Abstract
The principal objective of the present paper is to meticulously review the family of biomaterials used in implants. A spectrum of applications of biomaterials in the perspective of prosthesis is also presented. This paper also emphasises on the review of the recent advancements in the field of biomedical implants with respect to mechanical engineering perspective. The latest technologies such as finite element modelling of prosthetic implants, additive manufacturing of implants and certain experimental methods adopted in the field of prosthesis are discussed. Moreover, various models were modelled using SOLIDWORKS® 2022 modelling software and analysed using ANSYS® 2021 R2 finite element analysing software and implant models were additive manufactured to make this review more interesting and for better understanding. Overall, the latest technology in the field of mechanical engineering that fuels its impact in life-saving biomedical engineering has been discussed briefly.
Graphical abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
A prosthesis or prosthetic implant is a manmade medical device that replaces a missing body part, which might be lost through injury, malady, or a condition present during childbirth. Prostheses are meant to re-establish the specific functions of the missing body part. The world of innovation [1] has led to the development of smart prosthetic implants with enhanced aesthetic and functional fronts. The utilisation of prosthesis has become significantly important recently, driven by increasing aging population.
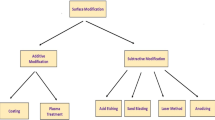
In the present medical research, different configurations of implants have been studied and implemented for various applications in the human body. The main objective of these implants (knee implant, hip implant, dental implant, bone plate, pacemaker, etc.) is focused towards the protection of human lives [2]. These applications differ in terms of their placement and positions subject to the biocompatibility of the human body. These implants are put in areas of high mechanical pressure, for example in the joints during bone replacement or in areas of high synthetic and electrical movement, for example the use of neuroprosthetics [3]. Normally, the implants join the fractured bone as well as deliver good strength to the human body as a principal load-bearing member. Prostheses are broadly classified into external and internal prosthesis; the former deals with the artificial limbs and is employed externally; the latter deals with internal body implants (Fig. 1).
The perfect prosthesis should possess the characteristic features, viz. biocompatible to the human biological environment, corrosion resistance and wear resistance, acceptable solidarity to continue fatigue loading encountered by the joint and low moduli to limit bone resorption [4]. Modern composite materials like carbon fibre are making prosthetics both lighter and more grounded. Modern mechanical engineering tools play a vital role in the field of prosthesis. The modern tools include finite element analysis of implants, modern fabrication processes like additive manufacturing and experimentation techniques. Progressions in additive manufacturing and biometrics have upgraded the lives of amputees. In the field of biomechanics, mechanical testing is a very helpful tool. Mechanical testing is used in traditional biomechanics for a variety of purposes. For example, testing may be used to identify the mechanical characteristics of bone under various loading modes and diverse circumstances, such as age and disease status [5, 6]. Testing can also be used to evaluate fracture fixation methods and support clinical techniques. Implants and biomaterials can be tested mechanically to verify their strength and suitability for therapeutic applications. There are several fundamentals that must be understood in order to do mechanical testing correctly, even though the information from a mechanical test will vary [7,8,9].
The nexus of biomedical innovations and mechanical engineering has grown more and more significant in recent years, providing a bright future for implants. A breakthrough method in mechanical engineering called additive manufacturing (AM) is essential to creating complex, tailored structures that meet the particular requirements of biomedical applications. In order to help with design optimisation, finite element analysis (FEA) offers a potent tool for simulating the intricate mechanical behaviour of implants and biomaterials. In addition to these simulations, experimental techniques offer useful empirical data that helps to validate and improve the models. The combination of mechanical engineering perspectives and biomedical engineering knowledge not only improves the accuracy and productivity of implant development, but it also creates new opportunities for innovation and the application of state-of-the-art technologies in healthcare. The potential for ground-breaking discoveries and game-changing solutions in the field of implants is becoming more and more apparent as these two disciplines come together. This review deals with an introduction to different biomaterials used for fabrication of internal prosthesis and their recent advancement technologies in the field of mechanical engineering (Fig. 2).
Biomaterials
Biomaterials are engineering materials which are compatible to the human body. Every biomaterial is triggered to execute the assigned specific function for versatile applications [10]. Additionally, biomaterials possess the properties, viz. nontoxic, biocompatible, biotough, and ease in manufacturing. Biomaterials are either naturally available or synthesised in the research facility using metallic parts, polymers, ceramics or composite materials. Biomaterials are regularly utilised or potentially adjusted for clinical applications and in this way include part of a human body or biomedical devices which performs, increases, or replaces a characteristic capacity [11]. The family of biomaterials is indicated in Fig. 3.
Metals and alloys
Metallic biomaterials are mainly used in situations where maximum amount of load-bearing acts. Metallic biomaterials can be widely used in the field of prosthesis as they possess very good mechanical properties. But their biocompatibility is low; corrosion is possible in the physiological environment and mechanical properties differ from human tissues [12]. Stainless steel, cobalt-chromium alloys and titanium alloys are the metals and alloys mostly used in the field of prosthesis [13,14,15]. In joint prosthesis, metallic implants are used in the knee, hip and shoulder. Metals are mainly used hip replacement as they are capable of bearing load [16, 17]. A hip implant assembled model of acetabular component, plastic liner, femoral head and femoral stem has been attempted in the present paper using SOLIDWORKS® 3D modelling software version 2022 (Fig. 4a).
In knee replacement, metallic implants replace the load-bearing surfaces of the knee joint to cure the pain and disability [18, 19]. A knee implant assembly model of femoral component, plastic spacer and tibia component has been undertaken in the present paper using SOLIDWORKS® 2022 3D modelling software (Fig. 4b). In fracture fixation plates, metallic bone plates are used to heal the fractured bone [20, 21].
A distal femur locking bone plate model has been endeavoured in the present paper using SOLIDWORKS® 2022 3D modelling software (Fig. 4c). In dental implants, metals are used as tooth replacement [22, 23]. In heart valves, implants are used to replace damaged heart valves. Metallic implants are used in caged disc and hinged leaflet valves [24].
Ceramics
Ceramic biomaterials are highly biocompatible, corrosion resistant, good compression resistant and low to thermal conductivity. Ceramics show numerous applications as biomaterials due to their physicochemical properties. They have the advantage of being inert in the human body, and their hardness and resistance to abrasion make them useful for bones and tooth replacement. But ceramic composites possess low impact resistance and have difficulties in manufacturing and fabrication. Aluminium oxides, zirconia, calcium aluminates and calcium phosphates are the good candidate materials for ceramic materials in prosthesis [25, 26]. Zirconia is used in dental implants. It is bioinert, which means that it will never trigger chemical reactions, migrate to other sites in the body or corrode. However, if properly cared for, they are expected to last for 15–20 years or more, similar to titanium implants [27, 28]. A dental implant assembled model of crown, abutment and screw has been explored in the present paper using SOLIDWORKS® 20223D modelling software (Fig. 4d).
