Abstract
Background
Mercaptopurine-induced neutropenia can interrupt chemotherapy and expose patients to infection during childhood acute lymphoblastic leukemia (ALL) treatment. Previously, six candidate gene variants associated with mercaptopurine intolerance were reported. Herein, we investigated the association between the mean tolerable dose of mercaptopurine and these genetic variants in Taiwanese patients.
Methods
In total, 294 children with ALL were treated at the National Taiwan University Hospital from April 1997 to December 2017. Germline variants were analyzed for NUDT15, SUCLA2, TPMT, ITPA, PACSIN2, and MRP4. Mean daily tolerable doses of mercaptopurine in the continuation phase of treatment were correlated with these genetic variants.
Results
Mercaptopurine intolerance was significantly associated with polymorphisms in NUDT15 (P value < 0.0001). Patients with SUCLA2 variants received lower mercaptopurine doses (P value = 0.0119). The mean mercaptopurine doses did not differ among patients with TPMT, ITPA, MRP4, and PACSIN2 polymorphisms (P value = 0.9461, 0.5818, and 0.7951, respectively). After multivariable linear regression analysis, only NUDT15 variants retained their clinically significant correlation with mercaptopurine intolerance (P value < 0.0001).
Conclusion
In this cohort, the major genetic determinant of mercaptopurine intolerance was NUDT15 in Taiwanese patients.
Impact
-
NUDT15 causes mercaptopurine intolerance in children with ALL.
-
The NUDT15 variant is a stronger predictor of mercaptopurine intolerance than TPMT in a Taiwanese cohort. This finding is similar with studies performed on Asian populations rather than Caucasians.
-
Pre-emptive genotyping of the patients’ NUDT15 before administering mercaptopurine may be more helpful than genotyping TPMT in Asians.
Similar content being viewed by others
Introduction
Thiopurines (e.g., 6-mercaptopurine (6MP), 6-thioguanine, and azathioprine) are important antimetabolites with different clinical indications. In childhood acute lymphoblastic leukemia (ALL), 6MP-based maintenance therapy is one of the most vital treatments, serving as the backbone of current clinical trials of pediatric ALL. Genetic variations in the genes responsible for thiopurine metabolism can directly influence drug toxicity and anti-leukemic efficacy. For example, genetic polymorphisms in thiopurine methyltransferase (TPMT) have been linked to the susceptibility of thiopurine-induced hematopoietic toxicity in patients, and pre-emptive TPMT genotype-guided dosing is a major example of genetics-based precision medicine in cancer treatment.1 TPMT variants are rare in Asian populations, with most studies indicating that there are no associations between TPMT variants and the tolerable dose of 6MP.2 Genome-wide association studies focusing on Asian populations have identified several new single-nucleotide polymorphisms (SNPs) associated with leukopenia after 6MP administration, including SNPs in inosine triphosphatase (ITPA)3 and multidrug resistance protein 4 (an ATP-binding cassette subfamily C member 4) (MRP4).4,5 However, these genes have not been subjected to thorough validations and have displayed controversial results.6,7
A genome-wide association study described a missense variant in the NUDT15 gene (rs116855232, referred to as c.415C > T or p.Arg139Cys) that is strongly associated with thiopurine-related myelosuppression in patients with inflammatory bowel disease in the Korean population.8 Jun Yang and co-workers2 also identified this variant in association with 6MP intolerance in childhood ALL in a genome-wide association study. Moriyama et al.9 identified four coding variants, c.415C > T, c.416G > A, c.52G > A, and c.36_37insGGAGTC that caused changes in the amino acid sequence of the NUDT15 protein. Additionally, three novel genetic variants of NUDT15 (p.R34T, p.K35E, and p.G17_V18del) have been identified and observed to account for 6MP intolerance.10 Subsequent studies have also validated its clinical significance in childhood ALL. NUDT15 encodes a nucleotide diphosphatase that inactivates thioguanosine triphosphate (TGTP) via conversion to thioguanosine monophosphate. Thus, NUDT15 functions as a negative regulator of intracellular TGTP, with dysfunctional NUDT15 variants leading to the accumulation of DNA-TG resulting in increased cytotoxicity.11 The NUDT15 variants have been subjected to intensive validation and confirmed to be a major genetic determinant of 6MP intolerance in the Asian population.9
In this study, we genotyped the SNPs associated with 6MP intolerance in a Taiwanese cohort treated with the TPOG-ALL (Taiwan Pediatric Oncology Group Acute lymphoblastic leukemia) protocol. The association of these genetic variants with the tolerable dose of 6MP was correlated. With this retrospective study, we hope to identify the most important genetic determination of 6MP intolerance in the Taiwanese cohort. This result can guide the pre-emptive genetic diagnosis before 6MP administration to avoid unwanted neutropenia in future ALL clinical trials.
