m depth) from which the present shark species were collected could explain the absence of this nematode19,70,71.
Regarding the present impoverished species richness of the parasite communities, a recent study, based on a comprehensive analysis of literature records and including several data from 91 different shark species, suggested that the diet breadth of a shark species measured by the diversity of prey families ingested can be the better predictor of cestode species richness5. Since the cestodes represent the most important group of parasites in sharks, this could suggest that the populations of the three shark species here studied may have a restricted diet than the same species from other geographical areas, showing in turn poorer parasite communities. Indeed, it is generally agreed that a host with broader diet is exposed to a greater number of potential intermediate host species, resulting in a greater richness of trophically transmitted parasites when compared to a consumer with narrower diet72,73,74.
Moreover, the parasite communities may be good indicators of environmental disturbance because they reflect the interactions between a possible stressor and either free-living larval stages or populations of their intermediate and final hosts2,6,8,9,63,64. In the present case, the Gulf of Naples is strongly affected by overfishing and different kinds of anthropic stressors23,24,25, and thus the poor values of parasite communities here found could suggest an unstable ecosystem with a decrease in biomass and richness of intermediate hosts with subsequently restricted diet in the three present sharks2,6,7,8,9. Indeed, a recent study from two Mediterranean coastal areas, contiguous between them but with different degree of anthropic pressure and deterioration (i.e., Gulf of Naples versus Gulf of Salerno), reported that the parasite communities of salema Sarpa salpa (Linnaeus, 1758) and white seabream Diplodus sargus (Linnaeus, 1758) responded differently to specific biological factors depending by locality, thus highlighting how environmental conditions may exert a strong influence on parasite infracommunities stressing different degrees of deterioration between sampling areas, with lower values of parasite communities found in the Gulf of Naples10. Similar differences were described by Derbel et al.7 comparing parasite communities of teleost fish from unprotected and protected areas of south-eastern coast of Tunisia.
Morphological and physiological variables were differently correlated to parasite community structure depending on shark species. Some morphological traits (i.e., FL in S. canicula and G. melastomus, and BCI in G. melastomus) were significantly correlated (albeit to a different extent) with parasite abundance. This could be explained by the ontogenetic changes in fish diets, suggesting cumulative infection with age, in particular for larval forms which accumulate in muscles, i.e., Grillotia sp.22,71. Indeed, it has been documented how individuals of these shark species, increasing in body length, also increase their consumption of cephalopods and mesopelagic fishes50, which could represent the main intermediate hosts for trypanorhynch cestodes that dominated numerically the parasite communities in all shark species (i.e., Grillotia sp.). Present results were congruent with those of Dallarés et al.20 from the north-western Mediterranean, where significant positive relationships with host size were detected at least for the total parasite abundance and the abundance of larvae of Grillotia adenoplusia (Pintner, 1903) Palm, 2004 in G. melastomus. The lack of correlation between morphological traits and parasite abundance in E. spinax could be explained by the fact that most (77%) of individuals of this shark species were juveniles, in contrast to the other two species which were mostly represented by adult individuals.
Regarding the physiological traits, a significant correlation with the similarity of parasite infracommunities was found for GSI and HSI in S. canicula. This significant correlation could be explained by the fact that most (73.5%) of individuals of this species were in reproductive stage, as the GSI is a good indicator of reproductive activity of fish while the HSI is strictly related to the gonadal maturation75. The present results suggest that large, sexually mature, individuals increasing their food intake in quality and amount could harbour a high number of trophically transmitted parasite species and individuals because they may also ingest more potential infected intermediate hosts76,77. The present hypothesis is supported by Capapé et al.78, who reported that both male and female GSI values increased with the total length of specimens in S. canicula, and HSI reached significantly high values in adults of both sex. A significant effect of sex on compositional similarity of parasite infracommunity was observed only in males of S. canicula, suggesting differences in composition of the diet and abundance of infected prey items among genders. The present result was congruent with Mnasri et al.54, who showed that preferred preys of S. canicula males from the coast of Tunisia were teleost fishes consumed in higher abundance and occurrence, while females preferred crustaceans. The analysis of the relationship between predictors and parasite abundance confirmed the hypothesis regarding the importance of some specific traits, namely HIS and sex in S. canicula, FL in G. melastomus, and GMS in E. spinax.