As ceramics can lessen wear and corrosion, they are employed as a covering material for other implants. Orthopaedic and dental implants are increasingly being coated with ceramic materials to increase their wear resistance and promote tissue integration, which will increase the implant’s stability over time. Ceramics are used as bone grafts to replace a missing bone by trauma. Ceramic-based bone grafts are synthetic products that have been widely utilised to reduce the need for iliac crest bone grafting. Ceramic matrices are inorganic, ionically bonded preparations that comprise a large collection of bone graft substitutes [29, 30]. Ceramics are also used as endoprobe for endoscopy treatments to examine a person’s digestive tract without surgery [31, 32]. Ceramics are used as otologic implants to treat injuries related to ear. It is a surgically implanted electronic device that is placed in the temporal bone, which is located behind the ear. This is connected to an electrode array that has been inserted into the cochlea (inner ear) [33, 34]. Ceramics are used in tissue engineering to replace the biological tissue. Bioceramics can be manufactured into a range of forms such as powder, coating, and bulk to serve various purposes in the repair or replacement of human tissue [35, 36].
Polymers
Polymeric biomaterials are easy to synthesise and possess low density. Application of polymers is unlimited when compared to metallic and ceramic polymers. Since they may be made to suit a variety of uses, polymers are frequently employed in the manufacturing of implants. They are simple to manufacture and customise [37]. Biodegradable polymers [38] start to degrade inside the body in certain time when their work is complete. Polypropylene, polyethylene, Teflon, polymethyl methacrylate, silicone, and nylon are the mostly used prosthetic polymers. Polymeric biomaterials are used for making contact lenses for the enhancement of vision. The contact lenses lie on the cornea and they are a replacement for eyeglasses [39, 40]. A contact lens model has been introduced in the present paper using SOLIDWORKS® 2022 3D modelling software (Fig. 4e).
Silicone is used in breast implants. These implants are used to change the size and shape of the breasts and also used to cure congenital defects and deformities of the chest wall. Most silicone and saline implants are FDA approved for 10–20 years, but this does not mean that you have to get them replaced every 10–20 years. You can safely go beyond these time frames, and most patients only have to have 1–2 replacements in their lifetime [41, 42]. Polymers are used in nasal implant which supports lateral cartilage in the nose. One implant may be used to correct one side, or two may be used to correct both sides of the nose to open the nasal passages. The material is gradually absorbed over a period of about 18 months. Supporting the cartilage reduces nasal airway obstruction [43, 44] and helps breathe better. Polymers are used as a coating material in cochlear implants that is used for improving the hearing [45]. Teflon is used for vascular implants, where they substitute an infected artery. Teflon has an excellent patency rate, is simple to apply, and has acceptable interactions with tissues. It also tolerates pressure and flows in medium and large arteries [46, 47]. An artificial stent model has been modelled in this paper using SOLIDWORKS® 2022 software (Fig. 4f).
Composite
Biomaterial using composites has a main advantage of flexibility in design, as their properties are direction dependant and can be optimised [48]. Composites possess greater specific strength, greater specific stiffness, and greater fatigue strength with lesser weight. Due to these advantages, composites replace other biomaterials in various prosthetic implants [49, 50]. In dental implants, composites are used in fixing the missing or damaged teeth. Different types of composite used since its introduction include macrofill composites, microfill composites, hybrid composites, and nanofill composites [51, 52]. Composites and hybrid composites are used in tissue engineering [53, 54]. The usages of composite materials in the orthopaedics field have been increased drastically in the recent years. Composites are used for joint prosthesis in hip, knee and shoulder replacement [55, 56]. Composites are used in bone plates for curing the fractured bone. Composites are also used for making artificial bones for scientific analysis. Due to their capacity to precisely mimic the properties of real bone when compared to first-generation and second-generation bone substitute materials, composites are currently regarded as third-generation orthopaedic biomaterials. Due to the higher stiffness and strength of the inorganic material’s intrinsic qualities, the combination of polymers and ceramic phases results in composite materials with superior mechanical capabilities [57, 58]. Composites are used in artificial tendons which connect muscle to bone and are capable of holding the pressure. To mimic the compliance of a natural anterior cruciate tendon, an artificial ligament must meet stringent specifications and possess at least three crucial characteristics, such as high tensile strength, high elongation, and the suitable stiffness [59, 60].
Advancements in the field of implants: mechanical engineering perspective
Finite element simulation in implants
The finite element analysis (FEA) is the simulation of a system utilising the numerical procedure so called finite element method (FEM). The idea of breaking intricate items down into smaller ones or building complicated objects out of simpler ones is the foundation of the finite element approach, also known as finite element analysis. Mathematics frequently does not provide sufficiently strong techniques for discovering the exact answer, and frequently not even an approximate one, to a practical problem. So, the fundamental principle behind the finite element approach is to use discrete components or pieces whose behaviour is completely known in order to solve a complex problem. In order to calculate and analyse complicated resistance constructions with more specialised qualities, such as biomechanical ones, for which analytical calculation methods are no longer useful, the finite element analysis approach is also evidently necessary [61,62,63].
In the present paper, the finite element analysis of hip implant and femur bone has been carried out in ANSYS® 2021 R2 (Fig. 5). Both were first modelled using SOLIDWORKS® 2022. Then, this 3D model is imported into ANSYS® 2021 R2 environment and simulated. FEA has numerous applications in the field of implants; some are mentioned in Table 1 [64,65,66,67,68,69,70,71,72].
Additive manufacturing in implants
Additive manufacturing is an emerging area in the field of medical prosthesis which is a revolutionary manufacturing process that involves building three-dimensional objects layer by layer from digital models. Unlike traditional subtractive manufacturing methods that involve cutting or shaping materials to create the desired object, additive manufacturing adds material gradually to create the final product. The process typically begins with the creation of a digital model using computer-aided design (CAD) software. This digital model is then sliced into thin cross-sectional layers. During the printing process, these layers are successively deposited, solidified, or fused to build up the final three-dimensional object. Various materials can be used in additive manufacturing, including plastics, metals, ceramics, and even biological materials. Different technologies exist within the realm of additive manufacturing, each with its own set of advantages and applications. Common techniques include fused deposition modelling (FDM), stereolithography (SLA), selective laser sintering (SLS), and electron beam melting (EBM). Additive manufacturing offers several benefits, such as the ability to produce complex and customised geometries, reduce material waste, and facilitate rapid prototyping and on-demand production. It has found applications across various industries, including aerospace, healthcare, automotive, and consumer goods [73]. Additive manufacturing is engineered to create both internal and external prosthesis. Additive manufactured prosthetics offer an economically and genuinely necessary assistance for the amputees. Additive manufacturing is becoming a major technique for the manufacturing of implants. It is a developing technology that creates a real-world 3D item from a 3D digital model. In 2012, a hand prosthesis known as “Robohand”—the first upper limb prosthesis to be 3D printed—was created. Since then, more additive manufactured prosthetic devices have become accessible, and the technology has continued to advance but is still in its early stages in several areas. In fact, more advancement is needed to enhance the comfort, strength, and functionality of additive manufactured prosthetics as well as to increase their anthropomorphism and cosmesis. The most creative and printable prosthesis is the hands. Also, the majority of additive manufactured upper limb prosthetics are made for kids. Their growth, which necessitates a recurring change in prosthesis, is the primary cause of this trend. For many families, having to continually buy new commercial equipment is a burden. Hence, prosthesis serves as a temporary or permanent solution for this population. Furthermore, the abandonment rate can be reduced compared to traditional commercial prostheses since additive manufactured prostheses can be customised with patterns selected by the young receivers and some of the devices are lightweight and simple to activate [74,75,76]. Figure 6a is an additive manufactured hip implant stem, and Fig. 6b is a bone plate both additive manufactured using FDM technology. Table 2 [5, 77,78,79,80,81,82,83,84] explains the additive manufactured implants for biomedical applications Fig. 7 [85, 86].
