Key Points
-
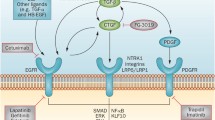
The epidermal growth factor receptor (EGFR) pathway has a crucial role in renal physiology, development, tissue repair and electrolyte handling
-
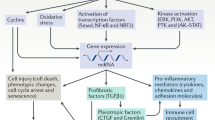
Experimental studies have provided convincing evidence that dysregulated EGFR signalling is involved in many chronic kidney diseases
-
Interventions in the EGFR pathway have been shown to improve renal outcomes in various models of chronic kidney disease (CKD)
-
Nonspecific interventions in the EGFR pathway might lead to a number of systemic adverse effects, as demonstrated in clinical trials in the field of oncology
-
A targeted approach might limit these adverse effects but still inhibit renal fibrosis, and might serve as a treatment strategy for patients with CKD
Abstract
The epidermal growth factor receptor (EGFR) pathway has a critical role in renal development, tissue repair and electrolyte handling. Numerous studies have reported an association between dysregulation of this pathway and the initiation and progression of various chronic kidney diseases such as diabetic nephropathy, chronic allograft nephropathy and polycystic kidney disease through the promotion of renal cell proliferation, fibrosis and inflammation. In the oncological setting, compounds that target the EGFR pathway are already in clinical use or have been evaluated in clinical trials; in the renal setting, therapeutic interventions targeting this pathway by decreasing ligand availability with disintegrin and metalloproteinase inhibitors or with ligand-neutralizing antibodies, or by inhibiting receptor activation with tyrosine kinase inhibitors or monoclonal antibodies are only just starting to be explored in animal models of chronic kidney disease and in patients with autosomal dominant polycystic kidney disease. In this Review we focus on the role of the EGFR signalling pathway in the kidney under physiological conditions and during the pathophysiology of chronic kidney diseases and explore the clinical potential of interventions in this pathway to treat chronic renal diseases.





Similar content being viewed by others
References
Cohen, S. The epidermal growth factor (EGF). Cancer 51, 1787–1791, (1983).
Carpenter, G. & Zendegui, J. G. Epidermal growth factor, its receptor, and related proteins. Exp. Cell Res. 164, 1–10 (1986).
Edwin, F. et al. A historical perspective of the EGF receptor and related systems. Methods Mol. Biol. 327, 1–24 (2006).
Threadgill, D. W. et al. Targeted disruption of mouse EGF receptor: effect of genetic background on mutant phenotype. Science 269, 230–234 (1995).
Tang, J., Liu, N. & Zhuang, S. Role of epidermal growth factor receptor in acute and chronic kidney injury. Kidney Int. 83, 804–810 (2013).
Wang, S. & Hirschberg, R. Role of growth factors in acute renal failure. Nephrol. Dial. Transplant. 12, 1560–1563 (1997).
Hammerman, M. R. Growth factors and apoptosis in acute renal injury. Curr. Opin. Nephrol. Hypertens. 7, 419–424 (1998).
Nouwen, E. J. & De Broe, M. E. EGF and TGF-α in the human kidney: identification of octopal cells in the collecting duct. Kidney Int. 45, 1510–1521 (1994).
Tsujioka, H. et al.: Emerging strategies for ErbB ligand-based targeted therapy for cancer. Anticancer Res. 30, 3107–3112 (2010).
Salomon, D. S., Brandt, R., Ciardiello, F. & Normanno, N. Epidermal growth factor-related peptides and their receptors in human malignancies. Crit. Rev. Oncol. Hematol. 19, 183–232 (1995).
Carter, P. et al. Humanization of an anti-p185HER2 antibody for human cancer therapy. Proc. Natl Acad. Sci. USA 89, 4285–4289 (1992).
Zeng, F., Singh, A. B. & Harris, R. C. The role of the EGF family of ligands and receptors in renal development, physiology and pathophysiology. Exp. Cell Res. 315, 602–610 (2009).
Melenhorst, W. B. et al. Epidermal growth factor receptor signaling in the kidney: key roles in physiology and disease. Hypertension 52, 987–993 (2008).