Methods
Participants and protocols
Participants younger than 18 years of age and diagnosed with ALL were selected at the National Taiwan University Hospital, Taipei, Taiwan. In total, 283 participants were enrolled in this study and treated with the TPOG-ALL protocol between April 1997 and December 2017. This study was approved (No: 201510016RIND) by the Institutional Review Board of the National Taiwan University Hospital. Informed consent was obtained from the parents or legal guardians of the patients, and, in total, 294 patients were enrolled at the study’s initiation. Participants were divided into three groups: very high risk (VHR), high risk (HR), and standard risk (SR), according to the TPOG-ALL-93,12,13 TPOG-ALL-2002,13,14,15 and TPOG-ALL-2013 protocols. Risk classification using the TPOG-ALL protocols was based on the age of onset, initial white blood cell (WBC) count, and cytogenetic alterations at diagnosis. Participants were prospectively assigned to each group (SR, HR, or VHR) based on clinical features and the biological features of their leukemic cells. However, the risk classification of the TPOG-ALL-2013 protocol was additionally determined by the level of minimal residual disease at the end of induction therapy, in addition to the above-mentioned clinical parameters. Maintenance therapy with methotrexate (MTX) was administered at 40 mg/m2 per week and 6MP was initially administered at a dosage of 50–75, 60, 50, or 40 mg/m2 per night depending on the protocol. Dosages were titrated to maintain a WBC count between 1800 and 3000/mm3, absolute neutrophil count (ANC) between 500 and 1200/mm3, and platelet count ≥50,000/mm3. If the counts were low, 6MP was the first to be reduced in dosage by a 25% decrement. If WBC or ANC failed to double at 1 week following dexamethasone pulse therapy, the 6MP and MTX dosages were reduced by 50% referring to reduction from the initial dose given. If WBC or ANC remained the same or decreased, 6MP and MTX doses were halted because the participant would be at a high risk of infection. Blood counts after 3–4 days were used to determine whether the 6MP therapy could be resumed.15
The genetic types of NUDT15, PACSIN2, SUCLA2, ITPA, MRP4, and TPMT
Germline DNA was extracted from remission peripheral blood or bone marrow using the phenol–chloroform method, and DNA concentrations were measured using a NanoDrop 1000 instrument (Thermo Fisher Scientific, Waltham, MA, USA). Three coding sequences of NUDT15 were amplified by polymerase chain reaction (PCR) and evaluated using Sanger sequencing. SNPs in PACSIN2, SUCLA2, ITPA, MRP4, and TPMT were evaluated via TaqMan quantitative PCR (qPCR) assays. Supplementary Table S1 contains the six genes and their polymorphisms. Primer sequences and SNPs are listed in Supplementary Table S2.
For PCR, 5 ng genomic DNA was mixed with 0.5 μM of each primer and 1× Phusion Hot Start II High-Fidelity PCR Master Mix (Thermo Fisher Scientific). PCR was performed via heating to 98 °C for 60 s, followed by 38 cycles at 98 °C for 10 s, maintenance at the annealing temperature for 30 s, and then 72 °C for 20 s; the final extension was performed at 72 °C for 5 min. PCR products were separated using gel electrophoresis, and bands were excised and extracted using a FavorPrep GEL purification kit (FavorGen, Ping-Tung, Taiwan).