Experimental methods for testing implants
Experimental validation plays a pivotal role in ensuring the accuracy, reliability, and real-world applicability of finite element analysis (FEA) simulations for implants. While FEA provides a powerful virtual platform to predict and analyse the mechanical behaviour of implants under various conditions, experimental validation serves as a crucial step in bridging the gap between simulation and reality. Experimental testing of prosthesis is a challenging task. Setup for testing the implants is crafted based upon the requirements.
When these orthopaedic implants are mechanically tested, the rigidity of the implant, the number of cycles it can withstand before breaking, how the implant affects the rest of the body around it, and a variety of other requirements may be examined. Regardless of the situation or requirement, it is crucial to understand that an implant’s testing process should always aim to simulate how it would be mechanically loaded in the body during clinical use. The anatomy around the implant, the biomechanics of the implant, the body, and the interaction between the body and implant must all be considered during appropriate testing of orthopaedic implants. At every stage, one should be aware of the implant’s design and its intended use in patients.
Testing implants involves a range of setups to evaluate their performance, durability, and safety. The specific setups can vary depending on the type of implant and the desired characteristics to be assessed. Mechanical testing rig is used in evaluating the mechanical strength, stability, and fatigue resistance of the implant. Instron or similar testing machines are often used to apply controlled forces, torque, or cyclic loading to simulate the physiological conditions the implant may experience [5]. Wear and friction testing apparatus is used in examining the wear resistance and frictional properties of materials used in the implant. Tribometers or pin-on-disk setups simulate the relative motion between implant components to assess wear rates and friction coefficients [87, 88]. Electrochemical corrosion testing is used for evaluating the corrosion resistance of metallic implants in physiological environments. Electrochemical cells are used to subject the implant to corrosive conditions, measuring parameters like corrosion potential and corrosion current [89, 90]. In vitro and in vivo testing chambers are used for assessing biological responses to implants in controlled environments. Cells or tissues are cultured in vitro on the implant surface or implanted into animal models to study factors like tissue integration, host response, and long-term stability [91, 92]. Radiographic testing setup is used for examining the structural integrity and positioning of implants. X-ray or CT imaging is used to visualise the implant within the body, allowing for assessment of its placement, alignment, and any potential issues [93, 94].
These setups collectively provide a comprehensive understanding of an implant’s performance, ensuring that it meets safety and efficacy standards before being introduced for clinical use. The combination of in silico (computational), in vitro (laboratory), and in vivo (animal or human) testing allows for a thorough evaluation of implant behaviour in different contexts. Many of these experimental methods followed in testing the implants by the past researchers (Table 3) [85, 95,96,97,98,99,100].
Future directions and challenges
Exploration of the potential of 4D printing and advanced biofabrication techniques for creating dynamic and self-assembling implants [101]. Investigation on the integration of nanotechnology for developing nanocomposite biomaterials with enhanced mechanical properties and targeted drug delivery capabilities. The introduction of nanoscale materials can improve the performance and longevity of implants [102, 103]. Usage of patient-specific data, such as medical imaging and genetic information, to create custom-designed implants that fit patients’ unique anatomical and biomechanical characteristics [104, 105]. The development of smart implants equipped with sensors and actuators that can monitor their own performance and respond to physiological changes [106, 107]. The advancement of biodegradable materials that can provide temporary mechanical support or drug delivery and then naturally degrade, reducing the need for additional surgeries for implant removal [108, 109]. Biomimetic design principles that mimic natural structures and mechanisms to create highly efficient and mechanically robust implants [110, 111]. The development of non-invasive or minimally invasive techniques for assessing implant performance in vivo, allowing for real-time monitoring and early detection of potential issues [112, 113].
The challenge of implants is ensuring the long-term durability and reliability of implants, especially in high-stress environments. Exploring the strategies for improving the mechanical stability of implants over their lifespan [114, 115]. Ongoing challenges related to biocompatibility, including the immune response to implants and potential allergic reactions. Investigating the novel surface modifications and coatings to enhance biocompatibility [116, 117]. Addressing regulatory challenges related to the approval and standardisation of new biomaterials and implant technologies [118, 119]. Examining balance between developing advanced biomaterials and implants and ensuring cost-effectiveness for widespread adoption. Exploring strategies to make cutting-edge technologies accessible and affordable [120, 121]. The challenges related to the integration of implants with the host tissue, such as minimising the risk of infection, promoting tissue regeneration, and preventing implant rejection [122]. Considering the ethical implications of implantable technologies, such as issues related to privacy, informed consent, and equitable access to advanced treatments [123, 124]. Examining the environmental impact of biomaterials, implant production, and implementing sustainable practices and materials to reduce the ecological footprint of the field [125, 126].
Conclusion
This review paper enumerates the internal prosthetic implant in the view of materials, applications, additive manufacturing, numerical and experimental modelling. The researches of biomaterials in synthesis of prosthesis are rapidly increasing day by day in the scope of optimising the best devices as it is being reinforced in the human body. On the other hand, composite material plays a vital role in prosthetic implants due to their excellent characteristic features. Finite element simulation gives the freedom of choosing the best lightweight materials, good mechanical behaviour and desired directional properties of the implants before being applied inside the human body. An attempt has been made in this paper for modelling hip implant, shoulder implant, bone plate, dental implant, contact lens, artificial stent models using SOLIDWORKS® 2022. A finite element model of a hip implant and femur bone is also explored in this paper using ANSYS® 2021 R2. 3D printing has been a remarkable technology in the prosthetic field as it has reduced the time and cost in the manufacturing implants. 3D printing of a hip implant stem has been fabricated in this paper. Different experimental setups are also being used for validating the performance of prosthesis. Various advancements are being made in the prosthetic field for the betterment of the properties of the biomaterials based on the desired applications. Thus, biomaterials are the future for the medical and engineering researchers for the design and development of optimal, sustainable, efficient and lightweight implants.
