Abstract
Objective
To examine the association between post-diagnostic metformin or statin use with all-cause and prostate cancer (PCa)-specific mortality in men with advanced prostate cancer.
Methods
Our study consisted of 4572 men (Black = 1352, White = 3192, Other Race = 28) diagnosed with advanced cancer (T4/M1/N1) between 1999 and 2013 in the Veteran Health Administration. The association between post-diagnostic (1) metformin and (2) statin use with all-cause and PCa-specific mortality was examined using multivariable, time-varying Cox Proportional Hazard Models. In a secondary analysis, models were stratified by race.
Results
Post-diagnostic metformin use was associated with a reduced risk of all-cause (Hazard Ratio (HR) 0.84, 95% Confidence Interval (CI): 0.73, 0.96) and PCa-specific death (HR: 0.76, 95% CI: 0.63, 0.91). In stratified analyses, the inverse association between post-diagnostic metformin use and both all-cause PCa-specific mortality was limited to White men. Post-diagnostic statin use was associated with a reduced risk of all-cause (HR: 0.75, 95% CI: 0.68, 0.83) and PCa-specific mortality (HR: 0.72; 95% CI: 0.64, 0.81). In stratified analyses, similar inverse associations were observed for post-diagnostic statin use and all-cause and PCa-specific mortality in both Black and White men.
Conclusion
Post diagnostic metformin and statin use may prevent progression to lethal prostate cancer in men with advanced prostate cancer.
Similar content being viewed by others
Introduction
In the United States, 18% of men are diagnosed with prostate cancer (PCa) that has spread to lymph nodes or metastasized [1]. Although the overall survival rate for PCa is high, the 5-year survival rate among men with distant metastases is only 30% [1]. In particular, Black men are almost twice as likely to be diagnosed with PCa that has metastasized distantly and more than twice as likely to die from PCa as compared to White men [2].
Traditional treatments for men with advanced PCa include Androgen Deprivation Therapy (ADT) or chemotherapy [3]. Both have significant side effects, but recent evidence has suggested the chemotherapeutic potential of commonly used medications including metformin and statins. Metformin acts first by reducing hepatic glucose production. The decreased circulating glucose levels and the associated decrease in insulin-like growth factor-1 are hypothesized to protect against cancer cell growth [4]. Statins are presumed to inhibit PCa growth by inhibiting production of tumorigenic molecules and by decreasing activation of oncoproteins Ras and Rho [5]. Both medications have an anti-inflammatory effect, which can further reduce tumor growth [4, 5].
Previous studies of metformin and statins have suggested that both medications individually are associated with a reduced risk of incident PCa, biochemical recurrence, and cancer mortality [6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21]. However, only a limited number of these studies have examined metformin or statin use in men with advanced or metastatic disease. Results from these studies are inconsistent and use varying definitions of advanced disease making comparisons across studies difficult [18, 19, 22,23,24,25]. A recent Surveillance Epidemiology End Results (SEER)-Medicare study among men with high-risk PCa study found that statin use was associated with a reduced risk of PCa mortality, but observed no association between metformin use alone and PCa death [25]. This study defined advanced diseased broadly, utilizing the D’Amico classification of high-risk disease (stage ≥T2c or prostate-specific antigen (PSA) level ≥20 or Gleason score ≥8) [25]. By contrast, a Canadian registry-based study found that metformin use was associated with a reduced risk of PCa mortality in men with advanced disease, but the authors defined advanced disease simply as men that received ADT as primary treatment [22]. Lastly, the Finnish Randomized Study of PCa Screening found that statin use was suggestive of a reduced risk of PCa mortality in men diagnosed with advanced disease (PSA above 20 ng/ml, Gleason 8–10, Stage T2c-T4 and Node+) or men that received ADT, but not in men specifically diagnosed with M1 metastatic disease [18, 26]. It is important to note that both the Canadian and Finnish study may have limited generalizability to men diagnosed with PCa in the United States [18, 22]. To our knowledge, no study has examined metformin or statin use specifically among Black men with advanced disease.
