Abstract
Background
Meniscal injuries frequently require surgical intervention to restore knee joint function and stability. Intraoperative platelet-rich plasma (PRP) injection has emerged as a potential adjunctive therapy to enhance tissue healing post-meniscal repair. This systematic review and meta-analysis aimed to evaluate the efficacy of PRP in terms of pain relief, functional recovery, and overall success rates in patients undergoing meniscal repair procedures.
Methods
A comprehensive search strategy was employed to identify relevant studies across Scopus, PubMed, Embase, and the Cochrane Library databases. The inclusion criteria encompassed human studies, including randomized controlled trials (RCTs), cohorts, and case–control studies, focusing on intraoperative platelet-rich plasma (PRP) use post-meniscal repair and reporting outcomes related to pain, functionality, and cure rates. Exclusion criteria comprised animal studies, non-English publications, studies lacking relevant outcome measures, and those with insufficient data. Two reviewers independently screened titles and abstracts, resolving disagreements through consensus or consultation with a third reviewer, followed by a full-text assessment for potentially eligible studies. Data extraction was conducted independently by two reviewers using a standardized form. The reliability of observational studies was evaluated using the Newcastle–Ottawa Scale. Subgroup analyses and pooled effect estimates for main outcomes were computed using RevMan 5.3, a meta-analysis tool.
Results
The demographic analysis revealed that the PRP group had an average age of 41.39 years, while the control group had an average age of 42.1 years. In terms of gender distribution, the PRP group consisted of 61 men and 29 women, while the control group had 62 men and 34 women. Pain ratings showed a preference for PRP with a mean difference of 4.83 (p = 0.13). However, there was no significant difference in Lysholm scores (mean difference: − 0.44, p = 0.91) or IKDC scores (mean difference: 2.80, p = 0.14) between the PRP and control groups. Similarly, ROM measures did not show a statistically significant difference, with a mean difference of 2.80 (p = 0.18). Additionally, there was no significant distinction in failure rates between the PRP and control groups, as indicated by a weighted mean difference of 0.71 (p = 0.52). These findings suggest that while PRP may offer some benefits in pain relief, its impact on functional recovery, range of motion, and failure rates following meniscal repair procedures is inconclusive.
Conclusion
The current evidence regarding the effect of intraoperative platelet-rich plasma (PRP) injection on patients undergoing meniscal repair remains inconclusive. While some studies suggest potential benefits in terms of pain relief and functional recovery, others show no significant differences compared to control groups. The impact of PRP therapy on overall success rates, including rates of re-tear and revision surgery, is also uncertain. Further well-designed randomized controlled trials with larger sample sizes are needed to provide more robust evidence and guide clinical practice in orthopedic surgery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Meniscal injuries are common in orthopedic practice, often necessitating surgical intervention to restore knee joint stability and function [1, 2]. Despite advancements in surgical techniques, optimizing postoperative healing and outcomes remains a significant concern [3]. Meniscal repair, a widely utilized surgical procedure, aims to preserve meniscal tissue and biomechanical function, thereby preventing degenerative changes in the knee joint [4, 5].
In recent years, there has been growing interest in adjunctive treatments aimed at enhancing tissue healing and recovery following meniscal repair. One such promising intervention is the use of platelet-rich plasma (PRP), which involves the intraoperative injection of autologous platelets, cytokines, and growth factors derived from the patient's own blood [6,7,8]. PRP has gained attention for its potential to augment tissue regeneration and modulate inflammatory processes, thereby accelerating healing and improving outcomes in various orthopedic conditions [9,10,11].
The rationale behind PRP therapy lies in its ability to deliver a concentrated source of bioactive molecules that play crucial roles in tissue repair and remodeling [9, 12, 13]. Platelets, as primary components of PRP, release growth factors such as platelet-derived growth factor (PDGF), transforming growth factor-beta (TGF-β), and vascular endothelial growth factor (VEGF), which promote angiogenesis, collagen synthesis, and cell proliferation [14, 15]. Additionally, PRP contains anti-inflammatory cytokines that may mitigate postoperative inflammation and pain, facilitating rehabilitation and functional recovery [16, 17].
Despite its theoretical benefits, the clinical efficacy of PRP in the context of meniscal repair remains a subject of debate. While some studies have reported favorable outcomes, others have found no significant difference in postoperative pain, functional improvement, or overall success rates between PRP-treated and control groups [18,19,20,21]. Moreover, the existing literature is characterized by heterogeneity in study designs, patient populations, PRP preparation techniques, and outcome measures, making it challenging to draw definitive conclusions [22,23,24].
Therefore, there is a need for a comprehensive review and meta-analysis of the available evidence to elucidate the impact of intraoperative PRP on pain relief, functional recovery, and surgical outcomes following meniscal repair. By synthesizing data from randomized controlled trials (RCTs), cohort studies, and case–control studies, this review aims to provide orthopedic surgeons, researchers, and healthcare providers with valuable insights into the potential benefits and limitations of PRP therapy in meniscal injuries.
In light of the aforementioned considerations, this systematic review and meta-analysis seek to address the following objectives:
-
1.
Evaluate the effect of intraoperative PRP injection on postoperative pain relief in patients undergoing meniscal repair.
-
2.
Assess the impact of PRP therapy on functional recovery, as measured by validated outcome measures such as the Lysholm score and International Knee Documentation Committee (IKDC) score.
-
3.
Determine the influence of PRP treatment on the overall success rates of meniscal repair procedures, including rates of re-tear and revision surgery.
By addressing these objectives, this study aims to provide a comprehensive analysis of the current evidence regarding the efficacy of PRP as an adjunctive therapy in meniscal repair procedures. The findings of this review may have important implications for clinical practice, guiding treatment decisions, and optimizing outcomes for patients with meniscal injuries.
Methodology
Study Design
This systematic review and meta-analysis aim to provide a comprehensive evaluation of the efficacy of intraoperative PRP administration following meniscal repair surgery. This study aims to employ a systematic approach to synthesize the available evidence and provide valuable insights into the effectiveness of PRP in meniscal repair.