References
Davis R, Singh A, Jackson MJ, et al. A comprehensive review on metallic implant biomaterials and their subtractive manufacturing. Int J Adv Manuf Technol. 2022;120(3):1473–530. https://doi.org/10.1007/S00170-022-08770-8.
Il IG. Biomaterials in orthopaedics: the past and future with immune modulation. Biomater Res. 2020;24(1):1–4. https://doi.org/10.1186/S40824-020-0185-7/METRICS.
Naleway SE, Thomas V, Restrepo D, Schniepp HC. Advanced manufacturing for biomaterials and biological materials, part II. JOM. 2020;72(4):1432–4. https://doi.org/10.1007/S11837-020-04060-4/METRICS.
Rokaya D. Advances in biomaterials for clinical applications. J Chitwan Med Coll. 2022;11(4):1–3. https://doi.org/10.54530/JCMC.593.
Wegner N, Klein M, Scholz R, Kotzem D, Macias Barrientos M, Walther F. Mechanical in vitro fatigue testing of implant materials and components using advanced characterization techniques. J Biomed Mater Res Part B Appl Biomater. 2022;110(4):898–909. https://doi.org/10.1002/JBM.B.34970.
Barros SE, Vanz V, Chiqueto K, Janson G, Ferreira E. Mechanical strength of stainless steel and titanium alloy mini-implants with different diameters: an experimental laboratory study. Prog Orthod. 2021;22(1):1–9. https://doi.org/10.1186/S40510-021-00352-W/TABLES/4.
Lewin S, Åberg J, Neuhaus D, et al. Mechanical behaviour of composite calcium phosphate–titanium cranial implants: effects of loading rate and design. J Mech Behav Biomed Mater. 2020:104. https://doi.org/10.1016/j.jmbbm.2020.103701.
Vakrčka P, Jíra A, Hájková P. Mechanical testing and numerical modelling of porous structures improving oseintegration of implants. Acta Polytech CTU Proc. 2020;26:126–32. https://doi.org/10.14311/APP.2020.26.0126.
Ortega-Martínez J, Delgado LM, Ortiz-Hernández M, et al. In vitro assessment of PEEK and titanium implant abutments: screw loosening and microleakage evaluations under dynamic mechanical testing. J Prosthet Dent. 2022;127(3):470–6. https://doi.org/10.1016/j.prosdent.2020.09.033.
Marin E, Boschetto F, Pezzotti G. Biomaterials and biocompatibility: an historical overview. J Biomed Mater Res - Part A. 2020;108(8):1617–33. https://doi.org/10.1002/JBM.A.36930.
Eftekhar Ashtiani R, Alam M, Tavakolizadeh S, Abbasi K. The role of biomaterials and biocompatible materials in implant-supported dental prosthesis. Evidence-based Complement Altern Med. 2021;2021. doi:https://doi.org/10.1155/2021/3349433
Thanigaivel S, Priya AK, Balakrishnan D, Dutta K, Rajendran S, Soto-Moscoso M. Insight on recent development in metallic biomaterials: strategies involving synthesis, types and surface modification for advanced therapeutic and biomedical applications. Biochem Eng J. 2022;187:108522. https://doi.org/10.1016/J.BEJ.2022.108522.
Lah NAC, Hussin MH. Titanium and titanium based alloys as metallic biomaterials in medical applications - spine implant case study. Pertanika J Sci Technol. 2019;27(1):459–72.
Annur D, Rokhmanto F, Thaha YN, et al. Processing and characterization of porous titanium for orthopedic implant prepared by argon-atmospheric sintering and arc plasma sintering. Mater Res. 2021;24(6):e20210122. https://doi.org/10.1590/1980-5373-MR-2021-0122.
Capellato P, Camargo SEA, Silva G, et al. Coated surface on Ti-30Ta alloy for biomedical application: mechanical and in-vitro characterization. Mater Res. 2020;23(6):e20200305. https://doi.org/10.1590/1980-5373-MR-2020-0305.
Kim T, See CW, Li X, Zhu D. Orthopedic implants and devices for bone fractures and defects: past, present and perspective. Eng Regen. 2020;1:6–18. https://doi.org/10.1016/J.ENGREG.2020.05.003.
Wang N, Fuh JYH, Dheen ST, Senthil KA. Functions and applications of metallic and metallic oxide nanoparticles in orthopedic implants and scaffolds. J Biomed Mater Res Part B Appl Biomater. 2021;109(2):160–79. https://doi.org/10.1002/JBM.B.34688.
Kelmers E, Szuba A, King SW, et al. Smart knee implants: an overview of current technologies and future possibilities. Indian J Orthop. 2022;28:1–8. https://doi.org/10.1007/S43465-022-00810-5/FIGURES/1.
Aggarwal AK, Baburaj V. Managing bone defects in primary total knee arthroplasty: options and current trends. Musculoskelet Surg. 2021;105(1):31–8. https://doi.org/10.1007/S12306-020-00683-7.
Olmos AAR, Fertuzinhos A, Campos TD, et al. Osteosynthesis metal plate system for bone fixation using bicortical screws: numerical–experimental characterization. Biology (Basel). 2022;11(6) https://doi.org/10.3390/BIOLOGY11060940.
Ma Z, Liu B, Li S, et al. A novel biomimetic trabecular bone metal plate for bone repair and osseointegration. Regen Biomater. 2023:10. https://doi.org/10.1093/RB/RBAD003.
Müller-Heupt LK, Schiegnitz E, Kaya S, Jacobi-Gresser E, Kämmerer PW, Al-Nawas B. Diagnostic tests for titanium hypersensitivity in implant dentistry: a systematic review of the literature. Int J Implant Dent. 2022;8(1):1–11. https://doi.org/10.1186/S40729-022-00428-0/FIGURES/4.
Ruiz Henao PA, Caneiro Queija L, Mareque S, Tasende Pereira A, Liñares González A, Blanco CJ. Titanium vs ceramic single dental implants in the anterior maxilla: a 12-month randomized clinical trial. Clin Oral Implants Res. 2021;32(8):951–61. https://doi.org/10.1111/CLR.13788.
Major R, Gawlikowski M, Sanak M, Lackner JM, Kapis A. Design, manufacturing technology and in-vitro evaluation of original, polyurethane, petal valves for application in pulsating ventricular assist devices. Polymers. 2020;12(12):2986. https://doi.org/10.3390/POLYM12122986.
Rafikova G, Piatnitskaia S, Shapovalova E, et al. Interaction of ceramic implant materials with immune system. Int J Mol Sci. 2023;24(4) https://doi.org/10.3390/IJMS24044200.