The objective of our study is to use the Veterans Health Administration (VHA) cohort (VA cohort) to assess the chemotherapeutic potential of metformin and statins in men with advanced PCa. Specifically, this study will examine association between post-diagnostic metformin and statin use and risk of PCa death in a racially-diverse cohort of men diagnosed with advanced PCa. The VA cohort is the ideal cohort to examine this association because it utilizes a nationally representative sample of men with PCa with a significant number of Black men. This allows us to examine the effect of metformin and statins among Black men, who are most likely to suffer from adverse PCa outcomes. Moreover, because the VA consists of both academic and non-academic medical centers across the country, the generalizability of our results is increased. Finally, because the VHA provides equal access to care to their enrollees, confounding by care access due to insurance status is removed from our study.
Methods
Data
Data from the VHA national medical care datasets were used. They include VA Clinical Cancer Registry (VACCR), Pharmacy Benefits Management, Clinical Data Warehouse (CDW), and Vital Statistics registry. Information on veterans diagnosed with cancer and treated at a VHA medical center is collected and stored in the VACCR, including information on patient demographics, cancer identification, stage, grade, and treatment [27]. Race is recorded independently by the VACCR and is available for 98% of men. The CDW stores clinical data, and includes additional variables such as PSA values and comorbid conditions.
IRB approval was received from the St. Louis VA Medical Center, Washington University, Department of Defense, and University of Delaware (reliance agreement with Washington University). A waiver of informed consent was received.
Study population
We identified 149,821 men diagnosed with PCa between 1999 and 2013. We utilized a comprehensive definition of advanced disease, defined as men diagnosed with T4, M1, or N1 PCa (n = 5718). We limited our cohort to those with known age, stage, comorbidity status, race, and locale. Our final analytic cohort consisted of 4572 men (80% of the cohort with a diagnosis of T4, M1, or N1 PCa). Of the 4572 men, 1352 men were Black, 3192 were White, and 28 were of another or unknown race.
Outcome
The outcomes of interest were all-cause death and PCa-specific death. VA Vital Status data was used to determine death status, and has been shown previously to capture 97% of all deaths [28, 29]. If the cause of death was noted as ICD-9 code C61, malignant neoplasm of the prostate, the cause of death was attributed to PCa. Death status was determined on December 28, 2014.
Exposure
Our primary exposures were post-diagnostic metformin and statin use. Metformin and statin prescriptions were identified using the Pharmacy Benefit System. Pharmacy records were available through 2014. Metformin use was identified by metformin prescriptions after PCa diagnosis. Statin use was identified in a similar fashion.
Covariates
Model covariates included age at diagnosis, race, locale, PCa grade and stage, Charlson comorbidity index (CCI), diabetes status, pre-diagnostic metformin use, and pre-diagnostic statin use. Age at diagnosis was analyzed as a continuous variable. Race (black, white, other), locale (urban, rural), grade (grade 3–4 vs. 1–2), stage (stage 3–4 vs. 1–2), CCI (none, 1–2, and ≥3), diabetes with complications (yes/no) (Supplementary Material), diabetes without complications (yes/no), pre-diagnostic metformin use (yes/no), and pre-diagnostic statin use (yes/no) were analyzed as categorical variables. Urban and rural status was determined using Rural-urban commuting area (RUCA) codes, with RUCA ≥ 7 defined as rural. In analyses of post-diagnostic statin use, we used a modified CCI score that did not include malignancy as all men in our cohort were diagnosed with PCa [30]. For analyses of post diagnostic metformin use, we additionally removed diabetes from the CCI. We chose to adjust for diabetes status separately in these analyses, and not as a component of the CCI, because diabetes is a potentially important confounder that is strongly associated with metformin use and survival. Patients that had diabetes with (ICD-9: 250.1×–250.9×) or without (ICD-9: 250.0×) complications were identified using ICD-9 codes.