Privalsky, M. L., Sealy, L., Bishop, J. M., McGrath, J. P. & Levinson, A. D. The product of the avian erythroblastosis virus erbB locus is a glycoprotein. Cell 32, 1257–1267 (1983).
Zheleznova, N. N., Wilson, P. D. & Staruschenko, A. Epidermal growth factor-mediated proliferation and sodium transport in normal and PKD epithelial cells. Biochim. Biophys. Acta 1812, 1301–1313 (2011).
Holbro, T. & Hynes, N. E. ErbB receptors: directing key signaling networks throughout life. Annu. Rev. Pharmacol. Toxicol. 44, 195–217 (2004).
Yoshioka, K. et al. Identification and localization of epidermal growth factor and its receptor in the human glomerulus. Lab. Invest. 63, 189–196 (1990).
Nakopoulou, L. et al. Immunohistochemical study of epidermal growth factor receptor (EGFR) in various types of renal injury. Nephrol. Dial. Transplant. 9, 764–769 (1994).
Gesualdo, L. et al. Expression of epidermal growth factor and its receptor in normal and diseased human kidney: an immunohistochemical and in situ hybridization study. Kidney Int. 49, 656–665 (1996).
Bollee, G. et al. Epidermal growth factor receptor promotes glomerular injury and renal failure in rapidly progressive crescentic glomerulonephritis. Nat. Med. 17, 1242–1250 (2011).
Press, M. F., Cordon-Cardo, C. & Slamon, D. J. Expression of the HER-2/neu proto-oncogene in normal human adult and fetal tissues. Oncogene 5, 953–962 (1990).
Wilson, S. J. et al. Inhibition of HER-2(neu/ErbB2) restores normal function and structure to polycystic kidney disease (PKD) epithelia. Biochim. Biophys. Acta 1762, 647–655 (2006).
Prigent, S. A. et al. Expression of the c-erbB-3 protein in normal human adult and fetal tissues. Oncogene 7, 1273–1278 (1992).
Veikkolainen, V. et al. ErbB4 modulates tubular cell polarity and lumen diameter during kidney development. J. Am. Soc. Nephrol. 23, 112–122 (2012).
Srinivasan, R., Poulsom, R., Hurst, H. C. & Gullick, W. J. Expression of the c-erbB-4/HER4 protein and mRNA in normal human fetal and adult tissues and in a survey of nine solid tumour types. J. Pathol. 185, 236–245 (1998).
Aguiari, G. et al. Polycystin-1 regulates amphiregulin expression through CREB and AP1 signalling: implications in ADPKD cell proliferation. J. Mol. Med. (Berl.) 90, 1267–1282 (2012).
Seno, M. et al. Human betacellulin, a member of the EGF family dominantly expressed in pancreas and small intestine, is fully active in a monomeric form. Growth Factors 13, 181–191 (1996).
Oh, Y. S., Shin, S., Lee, Y. J., Kim, E. H. & Jun, H. S. Betacellulin-induced beta cell proliferation and regeneration is mediated by activation of ErbB-1 and ErbB-2 receptors. PLoS ONE 6, e23894 (2011).
Strachan, L. et al. Cloning and biological activity of epigen, a novel member of the epidermal growth factor superfamily. J. Biol. Chem. 276, 18265–18271 (2001).
Singh, B., Bogatcheva, G., Washington, M. K. & Coffey, R. J. Transformation of polarized epithelial cells by apical mistrafficking of epiregulin. Proc. Natl Acad. Sci. USA 110, 8960–8965 (2013).
Zhuang, S. & Liu, N. EGFR signaling in renal fibrosis. Kidney Int. Suppl. 4, 70–74 (2014).
Salido, E. C., Lakshmanan, J., Fisher, D. A., Shapiro, L. J. & Barajas, L. Expression of epidermal growth factor in the rat kidney. an immunocytochemical and in situ hybridization study. Histochemistry 96, 65–72 (1991).
Salido, E. C., Yen, P. H., Shapiro, L. J., Fisher, D. A. & Barajas, L. In situ hybridization of prepro-epidermal growth factor mRNA in the mouse kidney. Am. J. Physiol. 256, F632–F638 (1989).