Qu Y, Liu L. Zirconia materials for dental implants: a literature review. Front Dent Med. 2021;2:57. https://doi.org/10.3389/FDMED.2021.687983.
da Hora Sales PH, Barros AW, de Oliveira-Neto OB, de Lima FJ, Carvalho AD, Leao JC. Do zirconia dental implants present better clinical results than titanium dental implants? A systematic review and meta-analysis. J Stomatol Oral Maxillofac Surg. 2023;124(1):101324. https://doi.org/10.1016/J.JORMAS.2022.10.023.
Borgonovo AE, Ferrario S, Maiorana C, Vavassori V, Censi R, Re D. A clinical and radiographic evaluation of zirconia dental implants: 10-year follow-up. Int J Dent. 2021;2021 https://doi.org/10.1155/2021/7534607.
Sadeghzade S, Liu J, Wang H, et al. Recent advances on bioactive baghdadite ceramic for bone tissue engineering applications: 20 years of research and innovation (a review). Mater Today Bio. 2022;17:100473. https://doi.org/10.1016/J.MTBIO.2022.100473.
Ene R, Nica M, Ene D, Cursaru A, Cirstoiu C. Review of calcium-sulphate-based ceramics and synthetic bone substitutes used for antibiotic delivery in PJI and osteomyelitis treatment. EFORT Open Rev. 2021;6(5):297–304. https://doi.org/10.1302/2058-5241.6.200083.
Al-Nahlawi T, Ala Rachi M, Abu HA. Endodontic perforation closure by five mineral oxides silicate-based cement with/without collagen sponge matrix. Int J Dent. 2021;2021 https://doi.org/10.1155/2021/4683689.
Stukan M, Buderath P, Szulczyński B, Gębicki J, Kimmig R. Accuracy of ultrasonography and magnetic resonance imaging for preoperative staging of cervical cancer—analysis of patients from the prospective study on total mesometrial resection. Diagnostics. 2021;11(10):1749. https://doi.org/10.3390/DIAGNOSTICS11101749/S1.
Spałek J, Ociepa P, Deptuła P, et al. Biocompatible materials in otorhinolaryngology and their antibacterial properties. Int J Mol Sci. 2022;23(5):2575. https://doi.org/10.3390/IJMS23052575.
Al-Dujaili AMA, Aswad AM, Oribi IM. Preparation and characterization of hydroxyapatite and optimizing its properties using regression model. J Phys Conf Ser. 2021;1973(1) https://doi.org/10.1088/1742-6596/1973/1/012141.
Kumar P, Dehiya BS, Sindhu A. Bioceramics for hard tissue engineering applications: a review. Int J Appl Eng Res. 2018;13(5):2744–52. Accessed April 18, 2023. http://www.ripublication.com
Huang J, Best S. Ceramic biomaterials for tissue engineering. Tissue Eng Using Ceram Polym Second Ed. 2014;1:3–34. https://doi.org/10.1533/9780857097163.1.3.
Kovylin RS, Aleynik DY, Fedushkin IL. Modern porous polymer implants: synthesis, properties, and application. Polym Sci Ser C. 2021;63(1):29–46. https://doi.org/10.1134/S1811238221010033.
Panchal SS, Vasava DV. Biodegradable polymeric materials: synthetic approach. ACS Omega. 2020;5(9):4370–9. https://doi.org/10.1021/ACSOMEGA.9B04422/ASSET/IMAGES/LARGE/AO9B04422_0002.JPEG.
Karayilan M, Clamen L, Becker ML. Polymeric materials for eye surface and intraocular applications. Biomacromolecules. 2021;22(2):223–61. https://doi.org/10.1021/ACS.BIOMAC.0C01525/ASSET/IMAGES/MEDIUM/BM0C01525_0038.GIF.
Tran NPD, Yang MC. Synthesis and characterization of silicone contact lenses based on TRIS-DMA-NVP-HEMA hydrogels. Polymers (Basel). 2019;11(6) https://doi.org/10.3390/POLYM11060944.
Kappel RM, Pruijn JM. Explantation of silicone breast implants ameliorates gel bleed related health complaints in women with breast implant illness. Clin Med Rev Case Rep. 2020;2020:301. https://doi.org/10.23937/2378-3656/1410301.
Alcocer P. Presentation from FELAQ. Cir Plast Ibero-Latinoamericana. 2020;46:S5–6. https://doi.org/10.4321/S0376-78922020000200003.
Sidle DM, Stolovitzky P, O’Malley EM, Ow RA, Nachlas NE, Silvers S. Bioabsorbable implant for treatment of nasal valve collapse with or without concomitant procedures. Facial Plast Surg. 2021;37(5):673–80. https://doi.org/10.1055/s-0041-1726464.
Villanueva K, Martin D, Martinkovich S, Blomain EW. Treatment of a chronically infected nasal silicone prosthesis with continuous antibiotic irrigation and gentamicin-impregnated polymethylmethacrylate beads. JPRAS Open. 2018;15:18–24. https://doi.org/10.1016/J.JPRA.2017.10.001.
Dhanasingh A. Design of a cochlear implant electrode. ENT Updat. 2022;12(2):97–109. https://doi.org/10.5152/entupdates.2022.22213.
Blume C, Kraus X, Heene S, et al. Vascular implants – new aspects for in situ tissue engineering. Eng Life Sci. 2022;22(3-4, 344):–360. https://doi.org/10.1002/ELSC.202100100.
Leal BBJ, Wakabayashi N, Oyama K, Kamiya H, Braghirolli DI, Pranke P. Vascular tissue engineering: polymers and methodologies for small caliber vascular grafts. Front Cardiovasc Med. 2020;7:592361. https://doi.org/10.3389/FCVM.2020.592361.
Kennedy SM, Vasanthanathan A, RB JR, Amudhan K. Advancements and prospects of polymer-based hybrid composites for bone plate applications. Polym Technol Mater. 2023;63(1):1–20. https://doi.org/10.1080/25740881.2023.2274564.
Egbo MK. A fundamental review on composite materials and some of their applications in biomedical engineering. J King Saud Univ - Eng Sci. 2021;33(8):557–68. https://doi.org/10.1016/J.JKSUES.2020.07.007.
Guo Z, Poot AA, Grijpma DW. Advanced polymer-based composites and structures for biomedical applications. Eur Polym J. 2021;149:110388. https://doi.org/10.1016/j.eurpolymj.2021.110388.
Yadav R, Meena A, Patnaik A. Biomaterials for dental composite applications: a comprehensive review of physical, chemical, mechanical, thermal, tribological, and biological properties. Polym Adv Technol. 2022;33(6):1762–81. https://doi.org/10.1002/PAT.5648.
Kavyashree S, Savitha PN, DMD SP. An overview of carbon fiber reinforced composites in prosthetic restoration. Int J Sci Res. 2022;11(6):6607–16. https://doi.org/10.21275/SR22608122651.