Wild-type genotypes were considered to be two alleles that were the same as the reference genotype. Mono-allelic variants had an allele with a polymorphism and an allele with the wild-type genotype. Bi-allelic variants had two alleles that differed from the reference genotype. The NUDT15 genotype was based on a study conducted by Moriyama et al.9,10 and Yang et al.16 Patients with NUDT15 *1/*2 and *3/*6 showed the same sequence result. To determine the diplotype of these patients, we sequenced their parents’ NUDT15 genotype (data not shown).
6MP dosage collection
The mean doses of 6MP for children with ALL treated with the TPOG-ALL protocols in the continuation phase were collected by reviewing their medical records. The 6MP dosage was obtained from the continuation phase to the end of the therapy, completion of trial, or date of relapse or death.
Statistical analysis
Genotype and allelic frequencies for all polymorphisms were estimated by direct counting. The difference in mean daily 6MP dose from the diplotype of each gene was determined by one-way analysis of variance and subsequent P values. P values < 0.05 after Tukey’s method for multiple testing were considered statistically significant. Exploratory analyses to assess NUDT15 and TPMT were based on tests for trends. Cohen κ coefficient was employed to ascertain inter-gene agreement between NUDT15 and SUCLA2. Cohen κ coefficient (κ, ranging from 0 to 1) was interpreted as almost perfect (κ > 0.80), substantial (0.61 ≤ κ ≤ 0.80), moderate (0.41 ≤ κ ≤ 0.60), fair (0.21 ≤ κ ≤ 0.40), slight (0.00 ≤ κ ≤ 0.20), and poor (κ < 0.00). A multivariable linear regression model was used to assess the mean daily 6MP dose of the different diplotypes among NUDT15, TPMT, SUCLA2, ITPA, MRP4, and PACSIN2. All analyses were performed using the SAS software version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Genotypes of NUDT15, TPMT, SUCLA2, ITPA, MRP4, and PACSIN2
In the present study, nine SNPs in five genes and all coding regions of NUDT15 were analyzed in 294 children diagnosed with ALL. Three patients (1.1%) had bi-allelic variants in NUDT15, 67 (24%) had mono-allelic variants, and 213 patients (75%) had wild-type NUDT15 (Table 1). No patient had bi-allelic variants in TPMT; 11 (4.1%) had mono-allelic variants; and 256 patients (96%) had wild-type TPMT. The minor allele frequency of TPMT was 0.02% in this cohort. ITPA (94C > A) polymorphisms were observed in 101 (34%) cases, 91 of which were mono-allelic and 10 of which displayed homozygosity. Fifteen patients (13%) had PACSIN2 polymorphisms, all of which were heterozygous. Ninety-eight patients had SUCLA2 polymorphisms, 88 of which were heterozygous, with 10 displaying homozygosity. MRP4 polymorphisms were detected in 234 (34%) cases, 134 of which were mono-allelic, with 36 displaying homozygosity. The diplotype and minor allele frequency of these genes are listed in Table 1.
Mean dose of 6MP according to the genotypes of NUDT15 and TPMT
In the analysis of the mean tolerable dose of 6MP, 70 patients with very high-risk ALL were excluded as 6MP and MTX constituted only one of the four pairs of drugs administered in rotation every 4 weeks and 6MP was scheduled to be administered for only 7 days every 4 weeks.
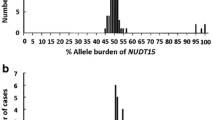
The mean daily doses of 6MP in the NUDT15 wild-type (*1/*1), mono-allelic variant (*1/*2, *1/*3, *1/*5, *1/*6, *1/*7), and bi-allelic variant groups (*2/*2, *2/*7, *3/*6) were 36.8 ± 19.9 (mean ± s.d.), 19.0 ± 13.3, and 4.4 ± 2.3 mg/m2, respectively (P < 0.0001). Patients with the bi-allelic variant NUDT15 tolerated a lower 6MP dose than those with mono-allelic variants and wild-type NUDT15 (Fig. 1).