Statistical analysis
Follow-up period began on date of PCa diagnosis and continued until date of death or censoring. All alive men were censored on December 28, 2014. Time-varying Cox Proportional Hazard Models were used to assess the association between metformin use and (1) all-cause and (2) PCa-specific mortality.
Metformin use was analyzed as a time-varying covariate. Men were considered metformin non-users from diagnosis until the first post-diagnostic metformin prescription and metformin-users from the first post-diagnostic metformin prescription until death or censoring. This analytic method has been used previously in similar analyses [31, 32] and allowed us to minimize any potential immortal time bias- i.e. the artificial survival benefit that occurs if men are considered metformin users before the first post-diagnostic presecription [33]. Analyses for all models utilized the time-varying approach. Adjusted models included age at diagnosis, locale, race, tumor grade, tumor stage, CCI (excluding malignancy and diabetes), diabetes status, and pre-diagnostic metformin use. In a sensitivity analysis, we additionally adjusted for income. For all analyses, all-cause and PCa-specific mortality were analyzed in separated models.
Given that we had an a-priori objective to examine associations in Black and White men, we repeated all analyses stratifying by race. In race-stratified models, race was not included in the multi-variable model.
Analyses for statin use were consistent with previously described models. Adjusted models included age at diagnosis, locale, race, tumor grade, tumor stage, CCI (excluding malignancy), diabetes status, and pre-diagnostic statin use.
In a supplementary analysis, we alternatively modeled metformin and statin duration. For both medications, duration was defined as the total sum of days with a prescription post-diagnosis. Any periods of non-use were not included in the duration sum. All analyses were conducted using SAS 9.2 (Cary, NC).
Results
Characteristics of study population
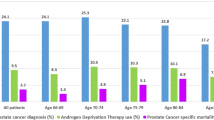
The characteristics of our cohort can be seen in Table 1. Among the analytic cohort (n = 4572), 16.6% had post-diagnostic metformin use, while 52.6% of the cohort had post-diagnostic statin use. The mean age of diagnosis was 68 years. The majority of men were from an urban locale (74.0%), White (69.8%), and diagnosed with grade 3–4 (84.5%) and stage 3–4 (82.3%) tumors. Under half of the cohort had a CCI (excluding malignancy) of zero (43.1%), while 34.7% had a CCI of 1–2. About a quarter of the cohort was diabetic, with 9.4% of men having diabetes with complications. Pre-diagnostic statin use (59.8%) was more prevalent in the cohort than pre-diagnostic metformin use (12.5%).
Ever use of post-diagnostic metformin was similar between Black (18.3%) and White (15.7%) men. However, ever use of post-diagnostic statin was less prevalent in Black men (46.9%) than White (54.9%) men. Black men were slightly younger at diagnosis (Mean age: 66.2) than White (68.5) men. Black men were more likely to be from an urban locale (88.5%) than White men (67.7%). The distribution of tumor grade, tumor stage, the CCI, and diabetes prevalence were similar between Black and White men. Pre-diagnostic metformin use was similar between Black and White men. However, Black men were less likely to be using statins in the pre-diagnostic period (30.5%) as compared to White men (44.1%).
Overall mortality and follow-up
The average follow-up time was 4.5 years. During follow-up, there were 2762 deaths in the analytic cohort. Over half of the deaths (n = 1794, 65.0%) were deaths attributable to PCa. Among Black men there were 813 deaths during follow-up; 530 (65.2%) of these deaths were attributable to PCa. Similarly, among white men there 1930 deaths of which 1249 (64.7%) were attributable to PCa.
Post-diagnostic metformin use and mortality
In multivariable, time-varying analyses (Table 2) post-diagnostic metformin use was associated with a reduction of all-cause mortality in the overall analytic cohort (multivariable-adjusted Hazard Ratio (aHR): 0.84, 95% Confidence Interval (CI): 0.73, 0.96) and a reduction in all-cause mortality among White men (aHR: 0.74, 95% CI: 0.62, 0.88. No statistically significant association was seen among Black men
Similar associations were seen for PCa-specific death. Post-diagnostic metformin use was associated with a reduction of PCa-specific mortality in the overall analytic cohort (aHR: 0.76, 95% CI: 0.63, 0.91) and among White men (aHR: 0.65, 95% CI: 0.52, 0.81). No statistically significant association was observed among Black men.