In conclusion, this represents the second comparative study of parasite community of Mediterranean shark species, being the only data for this basin known from the north-western Mediterranean18,19,20. Depending by host species, both morphological and physiological traits were able to predict the features of parasite communities at component and infracommunity levels. Impoverished parasite communities were found in the three shark species investigated (i.e., E. spinax, G. melastomus and S. canicula). This latter finding could be related to the restricted host diet and/or abiotic factors and decrease in prey biomass and richness resulting in lower probabilities of parasite transmission2,5,6,7,8,9,63,64,79 in the Gulf of Naples, an area characterized by overfishing and anthropic pressure23,24,25. Indeed, in agreement to that demonstrated previously5,63,64 the parasite fauna of deep-sea fish reflect their diet and the probability of infection and their rates are directly linked to the abundance and richness of free-living prey (i.e., intermediate/paratenic hosts).
References
Marcogliese, D. J. & Cone, D. K. Food webs: A plea for parasites. Trends Ecol. Evol. 12, 320–325. https://doi.org/10.1016/S0169-5347(97)01080-X (1997).
Marcogliese, D. J. Food webs and the transmission of parasites to marine fish. Parasitology 124(7), 83–99. https://doi.org/10.1017/s003118200200149x (2002).
Marcogliese, D. J. Parasites: Small players with crucial roles in the ecological theater. EcoHealth 1, 151–164. https://doi.org/10.1007/s10393-004-0028-3 (2004).
Marcogliese, D. J. Parasites of the superorganism: Are they indicators of ecosystem health?. Int. J. Parasitol. 35(7), 705–716. https://doi.org/10.1016/j.ijpara.2005.01.015 (2005).
Rasmussen, T. K. & Randhawa, H. S. Host diet influences parasite diversity: A case study looking at tapeworm diversity among sharks. Mar. Ecol. Prog. Ser. 605, 1–16. https://doi.org/10.3354/meps12751 (2018).
Vidal-Martínez, V. M., Pech, D., Sures, B., Purucker, S. T. & Poulin, R. Can parasites really reveal environmental impact?. Trends Parasitol. 26(1), 44–51. https://doi.org/10.1016/j.pt.2009.11.001 (2010).
Derbel, H., Châari, M. & Neifar, L. Digenean species diversity in teleost fishes from the Gulf of Gabes, Tunisia (Western Mediterranean). Parasite 19(2), 129–135. https://doi.org/10.1051/parasite/2012192129 (2012).
Mattiucci, S. et al. Temporal stability of parasite distribution and genetic variability values of Contracaecum osculatum sp. D and C. osculatum sp. E (Nematoda: Anisakidae) from fish of the Ross Sea (Antarctica). Int. J. Parasitol. Parasites Wildl. 4(3), 356–367. https://doi.org/10.1016/j.ijppaw.2015.10.004 (2015).
Sures, B., Nachev, M., Selbach, C. & Marcogliese, D. J. Parasite responses to pollution: what we know and where we go in ‘Environmental Parasitology’. Parasit. Vectors 10, 65. https://doi.org/10.1186/s13071-017-2001-3 (2017).
Santoro, M., Iaccarino, D. & Bellisario, B. Host biological factors and geographic locality influence predictors of parasite communities in sympatric sparid fishes off the southern Italian coast. Sci. Rep. 10(1), 13283. https://doi.org/10.1038/s41598-020-69628-1 (2020).
Thomas, F., Poulin, R., de Meeüs, T., Guégan, J. F. & Renaud, F. Parasites and ecosystem engineering: What roles could they play?. Oikos 84, 167–171. https://doi.org/10.2307/3546879 (1999).