Ju, W. et al. Tissue transcriptome-driven identification of epidermal growth factor as a chronic kidney disease biomarker. Sci. Transl. Med. 7, 316ra193 (2015).
Nakagawa, T. et al. Distribution of heparin-binding EGF-like growth factor protein and mRNA in the normal rat kidneys. Kidney Int. 51, 1774–1779 (1997).
Homma, T. et al. Induction of heparin-binding epidermal growth factor-like growth factor mRNA in rat kidney after acute injury. J. Clin. Invest. 96, 1018–1025 (1995).
Harskamp, L. R. et al. Urinary EGF receptor ligand excretion in patients with autosomal dominant polycystic kidney disease and response to tolvaptan. Clin. J. Am. Soc. Nephrol. 10, 1749–1756 (2015).
Klein, T. & Bischoff, R. Active metalloproteases of the A disintegrin and metalloprotease (ADAM) family: biological function and structure. J. Proteome Res. 10, 17–33 (2011).
Schneider, M. R. & Wolf, E. The epidermal growth factor receptor ligands at a glance. J. Cell. Physiol. 218, 460–466 (2009).
Nose, A. et al. Regulation of glucose transporter (GLUT1) gene expression by angiotensin II in mesangial cells: Involvement of HB-EGF and EGF receptor transactivation. Hypertens. Res. 26, 67–73 (2003).
Wang, Z. Transactivation of epidermal growth factor receptor by G protein-coupled receptors: recent progress, challenges and future research. Int. J. Mol. Sci. 17, 95 (2016).
Schlessinger, J. Ligand-induced, receptor-mediated dimerization and activation of EGF receptor. Cell 110, 669–672 (2002).
Sakurai, H., Tsukamoto, T., Kjelsberg, C. A., Cantley, L. G. & Nigam, S. K. EGF receptor ligands are a large fraction of in vitro branching morphogens secreted by embryonic kidney. Am. J. Physiol. 273, F463–F472 (1997).
Rogers, S. A., Ryan, G. & Hammerman, M. R. Metanephric transforming growth factor-alpha is required for renal organogenesis in vitro. Am. J. Physiol. 262, F533–F539 (1992).
Groenestege, W. M. et al. Impaired basolateral sorting of pro-EGF causes isolated recessive renal hypomagnesemia. J. Clin. Invest. 117, 2260–2267 (2007).
Tejpar, S. et al. Magnesium wasting associated with epidermal-growth-factor receptor-targeting antibodies in colorectal cancer: a prospective study. Lancet Oncol. 8, 387–394 (2007).
Kaissling, B., Lehir, M. & Kriz, W. Renal epithelial injury and fibrosis. Biochim. Biophys. Acta 1832, 931–939 (2013).
Liu, N. et al. Blocking the class I histone deacetylase ameliorates renal fibrosis and inhibits renal fibroblast activation via modulating TGF-beta and EGFR signaling. PLoS ONE 8, e54001 (2013).
Lautrette, A. et al. Angiotensin II and EGF receptor cross-talk in chronic kidney diseases: a new therapeutic approach. Nat. Med. 11, 867–874 (2005).
Chen, J. et al. EGFR signaling promotes TGFβ-dependent renal fibrosis. J. Am. Soc. Nephrol. 23, 215–224 (2012).
Liu, N. et al. Genetic or pharmacologic blockade of EGFR inhibits renal fibrosis. J. Am. Soc. Nephrol. 23, 854–867 (2012).
Tang, J. et al. Sustained activation of EGFR triggers renal fibrogenesis after acute kidney injury. Am. J. Pathol. 183, 160–172 (2013).
Zeng, F., Kloepfer, L. A., Finney, C., Diedrich, A., Harris, R. C. Specific endothelial heparin-binding EGF-like growth factor deletion ameliorates renal injury induced by chronic angiotensin II infusion. Am. J Physiol Renal Physiol. http://dx.doi.org/10.1152/ajprenal.00377.2015 (2016).
Qian, Y. et al. Novel epidermal growth factor receptor inhibitor attenuates angiotensin II-induced kidney fibrosis. J. Pharmacol. Exp. Ther. 356, 32–42 (2016).
