Abstract
Extended synaptotagmins (E-Syts) mediate lipid exchange between the endoplasmic reticulum (ER) and the plasma membrane (PM). Anchored on the ER, E-Syts bind the PM via an array of C2 domains in a Ca2+- and lipid-dependent manner, drawing the two membranes close to facilitate lipid exchange. How these C2 domains bind the PM and regulate the ER–PM distance is not well understood. Here, we applied optical tweezers to dissect PM binding by E-Syt1 and E-Syt2. We detected Ca2+- and lipid-dependent membrane-binding kinetics of both E-Syts and determined the binding energies and rates of individual C2 domains or pairs. We incorporated these parameters in a theoretical model to recapitulate salient features of E-Syt-mediated membrane contacts observed in vivo, including their equilibrium distances and probabilities. Our methods can be applied to study other proteins containing multiple membrane-binding domains linked by disordered polypeptides.







Similar content being viewed by others
Data availability
Data supporting the findings of this work were deposited at https://github.com/zhanglabyale/E-Syt_membrane_tethering_simulation.
References
Lemmon, M. A. Membrane recognition by phospholipid-binding domains. Nat. Rev. Mol. Cell Biol. 9, 99–111 (2008).
Hurley, J. H. Membrane binding domains. Biochim. Biophys. Acta 1761, 805–811 (2006).
Pinheiro, P. S., Houy, S. & Sorensen, J. B. C2-domain containing calcium sensors in neuroendocrine secretion. J. Neurochem. 139, 943–958 (2016).
Bian, X., Saheki, Y. & De Camilli, P. Ca2+ releases E-Syt1 autoinhibition to couple ER–plasma membrane tethering with lipid transport. EMBO J. 37, 219–234 (2018).
Xu, J. J. et al. Structure and Ca2+-binding properties of the tandem C2 domains of E-Syt2. Structure 22, 269–280 (2014).
Pangrsic, T., Reisinger, E. & Moser, T. Otoferlin: a multi-C2 domain protein essential for hearing. Trends Neurosci. 35, 671–680 (2012).
Min, S. W., Chang, W. P. & Sudhof, T. C. E-Syts, a family of membranous Ca2+-sensor proteins with multiple C2 domains. Proc. Natl Acad. Sci. USA 104, 3823–3828 (2007).
Saheki, Y. & De Camilli, P. The extended-synaptotagmins. Biochim. Biophys. Acta 1864, 1490–1493 (2017).
Lek, A., Evesson, F. J., Sutton, R. B., North, K. N. & Cooper, S. T. Ferlins: regulators of vesicle fusion for auditory neurotransmission, receptor trafficking and membrane repair. Traffic 13, 185–194 (2012).
Giordano, F. et al. PI(4,5)P2-dependent and Ca2+-regulated ER–PM interactions mediated by the extended synaptotagmins. Cell 153, 1494–1509 (2013).
Chang, C. L. et al. Feedback regulation of receptor-induced Ca2+ signaling mediated by E-Syt1 and Nir2 at endoplasmic reticulum–plasma membrane junctions. Cell Rep. 5, 813–825 (2013).
Saheki, Y. & De Camilli, P. Endoplasmic reticulum–plasma membrane contact sites. Annu. Rev. Biochem. 86, 659–684 (2017).
Saheki, Y. et al. Control of plasma membrane lipid homeostasis by the extended synaptotagmins. Nat. Cell Biol. 18, 504–515 (2016).
Yu, H. J. et al. Extended synaptotagmins are Ca2+-dependent lipid transfer proteins at membrane contact sites. Proc. Natl Acad. Sci. USA 113, 4362–4367 (2016).
Zhao, H. X. & Lappalainen, P. A simple guide to biochemical approaches for analyzing protein–lipid interactions. Mol. Biol. Cell 23, 2823–2830 (2012).
Knight, J. D., Lerner, M. G., Marcano-Velazquez, J. G., Pastor, R. W. & Falke, J. J. Single molecule diffusion of membrane-bound proteins: window into lipid contacts and bilayer dynamics. Biophys. J. 99, 2879–2887 (2010).