Eldeeb AE, Salah S, Elkasabgy NA. Biomaterials for tissue engineering applications and current updates in the field: a comprehensive review. AAPS PharmSciTech. 2022;23:3. https://doi.org/10.1208/s12249-022-02419-1.
Petre DG, Leeuwenburgh SCG. The use of fibers in bone tissue engineering. https://home.liebertpub.com/teb. 2022;28(1):141–59. https://doi.org/10.1089/TEN.TEB.2020.0252.
Maharaj PSRS, Vasanthanathan A, Ebenezer FBD, Giriharan R, Athithiyan M. In situ bio printing of carbon fiber reinforced PEEK hip implant stem. AIP Conf Proc. 2022;2653(1):030008. https://doi.org/10.1063/5.0110578.
Diabb Zavala JM, Leija Gutiérrez HM, Segura-Cárdenas E, et al. Manufacture and mechanical properties of knee implants using SWCNTs/UHMWPE composites. J Mech Behav Biomed Mater. 2021;120:104554. https://doi.org/10.1016/J.JMBBM.2021.104554.
Kabiri A, Liaghat G, Alavi F, et al. Glass fiber/polypropylene composites with potential of bone fracture fixation plates: manufacturing process and mechanical characterization. J Compos Mater. 2020;54(30):4903–19. https://doi.org/10.1177/0021998320940367.
Hutchinson DJ, Granskog V, von Kieseritzky J, et al. Highly customizable bone fracture fixation through the marriage of composites and screws. Adv Funct Mater. 2021;31(41):2105187. https://doi.org/10.1002/ADFM.202105187.
Shiroud Heidari B, Ruan R, Vahabli E, et al. Natural, synthetic and commercially-available biopolymers used to regenerate tendons and ligaments. Bioact Mater. 2023;19:179–97. https://doi.org/10.1016/J.BIOACTMAT.2022.04.003.
Pan L, Wang F, Cheng Y, et al. A supertough electro-tendon based on spider silk composites. Nat Commun. 2020;11(1):1–9. https://doi.org/10.1038/s41467-020-14988-5.
Leblebicioğlu Kurtuluş I, Kilic K, Bal B, Kilavuz A. Finite element analysis of the stress distribution associated with different implant designs for different bone densities. J Prosthodont. 2022;31(7):614–22. https://doi.org/10.1111/JOPR.13539.
Zupancic Cepic L, Frank M, Reisinger A, Pahr D, Zechner W, Schedle A. Biomechanical finite element analysis of short-implant-supported, 3-unit, fixed CAD/CAM prostheses in the posterior mandible. Int. J Implant Dent. 2022;8(1):1–13. https://doi.org/10.1186/S40729-022-00404-8.
Ausiello P, Tribst JPM, Ventre M, et al. The role of cortical zone level and prosthetic platform angle in dental implant mechanical response: a 3D finite element analysis. Dent Mater. 2021;37(11):1688–97. https://doi.org/10.1016/J.DENTAL.2021.08.022.
Colic K, Sedmak A, Grbovic A, Tatic U, Sedmak S, Djordjevic B. Finite element modeling of hip implant static loading. Procedia Eng. 2016;149(June):257–62. https://doi.org/10.1016/j.proeng.2016.06.664.
Shashishekar DC, Sanjay SJ. Finite element modeling and analysis of prosthetic knee joint- a review. Ijireeice. 2021;9(10):264–9. https://doi.org/10.17148/ijireeice.2021.91012.
Denard PJ, Lederman E, Parsons BO, Romeo AA. Finite element analysis of glenoid-sided lateralization in reverse shoulder arthroplasty. J Orthop Res. 2017;35(7):1548–55. https://doi.org/10.1002/jor.23394.
Maharaj PSRS, Maheswaran R, Vasanthanathan A. Numerical analysis of fractured femur bone with prosthetic bone plates. Procedia Eng. 2013;64:1242–51. https://doi.org/10.1016/j.proeng.2013.09.204.
Rubo JH, Capello Souza EA. Finite-element analysis of stress on dental implant prosthesis. Clin Implant Dent Relat Res. 2010;12(2):105–13. https://doi.org/10.1111/j.1708-8208.2008.00142.x.
Myung Y, Lee JG, Cho M, Heo CY. Finite element analysis of long-term changes of the breast after augmentation mammoplasty: implications for implant design. Arch Plast Surg. 2019;46(4):386–9. https://doi.org/10.5999/aps.2019.00346.
Batalu ND, Semenescu A, Mates IM, Negoita OD, Purcarea VL, Badica P. Computer assisted design and finite element analysis of contact lenses. Rom J Ophthalmol. 2016;60(3):132–7.
Huang HL, Fuh LJ, Ko CC, Hsu JT, Chen CC. Biomechanical effects of a maxillary implant in the augmented sinus: a three-dimensional finite element analysis. Int J Oral Maxillofac Implants. 2009;24(3):455–62. http://www.ncbi.nlm.nih.gov/pubmed/19587867. Accessed 10-01-2024
Xu J, Yang J, Sohrabi S, Zhou Y, Liu Y. Finite element analysis of the implantation process of overlapping stents. J Med Devices, Trans ASME. 2017;11(2):1–9. https://doi.org/10.1115/1.4036391.
Kennedy SM, Robert RBJ, Seenikannan P, Arunachalam V, Amudhan K. An investigation on mechanical properties of 3D pen fused zones for additive manufactured parts. Eng Solid Mech. 2023;11(3):263–70. https://doi.org/10.5267/j.esm.2023.3.003.
Al-Hamdan K. Accuracy of implant placement utilizing 3D printed and thermoplastic surgical guides: a CBCT-analysis. Biosci Biotechnol Res Commun. 2020;13(3):1210–3. https://doi.org/10.21786/bbrc/13.3/34.
Attarilar S, Ebrahimi M, Djavanroodi F, Fu Y, Wang L, Yang J. 3D Printing technologies in metallic implants: a thematic review on the techniques and procedures. Int J Bioprinting. 2021;7(1):21–46. https://doi.org/10.18063/ijb.v7i1.306.
Singh S, Urooj S, Batra N, Kalathil S. Healthcare applications of 3D printing in human implants: a review. In: 2020 IEEE 17th India Counc Int Conf INDICON 2020. Published online December, vol. 10. p. 2020. https://doi.org/10.1109/INDICON49873.2020.9342316.
Reddy RJ, Reddy VV, Student MT, Pradesh A, Pradesh A, Pradesh A. Hip joinT CT scan data to fabrication process using 3D printing machine for demonstration of medical purpose. International Journal of Technical Innovation in Modern Engineering & Science (IJTIMES). 2018;4(12):208–15.
Harish S, Devadath VR. Additive manufacturing and analysis of tibial insert in total knee replacement implant. Int Res J Eng Technol. 2015:2395–56. https://irjet.net/archives/V2/i4/Irjet-v2i4110.pdf. Accessed 10-01-2024
Al Najjar M, Mehta SS, Monga P. Three dimensional scapular prints for evaluating glenoid morphology: an exploratory study. J Clin Orthop Trauma. 2018;9(3):230–5. https://doi.org/10.1016/j.jcot.2018.06.001.