Timi, J. T. & Poulin, R. Why ignoring parasites in fish ecology is a mistake. Int. J. Parasitol. 50(10–11), 755–761. https://doi.org/10.1016/j.ijpara.2020.04.007 (2020).
Lafferty, et al. Parasites in food webs: The ultimate missing links. Ecol. Lett. 11, 533–546. https://doi.org/10.1111/j.1461-0248.2008.01174.x (2008).
Heithaus, M. R., Frid, A., Wirsing, A. J. & Worm, B. Predicting ecological consequences of marine top predator declines. Trends Ecol. Evol. 23(4), 202–210. https://doi.org/10.1016/j.tree.2008.01.003 (2008).
Stevens, J. D., Bonfil, R., Dulvy, N. K. & Walker, P. A. The effects of fishing on sharks, rays, and chimaeras (chondrichthyans), and the implications for marine ecosystems. ICES J. Mar. Sci. 57(3), 476–494. https://doi.org/10.1006/jmsc.2000.0724 (2000).
Myers, R. A., Baum, J. K., Shepherd, T. D., Powers, S. P. & Peterson, C. H. Cascading effects of the loss of apex predatory sharks from a coastal ocean. Science 315(5820), 1846–1850. https://doi.org/10.1126/science.1138657 (2007).
Palm, H. W. Fish parasites as biological indicators in a changing world: can we monitor environmental impact and climate change? In: Progress in Parasitology (ed. Melhorn, H.) 223–250 (Berlin, 2011).
Dallarés, S. Twenty thousand parasites under the sea: A multidisciplinary approach to parasite communities of deep-dwelling fishes from the slopes of the Balearic Sea (NW Mediterranean). PhD Thesis. Universitat Autònoma de Barcelona. (2016).
Dallarés, S., Pérez-del-Olmo, A., Montero, F. E. & Carrassón, M. Composition and seasonal dynamics of the parasite communities of Scyliorhinus canicula (L., 1758) and Galeus melastomus Rafinesque. (Elasmobranchii) from the NW Mediterranean Sea in relation to host biology and ecological features. Hydrobiologia 799(275–291). https://doi.org/10.1007/s10750-017-3226-z (2017).
Dallarés, S., Padrós, F., Cartes, J. E., Solé, M. & Carrassón, M. The parasite community of the sharks Galeus melastomus, Etmopterus spinax and Centroscymnus coelolepis from the NW Mediterranean deep-sea in relation to feeding ecology and health condition of the host and environmental gradients and variables. Deep Sea Res. Part I Oceanogr. Res. Pap. 129, 41–58. https://doi.org/10.1016/j.dsr.2017.09.007 (2017).
Ebert, D. A. & Dando, M. Field Guide to Sharks, Rays & Chimaeras of Europe and the Mediterranean. 385 (Princeton University Press, 2021).
Santoro, M., Bellisario, B., Crocetta, F., Degli Uberti, B. & Palomba, M. A molecular and ecological study of Grillotia (Cestoda: Trypanorhyncha) larval infection in small to mid-sized benthonic sharks in the Gulf of Naples, Mediterranean Sea. Ecol. Evol. 11(20), 13744–13755. https://doi.org/10.1002/ece3.7933 (2021).
Crocetta, F. et al. Bottom-trawl catch composition in a highly polluted coastal area reveals multifaceted native biodiversity and complex communities of fouling organisms on litter discharge. Mar. Environ. Res. 155, 104875. https://doi.org/10.1016/j.marenvres.2020.104875 (2020).
Hay Mele, B. et al. Ecological assessment of anthropogenic impact in marine ecosystems: the case of Bagnoli Bay. Mar. Environ. Res. 158, 104953. https://doi.org/10.1016/j.marenvres.2020.104953 (2020).
Rizzo, L., Musco, L. & Crocetta, F. Cohabiting with litter: Fish and benthic assemblages in coastal habitats of a heavily urbanized area. Mar. Poll. Bull. 164, 112077. https://doi.org/10.1016/j.marpolbul.2021.112077 (2021).
