Abstract
Background
Previous clinical and preclinical studies have indicated that the N-methyl-d-aspartate (NMDA) receptor antagonist, memantine, has neuroprotective properties as well as antidepressant effects. The present study was designed to examine behavioral and molecular effects of memantine administration in rats subjected to the repeated unpredictable stress (RUS) paradigm.
Methods
Rats were split into four groups at random including control + saline, control + memantine, stressed + saline and stressed + memantine. After 10 days of exposure to the RUS paradigm, rats were administered memantine (20 mg/kg) intraperitoneally (ip) for 14 days. Depression-like behavior and memory performance were assessed by measuring immobility time in the forced swim test and passive avoidance test, respectively. The mRNA levels of BDNF and TrkB in the prefrontal cortex and hippocampus were measured by real-time quantitative PCR.
Results
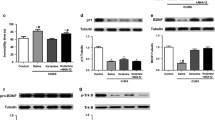
Our results demonstrated that the RUS paradigm caused depression-like behavior and impairment of memory retrieval in rats. We did not find significant changes in BDNF or TrkB mRNA levels in hippocampus, but mRNA levels of TrkB in the prefrontal cortex showed a significant downregulation. Administration of memantine reversed depression-like behavior and memory impairment and significantly increased BDNF and TrkB mRNA levels in both prefrontal cortex and hippocampus of stress exposed rats.
Conclusions
Our study supports the hypothesis that drugs with antagonistic properties on the NMDA receptor, such as memantine, might be efficient in treatment of major depression. Our results also suggest that upregulated mRNA levels of BDNF and TrkB in the brain might be essential for antidepressant-like activity of memantine in stress exposed rats.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Cuijpers P., Smit F. Excess mortality in depression: a meta-analysis of community studies. J Affect Disord 2002;72:227–36.
Kessler RC, Berglund P, Demler O, Jin R, Merikangas KR, Walters EE. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the national comorbidity survey replication. Arch Gen Psychiatry 2005;62:593–602.
Murray CJL, Lopez AD. Global mortality, disability, and the contribution of risk factors: global burden of disease study. Lancet 1997;349:1436–42.
Mutlu O, Ulak G, Çelikyurt IK, Akar FY, Erden F. Effects of citalopram on cognitive performance in passive avoidance, elevated plus-maze and three-panel runway tasks in naïve rats. Chin J Physiol 2011;54:36–46.
Ulak G, Göçmez S, Erden F, Tanyeri P, Utkan T, Yildiz F, et al. Chronic administration of fluoxetine or venlafaxine induces memory deterioration in an inhibitory avoidance task in rats. Drug Dev Res 2006;67:456–61.
McEwen BS. Glucocorticoids, depression, and mood disorders: structural remodeling in the brain. Metabolism 2005;54:20–3.
Van Praag HM. Can stress cause depression? Prog Neuro-Psychopharmacology Biol Psychiatry 2004;28:891–907.
Pariante CM. Depression, stress and the adrenal axis. J Neuroendocrinol 2003;15:811–2.
Kendler KS, Thornton LM, Gardner CO. Genetic risk, number of previous depressive episodes, and stressful life events in predicting onset of major depression. Am J Psychiatry 2001;158:582–6.
Charney DS, Manji HK. Life stress, genes, and depression: multiple pathways lead to increased risk and new opportunities for intervention. Sci Stke 2004;225.
Holsboer F, Barden N. Antidepressants and hypothalamic-pituitary-adrenocortical regulation. Endocr Rev 1996;17:187–205.
Linkowski P, Mendlewicz J, Leclercq R, Brasseur M, Hubain P, Golstein J, et al. The 24-hour profile of adrenocorticotropin and cortisol in major depressive illness. J Clin Endocrinol Metab 1985;61:429–38.
Holsboer F, Gerken A, Stalla GK, Müller OA. ACTH, cortisol, and corticosterone output after ovine corticotropin-releasing factor challenge during depression and after recovery. Biol Psychiatry 1985;20:276–86.
Kosten TA, Galloway MP, Duman RS, Russell DS, D’sa C. Repeated unpredictable stress and antidepressants differentially regulate expression of the bcl-2 family of apoptotic genes in rat cortical, hippocampal, and limbic brain structures. Neuropsychopharmacology 2008;33:1545–58.
Perrotti LI, Hadeishi Y, Ulery PG, Barrot M, Monteggia L, Duman RS, et al. Induction of (FosB in reward-related brain structures after chronic stress. J Neurosci 2004;24:10594–602.