Tappa K, Jammalamadaka U, Weisman JA, et al. 3D printing custom bioactive and absorbable surgical screws, pins, and bone plates for localized drug delivery. J Funct Biomater. 2019;10(2) https://doi.org/10.3390/jfb10020017.
Chung YJ, Park JM, Kim TH, Ahn JS, Cha HS, Lee JH. 3D printing of resin material for denture artificial teeth: chipping and indirect tensile fracture resistance. Materials (Basel). 2018;11(10):1–13. https://doi.org/10.3390/ma11101798.
Assefa BG, Saastamoinen T, Pekkarinen M, et al. Realizing freeform lenses using an optics 3D-printer for industrial based tailored irradiance distribution. OSA Contin. 2019;2(3):690. https://doi.org/10.1364/osac.2.000690.
Yi HG, Choi YJ, Jung JW, et al. Three-dimensional printing of a patient-specific engineered nasal cartilage for augmentative rhinoplasty. J Tissue Eng. 2019:10. https://doi.org/10.1177/2041731418824797.
Wu Y, Fuh JYH, Wong YS, Sun J. Fabrication of 3D scaffolds via E-jet printing for tendon tissue repair. Int Manuf Sci Eng Conf. 2016;2015:3–8. https://doi.org/10.1115/msec2015-9367.
Xu H, Zhang D, Chen K, Zhang X. Micromotion analysis of the head-neck interface of a PEEK artificial hip prosthesis Micromotion analysis of the head-neck interface of a PEEK arti ficial hip prosthesis. Mater Res Express. 2019:6.
Vasanthanathan A, Kennedy SM. Bio-printing of femur model: a bone substitute for biomedical research. Mater Tehnol. 2023;57(3):283–9. https://doi.org/10.17222/mit.2023.831.
Korkmaz ME, Gupta MK, Singh G, et al. Machine learning models for online detection of wear and friction behaviour of biomedical graded stainless steel 316L under lubricating conditions. Int J Adv Manuf Technol. 2023;128(5-6):2671–88. https://doi.org/10.1007/S00170-023-12108-3/FIGURES/15.
Hussain O, Saleem SS, Ahmad B. Friction and wear performance evaluation of UHMWPE using Taguchi based grey approach: a study on the influence of load and bio-serum lubrication. Mater Chem Phys. 2020;239:121918. https://doi.org/10.1016/J.MATCHEMPHYS.2019.121918.
Silva A, Plaine AH. Electrochemical corrosion study of biomaterials: a bibliometric study based on co-word analysis. Results Eng. 2023;20:101489. https://doi.org/10.1016/J.RINENG.2023.101489.
Welles TS, Ahn J. Driving electrochemical corrosion of implanted CoCrMo metal via oscillatory electric fields without mechanical wear. Sci Rep. 2021;11(1):1-16. doi:https://doi.org/10.1038/s41598-021-01810-5
Chen IC, Su CY, Tu JJ, Kao DW, Fang HW. In vitro studies of factors affecting debridement of dental implants by tricalcium phosphate powder abrasive treatment. Sci Rep. 2023;13(1):1–10. https://doi.org/10.1038/s41598-023-35053-3.
Chen CS, Chang JH, Srimaneepong V, et al. Improving the in vitro cell differentiation and in vivo osseointegration of titanium dental implant through oxygen plasma immersion ion implantation treatment. Surf Coatings Technol. 2020;399:126125. https://doi.org/10.1016/J.SURFCOAT.2020.126125.
Trochim B, Borowska M, Szarmach J. Analysis of X-rays in bone remodelling around active and replace dental implants. Signal, Image Video Process. 2022;16(1):111–8. https://doi.org/10.1007/S11760-021-01971-W/TABLES/9.
Khosravifard N, Saberi BV, Khosravifard A, Zakerjafari H, Vafaei R, Ghaffari ME. A diagnostic accuracy study on an innovative auto-edge detection technique for identifying simulated implant fractures on radiographic images. Sci Rep. 2022;12(1):1–9. https://doi.org/10.1038/s41598-022-24266-7.
Brockett CL, Carbone S, Fisher J, Jennings LM. Influence of conformity on the wear of total knee replacement: an experimental study. Proc Inst Mech Eng Part H J Eng Med. 2018;232(2):127–34. https://doi.org/10.1177/0954411917746433.
Anglin C, Wyss UP, Pichora DR. Mechanical testing of shoulder prostheses and recommendations for glenoid design. J Shoulder Elb Surg. 2000;9(4):323–31. https://doi.org/10.1067/mse.2000.105451.
Senthil Maharaj PSR, Vasanthanathan A. An insight into the mechanical and tribological behavior of carbon-flax reinforced bioepoxy hybrid composite bone plates for orthopedic applications. Polym Polym Compos. 2023:31. https://doi.org/10.1177/09673911231178444/ASSET/IMAGES/LARGE/10.1177_09673911231178444-FIG8.JPEG.
Chakladar ND, Harper LT, Parsons AJ. Optimisation of composite bone plates for ulnar transverse fractures Optimisation of composite bone plates for ulnar transverse fractures. J Mech Behav Biomed Mater. 2016;57(February):334–46. https://doi.org/10.1016/j.jmbbm.2016.01.029.
Mura MD, Dini G, Lanzetta M, Rossi A. An experimental analysis of laser machining for dental implants. Procedia CIRP. 2018;67:356–61. https://doi.org/10.1016/J.PROCIR.2017.12.226.
Ramião NG, Martins PS, Barroso ML, Santos DC, Fernandes AA. An experimental analysis of shell failure in breast implants. J Mech Behav Biomed Mater. 2017;72(February):22–8. https://doi.org/10.1016/j.jmbbm.2017.04.005.
Kennedy SM, Amudhan K, Jeen Robert RB, Vasanthanathan A, Pandian VM, A. Experimental and finite element analysis on the effect of pores on bio-printed polycaprolactone bone scaffolds. Bioprinting. 2023;34(August):e00301. https://doi.org/10.1016/j.bprint.2023.e00301.
Gulati K, Chopra D, Kocak-Oztug NA, Verron E. Fit and forget: the future of dental implant therapy via nanotechnology. Adv Drug Deliv Rev. 2023;199:114900. https://doi.org/10.1016/J.ADDR.2023.114900.
Hossain N, Islam MA, Chowdhury MA, Alam A. Advances of nanoparticles employment in dental implant applications. Appl Surf Sci Adv. 2022;12:100341. https://doi.org/10.1016/J.APSADV.2022.100341.
Paxton NC, Tetsworth K, Woodruff MA. Personalization for surgical implants. Digit Hum Model Med Digit Twin. 2023;1:849–74. https://doi.org/10.1016/B978-0-12-823913-1.00019-1.