Tanduo, V., Osca, D. & Crocetta, F. A bycatch surprise: Scyllarus subarctus Crosnier, 1970 (Decapoda: Achelata: Scyllaridae) in the Mediterranean Sea. J. Crust. Biol. 41(2), ruab010. https://doi.org/10.1093/jcbiol/ruab010 (2021).
Follesa, M. C. & Carbonara, P. Atlas of the maturity stages of Mediterranean fishery resources in General Fisheries Commission for the Mediterranean.Studies and Reviews (ed. FAO) 259 (FAO, 2019).
Le Cren, E. D. The length-weight relationship and seasonal cycle in gonad weight and condition in the perch (Perca fuviatilis). J. Anim. Ecol. 20, 201–219 (1951).
Mouine, N., Francour, P., Ktari, M. H. & Chakroun-Marzouk, N. The reproductive biology of Diplodus sargus sargus in the Gulf of Tunis (Central Mediterranean). Sci. Mar. 71(3), 461–469. https://doi.org/10.3989/scimar.2007.71n3461 (2007).
Santoro, M., Palomba, M., Mattiucci, S., Osca, D. & Crocetta, F. New parasite records for the sunfish Mola mola in the Mediterranean Sea and their potential use as biological tags for long-distance host migration. Front. Vet. Sci. 7, 579728. https://doi.org/10.3389/fvets.2020.579728 (2020).
Kabata, Z. Parasitic Copepoda of British Fishes. 468 (The Ray Society, British Museum, 1979).
Bray, R. A. & Moore, A. B. M. The first record of the elasmobranch parasite Diphterostomum betencourti (Monticelli, 1893) (Digenea, Zoogonidae) in the coastal waters of southern England. Acta Parasitol. 45(4), 299–302 (2000).
Gibson, D. I. Superfamily Azygioidea Lühe, 1909 in Keys to the Trematoda. Vol. 1(eds. Gibson, D. I., Jones, A. & Bray, R. A.) 19–24 (CAB International, 2002).
Anderson, R. C., Chabaud, A. G. & Willmott, S. CIH keys to the nematode parasites of vertebrates: Archival volume (eds. Anderson, R. C., Chabaud, A. G. & Willmott, S.) 463 (CAB International, 2009).
Palm, H. W. The Trypanorhyncha Diesing, 1863 (IPB-PKSPL Press, 2004).
Dallarés, S., Pérez-del-Olmo, A., Carrassón, M. & Kuchta, R. Morphological and molecular characterisation of Ditrachybothridium macrocephalum Rees, 1959 (Cestoda: Diphyllidea) from Galeus melastomus Rafinesque in the Western Mediterranean. Syst. Parasitol. 92, 45–55. https://doi.org/10.1007/s11230-015-9586-8 (2015).
Zhu, X., Gasser, R. B., Podolska, M. & Chilton, N. B. Characterisation of anisakid nematodes with zoonotic potential by nuclear ribosomal DNA sequences. Int. J. Parasitol. 28(12), 1911–1921. https://doi.org/10.1016/s0020-7519(98)00150-7 (1998).
Van der Auwera, G., Chapelle, S. & De Wachter, R. Structure of the large ribosomal subunit RNA of Phytophthora megasperma, and phylogeny of the oomycetes. FEBS Lett. 338(2), 133–136. https://doi.org/10.1016/0014-5793(94)80350-1 (1994).
Palm, H. W., Waeschenbach, A., Olson, P. D. & Littlewood, D. T. Molecular phylogeny and evolution of the Trypanorhyncha Diesing, 1863 (Platyhelminthes: Cestoda). Mol. Phyl. Evol. 52(2), 351–367. https://doi.org/10.1016/j.ympev.2009.01.019 (2009).
Kumar, S., Stecher, G., Li, M., Knyaz, C. & Tamura, K. MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 35, 1547–1549. https://doi.org/10.1093/molbev/msy096 (2018).