Data points are represented by a dot. The lower and upper boundaries of the box correspond to the first and third quartiles, respectively. The line within the box represents the median. The upper whisker extends from the upper boundary of the box to the largest value, no further than 1.5× the interquartile range (IQR). The lower whisker extends from the lower boundary of the box to the lowest value, at most no further than 1.5× the IQR. The mean daily mercaptopurine dose was collected by reviewing the medical records. Median values and IQRs are provided to facilitate interpretation of mean daily mercaptopurine dose. Compared to the leftmost group, Kruskal–Wallis P values for the differences in mean daily mercaptopurine dose were <0.0001. Star alleles were assigned by the PharmGKB haplotype set translational table (https://www.pharmgkb.org/).
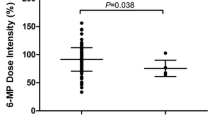
The mean doses of 6MP in the TPMT (rs1142345, rs1800460, and rs1800462) wild-type and mono-allelic variant groups were 31.7 ± 20.3 and 17.7 ± 1.3 mg/m2, respectively (P = 0.4754). No patient in this cohort had double bi-allelic variants of NUDT15 and TPMT. There were two groups—wild-type TPMT with mono-allelic NUDT15 group and mono-allelic TPMT with mono-allelic NUDT15 group. The corresponding daily dose in the two groups were 19.6 ± 13.7 and 13.0 ± 0.9 mg/m2 per day, respectively. Supplementary Figure S1 contains the 6MP dose and the variant alleles of NUDT15 and TPMT. NUDT15 variants remained as an independent genetic determinant of 6MP intolerance in this cohort, irrespective of the TPMT variants. A trend was also found, where the intermediate metabolizers of these compounds were found to tolerate a lower dose of 6MP than the NUDT15 or TPMT variants alone (Supplementary Fig. S1).
Mean dose of 6MP according to genotypes of SUCLA2, ITPA, MRP4, and PACSIN2 individually
Patients with SUCLA2 mono-allelic and bi-allelic variants tolerated less 6MP than those with wild-type SUCLA2 (Table 2, P = 0.0119). However, the mean 6MP dose did not differ among patients with ITPA, MRP4, and PACSIN2 polymorphisms (P = 0.9416, 0.5818, 0.7951, respectively, Table 2).
Inter-gene agreement and multivariable linear regression model
We examined the correlation coefficient among these patients and the association between NUDT15 and SUCLA2 by the Cohen κ method. The level of NUDT15 and SUCLA2 agreement was moderate (K = 0.5001, 95% confident interval was 0.3971–0.6031, P < 0.0001, Supplementary Table S3). By examining NUDT15, TPMT, SUCLA2, ITPA, MRP4, and PACSIN2 using a multivariable linear regression model, we found that only NUDT15 exhibited significance relative to the mean daily 6MP dose (Table 3). SUCLA2 completely lost its significance as revealed by the multivariable linear regression model after adjusting for NUDT15.
Discussion
This retrospective study in Taiwan revealed that NUDT15 variants are the most significant genetic determinants of 6MP intolerance. Patient with TPMT variants and the mono-allelic variant NUDT15 tolerated less 6MP than patients with the mono-allelic variant of NUDT15, but without the TPMT variant. The study also reveals an additional effect of TPMT variants on the mono-allelic NUDT15 variants. Although patients with SUCLA2 variants tolerated less 6MP (P = 0.0119) than those with wild-type SUCLA2, this was identified as a genetic linkage effect by statistical analysis. Other reported candidate gene polymorphisms, including ITPA, MRP4, SUCLA2, and PACSIN2, had no effect on 6MP tolerance in this cohort.