Heine VM, Maslam S, Zareno J, Joëls M, Lucassen PJ. Suppressed proliferation and apoptotic changes in the rat dentate gyrus after acute and chronic stress are reversible. Eur J Neurosci 2004;19:131–44.
Ortiz J, Fitzgerald LW, Lane S, Terwilliger R, Nestler EJ. Biochemical adaptations in the mesolimbic dopamine system in response to repeated stress. Neuropsychopharmacology 1996;14:443–52.
Willner P, Muscat R, Papp M. Chronic mild stress-induced anhedonia: a realistic animal model of depression. Neurosci Biobehav Rev 1992;16:525–34.
Ferguson JM, Shingleton RN. An open-label, flexible-dose study of memantine in major depressive disorder. Clin Neuropharmacol 2007;30:136–44, doi:http://dx.doi.org/10.1097/WNF.0b013e3180314ae7.
Kim Y-K, Na K-S. Role of glutamate receptors and glial cells in the pathophysiology of treatment-resistant depression. Prog Neuro-Psychopharmacol Biol Psychiatry 2016;70:117–26.
Hashimoto K. Emerging role of glutamate in the pathophysiology of major depressive disorder. Brain Res Rev 2009;61:105–23, doi:http://dx.doi.org/10.1016/j.brainresrev.2009.05.005.
Xia P, Chen HV, Zhang D, Lipton SA. Memantine preferentially blocks extrasynaptic over synaptic NMDA receptor currents in hippocampal autapses. J Neurosci 2010;30:11246–50, doi:http://dx.doi.org/10.1523/JNEUROSCI.2488-10.2010.
Berman RM, Cappiello A, Anand A, Oren DA, Heninger GR, Charney DS, et al. Antidepressant effects of ketamine in depressed patients. Biol Psychiatry 2000;47:351–4.
Garcia LSB, Comim CM, Valvassori SS, Réus GZ, Barbosa LM, Andreazza AC, et al. Acute administration of ketamine induces antidepressant-like effects in the forced swimming test and increases BDNF levels in the rat hippocampus. Prog Neuro-Psychopharmacol Biol Psychiatry 2008;32:140–4, doi:http://dx.doi.org/10.1016/j.pnpbp.2007.07.027.
Garcia LSB, Comim CM, Valvassori SS, Réus GZ, Stertz L, Kapczinski F, et al. Ketamine treatment reverses behavioral and physiological alterations induced by chronic mild stress in rats. Prog Neuro-Psychopharmacol Biol Psychiatry 2009;33:450–5.
Garcia LS, Comim CM, Valvassori SS, Réus GZ, Andreazza AC, Stertz L, et al. Chronic administration of ketamine elicits antidepressant-like effects in rats without affecting hippocampal brain-derived neurotrophic factor protein levels. Basic Clin Pharmacol Toxicol 2008;103:502–6.
Moryl E, Danysz W, Quack G. Potential antidepressive properties of amantadine, memantine and bifemelane. Pharmacol Toxicol 1993;72:394–7, doi:http://dx.doi.org/10.1111/j.1600-0773.1993.tb01351.x.
Quan M-N, Zhang N, Wang Y-Y, Zhang T, Yang Z. Possible antidepressant effects and mechanisms of memantine in behaviors and synaptic plasticity of a depression rat model. Neuroscience 2011;182:88–97.
Réus GZ, Stringari RB, Kirsch TR, Fries GR, Kapczinski F, Roesler R, et al. Neurochemical and behavioural effects of acute and chronic memantine administration in rats: further support for NMDA as a new pharmacological target for the treatment of depression? Brain Res Bull 2010;81:585–9, doi:http://dx.doi.org/10.1016/j.brainresbull.2009.11.013.
Zarate CA, Singh JB, Carlson PJ, Brutsche NE, Ameli R, Luckenbaugh DA, et al. A Randomized Trial of an NMDA antagonist in treatment-resistant major depression. Arch Gen Psychiatry 2006;63:856–64, doi:http://dx.doi.org/10.1001/archpsyc.63.8.856.
Amidfar M, Khiabany M, Kohi A, Salardini E, Arbabi M, Roohi Azizi M, et al. Effect of memantine combination therapy on symptoms in patients with moderate-to-severe depressive disorder: randomized, double-blind, placebo-controlled study. J Clin Pharm Ther 2017;42:44–50.