Kok, H. M., Falke, L. L., Goldschmeding, R. & Nguyen, T. Q. Targeting CTGF, EGF and PDGF pathways to prevent progression of kidney disease. Nat. Rev. Nephrol. 10, 700–711 (2014).
Rayego-Mateos, S. et al. TWEAK transactivation of the epidermal growth factor receptor mediates renal inflammation. J. Pathol. 231, 480–494 (2013).
Harris, R. EGFR signaling in podocytes at the root of glomerular disease. Nat. Med. 17, 1188–1189 (2011).
Paizis, K. et al. Heparin-binding epidermal growth factor-like growth factor is expressed in the adhesive lesions of experimental focal glomerular sclerosis. Kidney Int. 55, 2310–2321 (1999).
Rintala, J. M., Savikko, J., Rintala, S. E., Palin, N. & Koskinen, P. K. Epidermal growth factor receptor inhibition with erlotinib ameliorates anti-thy 1.1-induced experimental glomerulonephritis. J. Nephrol. 29, 359–365 (2015).
Advani, A. et al. Inhibition of the epidermal growth factor receptor preserves podocytes and attenuates albuminuria in experimental diabetic nephropathy. Nephrology (Carlton) 16, 573–581 (2011).
Panchapakesan, U., Pollock, C. & Saad, S. Renal epidermal growth factor receptor: Its role in sodium and water homeostasis in diabetic nephropathy. Clin. Exp. Pharmacol. Physiol. 38, 84–88 (2011).
Wassef, L., Kelly, D. J. & Gilbert, R. E. Epidermal growth factor receptor inhibition attenuates early kidney enlargement in experimental diabetes. Kidney Int. 66, 1805–1814 (2004).
Saad, S. et al. High glucose transactivates the EGF receptor and up-regulates serum glucocorticoid kinase in the proximal tubule. Kidney Int. 68, 985–997 (2005).
Uttarwar, L. et al. HB-EGF release mediates glucose-induced activation of the epidermal growth factor receptor in mesangial cells. Am. J. Physiol. Renal Physiol. 300, F921–F931 (2011).
Wu, D. et al. PKC-β1 mediates glucose-induced akt activation and TGF-β1 upregulation in mesangial cells. J. Am. Soc. Nephrol. 20, 554–566 (2009).
Perlman, A. S. et al. Serum inflammatory and immune mediators are elevated in early stage diabetic nephropathy. Ann. Clin. Lab. Sci. 45, 256–263 (2015).
Gilbert, R. E. et al. Increased epidermal growth factor in experimental diabetes related kidney growth in rats. Diabetologia 40, 778–785 (1997).
Miyazawa, T. et al. Low nitric oxide bioavailability upregulates renal heparin binding EGF-like growth factor expression. Kidney Int. 84, 1176–1188 (2013).
Chen, J., Chen, J. K. & Harris, R. C. EGF receptor deletion in podocytes attenuates diabetic nephropathy. J. Am. Soc. Nephrol. 26, 1115–1125 (2015).
Lindenmeyer, M. T. et al. Interstitial vascular rarefaction and reduced VEGF-A expression in human diabetic nephropathy. J. Am. Soc. Nephrol. 18, 1765–1776 (2007).
Rintala, J. M. et al. Epidermal growth factor inhibition, a novel pathway to prevent chronic allograft injury. Transplantation 98, 821–827 (2014).
Solez, K. et al. International standardization of criteria for the histologic diagnosis of renal allograft rejection: the banff working classification of kidney transplant pathology. Kidney Int. 44, 411–422 (1993).
Sis, B. et al. Epidermal growth factor receptor expression in human renal allograft biopsies: an immunohistochemical study. Transpl. Immunol. 13, 229–232 (2004).
Rizk, D. & Chapman, A. B. Cystic and inherited kidney diseases. Am. J. Kidney Dis. 42, 1305–1317 (2003).
Torres, V. E., Bankir, L. & Grantham, J. J. A case for water in the treatment of polycystic kidney disease. Clin. J. Am. Soc. Nephrol. 4, 1140–1150 (2009).
Neufeld, T. K. et al. In vitro formation and expansion of cysts derived from human renal cortex epithelial cells. Kidney Int. 41, 1222–1236 (1992).