Search Strategy
Databases
A systematic literature search was conducted across multiple electronic databases, including PubMed, Embase, Scopus, and the Cochrane Library. The search strategy was developed using a combination of medical subject headings (MeSH) terms and keywords such as platelet-rich plasma, Meniscal injuries, Surgery, tissue recovery, and orthopaedic. The search strategy was designed to capture relevant studies published in English. The search strategy was peer-reviewed to ensure comprehensiveness and accuracy.
Inclusion Criteria
-
(i)
Studies conducted on human subjects.
-
(ii)
Analyses include case–control studies, cohorts, and randomized controlled trials (RCTs).
-
(iii)
Research examining the use of PRP intraoperatively after meniscal repair.
-
(iv)
Studies reporting outcomes related to pain reduction, improved functionality, and cure rates.
Exclusion Criteria
-
(i)
Animal studies.
-
(ii)
Studies not published in English.
-
(iii)
Studies lacking relevant outcome measures.
-
(iv)
Studies with insufficient data for analysis.
Study Selection
Two independent reviewers will screen titles and abstracts of retrieved articles to identify potentially eligible studies based on the predefined inclusion and exclusion criteria. Full-text articles of potentially relevant studies will then be retrieved and assessed for eligibility. Any discrepancies in study selection was resolved through discussion between the reviewers or consultation with a third reviewer if necessary.
Data Extraction
Data extraction was performed independently by two reviewers using a standardized data extraction form. The following information was extracted from each included study:
-
(a)
Study characteristics: authors, publication year, study design.
-
(b)
Patient demographics: age, gender.
-
(c)
Intervention details: PRP administration protocol, meniscal repair techniques.
-
(d)
Outcome measures: pain scores, functional assessments, cure rates.
Any discrepancies in data extraction were resolved through discussion between the reviewers or consultation with a third reviewer if necessary.
Quality Assessment
Risk of Bias Assessment
The risk of bias in included studies was assessed using appropriate tools depending on the study design. For RCTs, the Cochrane Risk of Bias tool was employed, while the Newcastle–Ottawa Scale was used for observational studies. Quality assessment will guide the interpretation of study findings and inform the overall strength of evidence.
Data Synthesis
Statistical Analysis
Statistical analysis was performed using RevMan 5.3 software. Meta-analysis was conducted to determine pooled effect sizes for the main outcomes, including pain reduction, improved functionality, and cure rates. Subgroup analyses was conducted based on primary research, PRP delivery protocol, and methods for meniscal healing to identify potential sources of heterogeneity.
Heterogeneity and Sensitivity Analysis
Heterogeneity Assessment: statistical heterogeneity was evaluated using I2 statistics. In the event that significant heterogeneity is discovered, a model which utilizes random effects was utilized.
Sensitivity Analysis: the effect of individual studies on the overall results was evaluated using sensitivity analysis. To test how solid the results are, we will not include studies that are prone to bias.
Publication Bias: publication bias was evaluated using funnel plots and Egger's test if a sufficient number of studies are included. Publication bias assessment is important to ensure that the results of the meta-analysis are not influenced by the selective publication of studies with positive results.
Subgroup analysis: subgroup analyses was conducted based on study features and patient demographics to identify potential sources of heterogeneity. Subgroup analysis allows for a more nuanced understanding of the effectiveness of PRP in different patient populations and treatment settings.
Grading of Recommendations Assessment, Development, and Evaluation (GRADE): the overall quality of evidence for each outcome was assessed using the GRADE methodology. GRADE provides a structured approach to evaluating the quality of evidence and strength of recommendations, considering factors such as study design, risk of bias, consistency, and precision.
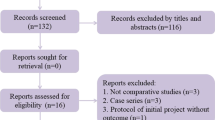
Reporting: this systematic review and meta-analysis will adhere to the reporting requirements outlined by the PRISMA guidelines to ensure transparency and accuracy in reporting. The PRISMA checklist was used to guide the reporting of methods, results, and conclusions. Clear and concise reporting is essential for ensuring the reproducibility and credibility of the study findings (Fig. 1).
Results
In Table 1, the participants who participated in the PRP and Control groups across all four studies are shown, along with their demographic features. The average age of the 25 male and 5 female participants in the PRP group was 37.3 years. This group had individuals with a body mass index (BMI) of 25.7 and an average pretreatment duration of 105.6 weeks. In comparison, the Control group took a different approach; its members had an average age of 35.6 years, with 19 men and 12 women, and a body mass index (BMI) of 24.8. The duration between their surgeries was 58.3 weeks on average. Across all studies, the PRP group had an average age of 41.39 years and a mean body mass index of 25.44, comprising 90 individuals—61 males and 29 females. These data provide a comprehensive summary of the characteristics of participants in the PRP group. In contrast, the Control group comprised 96 individuals (62 men and 34 women) with an average age of 42.1 years and a mean BMI of 26.82. These values offer insights into the demographic distribution of participants across the studies, facilitating an understanding of the characteristics of individuals in the PRP and Control groups.
Table 2 in the previously discussed research presents the pain ratings reported by both the Control group and the PRP group in various studies. In the study by Yang et al. [21]), the mean pain reported by participants in the PRP group (3.7, standard deviation 1.1) was significantly higher than that experienced by participants in the Control group (2.7, standard deviation 0.8). The study included a total of 60 individuals, with 31 assigned to the PRP group and 31 to the control group. The weight column indicates the overall analytical contribution of each study. The PRP group might have experienced a statistically significant difference, as the 95% confidence interval (CI) is 1.00 [0.52, 1.48]. Consequently, the Mean Difference column displays the disparity in mean pain levels between the Control group and the PRP group. In Pujol et al. [25], the findings revealed that the PRP group reported considerably less pain (93.3) with a significantly lower standard deviation (SD) of 1.12 compared to the Control group (mean 78.4, SD 1.7). Thirteen participants were included in each group. The mean difference for pain ratings was 14.90 [13.93, 15.87], indicating that the PRP group fared better than the control group. In Yi et al. [26], the mean difference for the control group was − 4.49, while the PRP group showed a negative mean difference of − 5.84, indicating a lower level of pain. Each group consisted of 28 participants. The statistical significance of the difference favoring the PRP group is indicated by the reported mean difference of − 1.35 [− 1.53, − 1.17]. There were 75 participants in the PRP group and 76 in the control group for these studies, with a 95 percent confidence interval. The overall analysis indicates a mean difference of 4.83 [− 1.42, 11.08], although the p value (p = 0.13) suggests that the difference is not statistically significant. Heterogeneity and test for overall effect: the statistics for heterogeneity (τ2, χ2, I2) indicate significant variation across the studies. Given the p value of 0.13 and Z value of 1.51 for the overall effect test, it is reasonable to conclude that the overall impact does not meet the criteria for statistical significance (Fig. 2).