Katalinic N, Lai R, Somogyi A, Mitchell PB, Glue P, Loo CK. Ketamine as a new treatment for depression: a review of its efficacy and adverse effects. Aust New Zeal J Psychiatry 2013;47:710–27.
Kavirajan H. Memantine: a comprehensive review of safety and efficacy. Expert Opin Drug Saf 2009;8:89–109.
Reisberg B, Doody R, Stöffler A, Schmitt F, Ferris S, Möbius HJ. Memantine in moderate-to-severe Alzheimer’s disease. N Engl J Med 2003;348:1333–41.
Volbracht C, Van Beek J, Zhu C, Blomgren K, Leist M. Neuroprotective properties of memantine in different in vitro and in vivo models of excitotoxicity. Eur J Neurosci 2006;23:2611–22, doi:http://dx.doi.org/10.1111/j.1460-9568.2006.04787.x.
Réus GZ, Abelaira HM, Stringari RB, Fries GR, Kapczinski F, Quevedo J. Memantine treatment reverses anhedonia, normalizes corticosterone levels and increases BDNF levels in the prefrontal cortex induced by chronic mild stress in rats. Metab Brain Dis 2012;27:175–82.
Rogóz Z, Skuza G, Maj J, Danysz W. Synergistic effect of uncompetitive NMDA receptor antagonists and antidepressant drugs in the forced swimming test in rats. Neuropharmacology 2002;42:1024–30, doi:http://dx.doi.org/10.1016/S0028-3908(02)00055-2.
Nibuya M, Morinobu S, Duman RS. Regulation of BDNF and trkB mRNA in rat brain by chronic electroconvulsive seizure and antidepressant drug treatments. J Neurosci 1995;15:7539–47.
Duman RS, Monteggia LM. A neurotrophic model for stress-related mood disorders. Biol Psychiatry 2006;59:1116–27.
Nestler EJ, Barrot M, DiLeone RJ, Eisch AJ, Gold SJ, Monteggia LM. Neurobiology of depression. Neuron 2002;34:13–25.
Sandi C, Woodson JC, Haynes VF, Park CR, Touyarot K, Lopez-Fernandez MA, et al. Acute stress-induced impairment of spatial memory is associated with decreased expression of neural cell adhesion molecule in the hippocampus and prefrontal cortex. Biol Psychiatry 2005;57:856–64.
Porsolt RD, Le Pichon M, Jalfre M. Depression: a new animal model sensitive to antidepressant treatments. Nature 1977;266:730–2, doi:http://dx.doi.org/10.1038/266730a0.
Monleón S, Vinader-Caerols C, Arenas MC, Parra A. Antidepressant drugs and memory: insights from animal studies. Eur Neuropsychopharmacol 2008;18:235–48.
Schmittgen TD, Livak KJ. Analyzing real-time PCR data by the comparative CT method. Nat Protoc 2008;3:1101–8.
Marvanová M, Lakso M, Pirhonen J, Nawa H, Wong G, Castrén E. The neuroprotective agent memantine induces brain-derived neurotrophic factor and trkB receptor expression in rat brain. Mol Cell Neurosci 2001;18:247–58, doi:http://dx.doi.org/10.1006/mcne.2001.1027.
Réus GZ, Stringari RB, Ribeiro KF, Ferraro AK, Vitto MF, Cesconetto P, et al. Ketamine plus imipramine treatment induces antidepressant-like behavior and increases CREB and BDNF protein levels and PKA and PKC phosphorylation in rat brain. Behav Brain Res 2011;221:166–71, doi:http://dx.doi.org/10.1016/j.bbr.2011.02.024.
Rogoz Z, Skuza G, Legutko B. Repeated co-treatment with fluoxetine and amantadine induces brain-derived neurotrophic factor gene expression in rats. Pharmacol Rep 2008;60:817–26.
Amidfar M, Réus GZ, Quevedo J, Kim Y-K, Arbabi M. Effect of co-administration of memantine and sertraline on the antidepressant-like activity and brain-derived neurotrophic factor (BDNF) levels in the rat brain. Brain Res Bull 2017;128:29–33.
Kuroda Y, McEwen BS. Effect of chronic restraint stress and tianeptine on growth factors, growth-associated protein-43 and microtubule-associated protein 2 mRNA expression in the rat hippocampus. Mol Brain Res 1998;59:35–9.
Rasmusson AM, Shi L, Duman R. Downregulation of BDNF mRNA in the hippocampal dentate gyrus after re-exposure to cues previously associated with footshock. Neuropsychopharmacology 2002;27:133–42.