Post-diagnostic statin use and mortality
In multivariate analyses, post-diagnostic statin use (Table 3) was associated with a reduction of all-cause mortality in the overall analytic cohort (aHR: 0.75, 95% CI: 0.68, 0.83). Similarly, post-diagnostic statin-use was associated with a reduced risk of all-cause death in both Black (aHR: 0.76, 95% CI: 0.64, 0.90) and White men (aHR: 0.75, 95% CI: 0.67, 0.83).
Post-diagnostic statin use was also associated with reductions in PCa-specific mortality. In the overall cohort, post-diagnostic statin use was associated with a reduction in prostate-cancer specific death (aHR: 0.72, 95% CI: 0.64, 0.81). Consistent reductions in the risk of prostate-cancer specific death were seen for both Black (aHR: 0.71, 95% CI: 0.57, 0.88) and White (aHR: 0.72, 95% CI: 0.63, 0.83) men.
Supplementary analyses
Results for both metformin and statin were consistent with additional adjustment for income (data not shown). Analyses of metformin and statin duration indicated that increased duration was associated with a reduced risk of all-cause and PCa-specific mortality in both Black and white men (Supplemental Tables 1 and 2).
Discussion
In this large, nationally representative study of veteran men with advanced PCa, we observed that post-diagnostic metformin use was associated with a reduced risk of all-cause and PCa-specific mortality. In analyses stratified by race, post-diagnostic metformin use was associated with a reduced risk of both all-cause and PCa-specific death in White men, however, no association was observed in Black men. We further observed that post-diagnostic statin use was associated with a reduced risk of both all-cause and PCa-specific death in the full analytic cohort, Black, and White men. To our knowledge, we are one of the first studies to examine post-diagnostic metformin and statin use specifically in Black men with advanced PCa.
Our results are largely consistent with the limited number of studies among men with advanced PCa. Despite variable definitions of advanced disease, consistent with our findings, metformin use has been previously associated with a reduced risk of PCa-specific death [20, 24]. However, a SEER-Medicare study observed no association between metformin use alone and PCa-specific mortality [25]. One possible explanation for this could be that 39% of our cohort ultimately had a death attributable to PCa while only 9% of the SEER-Medicare cohort had a death attributable to PCa [25]. Moreover, unlike our study, other studies consisted primarily of White men.
Importantly, our results expand findings to date by examining associations of post-diagnostic metformin and statin use and mortality specifically among Black men. This is of clinical importance as Black men are more likely to be diagnosed with advanced PCa and die from PCa [2], yet remain underrepresented in epidemiological studies. We did not observe any association between metformin use and PCa-specific death among Black men in time-varying analyses, however, we did observe an inverse association between metformin duration and PCa-specific death in Black and White men. This is perhaps driven by the fact that mean post-diagnostic metformin duration was similar in White and Black men. Moreover, previous research suggests that Black patients are less adherent and more likely to discontinue metformin over time [34]. This could have potentially biased our time-varying analyses towards the null.
We further observed that statin use was inversely associated with both all-cause and PCa-specific mortality among Black and White men. Yet, we also observed that post-diagnostic statin use was lower in Black men (46.9%) than White men (54.9%). This observation is consistent with previous findings that have indicated that Black patients are less likely than White patients to receive guideline-concordant statin therapy [35]. Thus our results suggest that less than ideal levels of statin-therapy could play a role in the persistent disparities observed in PCa outcomes given the strong inverse association between statin use and mortality observed in our study.
More broadly, our results suggest the importance of comorbidity management in men with advanced PCa. Treatment for chronic conditions is commonly siloed from PCa treatment and care, as typically, specialists such as urologists, radiation oncologists, and medical oncologists are responsible for managing PCa treatment, while management of comorbid chronic diseases among PCa survivors fall largely under the purview of primary care physicians [36]. This separation of care can cause cancer survivors to receive less than the recommended care across a broad range of chronic conditions [37].
