Abstract
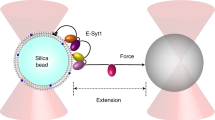
The three extended synaptotagmins (E-Syts) are endoplasmic reticulum (ER)-localized membrane proteins that mediate tethering of the ER to the plasma membrane (PM) via C2 domain-dependent interactions regulated by Ca2+ and/or PI(4,5)P2. The E-Syts also contains a Synaptotagmin-like Mitochondrial lipid-binding Protein (SMP) domain, a lipid-harboring module through which they mediate lipid transport between the two adjacent membranes. Here, we describe in vitro liposome-based methods to study the membrane tethering and lipid transport functions of E-Syt1. Its membrane tethering activity is monitored through a turbidity-based assay, and its lipid transport property is analyzed via fluorescence resonance energy transfer (FRET)-based assay. These in vitro methods have enabled us to gain insight into the mechanism of action and regulation of E-Syt1, such as the role of Ca2+ in releasing E-Syt1 from an autoinhibitory conformation. The same methods could be adapted to the study of other lipid transport proteins that function at membrane contact sites.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Friedman JR, Voeltz GK (2011) The ER in 3D: a multifunctional dynamic membrane network. Trends Cell Biol 21:709–717
Gallo A, Vannier C, Galli T (2016) Endoplasmic reticulum-plasma membrane associations:structures and functions. Annu Rev Cell Dev Biol 32:279–301
Saheki Y, De Camilli P (2017) Endoplasmic reticulum-plasma membrane contact sites. Annu Rev Biochem 86:659–684
Stefan CJ, Manford AG, Emr SD (2013) ER-PM connections: sites of information transfer and inter-organelle communication. Curr Opin Cell Biol 25:434–442
Bian X, Saheki Y, De Camilli P (2018) Ca(2+) releases E-Syt1 autoinhibition to couple ER-plasma membrane tethering with lipid transport. EMBO J 37:219–234
Giordano F, Saheki Y, Idevall-Hagren O et al (2013) PI(4,5)P(2)-dependent and Ca(2+)-regulated ER-PM interactions mediated by the extended synaptotagmins. Cell 153:1494–1509
Manford AG, Stefan CJ, Yuan HL et al (2012) ER-to-plasma membrane tethering proteins regulate cell signaling and ER morphology. Dev Cell 23:1129–1140
Min SW, Chang WP, Sudhof TC (2007) E-Syts, a family of membranous Ca2+−sensor proteins with multiple C2 domains. Proc Natl Acad Sci U S A 104:3823–3828
Toulmay A, Prinz WA (2012) A conserved membrane-binding domain targets proteins to organelle contact sites. J Cell Sci 125:49–58
Saheki Y, Bian X, Schauder CM et al (2016) Control of plasma membrane lipid homeostasis by the extended synaptotagmins. Nat Cell Biol 18:504–515
Kopec KO, Alva V, Lupas AN (2010) Homology of SMP domains to the TULIP superfamily of lipid-binding proteins provides a structural basis for lipid exchange between ER and mitochondria. Bioinformatics 26:1927–1931
Kopec KO, Alva V, Lupas AN (2011) Bioinformatics of the TULIP domain superfamily. Biochem Soc Trans 39:1033–1038
Lee I, Hong W (2006) Diverse membrane-associated proteins contain a novel SMP domain. FASEB J 20:202–206
Schauder CM, Wu X, Saheki Y et al (2014) Structure of a lipid-bound extended synaptotagmin indicates a role in lipid transfer. Nature 510:552–555
Chang CL, Hsieh TS, Yang TT et al (2013) Feedback regulation of receptor-induced Ca2+ signaling mediated by E-Syt1 and Nir2 at endoplasmic reticulum-plasma membrane junctions. Cell Rep 5:813–825
Fernandez-Busnadiego R, Saheki Y, De Camilli P (2015) Three-dimensional architecture of extended synaptotagmin-mediated endoplasmic reticulum-plasma membrane contact sites. Proc Natl Acad Sci U S A 112:E2004–E2013
Idevall-Hagren O, Lu A, Xie B et al (2015) Triggered Ca2+ influx is required for extended synaptotagmin 1-induced ER-plasma membrane tethering. EMBO J 34:2291–2305
Kornmann B, Currie E, Collins SR et al (2009) An ER-mitochondria tethering complex revealed by a synthetic biology screen. Science 325:477–481
Lees JA, Messa M, Sun EW et al (2017) Lipid transport by TMEM24 at ER-plasma membrane contacts regulates pulsatile insulin secretion. Science 355:709
Liu LK, Choudhary V, Toulmay A et al (2017) An inducible ER-Golgi tether facilitates ceramide transport to alleviate lipotoxicity. J Cell Biol 216:131–147
Reinisch KM, De Camilli P (2016) SMP-domain proteins at membrane contact sites: structure and function. Biochim Biophys Acta 1861:924–927
Yu H, Liu Y, Gulbranson DR et al (2016) Extended synaptotagmins are Ca2+−dependent lipid transfer proteins at membrane contact sites. Proc Natl Acad Sci U S A 113:4362–4367
Bornhorst JA, Falke JJ (2000) Purification of proteins using polyhistidine affinity tags. Methods Enzymol 326:245–254
Nichols JW, Pagano RE (1983) Resonance energy transfer assay of protein-mediated lipid transfer between vesicles. J Biol Chem 258:5368–5371
Angeletti C, Nichols JW (1998) Dithionite quenching rate measurement of the inside-outside membrane bilayer distribution of 7-nitrobenz-2-oxa-1,3-diazol-4-yl-labeled phospholipids. Biochemistry 37:15114–15119
Acknowledgments
We thank Frederic Pincet, Joshua Lees, Frank Wilson, Heather Wheeler, Louise Lucast, and Yiying Cai for technical assistance. P.D.C. was supported in part by NIH grants NS036251 and DA018343. X.B. was supported by a Human Frontier Science Program long-term fellowship.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Bian, X., De Camilli, P. (2019). In Vitro Assays to Measure the Membrane Tethering and Lipid Transport Activities of the Extended Synaptotagmins. In: Drin, G. (eds) Intracellular Lipid Transport. Methods in Molecular Biology, vol 1949. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-9136-5_15
Download citation
DOI: https://doi.org/10.1007/978-1-4939-9136-5_15
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-9135-8
Online ISBN: 978-1-4939-9136-5
eBook Packages: Springer Protocols