Xu H, Chen Z, He J, Haimanot S, Li X, Dyck L, et al. Synergetic effects of quetiapine and venlafaxine in preventing the chronic restraint stress-induced decrease in cell proliferation and BDNF expression in rat hippocampus. Hippocampus 2006;16:551–9.
Shi S-S, Shao S, Yuan B, Pan F, Li Z-L. Acute stress and chronic stress change brain-derived neurotrophic factor (BDNF) and tyrosine kinase-coupled receptor (TrkB) expression in both young and aged rat hippocampus. Yonsei Med J 2010;51:661–71.
Siuciak JA, Lewis DR, Wiegand SJ, Lindsay RM. Antidepressant-like effect of brain-derived neurotrophic factor (BDNF). Pharmacol Biochem Behav 1997;56:131–7, doi:http://dx.doi.org/10.1016/S0091-3057(96)00169-4.
Shirayama Y, AC-H Chen, Nakagawa S, Russell DS, Duman RS. Brain-derived neurotrophic factor produces antidepressant effects in behavioral models of depression. J Neurosci 2002;22:3251–61 20026292.
Duman RS, Malberg J, Nakagawa S, D’Sa C. Neuronal plasticity and survival in mood disorders. Biol Psychiatry 2000;48:732–9.
Duman RS, Heninger GR, Nestler EJ. A molecular and cellular theory of depression. Arch Gen Psychiatry 1997;54:597–606.
Lewin GR, Barde Y-A. Physiology of the neurotrophins. Annu Rev Neurosci 1996;19:289–317.
Watanabe Y, Gould E, Daniels DC, Cameron H, McEwen BS. Tianeptine attenuates stress-induced morphological changes in the hippocampus. Eur J Pharmacol 1992;222:157–62.
Watanabe Y, Gould E, McEwen BS. Stress induces atrophy of apical dendrites of hippocampal CA3 pyramidal neurons. Brain Res 1992;588:341–5.
Stein-Behrens B, Mattson MP, Chang I, Yeh M, Sapolsky R. Stress exacerbates neuron loss and cytoskeletal pathology in the hippocampus. J Neurosci 1994;14:5373–80.
Henningsen K, Andreasen JT, Bouzinova EV, Jayatissa MN, Jensen MS, Redrobe JP, et al. Cognitive deficits in the rat chronic mild stress model for depression: relation to anhedonic-like responses. Behav Brain Res 2009;198:136–41.
Mahableshwarkar AR, Zajecka J, Jacobson W, Chen Y, Keefe RSE. A randomized, placebo-controlled, active-reference, double-blind, flexible-dose study of the efficacy of vortioxetine on cognitive function in major depressive disorder. Neuropsychopharmacology 2015;40:2025.
Patterson SL, Abel T, Deuel TAS, Martin KC, Rose JC, Kandel ER. Recombinant BDNF rescues deficits in basal synaptic transmission and hippocampal LTP in BDNF knockout mice. Neuron 1996;16:1137–45.
Popoli M, Gennarelli M, Racagni G. Modulation of synaptic plasticity by stress and antidepressants. Bipolar Disord 2002;4:166–82.
Arenas MC, Vinader-Caerols C, Monleón S, Martos AJ, Everss E, Ferrer-Añó A, et al. Are the effects of the antidepressants amitriptyline, maprotiline, and fluoxetine on inhibitory avoidance state-dependent? Behav Brain Res 2006;166:150–8.
Hickie I, Naismith S, Ward PB, Turner K, Scott E, Mitchell P, et al. Reduced hippocampal volumes and memory loss in patients with early-and late-onset depression. Br J Psychiatry 2005;186:197–202.
Bremner JD, Vythilingam M, Vermetten E, Nazeer A, Adil J, Khan S, et al. Reduced volume of orbitofrontal cortex in major depression. Biol Psychiatry 2002;51:273–9.
Bennett Ao MR. Stress and anxiety in schizophrenia and depression: glucocorticoids, corticotropin-releasing hormone and synapse regression. Aust New Zeal J Psychiatry 2008;42:995–1002.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Amidfar, M., Kim, YK. & Wiborg, O. Effectiveness of memantine on depression-like behavior, memory deficits and brain mRNA levels of BDNF and TrkB in rats subjected to repeated unpredictable stress. Pharmacol. Rep 70, 600–606 (2018). https://doi.org/10.1016/j.pharep.2017.12.007
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2017.12.007