Beyond just treatment for existing comorbidities, our results provide support for the chemotherapeutic role of metformin and statins in preventing progression to lethal PCa in men diagnosed advanced PCa. This is important as traditional treatments for PCa have a wide range of potential side effects that can have significant impacts on quality of life including urinary, bowel, or sexual dysfunction, lower testosterone, and proctitis [38]. By contrast, metformin and statins are readily available, low-cost, commonly prescribed medications that are generally well-tolerated with low toxicity. The broader use of these medications in advanced PCa patients has the potential to reduce PCa disparities.
Our study is limited by lack of detailed information on medication adherence as we cannot assess whether the men in our study used medications as prescribed. However, we would expect non-adherence to be non-differential across mortality status and this would bias our results toward the null. Despite this, we still observed significant inverse associations. It is also possible that a study among U.S. Veterans may not be fully generalizable to all U.S. men. A key strength is that our study population consisted of a large, nationally representative, ethnically diverse, cohort of US Veterans. Importantly, our study population included all men diagnosed with PCa over a 14-year period at any VA hospital. Moreover, in other studies, access and adherence to prescription medications as well as receipt of adequate PCa care is likely confounded by access to care. We are uniquely able to account for this because the VHA provides care to all Veterans, and thus biases attributable to unequal access to care should be minimized. Additionally, by using a time-varying definition of medication use, we minimized any potential immortal time bias. Finally, to our knowledge we are one of the first studies to examine the impact of metformin and statins specifically in Black men with advanced disease.
Conclusion
Metformin and statins may play a chemotherapeutic role in preventing progression to lethal PCa Future clinical trials should examine the impact of incorporating metformin or statins as a part of the stand of care for men with advanced PCa.
Data availability
Access to VA data is governed by the VA and requires a Without Compensation (WOC) appointment through the VA.
References
SEER. Surveillance, Epidemiology, and end results program. SEER Stat Fact Sheets: Prostate Cancer. https://seer.cancer.gov/statfacts/html/prost.html. Accessed 2 Mar 2021.
SEER. Surveillance, Epidemiology, and End Results Program. Prostate cancer recent trends in SEER age-adjusted incidence rates, 2000–2017. https://seer.cancer.gov/explorer/application.html?site=66&data_type=1&graph_type=2&compareBy=race&chk_race_5=5&chk_race_4=4&chk_race_3=3&chk_race_6=6&chk_race_2=2&hdn_sex=2&age_range=1&stage=106&rate_type=1&advopt_precision=1&advopt_display=2. Accessed 2 Mar 2021.
Teo MY, Rathkopf DE, Kantoff P. Treatment of advanced prostate cancer. Annu Rev Med. 2019;70:479–99.
Papanas N, Maltezos E, Mikhailidis DP. Metformin and cancer: licence to heal? Expert Opin Investig Drugs. 2010;19:913–7.
Demierre MF, Higgins PD, Gruber SB, Hawk E, Lippman SM. Statins and cancer prevention. Nat Rev Cancer. 2005;5:930–42.
Allott EH, Farnan L, Steck SE, Arab L, Su LJ, Mishel M, et al. Statin use and prostate cancer aggressiveness: results from the population-based North Carolina-Louisiana Prostate Cancer Project. Cancer Epidemiol Biomark Prev. 2016;25:670–7.
Allott EH, Howard LE, Cooperberg MR, Kane CJ, Aronson WJ, Terris MK, et al. Postoperative statin use and risk of biochemical recurrence following radical prostatectomy: results from the Shared Equal Access Regional Cancer Hospital (SEARCH) database. BJU Int. 2014;114:661–6.
Bansal D, Undela K, D'Cruz S, Schifano F. Statin use and risk of prostate cancer: a meta-analysis of observational studies. PLoS ONE. 2012;7:e46691.