Ma, L. et al. Single-molecule force spectroscopy of protein–membrane interactions. eLife 6, e30493 (2017).
Nath, V. R., Mishra, S., Basak, B., Trivedi, D. & Raghu, P. Extended synaptotagmin regulates membrane contact site structure and lipid transfer function in vivo. EMBO Rep. 21, e50264 (2020).
Fernandez-Busnadiego, R., Saheki, Y. & De Camilli, P. Three-dimensional architecture of extended synaptotagmin-mediated endoplasmic reticulum–plasma membrane contact sites. Proc. Natl Acad. Sci. USA 112, E2004–E2013 (2015).
Idevall-Hagren, O., Lu, A., Xie, B. & De Camilli, P. Triggered Ca2+ influx is required for extended synaptotagmin 1-induced ER–plasma membrane tethering. EMBO J. 34, 2291–2305 (2015).
Kang, F. et al. E-Syt1 re-arranges STIM1 clusters to stabilize ring-shaped ER–PM contact sites and accelerate Ca2+ store replenishment. Sci Rep. 9, 3975 (2019).
Schauder, C. M. et al. Structure of a lipid-bound extended synaptotagmin indicates a role in lipid transfer. Nature 510, 552–555 (2014).
Bian, X., Zhang, Z., Xiong, Q. C., De Camilli, P. & Lin, C. X. A programmable DNA-origami platform for studying lipid transfer between bilayers. Nat. Chem. Biol. 15, 830–837 (2019).
Li, P. Q., Lees, J. A., Lusk, C. P. & Reinisch, K. M. Cryo-EM reconstruction of a VPS13 fragment reveals a long groove to channel lipids between membranes. J. Cell Biol. 219, e202001161 (2020).
Wong, L. H., Gatta, A. T. & Levine, T. P. Lipid transfer proteins: the lipid commute via shuttles, bridges and tubes. Nat. Rev. Mol. Cell Biol. 20, 85–101 (2019).
Corbalan-Garcia, S. & Gomez-Fernandez, J. C. Signaling through C2 domains: more than one lipid target. Biochim. Biophys. Acta 1838, 1536–1547 (2014).
Veggiani, G. et al. Programmable polyproteams built using twin peptide superglues. Proc. Natl Acad. Sci. USA 113, 1202–1207 (2016).
Min, D., Jefferson, R. E., Bowie, J. U. & Yoon, T. Y. Mapping the energy landscape for second-stage folding of a single membrane protein. Nat. Chem. Biol. 11, 981–987 (2015).
Zhang, Y. L., Jiao, J. & Rebane, A. A. Hidden Markov modeling with detailed balance and its application to single protein folding. Biophys. J. 111, 2110–2124 (2016).
Rebane, A. A., Ma, L. & Zhang, Y. L. Structure-based derivation of protein folding intermediates and energies from optical tweezers. Biophys. J. 110, 441–454 (2016).
Steinkuhler, J. et al. Membrane fluctuations and acidosis regulate cooperative binding of ‘marker of self’ protein CD47 with the macrophage checkpoint receptor SIRPα. J. Cell Sci. 132, jcs216770 (2018).
Weikl, T. R., Hu, J. L., Kav, B. & Rozycki, B. Binding and segregation of proteins in membrane adhesion: theory, modeling, and simulations. Adv. Biomembr. Lipid Self-Assem. 30, 159–194 (2019).
Marko, J. F. & Siggia, E. D. Stretching DNA. Macromolecules 28, 8759–8770 (1995).
Krishnamurthy, V. M., Semetey, V., Bracher, P. J., Shen, N. & Whitesides, G. M. Dependence of effective molarity on linker length for an intramolecular protein–ligand system. J. Am. Chem. Soc. 129, 1312–1320 (2007).
Shen, H., Pirruccello, M. & De Camilli, P. SnapShot: membrane curvature sensors and generators. Cell 150, 1300.e1–1300.e2 (2012).
Ross, T. D. et al. Integrins in mechanotransduction. Curr. Opin. Cell Biol. 25, 613–618 (2013).