Table 3 displays the Lysholm scores for the Control group and the platelet-rich plasma (PRP) group from various studies. In the study conducted by Yang et al., the PRP group had a mean Lysholm score of 27.8 (SD = 11.7), while the control group had a mean score of 24.4 (SD = 11.1). There were 31 participants in the control group and 30 in the PRP group. The Mean Difference column indicates how the PRP group, and the Control group performed on the Lysholm test. A 95% confidence interval (CI) of 3.40 [− 2.33, 9.13] suggests a statistically significant difference in favor of the PRP group. In the research conducted by Justin W et al. in 2015 [27], the control group had a Lysholm score considerably higher than the PRP group, with a mean score of 89 (standard deviation: 9.7) compared to 66 (SD: 38.9) in the PRP group. There were two groups in the study: 20 participants in the control group and 15 participants in the PRP group. Compared to the Control group, the PRP group had significantly lower Lysholm scores (− 23.00 [− 39.69, − 6.31]), indicating a lower probability of success for the PRP group. According to Yi et al., each group consisted of twenty-eight participants. The PRP group had an average Lysholm score of 31.98 (standard deviation = 1.88), while the control group had a score of 28.93 (standard deviation = 1.92). The mean difference was 3.05 [2.05, 4.05], indicating a statistically significant difference in favor of the PRP group. During the study, 73 individuals were part of the PRP group, and 79 individuals were part of the control group (95 percent CI). The mean difference of − 0.44 [− 7.80, 6.92] shows that there was no statistically significant change in the Lysholm scores between the PRP and Control groups. Test for overall effect and heterogeneity: there is considerable variability across trials, as indicated by the heterogeneity statistics (τ2, χ2, I2). The test’s Z-value of 0.12 and p value of 0.91 suggest no statistically significant overall impact (Fig. 3).
In the following research, the International Knee Documentation Committee (IKDC) conducted tests on both the PRP and Control groups, and the results are displayed in Table 4. In the study conducted by Yang et al., the mean IKDC score for the PRP group was 25.5, with a standard deviation of 10.4, which was identical to the control group's mean score of 25.7. Thirty participants were included in the PRP group, while 31 participated in the control group. As depicted in the Mean Difference column, the IKDC scores of the Control group and the PRP group showed a significant difference. The mean difference has a 95% confidence interval of 2.80 [− 2.32, 7.92]. Although the difference is not statistically significant, it does suggest that the PRP group had somewhat higher IKDC ratings compared to the Control group. According to Pujol et al. [25], the control group had an average IKDC score of 87.9 with a standard deviation of 7.9, while the PRP group had an average score of 90.7 with an unusually low standard deviation of 8.5. Both groups consisted of 17 participants each. The PRP group demonstrated a comparable differential in IKDC scores, as indicated by the mean difference of 2.80 [− 2.72, 8.32]; however, this difference did not meet the threshold for statistical significance. Overall (95% CI): Throughout the duration of the study, there were 47 participants in the PRP group and 48 in the control group. The mean difference of 2.80 [− 0.95, 6.55] in the overall analysis suggests a significant difference in IKDC scores favoring the PRP group; however, this difference did not reach statistical significance. Heterogeneity and yest for overall effect: heterogeneity statistics, such as τ2 = 0.00, χ2 = 0.00, df = 1, p = 1.00, and I2 = 0 percent, demonstrate the absence of trial-to-trial variability. The overall effect test yielded a p value of 0.14 and a Z value of 1.46, suggesting that the overall influence is not statistically significant (Fig. 4).
Table 5 presents the results of range of motion (ROM) tests conducted in several trials with both the control and PRP groups. In the study conducted by Griffin et al. [27], the average ROM in the PRP group was 125°, with a standard deviation (SD) of 22.5°, while the control group's mean ROM was slightly lower at 119°, with a standard deviation of 34.75°. Fifteen participants represented the PRP group and 20 represented the Control group out of a total of 30 participants in the study. The “Mean Difference” column indicates the variation in ROM between the control group and the PRP group, with a confidence interval (CI) for this difference ranging from 13 to 25. Although there may not have been a statistically significant difference, the PRP group appeared to have slightly more ROM than the control group. According to Pujol et al., the average ROM for the PRP group was 135.0°, with a small standard deviation (SD) of 2.5°, while the ROM in the control group was slightly lower at 130.0°, with the same SD of 2.5°. Both groups consisted of 17 participants each. The Mean Difference was reported as 5.00 [3.32, 6.68], indicating a moderate difference in ROM favoring the PRP group, and this difference was statistically significant. In Yi et al., the PRP group's average ROM was 118.55°, with a standard deviation (SD) of 7.87°, whereas the control group's ROM was lower at 109.81°, with a lower SD of 5.51°. There were 28 individuals in each of the two groups. A statistically significant improvement in ROM for the PRP group is indicated by a mean difference of 8.74 [5.18, 12.30]. Total (95% CI): the total number of participants in all investigations was 65 for the Control group and 60 for the PRP group. An overall mean difference of 2.80 [− 0.95, 6.55] was found in the study, suggesting a small but non-significant difference in ROM favoring the PRP group. Heterogeneity and test for overall effect: heterogeneity statistics (τ2 = 2.86, χ2 = 3.47, df = 2, p = 0.18, I2 = 42%) indicate considerable diversity across trials. The overall effect test's Z-value of 4.28 and p value of less than 0.0001 suggest a clear advantage for the PRP group in terms of ROM development when considering the entire impact (Fig. 5).