Morgulis, A. et al. Database indexing for production MegaBLAST searches. Bioinformatics 24(16), 1757–1764. https://doi.org/10.1093/bioinformatics/btn322 (2008).
Bush, A. O., Lafferty, K. D., Lotz, J. M. & Shostak, A. W. Parasitology meets ecology on its own terms: Margolis et al. revisited. J. Parasitol. 83, 575–583. https://doi.org/10.2307/3284227 (1997).
Euzet, L. & Combes, C. Les problèmes de l’espèces chez les animaux parasites. Bull. Soc. Zool. Fr. 40, 239–285 (1980).
Anderson, M. J. A new method for non-parametric multivariate analysis of variance. Austral. Ecol. 26(1), 32–46. https://doi.org/10.1111/j.1442-9993.2001.01070.pp.x (2001).
McArdle, B. H. & Anderson, M. J. Fitting multivariate models to community data: .a comment on distance-based redundancy analysis. Ecology 82(1), 290–297. https://doi.org/10.1890/0012-9658(2001)082[0290:FMMTCD]2.0.CO;2 (2001).
Locke, S. A., McLaughlin, J. D. & Marcogliese, D. J. Predicting the similarity of parasite communities in freshwater fishes using the phylogeny, ecology and proximity of hosts. Oikos 122(1), 73–83. https://doi.org/10.1111/j.1600-0706.2012.20211.x (2013).
McArtor, D. B., Lubke, G. H. & Bergeman, C. S. Extending multivariate distance matrix regression with an effect size measure and the asymptotic null distribution of the test statistic. Psychometrika 82, 1052–1077. https://doi.org/10.1007/s11336-016-9527-8 (2018).
Core Team. R: a language and environment for statistical computing. https://www.R-project.org/ (2017).
Caira, J. N., Bueno, V. & Jensen, K. Emerging global novelty in phyllobothriidean tapeworms (Cestoda: Phyllobothriidea) from sharks and skates (Elasmobranchii). Zool. J. Linnean Soc. 193, 1336–1363. https://doi.org/10.1093/zoolinnean/zlaa185 (2021).
Fanelli, E., Rey, J., Torres, P. & Gil de Sola, L. Feeding habits of blackmouth catshark Galeus melastomus Rafinesque, 1810 and velvet belly lantern shark Etmopterus spinax (Linnaeus, 1758) in the western Mediterranean. J. Appl. Ichthyol. 25(1), 83–93. https://doi.org/10.1111/j.1439-0426.2008.01112.x (2009).
Anastasopoulou, A. et al. Diet and feeding strategy of blackmouth catshark Galeus melastomus. J. Fish Biol. 83(6), 1637–1655. https://doi.org/10.1111/jfb.12269 (2013).
D’Iglio, C. et al. Biological and ecological aspects of the blackmouth catshark (Galeus melastomus Rafinesque, 1810) in the Southern Tyrrhenian Sea. J. Mar. Sci. Eng. 9(9), 967. https://doi.org/10.3390/jmse9090967 (2021).
Saldanha, L., Almeida, A. J., Andrade, F. & Guerreiro, J. Observations on the diet of some slope dwelling fishes of Southern Portugal. Int. Rev. der Gesamten Hydrobiol. 80(2), 217–234. https://doi.org/10.1002/iroh.19950800210 (1995).
Mnasri, N., El Kamel, O., Boumaïza, M., Reynaud, C. & Capapé, C. Food and feeding habits of the small-spotted catshark, Scyliorhinus canicula (Chondrichthyes: Scyliorhinidae) from the northern coast of Tunisia (central Mediterranean). Cah. Biol. Mar. 53(1), 139–150 (2012).
Šantić, M., Rađa, B. & Pallaoro, A. Feeding habits of small-spotted catshark (Scyliorhinus canicula Linnaeus, 1758) from the eastern central Adriatic Sea. Mar. Biol. Res. 8(10), 1003–1011. https://doi.org/10.1080/17451000.2012.702912 (2012).