Basu, R. et al. Cytotoxic T cells use mechanical force to potentiate target cell killing. Cell 165, 100–110 (2016).
Weikl, T. R. Membrane-mediated cooperativity of proteins. Annu. Rev. Phys. Chem. 69, 521–539 (2018).
Sheetz, M. P. Cell control by membrane–cytoskeleton adhesion. Nat. Rev. Mol. Cell Biol. 2, 392–396 (2001).
Brownell, W. E., Qian, F. & Anvari, B. Cell membrane tethers generate mechanical force in response to electrical stimulation. Biophys. J. 99, 845–852 (2010).
Jiao, J. Y., Rebane, A. A., Ma, L. & Zhang, Y. L. Single-molecule protein folding experiments using high-resolution optical tweezers. Methods Mol. Biol. 1486, 357–390 (2017).
Moffitt, J. R., Chemla, Y. R., Izhaky, D. & Bustamante, C. Differential detection of dual traps improves the spatial resolution of optical tweezers. Proc. Natl Acad. Sci. USA 103, 9006–9011 (2006).
Sirinakis, G., Ren, Y. X., Gao, Y., Xi, Z. Q. & Zhang, Y. L. Combined and versatile high-resolution optical tweezers and single-molecule fluorescence microscopy. Rev. Sci. Instrum. 83, 093708 (2012).
Zhang, Y. L., Sirinakis, G., Gundersen, G., Xi, Z. Q. & Gao, Y. DNA translocation of ATP-dependent chromatin remodelling factors revealed by high-resolution optical tweezers. Methods Enzymol. 513, 3–28 (2012).
Gittes, F. & Schmidt, C. F. Interference model for back-focal-plane displacement detection in optical tweezers. Opt. Lett. 23, 7–9 (1998).
Gao, Y. et al. Single reconstituted neuronal SNARE complexes zipper in three distinct stages. Science 337, 1340–1343 (2012).
Cowley, A. C., Fuller, N. L., Rand, R. P. & Parsegian, V. A. Measurement of repulsive forces between charged phospholipid bilayers. Biochemistry 17, 3163–3168 (1978).
Zorman, S. et al. Common intermediates and kinetics, but different energetics, in the assembly of SNARE proteins. eLife 3, e03348 (2014).
Acknowledgements
We thank J. Jiao and A. Rebane for technical assistance. This work was supported by NIH grants R35GM131714, R01GM093341 and R01GM120193 to Y.Z.; R01NS113236 to E.K.; and NS36251 and DA018343 to P.D.C.
Author information
Authors and Affiliations
Contributions
All authors designed experiments and analyzed data; J.G., L.M. and Y.L. performed single-molecule experiments; J.G., X.B., L.M. and Y.C. prepared protein and DNA samples; Y.Z. performed the calculations shown in Fig. 6. J.G., X.B., E.K., P.D.C. and Y.Z. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Chemical Biology thanks Raul Perez-Jimenez and other anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Representative proteins that contain multiple C2 domains.
The lengths (a.a.) of the predicted disordered linkers that join different protein domains or membranes are indicated.
Extended Data Fig. 2 Two conserved membrane binding motifs in the C2 domains of synaptotagmins (Syts) and extended synaptotagmins (E-Syts).
Alignments of the C2 amino acid sequences showing two conserved membrane binding motifs highlighted in bold and color: the Ca2+-binding motif (blue) and the basic patch (red).
Extended Data Fig. 3 The purified E-Syt1 C2ABCDE was sufficiently pure and biotinylated.
(a) SDS-PAGE of the purified E-Syt1 C2ABCDE stained with Coomassie blue. Streptavidin maintained its tetrameric structure in the SDS gel when it was loaded to the gel without heating to high temperature. Binding of streptavidin to the biotinylated E-Syt1 C2ABCDE shifted the migration of C2ABCDE to a position with a higher molecular weight. (b) Gel filtration chromatogram of E-Syt1 C2ABCDE in a Superdex-200 column. Similar results were obtained from three batches of purified proteins.