Table 6 presents a comparison of the failure rates in the PRP group and the control group based on an inverse variance (IV) random-effects model. Additionally, it shows the weighted mean difference as well as its 95 percent confidence interval (CI). In the study by Yang et al., out of 31 patients treated with the control group, 4 experienced failures, while 2 out of 30 patients received PRP treatment. Despite a weighted mean difference of 0.48 in favor of the PRP group, it was not statistically significant (95% CI 0.08–2.85). In Griffin et al., failure occurred in 4% of the PRP group's 15 patients and 5% of the control group's 20 patients. Again, the PRP group had a lead, with a weighted mean difference of 1.09 (95% CI 0.24–5.03); however, the difference did not meet the criteria for statistical significance. According to Pujol et al., the PRP group had one patient out of seventeen who experienced failure, while the control group had two patients out of seventeen who experienced failure. Although this difference was not statistically significant, the PRP group benefited from a weighted mean difference of 0.47 (with a 95% CI ranging from 0.25 to 5.72). Total (95% CI): across all studies, a total of 62 events were observed in the PRP group out of 68 total patients, compared to 100 events in the control group out of 100 total patients. The overall weighted mean difference in terms of failure rates is 0.71 (95% CI 0.25–2.02), suggesting that the PRP group had a minor but non-significant advantage. The results of the trials show no substantial heterogeneity (I2 = 0%, p = 0.74) and the overall effect test confirms that the PRP group and control group had similar failure rates (Z = 0.65, p = 0.52) (Fig. 6). The risk of bias chart is shown in the Fig. 7.
Discussion
Several medical issues were the subject of a thorough evaluation and meta-analysis to determine the relative effectiveness of control treatments and platelet-rich plasma (PRP) therapy. The results on the efficacy of PRP treatment were derived from meta-analyses that drew from a large number of clinical trials.
Demographic Characteristics
The analysis revealed significant variations in demographic characteristics between the PRP and control groups across the studies (Table 1). These differences, including age, gender distribution, body mass index (BMI), smoking status, and time to surgery, underscore the importance of considering potential confounding factors when interpreting the results.
Pain Scores
The meta-analysis showed mixed results regarding pain reduction between the PRP and control groups (Table 2). While some studies demonstrated statistically significant improvements in pain scores for the PRP group, others did not reach statistical significance. Nonetheless, the overall trend suggested a potential benefit of PRP therapy in reducing knee discomfort. The observed heterogeneity highlights the need for cautious interpretation of these findings and further investigation into the factors influencing pain outcomes following PRP treatment.
Lysholm Scores
The analysis of Lysholm scores revealed conflicting outcomes across studies, with some showing no significant difference between the PRP and control groups (Table 3), while others demonstrated superiority of PRP therapy. Despite the variability, the overall analysis did not find a statistically significant difference in Lysholm ratings between the groups.
IKDC Scores
Evaluation of IKDC scores indicated a moderate improvement favoring the PRP group, although the difference was not statistically significant (Table 4). The consistency of these findings across studies suggests a potential benefit of PRP therapy in enhancing knee function and symptoms.
Range of Motion (ROM)
ROM measurements exhibited varied outcomes, with some studies showing significant improvement following PRP therapy, while others did not demonstrate a significant difference (Table 5). Despite the overall analysis suggesting a slight improvement in ROM favoring the PRP group, significant heterogeneity was observed, highlighting the need for further research to elucidate the factors influencing ROM outcomes following PRP treatment.
Failure Rates
The analysis of failure rates did not reveal a statistically significant difference between the PRP and control groups (Table 6), despite some individual studies suggesting a trend towards lower failure rates in the PRP group. These findings underscore the importance of considering various factors, such as patient selection criteria and treatment protocols, in assessing the efficacy of PRP therapy in preventing treatment failure.
Quality of Evidence
Assessment of the quality of evidence using the GRADE framework highlighted the need for well-designed clinical studies to strengthen the evidence base for PRP therapy. Future research should focus on conducting prospective trials with robust methodologies to provide more conclusive evidence regarding the effectiveness of PRP therapy for musculoskeletal conditions.
Healthcare professionals can consider PRP therapy as part of a comprehensive treatment approach for patients with musculoskeletal conditions, such as osteoarthritis, tendon injuries, and ligament tears [28]. The results from this analysis indicate that PRP treatment has the potential to alleviate knee discomfort while simultaneously enhancing knee function and mobility. According to the results, PRP treatment may be more effective for certain patient groups than others. PRP treatment may be appropriate for some individuals, including those with mild to moderate osteoarthritis, tendon injuries without severe degeneration, and ligament tears [6, 29, 30]. This may determine which patients might gain the most from platelet-rich plasma therapy by combining clinical judgment with evidence-based recommendations. Patients should be informed about the level of evidence supporting PRP therapy, the expected outcomes, potential risks or side effects, and alternative treatment options [31]. This shared decision-making process empowers patients to make informed choices about their healthcare. This can guide in optimizing the delivery of PRP therapy [32]. This includes considerations such as the selection of PRP preparation methods, injection techniques, dosing regimens, and rehabilitation protocols [33]. Additional research is needed to strengthen the evidence base for the therapy of PRP. This can be accomplished by conducting clinical studies that are well designed, conducting prospective trials, and working together with research teams that come from a variety of disciplines.
By having a deeper comprehension of the molecular mechanisms that are responsible for the effects of PRP therapy, clinical decision-making and therapeutic potential may be improved [34]. A concentrated blend of bioactive chemicals, including platelets, growth factors, cytokines, and others, interact with different kinds of cells and signaling pathways in the body; this is what makes PRP so interesting [35]. To assist in tissue repair and regeneration, PRP contains growth factors such as transforming growth factor-beta (TGF-β), platelet-derived growth factor (PDGF), and vascular endothelial growth factor (VEGF). To complete the process, certain growth factors are necessary [36]. To alleviate pain and speed up the healing process for damaged tissue, these growth factors stimulate angiogenesis, collagen synthesis, and cell division. Furthermore, by reducing inflammation, PRP improves joint function and lessens discomfort. This is accomplished by increasing the synthesis of anti-inflammatory chemicals and inhibiting the release of cytokines that promote inflammation [13].