Mattiucci, S., Cipriani, P., Levsen, A., Paoletti, M. & Nascetti, G. Molecular epidemiology of Anisakis and Anisakiasis: an ecological and evolutionary road map. Adv. Parasitol. 99, 93–263. https://doi.org/10.1016/bs.apar.2017.12.001 (2018).
Santoro, M. et al. Helminth parasites of the dwarf sperm whale Kogia sima (Cetacea: Kogiidae) from the Mediterranean Sea, with implications on host ecology. Dis. Aquat. Org. 129(3), 175–182. https://doi.org/10.3354/dao03251 (2018).
Cipriani, P. et al. The Mediterranean European hake, Merluccius merluccius: detecting drivers influencing the Anisakis spp. larvae distribution. Fish. Res. 202, 79–89. https://doi.org/10.1016/j.fishres.2017.07.010 (2018).
Levsen, A. et al. Anisakis species composition and infection characteristics in Atlantic mackerel, Scomber scombrus, from major European fishing grounds—Reflecting changing fish host distribution and migration pattern. Fish. Res. 202, 112–121. https://doi.org/10.1016/j.fishres.2017.07.030 (2018).
Palomba, M., Mattiucci, S., Crocetta, F., Osca, D. & Santoro, M. Insights into the role of deep-sea squids of the genus Histioteuthis (Histioteuthidae) in the life cycle of ascaridoid parasites in the Central Mediterranean Sea waters. Sci. Rep. 11, 7135. https://doi.org/10.1038/s41598-021-86248-5 (2021).
Palombi, A. Il ciclo biologico di Diphterostomum brusinae Stossich (Trematode digenetico: fam. Zoogonidae Odhner). Considerazioni sui cicli evolutivi delle specie affini e dei trematodi in generale. Pubbl. Stn. Zool. 10, 111–149 (1930).
Gilardoni, C. et al. Cryptic speciation of the zoogonid digenean Diphterostomum flavum n. sp. demonstrated by morphological and molecular data. Parasite 27(44). https://doi.org/10.1051/parasite/2020040 (2020).
Campbell, R. A., Haedrich, R. L. & Munroe, T. A. Parasitism and ecological relationships among deep-sea benthic fishes. Mar. Biol. 57, 301–313. https://doi.org/10.1007/BF00387573 (1980).
Campbell R. A. Parasitism in the deep-sea in Deep-sea Biology, The Sea (ed. Rowe, G. T.) 473–552 (Wiley, 1983).
Isbert, W. et al. Metazoan parasite communities and diet of the velvet belly lantern shark Etmopterus spinax (Squaliformes: Etmopteridae): a comparison of two deep-sea ecosystems. J. Fish Biol. 86(2), 687–706. https://doi.org/10.1111/jfb.12591 (2015).
Klimpel, S., Palm, H. W. & Seehagen, A. Metazoan parasites and food composition of juvenile Etmopterus spinax (L., 1758) (Dalatiidae, Squaliformes) from the Norwegian Deep. Parasitol. Res. 89, 245–251. https://doi.org/10.1007/s00436-002-0741-1 (2003).
Moore, A. B. M. Metazoan parasites of the lesser-spotted dogfish Scyliorhinus canicula and their potential as stock discrimination tools. J. Mar. Biol. Assoc. UK 81(6), 1009–1013. https://doi.org/10.1017/S0025315401004982 (2001).
Silva, C., Veríssimo, A., Cardoso, P., Cable, J. & Xavier, R. Infection of the lesser spotted dogfish with Proleptus obtusus Dujardin, 1845 (Nematoda: Spirurida) reflects ontogenetic feeding behaviour and seasonal differences in prey availability. Acta Parasit. 62(2), 471–476. https://doi.org/10.1515/ap-2017-0055 (2017).