Aligning with previous reports, especially among the Asian population, NUDT15 variants are the most significant in the genetic determination of 6MP intolerance. In fact, TPMT variants were rare, with only 2.1% minor allele frequency; patients with the mono-allelic variant of NUDT15 were found to be less tolerant to 6MP than those with wild-type NUDT15. Since their identification, NUDT15 variants have proven to be a common factor associated with 6MP intolerance in a variety of diseases, especially in Asia. The frequency of NUDT15 mutants was 12% in this study. Liang et al.17 reported the association between 6MP intolerance and NUDT15 exon 3; however, these authors did not sequence NUDT15 in its entirety. In theory, they identified patients with (*1/*2) and (*1/*3), but missed an estimated 3% of patients with NUDT15 variants. We performed Sanger sequencing for all three exons of NUDT15, the products of which were comprehensive genetic variants of NUDT15 in the Taiwanese cohort. The allele frequency of NUDT15 c.415C > T was 16% and 13% in Thailand,18,19 0.37% in Lebanon,20 11% and 26% in Japan,21,22 16% in China,7 7.2% in Korea,23 8.3% in Guatemala, and 12% in Singapore.9 Our report of allele frequency is similar to other regional findings.7,9,18,19,21 The importance of NUDT15 variants on 6MP intolerance has received increasing attention in the Caucasian population as validated by several studies.24 These data provide a compelling rationale for additional pre-emptive testing of NUDT15 genetics, not only in Asians but also in Europeans.25
In contrast to NUDT15, the overall allele frequency of TPMT polymorphism was low in our Taiwanese population, only 2.1% in this cohort, whereas previously, Liang et al.17 demonstrated an allelic frequency of 0.6% in 249 Taiwanese individuals. TPMT demonstrates a minor contribution to the poor 6MP tolerance according to most reports in Asian populations, including one report from Taiwan.17 Only nine patients with mono-allelic mutations of TPMT and none with bi-allelic variants were identified in this cohort. However, a trend was observed, the compound intermediate metabolizers also tolerated a lower dose of 6MP than NUDT15 or TPMT variants alone. A recent study by Zhu et al.26 reported that a case with a combination of bi-allelic NUDT15 and mono-allelic TPMT genotype suffered more severe leukopenia than patient with the NUDT15 or TPMT variants alone. Another study in Asia failed to accurately discern the correlation between TPMT variants and 6MP tolerance.27 Three patients had both NUDT15 and TPMT mono-allelic variant, and these patients showed a low 6MP tolerability, similar to that in patients with the NUDT15 mono-allelic variant. The effect of inheriting both NUDT15 and TPMT variants has been reported in ALL patients.2,7
The relationship between SUCLA2 polymorphisms and 6MP intolerance was first reported in Crohn’s disease by Park et al.28 However, a previous study showed that its significance was completely nullified by relying on the linkage effect with NUDT15 (p.Arg139Cys), this is because both genes are located on the same chromosome. The SUCLA2 variants were associated with low mean tolerable doses in the initial analysis (P = 0.0119). However, after correlation coefficient and multivariable linear regression analysis, the association between SUCLA2 and the mean tolerable dose of 6MP was correlated with the status of NUDT15 and not that of SUCLA2 (Table 3).
Several studies have shown that ITPA genotypes significantly influence the metabolism of 6MP;29,30,31,32,33 however, studies from China7,34 have not confirmed this significance. A Japanese study revealed that polymorphisms in MRP4 affect the dose of 6MP in childhood ALL.4,35 Stocco et al.36 identified that PACSIN2 polymorphisms affected TPMT activity and 6MP-induced adverse effects in children with ALL. Smid et al.37 also identified that the PACSIN2 polymorphism was a significant risk factor for 6MP-induced toxicity in wild-type TPMT patients in Slovenia. These genetic polymorphisms in TPMT, ITPA, MRP4, and PACSIN2 did not affect the 6MP dosage in our cohort. This non-consistent data might suggest that these genetic variants may not be the major genetic determinants of 6MP intolerance in the Taiwanese populations.