Gandini S, Puntoni M, Heckman-Stoddard BM, Dunn BK, Ford L, DeCensi A, et al. Metformin and cancer risk and mortality: a systematic review and meta-analysis taking into account biases and confounders. Cancer Prev Res. 2014;7:867–85.
Park HS, Schoenfeld JD, Mailhot RB, Shive M, Hartman RI, Ogembo R, et al. Statins and prostate cancer recurrence following radical prostatectomy or radiotherapy: a systematic review and meta-analysis. Ann Oncol. 2013;24:1427–34.
Raval AD, Thakker D, Negi H, Vyas A, Salkini MW. Association between statins and clinical outcomes among men with prostate cancer: a systematic review and meta-analysis. Prostate Cancer Prostatic Dis. 2016;19:151–62.
Raval AD, Thakker D, Vyas A, Salkini M, Madhavan S, Sambamoorthi U. Impact of metformin on clinical outcomes among men with prostate cancer: a systematic review and meta-analysis. Prostate Cancer Prostatic Dis. 2015;18:110–21.
Wang CP, Lehman DM, Lam YF, Kuhn JG, Mahalingam D, Weitman S, et al. Metformin for reducing racial/ethnic difference in prostate cancer incidence for men with type II diabetes. Cancer Prev Res. 2016;9:779–87.
Yin M, Zhou J, Gorak EJ, Quddus F. Metformin is associated with survival benefit in cancer patients with concurrent type 2 diabetes: a systematic review and meta-analysis. Oncologist. 2013;18:1248–55.
Zhang P, Li H, Tan X, Chen L, Wang S. Association of metformin use with cancer incidence and mortality: a meta-analysis. Cancer Epidemiol. 2013;37:207–18.
Meng Y, Liao YB, Xu P, Wei WR, Wang J. Statin use and mortality of patients with prostate cancer: a meta-analysis. Onco Targets Ther. 2016;9:1689–96.
Stopsack KH, Ziehr DR, Rider JR, Giovannucci EL. Metformin and prostate cancer mortality: a meta-analysis. Cancer Causes Control. 2016;27:105–13.
Murtola TJ, Peltomaa AI, Talala K, Määttänen L, Taari K, Tammela TL, et al. Statin use and prostate cancer survival in the finnish randomized study of screening for prostate cancer. Eur Urol Focus. 2017;3:212–20.
Hamilton R, Ding K, Crook J, O'callaghan C, Higano C, Dearnaley D, et al. The association between statin use and outcomes in patients initiating androgen deprivation therapy. J Urol. 2015;193:e930.
Katz MS, Carroll PR, Cowan JE, Chan JM, D'Amico AV. Association of statin and nonsteroidal anti-inflammatory drug use with prostate cancer outcomes: results from CaPSURE. BJU Int. 2010;106:627–32.
Yu O, Eberg M, Benayoun S, Aprikian A, Batist G, Suissa S, et al. Use of statins and the risk of death in patients with prostate cancer. J Clin Oncol. 2014;32:5–11.
Margel D, Urbach DR, Lipscombe LL, Bell CM, Kulkarni G, Austin PC, et al. Metformin use and all-cause and prostate cancer-specific mortality among men with diabetes. J Clin Oncol. 2013;31:3069–75.
Niraula S, Pond G, de Wit R, Eisenberger M, Tannock IF, Joshua AM. Influence of concurrent medications on outcomes of men with prostate cancer included in the TAX 327 study. Can Urol Assoc J. 2013;7:E74–81.
Lu-Yao GL, Lin Y, Moore D, Graff J, Stroup A, McGuigan K, et al. Combination statin/metformin and prostate cancer specific mortality: a population-based study. J Clin Oncol. 2015;33:5018–5018.
Tan XL, E JY, Lin Y, Rebbeck TR, Lu SE, Shang M, et al. Individual and joint effects of metformin and statins on mortality among patients with high-risk prostate cancer. Cancer Med. 2020;9:2379–89.