Kim MK, Ham MJ, Kim WR, et al. Investigating the accuracy of mandibulectomy and reconstructive surgery using 3D customized implants and surgical guides in a rabbit model. Maxillofac Plast Reconstr Surg. 2023;45(1):1–17. https://doi.org/10.1186/S40902-023-00375-9/TABLES/5.
Baumann AP, O’Neill C, Owens MC, et al. FDA public workshop: orthopaedic sensing, measuring, and advanced reporting technology (SMART) devices. J Orthop Res. 2021;39(1):22–9. https://doi.org/10.1002/JOR.24833.
Gaobotse G, Mbunge E, Batani J, Muchemwa B. The future of smart implants towards personalized and pervasive healthcare in Sub-Saharan Africa: opportunities, barriers and policy recommendations. Sensors Int. 2022;3:100173. https://doi.org/10.1016/J.SINTL.2022.100173.
Drelich JW, Sikora-Jasinska M, Mostaed E, et al. Biodegradable materials for medical applications II. JOM. 2020;72(5):1830–2. https://doi.org/10.1007/S11837-020-04129-0/METRICS.
Chavda VP, Jogi G, Paiva-Santos AC, Kaushik A. Biodegradable and removable implants for controlled drug delivery and release application. Expert Opin Drug Deliv. 2022;19(10):1177–81. https://doi.org/10.1080/17425247.2022.2110065.
Li Y, Hu Y, Chen H, et al. A novel conceptual design of a biomimetic oral implant and its biomechanical effect on the repairment of a large mandibular defect. Med Nov Technol Devices. 2022;15:100147. https://doi.org/10.1016/J.MEDNTD.2022.100147.
Ustunel S, Prévôt ME, Clements RJ, Hegmann E. Cradle-to-cradle: designing biomaterials to fit as truly biomimetic cell scaffolds– a review. Liq Cryst Today. 2020;29(3):40–52. https://doi.org/10.1080/1358314X.2020.1855919.
Adrianto MF, Annuryanti F, Wilson CG, Sheshala R, Thakur RRS. In vitro dissolution testing models of ocular implants for posterior segment drug delivery. Drug Deliv Transl Res. 2022;12(6):1355–75. https://doi.org/10.1007/S13346-021-01043-Z/FIGURES/7.
Sekar P, S N, Desai V. Recent progress in in vivo studies and clinical applications of magnesium based biodegradable implants – a review. J Magnes Alloy. 2021;9(4):1147–63. https://doi.org/10.1016/J.JMA.2020.11.001.
French D, Ofec R, Levin L. Long term clinical performance of 10 871 dental implants with up to 22 years of follow-up: a cohort study in 4247 patients. Clin Implant Dent Relat Res. 2021;23(3):289–97. https://doi.org/10.1111/CID.12994.
Kohal RJ, Dennison DK. Clinical longevity of zirconia implants with the focus on biomechanical and biological outcome. Curr Oral Heal Reports. 2020;7(4):344–51. https://doi.org/10.1007/S40496-020-00289-9/TABLES/1.
Bandyopadhyay A, Mitra I, Goodman SB, Kumar M, Bose S. Improving biocompatibility for next generation of metallic implants. Prog Mater Sci. 2023;133:101053. https://doi.org/10.1016/J.PMATSCI.2022.101053.
Li H, Yao Z, Zhang J, et al. The progress on physicochemical properties and biocompatibility of tantalum-based metal bone implants. SN Appl Sci. 2020;2(4):1–14. https://doi.org/10.1007/S42452-020-2480-2/FIGURES/5.
Roohani I, Newsom E, Zreiqat H. High-resolution vat-photopolymerization of personalized bioceramic implants: new advances, regulatory hurdles, and key recommendations. Int Mater Rev. 2023; https://doi.org/10.1080/09506608.2023.2194744.
Favre P, Maquer G, Henderson A, Hertig D, Ciric D, Bischoff JE. In silico clinical trials in the orthopedic device industry: from fantasy to reality? Ann Biomed Eng. 2021;49(12):3213–26. https://doi.org/10.1007/S10439-021-02787-Y/FIGURES/6.
Kim CS, Lunde B, MacIsaac L, et al. Provision of contraceptive implants in school-based health centers: a cost-effectiveness analysis. Contraception. 2021;103(2):107–12. https://doi.org/10.1016/J.CONTRACEPTION.2020.11.009.
Matthys C, De Vijlder W, Besseler J, Glibert M, De Bruyn H. Cost-effectiveness analysis of two attachment systems for mandibular overdenture. Clin Oral Implants Res. 2020;31(7):615–24. https://doi.org/10.1111/CLR.13599.
Amin Yavari S, Castenmiller SM, van Strijp JA, et al. Combating implant infections: shifting focus from bacteria to host. Adv Mater. 2020;32(43):2002962. https://doi.org/10.1002/ADMA.202002962.
Rokhshad R, Ducret M, Chaurasia A, et al. Ethical considerations on artificial intelligence in dentistry: a framework and checklist. J Dent. 2023;135:104593. https://doi.org/10.1016/J.JDENT.2023.104593.
de Kanter AFJ, Jongsma KR, Bouten CVC, Bredenoord AL. How smart are smart materials? A conceptual and ethical analysis of smart lifelike materials for the design of regenerative valve implants. Sci Eng Ethics. 2023;29(5):1–18. https://doi.org/10.1007/S11948-023-00453-1/TABLES/3.
Agbeboh NI, Oladele IO, Daramola OO, Adediran AA, Olasukanmi OO, Tanimola MO. Environmentally sustainable processes for the synthesis of hydroxyapatite. Heliyon. 2020;6(4):e03765. https://doi.org/10.1016/J.HELIYON.2020.E03765.
Tomasi C, Derks J. Etiology, occurrence, and consequences of implant loss. Periodontol. 2000;88(1):13–35. https://doi.org/10.1111/PRD.12408.
Acknowledgements
The authors would like to thank and acknowledge the immense support and the facilities provided by Department of Mechanical Engineering, AAA College of Engineering and Technology and Mepco Schlenk Engineering College, Sivakasi, Tamil Nadu, India. The authors would also like to thank Dr. M. Sekar, Principal, AAA College of Engineering and Technology, Dr. P. Nagaraj, Senior Professor & Head, Department of Mechanical Engineering of Mepco Schlenk Engineering College, Dr. P. Seenikannan, Dean and Professor & Head, Department of Mechanical Engineering, AAA College of Engineering and Technology, and for their continuous support in writing this review paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kennedy, S.M., Vasanthanathan, A., Jeen Robert, R. et al. Impact of mechanical engineering innovations in biomedical advancements. In vitro models 3, 5–18 (2024). https://doi.org/10.1007/s44164-024-00065-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s44164-024-00065-4