The NUDT15 and TPMT variants are of absolute importance to the genetic determination of 6MP intolerance in childhood ALL; thus, pre-emptive genetic diagnosis before the administration of 6MP is suggested. In contrast to the higher TPMT allele frequency in Caucasian populations, NUDT15 has a higher allele frequency in Asian populations. However, we have come across increasing reports of NUDT15 with variants from Europe. In Taiwan, the pre-emptive diagnosis of NUDT15 is likely due to its high prevalence rate. TPMT, although low in frequency, leads to the development of 6MP intolerance when combined with NUDT15, similar to that observed in patients with bi-allelic NUDT15 variants. There have been reports of patients with an intermediate metabolizer status for both TPMT and NUDT15 (i.e., compound intermediate metabolizers), and there is a trend revealing lower thiopurine tolerance in these individuals than in those with intermediate metabolizer status, with TPMT or NUDT15 alone.1 Although there were only three compound intermediate metabolizers for the NUDT15 and TPMT variants in this cohort, the trend of lower 6MP tolerance in these patients seemed significant (Supplementary Fig. S1, trend P < 0.0001). Thus, to address its power due to the low frequency of TPMT variants in Asian patients with childhood ALL, this trend may require a larger clinical trial where more patients with TPMT variants are enrolled.
In conclusion, the most important genetic determinant of 6MP intolerance in this cohort was the NUDT15 variant. This finding is compatible with reports from other East-Asian countries. Although rare in Asia, TPMT polymorphisms might further reduce the mean tolerable dose for patients with NUDT15 variants. However, the impact of TPMT variants on 6MP intolerance may be overlooked in Taiwan and other Asian countries due to its low frequency. The clinical issue of the compound intermediate metabolizer status for both NUDT15 and TPMT variants might be worthy of further investigation.
References
Relling, M. V. et al. Clinical pharmacogenetics implementation consortium guideline for thiopurine dosing based on TPMT and NUDT15 genotypes: 2018 update. Clin. Pharmacol. Ther. 105, 1095–1105 (2019).
Sutiman, N. et al. Predictive role of NUDT15 variants on thiopurine-induced myelotoxicity in Asian inflammatory bowel disease patients. Pharmacogenom. J. 19, 31–43 (2018).
Gerbek, T. et al. Role of TPMT and ITPA variants in mercaptopurine disposition. Cancer Chemother. Pharmacol. 81, 579–586 (2018).
Tanaka, Y. et al. Interaction between NUDT15 and ABCC4 variants enhances intolerability of 6-mercaptopurine in Japanese patients with childhood acute lymphoblastic leukemia. Pharmacogenom. J. 18, 275–280 (2018).
Liu, C. et al. Differential effects of thiopurine methyltransferase (TPMT) and multidrug resistance-associated protein gene 4 (MRP4) on mercaptopurine toxicity. Cancer Chemother. Pharmacol. 80, 287–293 (2017).
Choi, R. et al. Pathway genes and metabolites in thiopurine therapy in Korean children with acute lymphoblastic leukaemia. Br. J. Clin. Pharmacol. 85, 1585–1597 (2019).
Zhou, H. et al. Optimal predictor for 6-mercaptopurine intolerance in Chinese children with acute lymphoblastic leukemia: NUDT15, TPMT, or ITPA genetic variants? BMC Cancer 18, 516 (2018).
Yang, S. K. et al. common missense variant in NUDT15 confers susceptibility to thiopurine-induced leukopenia. Nat. Genet. 46, 1017–1020 (2014).
Moriyama, T. et al. NUDT15 polymorphisms alter thiopurine metabolism and hematopoietic toxicity. Nat. Genet. 48, 367–373 (2016).
Moriyama, T. et al. Novel variants in NUDT15 and thiopurine intolerance in children with acute lymphoblastic leukemia from diverse ancestry. Blood 130, 1209–1212 (2017).
Lee, S. H. R. & Yang, J. J. Pharmacogenomics in acute lymphoblastic leukemia. Best. Pract. Res. Clin. Haematol. 30, 229–236 (2017).
Liang, D. C. Unexpected mortality from the use of E. coli l-asparaginase during remission induction therapy for childhood acute lymphoblastic leukemia: a report from the Taiwan Pediatric Oncology Group. Leukemia 13, 155–160 (1999).