Lowden, D. A. et al. Renal cysts in transgenic mice expressing transforming growth factor-alpha. J. Lab. Clin. Med. 124, 386–394 (1994).
MacRae Dell, K., Nemo, R., Sweeney, W. E. Jr & Avner, E. D. EGF-related growth factors in the pathogenesis of murine ARPKD. Kidney Int. 65, 2018–2029 (2004).
Nakanishi, K., Gattone, V. H. 2nd, Sweeney, W. E. & Avner, E. D. Renal dysfunction but not cystic change is ameliorated by neonatal epidermal growth factor in bpk mice. Pediatr. Nephrol. 16, 45–50 (2001).
Terzi, F. et al. Targeted expression of a dominant-negative EGF-R in the kidney reduces tubulo-interstitial lesions after renal injury. J. Clin. Invest. 106, 225–234 (2000).
Du, J. & Wilson, P. D. Abnormal polarization of EGF receptors and autocrine stimulation of cyst epithelial growth in human ADPKD. Am. J. Physiol. 269, C487–C495 (1995).
Stocklin, E., Botteri, F. & Groner, B. An activated allele of the c-erbB-2 oncogene impairs kidney and lung function and causes early death of transgenic mice. J. Cell Biol. 122, 199–208 (1993).
Zeng, F., Miyazawa, T., Kloepfer, L. A. & Harris, R. C. Deletion of ErbB4 accelerates polycystic kidney disease progression in cpk mice. Kidney Int. 86, 538–547 (2014).
Posner, I., Engel, M., Gazit, A. & Levitzki, A. Kinetics of inhibition by tyrphostins of the tyrosine kinase activity of the epidermal growth factor receptor and analysis by a new computer program. Mol. Pharmacol. 45, 673–683 (1994).
Hojjat-Farsangi, M. Small-molecule inhibitors of the receptor tyrosine kinases: promising tools for targeted cancer therapies. Int. J. Mol. Sci. 15, 13768–13801 (2014).
Adams, G. P. & Weiner, L. M. Monoclonal antibody therapy of cancer. Nat. Biotechnol. 23, 1147–1157 (2005).
Bailey, S. T. et al. mTOR inhibition induces compensatory, therapeutically targetable MEK activation in renal cell carcinoma. PLoS ONE 9, e104413 (2014).
Mulder, G. M. et al. ADAM17 up-regulation in renal transplant dysfunction and non-transplant-related renal fibrosis. Nephrol. Dial. Transplant. 27, 2114–2122 (2012).
Togashi, N., Ura, N., Higashiura, K., Murakami, H. & Shimamoto, K. Effect of TNF-α-converting enzyme inhibitor on insulin resistance in fructose-fed rats. Hypertension 39, 578–580 (2002).
Serino, M. et al. Mice heterozygous for tumor necrosis factor-α converting enzyme are protected from obesity-induced insulin resistance and diabetes. Diabetes 56, 2541–2546 (2007).
Souza, D. G. et al. Effects of PKF242-484 and PKF241-466, novel dual inhibitors of TNF-alpha converting enzyme and matrix metalloproteinases, in a model of intestinal reperfusion injury in mice. Eur. J. Pharmacol. 571, 72–80 (2007).
Sweeney, W. E. Jr et al. Combination treatment of PKD utilizing dual inhibition of EGF-receptor activity and ligand bioavailability. Kidney Int. 64, 1310–1319 (2003).
Nemo, R., Murcia, N. & Dell, K. M. Transforming growth factor alpha (TGF-α) and other targets of tumor necrosis factor-alpha converting enzyme (TACE) in murine polycystic kidney disease. Pediatr. Res. 57, 732–737 (2005).
Dell, K. M. et al. A novel inhibitor of tumor necrosis factor-α converting enzyme ameliorates polycystic kidney disease. Kidney Int. 60, 1240–1248 (2001).
Rose-John, S. ADAM17, shedding, TACE as therapeutic targets. Pharmacol. Res. 71, 19–22 (2013).