Development factors found in PRP promote the growth and differentiation of chondrocytes and mesenchymal stem cells (MSCs), two cell types essential for cartilage regeneration and repair [37]. By enhancing cartilage matrix synthesis and inhibiting matrix degradation, PRP supports the restoration of knee function and mobility [38]. Functional restoration of injured joint tissues is also aided by PRP-mediated stimulation of endogenous repair pathways, including the secretion of extracellular matrix proteins and tissue-specific growth factors [39]. By promoting the production of extracellular matrix components including hyaluronic acid, collagen, and glycosaminoglycans (GAGs), PRP treatment helps with tissue remodeling and repair [40]. These structural proteins provide mechanical support and lubrication to the joint, thereby improving joint flexibility and range of motion [41]. Additionally, PRP-induced neovascularization and tissue ingrowth promote the formation of new blood vessels and nerve fibers, which contribute to the restoration of normal joint biomechanics and proprioception [42].
By stimulating cell migration, proliferation, and differentiation at the injury site, PRP treatment improves tissue repair and regeneration [43]. Progenitor cells and stem cells are brought to injured tissue by the bioactive compounds in PRP, where they help with remodeling and repair. Moreover, PRP modulates the inflammatory response by regulating the activity of immune cells and cytokines, which helps to minimize tissue damage and prevent the progression of degenerative changes [44]. The therapeutic effects of PRP therapy are mediated by its ability to stimulate tissue repair, modulate inflammation, and promote regenerative processes in the musculoskeletal system [34]. By harnessing the biological mechanisms of PRP, we can optimize treatment strategies and improve outcomes for patients with musculoskeletal disorders [45]. Further research into the molecular pathways and cellular interactions underlying PRP therapy will continue to advance our understanding of its therapeutic potential and guide the development of novel interventions for joint-related conditions.
Clinical Implications
The PRP therapy has garnered significant interest in the field of orthopedics, particularly for its potential implications in improving knee function and alleviating symptoms associated with various knee conditions [46]. Previous studies have explored the effects of PRP on knee function in patients with conditions such as osteoarthritis, ligament injuries, meniscal tears, and tendonitis [6, 7, 46, 47]. Understanding the implications of PRP therapy on knee function requires consideration of findings from these studies and their clinical relevance. Numerous studies have investigated the use of PRP therapy for knee osteoarthritis, a degenerative joint condition characterized by cartilage deterioration and inflammation [48,49,50,51]. The implications of PRP therapy in knee OA focus on its ability to reduce pain, improve function, and potentially slow disease progression. A meta-analysis by McLarnon et al. [52] found that PRP injections significantly improved knee function and reduced pain compared to control treatments in patients with knee OA. These findings suggest that PRP therapy may have positive implications for enhancing knee function and mobility in individuals with OA.
PRP therapy has also been studied in the context of ligament injuries, such as anterior cruciate ligament (ACL) tears [53]. While the implications of PRP in ACL reconstruction are still under investigation, some studies have suggested potential benefits in promoting ligament healing and improving knee stability [54]. For example, a systematic review by Andriolo et al. [55] reported that PRP augmentation in ACL reconstruction may lead to better subjective and objective outcomes, including improved knee function and reduced laxity. These findings imply that PRP therapy could play a role in optimizing knee function following ligament injuries. Meniscal tears are common knee injuries that can significantly impact knee function and mobility. PRP therapy has been explored as a potential adjunctive treatment to enhance meniscal healing and preserve knee function [26]. While the evidence regarding the efficacy of PRP for meniscal tears is mixed, some studies have suggested promising outcomes. For instance, a randomized controlled trial by Barman et al. [56] found that PRP injection following meniscal repair resulted in better clinical outcomes and higher rates of meniscal healing compared to saline injection. These findings suggest that PRP therapy may have implications for improving knee function and reducing the risk of subsequent knee problems following meniscal tears. Chronic tendonitis or tendinopathy, such as patellar tendonitis (jumper’s knee) or Achilles tendinopathy can cause significant impairment in knee function and mobility [57]. PRP therapy has been investigated as a potential treatment option to promote tendon healing and improve knee function in patients with these conditions [58]. While the evidence regarding the efficacy of PRP for tendonitis is still evolving, some studies have shown promising results. For example, a systematic review by Charousset et al. [59] reported that PRP injections led to significant improvements in pain and function in patients with patellar tendinopathy. These findings suggest that PRP therapy may have implications for enhancing knee function and reducing symptoms associated with chronic tendonitis.
Conclusion
The analysis of postoperative pain relief in patients undergoing meniscal repair with intraoperative PRP injection yielded mixed results. While some studies demonstrated a statistically significant reduction in pain levels following PRP therapy, others did not observe significant differences compared to control groups. Therefore, the overall evidence regarding the effect of PRP on postoperative pain relief in meniscal repair patients is inconclusive.
In conclusion, while there is some evidence suggesting potential benefits of intraoperative PRP injection in patients undergoing meniscal repair, particularly in terms of pain relief and functional recovery, the overall findings are inconclusive and warrant further research. Future studies should focus on conducting well-designed randomized controlled trials with larger sample sizes to provide more robust evidence regarding the efficacy of PRP therapy in improving outcomes following meniscal repair procedures. Additionally, comprehensive assessments of functional outcomes and success rates, along with standardized protocols for PRP administration, are essential for accurately evaluating its clinical effectiveness and guiding evidence-based practice in orthopedic surgery.
Data availability
Not applicable.
References
Rodeo, S. A., Monibi, F., Dehghani, B., & Maher, S. (2020). Biological and mechanical predictors of meniscus function: Basic science to clinical translation. Journal of Orthopaedic Research, 38(5), 937–945. https://doi.org/10.1002/jor.24552
Smith, N. A., Parkinson, B., Hutchinson, C. E., Costa, M. L., & Spalding, T. (2016). Is meniscal allograft transplantation chondroprotective? A systematic review of radiological outcomes. Knee Surgery, Sports Traumatology, Arthroscopy, 24(9), 2923–2935. https://doi.org/10.1007/s00167-015-3573-0
Calanna, F., Duthon, V., & Menetrey, J. (2022). Rehabilitation and return to sports after isolated meniscal repairs: A new evidence-based protocol. Journal of Experimental Orthopaedics, 9(1), 80. https://doi.org/10.1186/s40634-022-00521-8
Perelli, S. (2023). Meniscal injuries of the knee. In Textbook of musculoskeletal disorders (pp. 451–9). Springer.