Lin, W. Y., Liu, H. C., Yeh, T. C., Wang, L. Y. & Liang, D. C. Triple intrathecal therapy without cranial irradiation for central nervous system preventive therapy in childhood acute lymphoblastic leukemia. Pediatr. Blood Cancer 50, 523–527 (2008).
Li, M. J. et al. Treatment for childhood acute lymphoblastic leukemia in Taiwan: Taiwan Pediatric Oncology Group ALL-2002 study emphasizing optimal reinduction therapy and central nervous system preventive therapy without cranial radiation. Pediatr. Blood Cancer 64, 234–241 (2017).
Liang, D. C. et al. Long-term results of Taiwan Pediatric Oncology Group studies 1997 and 2002 for childhood acute lymphoblastic leukemia. Leukemia 24, 397–405 (2010).
Yang, J. J. et al. Pharmacogene Variation Consortium Gene Introduction: NUDT15. Clin. Pharmacol. Ther. 105, 1091–1094 (2019).
Liang, D. C. et al. NUDT15 gene polymorphism related to mercaptopurine intolerance in Taiwan Chinese children with acute lymphoblastic leukemia. Pharmacogenom. J. 16, 536–539 (2016).
Chiengthong, K. et al. NUDT15 c.415C>T increases risk of 6-mercaptopurine induced myelosuppression during maintenance therapy in children with acute lymphoblastic leukemia. Haematologica 101, e24–e26 (2016).
Buaboonnam. J. et al. Effect of NUDT15 on incidence of neutropenia in children with acute lymphoblastic leukemia. Pediatr. Int. 61, 754–758 (2019).
Zgheib, N. K. et al. NUDT15 and TPMT genetic polymorphisms are related to 6-mercaptopurine intolerance in children treated for acute lymphoblastic leukemia at the Children’s Cancer Center of Lebanon. Pediatr. Blood Cancer 64, 146–150 (2017).
Tanaka. Y. et al. Susceptibility to 6-MP toxicity conferred by a NUDT15 variant in Japanese children with acute lymphoblastic leukaemia. Br. J. Haematol. 171, 109–115 (2015).
Suzuki, H. et al. Genotyping NUDT15 can predict the dose reduction of 6-MP for children with acute lymphoblastic leukemia especially at a preschool age. J. Hum. Genet. 61, 797–801 (2016).
Park, Y. et al. Star allele-based haplotyping versus gene-wise variant burden scoring for predicting 6-mercaptopurine intolerance in pediatric acute lymphoblastic leukemia patients. Front. Pharmacol. 10, 654 (2019).
Yang, J. J. et al. Inherited NUDT15 variant is a genetic determinant of mercaptopurine intolerance in children with acute lymphoblastic leukemia. J. Clin. Oncol. 33, 1235–1242 (2015).
Schaeffeler, E. et al. Impact of NUDT15 genetics on severe thiopurine-related hematotoxicity in patients with European ancestry. Genet. Med. 21, 2145–2150 (2019).
Zhu, Y. et al. Combination of common and novel rare NUDT15 variants improves predictive sensitivity of thiopurine-induced leukopenia in children with acute lymphoblastic leukemia. Haematologica 103, e293–e295 (2018).
Kim, J. H. et al. Influences of thiopurine methyltransferase genotype and activity on thiopurine-induced leukopenia in Korean patients with inflammatory bowel disease: a retrospective cohort study. J. Clin. Gastroenterol. 44, e242–e248 (2010).
Park, S. K. et al. Influences of XDH genotype by gene-gene interactions with SUCLA2 for thiopurine-induced leukopenia in Korean patients with Crohn’s disease. Scand. J. Gastroenterol. 51, 684–691 (2016).
Tanaka, Y. et al. The activity of the inosine triphosphate pyrophosphatase affects toxicity of 6-mercaptopurine during maintenance therapy for acute lymphoblastic leukemia in Japanese children. Leuk. Res. 36, 560–564 (2012).