Bakopoulos, V. et al. Parasites of Scyliorhinus canicula (Linnaeus, 1758) in the north-eastern Aegean Sea. J. Mar. Biol. Assoc. UK 98(8), 2133–2143. https://doi.org/10.1017/S0025315417001552 (2018).
Moravec, F., Van As, J. G. & Dyková, I. Proleptus obtusus Dujardin, 1845 (Nematoda: Physalopteridae) from the puffadder shyshark Haploblepharus edwardsii (Scyliorhinidae) from off South Africa. Syst. Parasitol. 53, 169–173. https://doi.org/10.1023/A:1021130825469 (2002).
Morris, T., Avenant-Oldewage, A., Lamberth, S. & Reed, C. Shark parasites as bio-indicators of metals in two South African embayments. Mar. Pollut. Bull. 104(1–2), 221–228. https://doi.org/10.1016/j.marpolbul.2016.01.027 (2016).
Kennedy, C. R., Bush, A. O. & Aho, J. M. Patterns in helminth communities: why are birds and fish different?. Parasitology 93(1), 205–215. https://doi.org/10.1017/S0031182000049945 (1986).
Bush, A. O., Lafferty, K. D., Lotz, J. M. & Shostak, A. W. Parasitology meets ecology on its own terms: Margolis et al. revisited. J. Parasitol. 83(4), 575–583. https://doi.org/10.2307/3284227 (1997).
Poulin, R. Species richness of parasite assemblages: Evolution and patterns. Annu. Rev. Ecol. Syst. 28, 341–358 (1997).
Rizzo, E. & Bazzoli, N. Reproduction and embryogenesis in Biology and physiology of freshwater neotropical fish (eds. Baldisserotto, B., Criscuolo Urbinati, E. & Cyrino J. E. P.) 287–313 (Academic Press, 2020).
Poulin, R. Variation in the intraspecific relationship between fish length and intensity of parasitic infection: Biological and statistical causes. J. Fish Biol. 56(1), 123–137. https://doi.org/10.1111/j.1095-8649.2000.tb02090.x (2005).
Poulin, R. & Morand, S. Parasite Biodiversity (eds.) 216 Smithsonian Institution Books, 2004.
Capapé, C. et al. Production, maturity, reproductive cycle and fecundity of small-spotted catshark, Scyliorhinus canicula (Chondrichthyes: Scyliorhinidae) from the northern coast of Tunisia (Central Mediterranean). J. Ichthyol. 54, 111–126. https://doi.org/10.1134/S0032945214010020 (2014).
Kennedy, C.R Helminth communities in freshwater fish: structured communities or stochastic assemblages? in Parasite Communities: Patterns and Processes (eds. Esch G. W., Bush A. O., Aho J. M.) 156 (Chapman and Hall, 1990).
Compagno, L. J. V. Sharks of the world in An annotated and illustrated catalogue of shark species known to date. Part 1 - Hexanchiformes to Lamniformes (ed. FAO) 249 (FAO, 1984).
Compagno, L. J. V. Sharks of the world in An annotated and illustrated catalogue of shark species known to date. Part 2 – Carcharhiniformes (ed. FAO) 486 (FAO, 1984).
Acknowledgements
Sampling in the Gulf of Naples was supported by the project ADViSE (PG/2018/0494374). The Maglione family (Fabrizio, Francesco, Giuseppe, Salvatore, and Vincenzo) (Giovanni Padre fishing vessel, Naples, Italy) offered the highest possible support during trawling activities.
Author information
Authors and Affiliations
Contributions
M.S. performed fish dissections, collected and identified parasites, analysed data, and wrote the main manuscript text; B.B. performed statistical analysis; V.T. and F.C. collected fish; M.P. performed fish dissection, collected parasites, and performed molecular analysis of parasites. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Santoro, M., Bellisario, B., Tanduo, V. et al. Drivers of parasite communities in three sympatric benthic sharks in the Gulf of Naples (central Mediterranean Sea). Sci Rep 12, 9969 (2022). https://doi.org/10.1038/s41598-022-14024-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-14024-0
- Springer Nature Limited