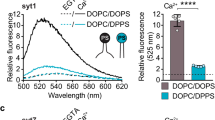
Extended Data Fig. 4 E-Syt1 C2AB only weakly binds to membranes enriched with negatively charged lipids.
(a, b) Diagrams showing no binding (a) and weak binding (b) of the E-Syt1 C2AB domain in the presence of 10% and 20% DOPS, respectively. (c) Force-extension curves showing no membrane binding of E-Syt1 C2AB domain in the absence of supported bilayer or in the presence of the supported bilayer containing 10% DOPS. Weak binding was detected in the presence of 20% DOPS, as indicated by the rip at low force (red arrow). (d) Extension-time trajectories at constant forces (black curves) and their idealized transitions derived from hidden-Markov modeling (red curves). (e) Unbinding probability and binding and unbinding rates (symbols) and their best model fits (lines). The fitting revealed an unbinding energy of 4.4 (±0.3) kBT for the E-Syt1 C2AB domain (Supplementary Table 2).
Extended Data Fig. 5 The microfluidic system to facilitate changes of the Ca2+ concentration in the single-molecule manipulation experiment.
(a) Schematics of the microfluidic system to change Ca2+ concentration when a single C2 repeat was being pulled. Two buffers containing 200 mM NaCl, 25 mM HEPES, pH 7.4, and 500 µM EGTA were prepared, one with CaCl2 (Ca2+ buffer) and another without CaCl2 (EGTA buffer). The two buffers were flowed through a mixer into the central flow cell. The two flows were independently controlled using computer-controlled pressure regulators (MS4-LR, Festo, NY) in combination with flow sensors (SLI-0430, Sensirion, Switzerland) that measure the flow rates. The constant flow rate was achieved by adjusting the pressure in the buffer vial through PID feedback control using a LabVIEW interface. The total calcium concentration in the flow cell ([Ca]), which consisted of both free and EGTA-chelated calcium, was determined by the total calcium concentration of the Ca2+ buffer ([Ca]=V1) and volume velocities of the two buffers (V1 and V2) before mixing. The free Ca2+ concentration ([Ca2+]) in the flow cell was calculated using Maxchelator (Web version v1.2) based on the total concentrations of calcium and EGTA. (b) The measured tracing dye concentration and predicted free Ca2+ concentration in the flow cell as the flow rate of the Ca2+ channel linearly increased from 0 to 12 µL/min while keeping the total flow rate of the two channels at 12 µL/min. To test the concentration change scheme, we added 100 nM rhodamine dye to the Ca2+ buffer and detected the concentration of the dye in the flow cell based on its fluorescence intensity measured by widefield fluorescence microscopy. We linearly increased the flow rate of the rhodamine-containing Ca2+ buffer from 0 to 12 µL/min and simultaneously decreased the flow rate of the EGTA buffer to keep the total flow rate of the two buffers to be 12 µL/min. The dye concentration linearly increased as expected, which justified our concentration change scheme. However, although the observation implied that the total calcium concentration in the flow cell varied linearly as predicted, the corresponding free Ca2+ concentration ([Ca2+]) responded in a nonlinear manner due to the buffering effect of EGTA. Combining with the flow control system, we detected C2 membrane binding transitions at constant force while changing Ca2+ concentration either continuously in the presence of a flow or stepwise in the absence of flow. While the former method allowed rapid [Ca2+] change at the expense of slight extra noise in force and extension measurements, the latter method permitted more accurate single-molecule measurement in the absence of flow after each [Ca2+] change (Fig. 4).
Extended Data Fig. 6 E-Syt1 C2ABCDE binds to the membrane containing 30% DOPS and 0% PI(4,5)P2 via its C2CD domain, but not its C2E domain.
C2CD unbinding probability (top panel) and binding and unbinding rates (bottom panel) as a function of force. The experimental data (symbols) were fit by a nonlinear model to yield the best-fits (lines).
Extended Data Fig. 7 E-Syt2 C2C undergoes a reversible force-dependent, but Ca2+-independent conformational change to inactivate its membrane binding.