Wan Rosalina, W. R. et al. Polymorphism of ITPA 94C>A and risk of adverse effects among patients with acute lymphoblastic leukaemia treated with 6-mercaptopurine. J. Clin. Pharm. Ther. 37, 237–241 (2012).
Farfan, M. J. et al. Prevalence of TPMT and ITPA gene polymorphisms and effect on mercaptopurine dosage in Chilean children with acute lymphoblastic leukemia. BMC Cancer 14, 299 (2014).
Khera, S. et al. Prevalence of TPMT, ITPA and NUDT 15 genetic polymorphisms and their relation to 6MP toxicity in north Indian children with acute lymphoblastic leukemia. Cancer Chemother. Pharmacol. 83, 341–348 (2019).
Moradveisi, B. et al. ITPA, TPMT, and NUDT15 genetic polymorphisms predict 6-mercaptopurine toxicity in Middle Eastern children with acute lymphoblastic leukemia. Front. Pharm. 10, 916 (2019).
Ma, X. et al. Inosine triphosphate pyrophosphohydrolase (ITPA) polymorphic sequence variants in Chinese ALL children and possible association with mercaptopurine related toxicity. Int. J. Clin. Exp. Pathol. 7, 4552–4556 (2014).
Tanaka, Y. et al. Multidrug resistance protein 4 (MRP4) polymorphisms impact the 6-mercaptopurine dose tolerance during maintenance therapy in Japanese childhood acute lymphoblastic leukemia. Pharmacogenom. J. 15, 380–384 (2015).
Stocco, G. et al. PACSIN2 polymorphism influences TPMT activity and mercaptopurine-related gastrointestinal toxicity. Hum. Mol. Genet 21, 4793–4804 (2012).
Smid, A. et al. PACSIN2 polymorphism is associated with thiopurine-induced hematological toxicity in children with acute lymphoblastic leukaemia undergoing maintenance therapy. Sci. Rep. 6, 30244 (2016).
Acknowledgements
We thank all the patients who participated in this study and their parents. We also acknowledge TPOG and the Childhood Cancer Foundation in Taiwan for their assistance in data collection and management. This work was supported by the Ministry of Science and Technology (No. 106-2314-B-002-199-) and the National Taiwan University Hospital (107-S3890). We gratefully acknowledge the financial support of the National Science Council of Taiwan, ROC.
Author information
Authors and Affiliations
Contributions
D.-S.W.: writing—original draft preparation, visualization, investigation, and data curation. C.-H.Y.: writing—original draft preparation, investigation, and data curation. Y.-H.C.: formal analysis, investigation, and data curation. C.-Y.L.: formal analysis. K.-H.L.: resources. S.-T.J.: resources. M.-Y.L.: resources. H.-H.C.: resources. S.-W.L.: supervision, writing—review and editing. D.-T.L.: Resources. H.-Y.C.: methodology, formal analysis. Y.-L.Y.: project administration, supervision, writing—review and editing, resources, conceptualization.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Informed consent
Informed consent was obtained from the parents or legal guardians of the patients, and, in total, 294 patients were enrolled at the study’s initiation.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, DS., Yu, CH., Lin, CY. et al. Childhood acute lymphoblastic leukemia mercaptopurine intolerance is associated with NUDT15 variants. Pediatr Res 89, 217–222 (2021). https://doi.org/10.1038/s41390-020-0868-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-020-0868-8
- Springer Nature America, Inc.
This article is cited by
-
Evaluation of FTO polymorphism in 6-mercaptopurine related intolerance in children with acute lymphoblastic leukemia
Cancer Chemotherapy and Pharmacology (2023)
-
Allele-specific polymerase chain reaction can determine the diplotype of NUDT15 variants in patients with childhood acute lymphoblastic Leukemia
Scientific Reports (2023)
-
Association of genetic variants in TPMT, ITPA, and NUDT15 with azathioprine-induced myelosuppression in southwest china patients with autoimmune hepatitis
Scientific Reports (2021)
-
Determination of NUDT15 variants by targeted sequencing can identify compound heterozygosity in pediatric acute lymphoblastic leukemia patients
Scientific Reports (2020)