Papalia, R., Del Buono, A., Osti, L., Denaro, V., & Maffulli, N. (2011). Meniscectomy as a risk factor for knee osteoarthritis: A systematic review. British Medical Bulletin, 99(1), 89–106. https://doi.org/10.1093/bmb/ldq043
Li, Z., & Weng, X. (2022). Platelet-rich plasma use in meniscus repair treatment: A systematic review and meta-analysis of clinical studies. Journal of Orthopaedic Surgery and Research, 17(1), 446. https://doi.org/10.1186/s13018-022-03293-0
Medina-Porqueres, I., Martin-Garcia, P., Sanz-De-Diego, S., Gomez-Caceres, A., Moya-Torrecilla, F., Reyes-Eldblom, M., et al. (2022). Clinical and functional outcome of meniscal injuries treated with platelet-rich plasma: A single-center case series. International Journal of Environmental Research and Public Health. https://doi.org/10.3390/ijerph19127118
Patil, P. (2023). Therapeutic uses of platelet-rich plasma (PRP) in sport injuries—A narrative review. Journal of Orthopaedic Reports 100287.
Dos Santos, R. G., Santos, G. S., Alkass, N., Chiesa, T. L., Azzini, G. O., da Fonseca, L. F., et al. (2021). The regenerative mechanisms of platelet-rich plasma: A review. Cytokine, 144(155560), 155560. https://doi.org/10.1016/j.cyto.2021.155560
Verma, R., Kumar, S., Garg, P., & Verma, Y. K. (2023). Platelet-rich plasma: A comparative and economical therapy for wound healing and tissue regeneration. Cell and Tissue Banking, 24(2), 285–306. https://doi.org/10.1007/s10561-022-10039-z
Dragoo, J. L., Braun, H. J., Durham, J. L., Ridley, B. A., Odegaard, J. I., Luong, R., et al. (2012). Comparison of the acute inflammatory response of two commercial platelet-rich plasma systems in healthy rabbit tendons. American Journal of Sports Medicine, 40(6), 1274–1281. https://doi.org/10.1177/0363546512442334
Laudy, A. B. M., Bakker, E. W. P., Rekers, M., & Moen, M. H. (2015). Efficacy of platelet-rich plasma injections in osteoarthritis of the knee: A systematic review and meta-analysis. British Journal of Sports Medicine, 49(10), 657–672. https://doi.org/10.1136/bjsports-2014-094036
Sundman, E. A., Cole, B. J., & Fortier, L. A. (2011). Growth factor and catabolic cytokine concentrations are influenced by the cellular composition of platelet-rich plasma. American Journal of Sports Medicine, 39(10), 2135–2140. https://doi.org/10.1177/0363546511417792
Cecerska-Heryć, E., Goszka, M., Serwin, N., Roszak, M., Grygorcewicz, B., Heryć, R., et al. (2022). Applications of the regenerative capacity of platelets in modern medicine. Cytokine & Growth Factor Reviews, 64, 84–94. https://doi.org/10.1016/j.cytogfr.2021.11.003
Mishra, A., Woodall, J., Jr., & Vieira, A. (2009). Treatment of tendon and muscle using platelet-rich plasma. Clinics in Sports Medicine, 28(1), 113–125. https://doi.org/10.1016/j.csm.2008.08.007
McRobb, J., Kamil, K. H., Ahmed, I., Dhaif, F., & Metcalfe, A. (2023). Influence of platelet-rich plasma (PRP) analogues on healing and clinical outcomes following anterior cruciate ligament (ACL) reconstructive surgery: A systematic review. European Journal of Orthopaedic Surgery & Traumatology, 33(2), 225–253. https://doi.org/10.1007/s00590-021-03198-4
Zhang, J., Middleton, K. K., Fu, F. H., Im, H.-J., & Wang, J.H.-C. (2013). HGF mediates the anti-inflammatory effects of PRP on injured tendons. PLoS ONE, 8(6), e67303. https://doi.org/10.1371/journal.pone.0067303
Chen, X., Jones, I. A., Togashi, R., Park, C., & Vangsness, C. T., Jr. (2020). Use of platelet-rich plasma for the improvement of pain and function in rotator cuff tears: A systematic review and meta-analysis with bias assessment. American Journal of Sports Medicine, 48(8), 2028–2041. https://doi.org/10.1177/0363546519881423
Everts, P. A., van Erp, A., DeSimone, A., Cohen, D. S., & Gardner, R. D. (2021). Platelet rich plasma in orthopedic surgical medicine. Platelets, 32(2), 163–174. https://doi.org/10.1080/09537104.2020.1869717
Mishra, A. K., Skrepnik, N. V., Edwards, S. G., Jones, G. L., Sampson, S., Vermillion, D. A., et al. (2014). Efficacy of platelet-rich plasma for chronic tennis elbow: A double-blind, prospective, multicenter, randomized controlled trial of 230 patients: A double-blind, prospective, multicenter, randomized controlled trial of 230 patients. American Journal of Sports Medicine, 42(2), 463–471. https://doi.org/10.1177/0363546513494359
Yang, C.-P., Hung, K.-T., Weng, C.-J., Chen, A.C.-Y., Hsu, K.-Y., & Chan, Y.-S. (2021). Clinical outcomes of meniscus repair with or without multiple intra-articular injections of platelet rich plasma after surgery. Journal of Clinical Medicine, 10(12), 2546. https://doi.org/10.3390/jcm10122546
Fabbro, M. D., Bortolin, M., Taschieri, S., Ceci, C., & Weinstein, R. L. (2016). Antimicrobial properties of platelet-rich preparations. A systematic review of the current pre-clinical evidence. Platelets, 27(4), 276–285. https://doi.org/10.3109/09537104.2015.1116686
Bennell, K. L., Paterson, K. L., Metcalf, B. R., Duong, V., Eyles, J., Kasza, J., et al. (2021). Effect of intra-articular platelet-rich plasma vs placebo injection on pain and medial tibial cartilage volume in patients with knee osteoarthritis: The RESTORE randomized clinical trial: The RESTORE randomized clinical trial. JAMA, 326(20), 2021–2030. https://doi.org/10.1001/jama.2021.19415
Sheth, U., Simunovic, N., Klein, G., Fu, F., Einhorn, T. A., Schemitsch, E., et al. (2012). Efficacy of autologous platelet-rich plasma use for orthopaedic indications: A meta-analysis. Journal of Bone and Joint Surgery. American Volume, 94(4), 298–307. https://doi.org/10.2106/JBJS.K.00154
Pujol, N., Salle De Chou, E., Boisrenoult, P., & Beaufils, P. (2015). Platelet-rich plasma for open meniscal repair in young patients: any benefit? Knee Surgery, Sports Traumatology, Arthroscopy, 23(1), 51–58. https://doi.org/10.1007/s00167-014-3417-3
Yi, X., Lee, J. E., Lee, Y. H., Yu, X., & Lee, H. S. (2023). Clinical efficacy of platelet-rich plasma combined with arthroscopic meniscal plasty on pain, function and physiologic indicators in elderly patients with knee meniscus injury: A retrospective observational study. American Journal of Translational Research, 15(6), 3806–3814.