Peltomaa AI, Raittinen P, Talala K, Taari K, Tammela TLJ, Auvinen A, et al. Prostate cancer prognosis after initiation of androgen deprivation therapy among statin users. A population-based cohort study. Prostate Cancer Prostatic Dis. 2021;24:917–24.
Department of Veterans Affairs. Veterans Affairs Central Cancer Registry (VACCR). https://www.data.va.gov/dataset/Veterans-Affairs-Central-Cancer-Registry-VACCR-/jvmd-8fgj. Accessed 15 Oct 2021.
Savas LS, del Junco DJ, Bastian LA, Vernon SW. Mortality ascertainment of women veterans: a comparison of sources of vital status information, 1979–2002. Med Care. 2009;47:125–8.
Sohn MW, Arnold N, Maynard C, Hynes DM. Accuracy and completeness of mortality data in the Department of Veterans Affairs. Popul Health Metr. 2006;4:2.
Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373–83.
Sanfilippo KM, Keller J, Gage BF, Luo S, Wang TF, Moskowitz G, et al. Statins are associated with reduced mortality in multiple myeloma. J Clin Oncol. 2016;34:4008–14.
Toriola AT, Luo S, Thomas TS, Drake BF, Chang SH, Sanfilippo KM, et al. Metformin use and pancreatic cancer survival among Non-Hispanic White and African American U.S. Veterans with diabetes mellitus. Cancer Epidemiol Biomark Prev. 2020;29:169–75.
Chaiteerakij R, Petersen GM, Bamlet WR, Chaffee KG, Zhen DB, Burch PA, et al. Metformin use and survival of patients with pancreatic cancer: a cautionary lesson. J Clin Oncol. 2016;34:1898–904.
Trinacty CM, Adams AS, Soumerai SB, Zhang F, Meigs JB, Piette JD, et al. Racial differences in long-term adherence to oral antidiabetic drug therapy: a longitudinal cohort study. BMC Health Serv Res. 2009;9:24.
Nanna MG, Navar AM, Zakroysky P, Xiang Q, Goldberg AC, Robinson J, et al. Association of patient perceptions of cardiovascular risk and beliefs on statin drugs with racial differences in statin use: insights from the patient and provider assessment of lipid management registry. JAMA Cardiol. 2018;3:739–48.
Skolarus TA, Wolf AM, Erb NL, Brooks DD, Rivers BM, Underwood W 3rd, et al. American Cancer Society prostate cancer survivorship care guidelines. CA Cancer J Clin. 2014;64:225–49.
Earle CC, Neville BA. Under use of necessary care among cancer survivors. Cancer. 2004;101:1712–9.
Prostate Cancer Foundation. Prostate cancer side effects. https://www.pcf.org/about-prostate-cancer/prostate-cancer-side-effects/. Accessed 21 Mar 2021.
Funding
SK is supported and by the DOD grant W81XWH-18-1-0168 (PC170130). BFD is supported by the DOD grant W81XWH-19-1-0214 (PC131967). SK, BFD, and S-HC were supported by the Foundation of Barnes-Jewish Hospital and Siteman Cancer Center.
Author information
Authors and Affiliations
Contributions
SK and BFD contributed to conception and design. S-HC contributed to the analytic strategy and made substantial contributions in interpreting the data. SK, MW, and VH conducted the data analyses. RLG provided clinical expertise and contributed to data interpretation. All authors assisted with drafting the manuscript and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and consent to participate
IRB approval was received from the St. Louis VA Medical Center, Washington University, the Department of Defense, and the University of Delaware (via a reliance agreement with Washington University). A waiver of informed consent was received. The study was performed in accordance with the Declaration of Helsinki.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Khan, S., Chang, SH., Hicks, V. et al. Improved survival with post-diagnostic metformin and statin use in a racially diverse cohort of US Veterans with advanced prostate cancer. Prostate Cancer Prostatic Dis 25, 707–712 (2022). https://doi.org/10.1038/s41391-021-00475-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-021-00475-5
- Springer Nature Limited
We’re sorry, something doesn't seem to be working properly.
Please try refreshing the page. If that doesn't work, please contact support so we can address the problem.