(a-c) Extension-time trajectories at constant force in the absence (a) and presence (b) of Ca2+ for E-Syt2 C2ABC or in the presence of Ca2+ for E-Syt2 C2C (c). The long gaps in the unbound state highlighted blue represent the binding inactive state. (d) Diagram of the conformational transition of the C2 domain in the binding active and inactive states.
Extended Data Fig. 8 Cytosolic E-Syt1 containing the SMP domain binds to membranes in a manner like its C2 repeat C2ABCDE.
(a) Schematic diagram showing different E-Syt1 binding states. (b) Force-extension curves obtained by pulling E-Syt1 under different conditions. (c) Extension-time trajectories of E-Syt1 at constant force in the presence or absence of Ca2+.
Extended Data Fig. 9 Calculations of equilibrium membrane distance and membrane binding energy and kinetics.
(a) Diagram illustrating the equilibrium membrane distance determined by the balanced membrane repulsive force (equation 6 in the main text) and E-Syt tethering force (equation 1). The equilibrium distance was solved as a solution for the system of equations \(\left\{ {\begin{array}{*{20}{l}} {\frac{{k_BT}}{P}\left[ {\frac{1}{{4\left( {1 - \frac{x}{L}} \right)^2}} + \frac{x}{L} - \frac{1}{4}} \right] = - V\prime (d),} \hfill \\ {d = x + h,} \hfill \end{array}} \right.\) (7). where V′ is the derivative of V(d) and h is the total length of the folded C2 modules in the pulling direction1 (estimated as 2 nm for each C2 module2,3). (b) Energy landscape corresponding to the C2-membrane interaction with the C2 module free in the solution (blue curve), tethered to the membrane by a flexible and relaxed polypeptide linker (black), or tethered to the membrane with a stretched linker (red). The three key parameters of the energy landscape associated with the free C2 module are determined from our single-molecule measurements (Supplementary Table 1). The energy landscape with the tethered and relaxed C2 module (corresponding to the force-unloaded C2 module) is determined by equation 3, with an effective concentration of the C2 module around the membrane estimated by equation 4. Note that the tethering does not affect the unbinding rate of the C2 module4. Stretching the bound C2 module by moving the PM membrane away from the ER membrane increases the energy barrier for C2 binding by Estretch and decreases the energy barrier for C2 unbinding by FΔx as indicated by equation 5.
Extended Data Fig. 10 Calculated state probabilities, forces, and energy as a function of membrane separation due to potential trans-membrane binding of E-Syt1 (left panel) and E-Syt2 (right panel) lacking a membrane-binding C-terminal C2 module.
(a, c) Schematics of different C2 binding and membrane tethering states in the absence and presence of Ca2+ for E-Syt1 (a) or E-Syt2 (c). The calculations were to simulate the results of membrane contact formation from in vivo imaging using E-Syts with mutant C-terminal C2 domains (C2E in E-Syt1 or C2C in E-Syt2 and E-Syt3) that did not bind to membranes or with the domains truncated. (b, d) Calculated probabilities (top panel), average stretching force (middle), and free energy (bottom) of different states for truncated E-Syt1 (b) or E-Syt2 (d). Calculations corresponding to the presence of Ca2+ or the absence of Ca2+ are indicated by solid and dashed lines, respectively, with their colors indicating different states as shown in a or b: red for the bound state I and black for the unbound state iii. Stable and unstable states are indicated by solid and hollow circles, respectively. The derived equilibrium distances and free energy are shown in Supplementary Table 3.
Supplementary information
Supplementary Information
Supplementary Tables 1–3 and Note
Rights and permissions
About this article
Cite this article
Ge, J., Bian, X., Ma, L. et al. Stepwise membrane binding of extended synaptotagmins revealed by optical tweezers. Nat Chem Biol 18, 313–320 (2022). https://doi.org/10.1038/s41589-021-00914-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41589-021-00914-3
- Springer Nature America, Inc.
This article is cited by
-
Insights into membrane association of the SMP domain of extended synaptotagmin
Nature Communications (2023)
-
Forces of Change: Optical Tweezers in Membrane Remodeling Studies
The Journal of Membrane Biology (2022)