Griffin, J. W., Hadeed, M. M., Werner, B. C., Diduch, D. R., Carson, E. W., & Miller, M. D. (2015). Platelet-rich plasma in meniscal repair: Does augmentation improve surgical outcomes? Clinical Orthopaedics and Related Research, 473(5), 1665–1672. https://doi.org/10.1007/s11999-015-4170-8
Filardo, G. (2012). Platelet-rich plasma intra-articular injections for cartilage degeneration and osteoarthritis: Single-versus double-spinning approach. Knee Surgery, Sports Traumatology, 20, 2082–2091.
Khoshbin, A., Leroux, T., Wasserstein, D., Marks, P., Theodoropoulos, J., Ogilvie-Harris, D., et al. (2013). The efficacy of platelet-rich plasma in the treatment of symptomatic knee osteoarthritis: A systematic review with quantitative synthesis. Arthroscopy, 29(12), 2037–2048. https://doi.org/10.1016/j.arthro.2013.09.006
Tomio, J., Yamana, H., Matsui, H., Yamashita, H., Yoshiyama, T., & Yasunaga, H. (2017). Tuberculosis screening prior to anti-tumor necrosis factor therapy among patients with immune-mediated inflammatory diseases in Japan: A longitudinal study using a large-scale health insurance claims database. International Journal of Rheumatic Diseases, 20(11), 1674–1683. https://doi.org/10.1111/1756-185x.13190
Moraes, V. Y., Lenza, M., Tamaoki, M. J., Faloppa, F., & Belloti, J. C. (2014). Platelet-rich therapies for musculoskeletal soft tissue injuries. Cochrane Database of Systematic Reviews, (4), CD010071. https://doi.org/10.1002/14651858.CD010071.pub3
Smith, H. C. (2012). Risk factors for anterior cruciate ligament injury: A review of the literature-part 1: Neuromuscular and anatomic risk. Sports Health, 4(1), 69–78.
Dai, W.-L., Zhou, A.-G., Zhang, H., & Zhang, J. (2017). Efficacy of platelet-rich plasma in the treatment of knee osteoarthritis: A meta-analysis of randomized controlled trials. Arthroscopy, 33(3), 659-670.e1. https://doi.org/10.1016/j.arthro.2016.09.024
Everts, P., Onishi, K., Jayaram, P., Lana, J. F., & Mautner, K. (2020). Platelet-rich plasma: New performance understandings and therapeutic considerations in 2020. International Journal of Molecular Sciences, 21(20), 7794. https://doi.org/10.3390/ijms21207794
Pavlovic, V., Ciric, M., Jovanovic, V., & Stojanovic, P. (2016). Platelet Rich Plasma: A short overview of certain bioactive components. Open Med (Warsz)., 11(1), 242–247. https://doi.org/10.1515/med-2016-0048
Foster, T. E., Puskas, B. L., Mandelbaum, B. R., Gerhardt, M. B., & Rodeo, S. A. (2009). Platelet-rich plasma: From basic science to clinical applications: From basic science to clinical applications. American Journal of Sports Medicine, 37(11), 2259–2272. https://doi.org/10.1177/0363546509349921
Tang, Y., Wang, H., Sun, Y., Jiang, Y., Fang, S., Kan, Z., et al. (2021). Using platelet-rich plasma hydrogel to deliver mesenchymal stem cells into three-dimensional PLGA scaffold for cartilage tissue engineering. ACS Applied Bio Materials, 4(12), 8607–8614. https://doi.org/10.1021/acsabm.1c01160
Nabavizadeh, S. S., Talaei-Khozani, T., Zarei, M., Zare, S., Hosseinabadi, O. K., Tanideh, N., et al. (2022). Attenuation of osteoarthritis progression through intra-articular injection of a combination of synovial membrane-derived MSCs (SMMSCs), platelet-rich plasma (PRP) and conditioned medium (secretome). Journal of Orthopaedic Surgery and Research, 17(1), 102. https://doi.org/10.1186/s13018-021-02851-2
Kon, E., Filardo, G., Di Martino, A., & Marcacci, M. (2011). Platelet-rich plasma (PRP) to treat sports injuries: Evidence to support its use. Knee Surgery, Sports Traumatology, Arthroscopy, 19(4), 516–527. https://doi.org/10.1007/s00167-010-1306-y
Potekaev, N. N., Borzykh, O. B., Medvedev, G. V., Pushkin, D. V., Petrova, M. M., Petrov, A. V., et al. (2021). The role of extracellular matrix in skin wound healing. Journal of Clinical Medicine, 10(24), 5947. https://doi.org/10.3390/jcm10245947
Blalock, D., Miller, A., Tilley, M., & Wang, J. (2015). Joint instability and osteoarthritis. Clinical Medicine Insights: Arthritis and Musculoskeletal Disorders, 8, 15–23. https://doi.org/10.4137/CMAMD.S22147
Laver, L., Marom, N., Dnyanesh, L., Mei-Dan, O., Espregueira-Mendes, J., & Gobbi, A. (2017). PRP for degenerative cartilage disease: A systematic review of clinical studies. Cartilage, 8(4), 341–364. https://doi.org/10.1177/1947603516670709
Sánchez, M., Anitua, E., Orive, G., Mujika, I., & Andia, I. (2009). Platelet-rich therapies in the treatment of orthopaedic sport injuries. Sports Medicine (Auckland, N. Z.), 39(5), 345–354. https://doi.org/10.2165/00007256-200939050-00002