Khong, T. F. et al. Inhibition of heparin-binding epidermal growth factor-like growth factor increases albuminuria in puromycin aminonucleoside nephrosis. Kidney Int. 58, 1098–1107 (2000).
Flamant, M. et al. Epidermal growth factor receptor trans-activation mediates the tonic and fibrogenic effects of endothelin in the aortic wall of transgenic mice. FASEB J. 17, 327–329 (2003).
Liu, N. et al. EGF receptor inhibition alleviates hyperuricemic nephropathy. J. Am. Soc. Nephrol. 26, 2716–2729 (2015).
Bou Matar, R. N., Klein, J. D. & Sands, J. M. Erlotinib preserves renal function and prevents salt retention in doxorubicin treated nephrotic rats. PLoS ONE 8, e54738 (2013).
Pugh, J. L., Sweeney, W. E. Jr & Avner, E. D. Tyrosine kinase activity of the EGF receptor in murine metanephric organ culture. Kidney Int. 47, 774–781 (1995).
Richards, W. G. et al. Epidermal growth factor receptor activity mediates renal cyst formation in polycystic kidney disease. J. Clin. Invest. 101, 935–939 (1998).
Torres, V. E. et al. EGF receptor tyrosine kinase inhibition attenuates the development of PKD in han:SPRD rats. Kidney Int. 64, 1573–1579 (2003).
Sweeney, W. E., Chen, Y., Nakanishi, K., Frost, P. & Avner, E. D. Treatment of polycystic kidney disease with a novel tyrosine kinase inhibitor. Kidney Int. 57, 33–40 (2000).
US National Libary of Science. ClinicalTrials.govhttp://www.clinicaltrials.gov/ct2/show/NCT01559363 (2016).
Rastogi, A. et al. Tyrosine kinase inhibitor tesevatinib for patients with autosomal dominant polycystic kidney disease [abstract]. J. Am. Soc. Nephrol. 26, SA-PO865 (2015).
US National Libary of Science. ClinicalTrials.govhttp://www.clinicaltrials.gov/ct2/show/NCT02616055 (2016).
US National Libary of Science. ClinicalTrials.govhttp://www.clinicaltrials.gov/ct2/show/NCT01233869 (2016).
Beyer, C. & Distler, J. H. Tyrosine kinase signaling in fibrotic disorders: translation of basic research to human disease. Biochim. Biophys. Acta 1832, 897–904 (2013).
Izzedine, H. et al. Electrolyte disorders related to EGFR-targeting drugs. Crit. Rev. Oncol. 73, 213–219 (2010).
Gooz, M. ADAM-17: the enzyme that does it all. Crit. Rev. Biochem. Mol. Biol. 45, 146–169 (2010).
Arribas, J. & Esselens, C. ADAM17 as a therapeutic target in multiple diseases. Curr. Pharm. Des. 15, 2319–2335 (2009).
Fang, Q. et al. EGFR mediates hyperlipidemia-induced renal injury via regulating inflammation and oxidative stress: the detrimental role and mechanism of EGFR activation. Oncotarget http://dx.doi.org/10.18632/oncotarget.8222 (2016).
Author information
Authors and Affiliations
Contributions
All authors researched data for the article, made substantial contributions to discussions of the content, wrote the article and reviewed and edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Harskamp, L., Gansevoort, R., van Goor, H. et al. The epidermal growth factor receptor pathway in chronic kidney diseases. Nat Rev Nephrol 12, 496–506 (2016). https://doi.org/10.1038/nrneph.2016.91
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2016.91
- Springer Nature Limited
This article is cited by
-
The disruptor of telomeric silencing 1-like (DOT1L) promotes peritoneal fibrosis through the upregulation and activation of protein tyrosine kinases
Molecular Biomedicine (2024)
-
Abnormal glycosylation in glioma: related changes in biology, biomarkers and targeted therapy
Biomarker Research (2023)
-
Urinary exosome proteins PAK6 and EGFR as noninvasive diagnostic biomarkers of diabetic nephropathy
BMC Nephrology (2023)
-
Renal plasticity revealed through reversal of polycystic kidney disease in mice
Nature Genetics (2021)
-
Key metalloproteinase-mediated pathways in the kidney
Nature Reviews Nephrology (2021)