Shen, L., Yuan, T., Chen, S., Xie, X., & Zhang, C. (2017). The temporal effect of platelet-rich plasma on pain and physical function in the treatment of knee osteoarthritis: Systematic review and meta-analysis of randomized controlled trials. Journal of Orthopaedic Surgery and Research, 12(1), 16. https://doi.org/10.1186/s13018-017-0521-3
Weber, A. E., Bolia, I. K., & Trasolini, N. A. (2021). Biological strategies for osteoarthritis: From early diagnosis to treatment. International Orthopaedics, 45(2), 335–344. https://doi.org/10.1007/s00264-020-04838-w
Kon, E., Di Matteo, B., Delgado, D., Cole, B. J., Dorotei, A., Dragoo, J. L., et al. (2020). Platelet-rich plasma for the treatment of knee osteoarthritis: An expert opinion and proposal for a novel classification and coding system. Expert Opinion on Biological Therapy, 20(12), 1447–1460. https://doi.org/10.1080/14712598.2020.1798925
Trams, E., Kulinski, K., Kozar-Kaminska, K., Pomianowski, S., & Kaminski, R. (2020). The clinical use of platelet-rich plasma in knee disorders and surgery—A systematic review and meta-analysis. Life, 10, 94.
Zhu, Y., Yuan, M., Meng, H. Y., Wang, A. Y., Guo, Q. Y., Wang, Y., et al. (2013). Basic science and clinical application of platelet-rich plasma for cartilage defects and osteoarthritis: A review. Osteoarthritis Cartilage, 21(11), 1627–1637. https://doi.org/10.1016/j.joca.2013.07.017
Szwedowski, D., Szczepanek, J., Paczesny, Ł, Zabrzyński, J., Gagat, M., Mobasheri, A., et al. (2021). The effect of platelet-rich plasma on the intra-articular microenvironment in knee osteoarthritis. International Journal of Molecular Sciences, 22(11), 5492. https://doi.org/10.3390/ijms22115492
Şen, E. İ, Yıldırım, M. A., Yeşilyurt, T., Kesiktaş, F. N., & Dıraçoğlu, D. (2020). Effects of platelet-rich plasma on the clinical outcomes and cartilage thickness in patients with knee osteoarthritis. Journal of Back and Musculoskeletal Rehabilitation, 33(4), 597–605. https://doi.org/10.3233/BMR-181209
Tucker, J. D. (2021). Randomized, placebo-controlled analysis of the knee synovial environment following platelet-rich plasma treatment for knee osteoarthritis. PM&R, 13, 707–719.
McLarnon, M., & Heron, N. (2021). Intra-articular platelet-rich plasma injections versus intra-articular corticosteroid injections for symptomatic management of knee osteoarthritis: Systematic review and meta-analysis. BMC Musculoskeletal Disorders, 22(1), 550. https://doi.org/10.1186/s12891-021-04308-3
Zicaro, J. P., Garcia-Mansilla, I., Zuain, A., Yacuzzi, C., & Costa-Paz, M. (2021). Has platelet-rich plasma any role in partial tears of the anterior cruciate ligament? Prospective comparative study. World Journal of Orthopedics, 12(6), 423–432. https://doi.org/10.5312/wjo.v12.i6.423
Zhu, T., Zhou, J., Hwang, J., & Xu, X. (2022). Effects of platelet-rich plasma on clinical outcomes after anterior cruciate ligament reconstruction: A systematic review and meta-analysis. Orthopaedic Journal of Sports Medicine, 10(1), 23259671211061536. https://doi.org/10.1177/23259671211061535
Andriolo, L., Matteo, D., Kon, B., Filardo, E., Venieri, G., & Marcacci, G. (2015). PRP augmentation for ACL reconstruction. BioMed Research International.
Barman, A., Bandyopadhyay, D., Mohakud, S., Sahoo, J., Maiti, R., Mukherjee, S., Prakash, S., Roy, S. S., & Viswanath, A. (2023). Comparison of clinical outcome, cartilage turnover, and inflammatory activity following either intra-articular or a combination of intra-articular with intra-osseous platelet-rich plasma injections in osteoarthritis knee: A randomized, clinical trial. Injury, 54(2), 728–737.
Walton, J., Kozina, E., Woo, F., & Jadidi, S. (2023). A review of patellar tendinopathy in athletes involved in jumping sports. Cureus, 15(10), e47459. https://doi.org/10.7759/cureus.47459
Tischer, T. (2020). Platelet-rich plasma (PRP) as therapy for cartilage, tendon and muscle damage-German working group position statement. Journal of Experimental Orthopaedics, 7(1), 1–11.
Charousset, C., Zaoui, A., Bellaiche, L., & Bouyer, B. (2014). Are multiple platelet-rich plasma injections useful for treatment of chronic patellar tendinopathy in athletes? A prospective study. American Journal of Sports Medicine, 42(4), 906–911. https://doi.org/10.1177/0363546513519964
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Thahir, M., Misbah, I., Bhaskaran, J. et al. Efficacy of Intraoperative Platelet-Rich Plasma After Meniscal Repair: Systematic Review and Meta-analysis. JOIO 58, 845–857 (2024). https://doi.org/10.1007/s43465-024-01155-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43465-024-01155-x