Abstract
Mining and excavation activities cause massive degradation of land, leading to complete loss of soil resources, vegetation, and biodiversity. Mine spoils support invasive weeds (predominantly Lantana) which can strive in these harsh conditions, causing allelopathy during plantation stage of reclamation. It is hypothesised that biochar produced from invasive weeds will enhance enzymatic activity, CO2 flux and overall fertility of coal mine spoil. A 6-month incubation study was conducted on the effect of biochar amendment (2 and 3%, w/w) on mine spoil enzymatic activities (dehydrogenase, invertase, amylase and cellulase), respiration and coal mine spoil fertility. The study showed that biochar significantly improved dehydrogenase (83%) and cellulase activity (78%) at 3% amendment. Geometric mean of enzymatic activities increased from 1.87 in control to 4.51 at 2% and 3.25 at 3% biochar amendment. Mine spoil physio-chemical properties such as soil organic carbon (65%), cation exchange capacity (54%), bulk density (25%) and water holding capacity (19%), were improved significantly compared to the unamended mine spoil. Biochar amendment reduced mine spoil CO2 flux at 2% (2.85 μmol CO2 m−2 s−1) and 3% (2.60 μmol CO2 m−2 s−1) compared to control (4.92 μmol CO2 m−2 s−1). The cost of biochar production and application (2%, w/w) in pit plantation during reclamation is estimated to be 844 USD t ha−1 (plantation density: 1600 trees ha−1). On the basis of present study, biochar preparation from invasive weeds can be used for sustainable reclamation of coal mine spoil.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Coal is an important contributor to worldwide energy generation but its environmental impact is quite catastrophic (Ahirwal and Maiti 2017). Coal ranks highest amongst the fossil fuels reserves in India and is responsible for 55% of energy requirement of the nation (Maiti 2013). 729 million tonnes (MT) of coal was produced in 2019–2020 in India, out of which > 90% of coal extraction was by surface mining. This causes loss of soil and vegetation cover and biodiversity, air and water pollution, complete disturbance of drainage and permanently alters natural landforms (Ahirwal et al. 2017). Mine spoil is nutrient deficient and characterised by low cation exchange capacity, nutrient unavailability, impoverished organic content and poor physical–chemical and biological characteristics (Frouz et al. 2008; Ussiri and Lal 2008). 2021–2030 is declared as the decade for ecosystem restoration by the UN General Assembly. Its main objective is the restoration of degraded land which provides essential ecosystem services. Therefore, the restoration of mine affected land is essential to fulfil the goals of global land-use policies. Biochar, a carbonaceous product of pyrolytic reaction of biomass has been reported to reclaim degraded land (Ghosh et al. 2020; Ghosh and Maiti 2020a, b). It is suitable for improving plant growth and microbial biodiversity (Pandey et al. 2020).
Enzymatic assays help quantify the microbial activity in soils which are responsible for important operations such as mineralization and humification (Palansooriya et al. 2019). This in turn influences the biogeochemical cycles of elements such as carbon, nitrogen, phosphorous and sulphur (Futa et al. 2020; García-Ruiz et al. 2008; Novak et al. 2018). Activities of these enzymes affect soil physio-chemical characteristics and support the growth of vegetative cover (Gascó et al. 2016; Ghosh and Maiti 2020a, b; Paz-Ferreiro et al. 2012). Thus, enzyme activity is a sensitive indicator for the evaluation of effect of an amendment in mine spoil reclamation (Ahirwal and Maiti 2018b; Palansooriya et al. 2019). Biochar application has been reported to increase activities of various extracellular and intracellular enzymes related to C, N, P and S cycles (Masto et al. 2013; Paz-Ferreiro et al. 2012; Gascó et al. 2016) and also improve enzymes such as dehydrogenase and catalase which are involved in the life process of microbes (Khadem and Raiesi 2017; Gascó et al. 2016). Other enzymatic activities such as urease, alkaline phosphatase, β-glucosidase, arylsulphatase, oxidase, fluorescein diacetate hydrolase has also been reported to improve by biochar application (Munir et al. 2021; Masto et al. 2013). The effect of biochar on enzymatic activity of coal mine spoil depends on the soil characteristics, microbial activities, types of enzymes and their interaction with biochar (Mohan et al. 2018). Sorption of enzyme molecules to the functional groups present on biochar surface influences the enzymatic reaction (Raiesi et al. 2019; Sandhu et al. 2019).
Surface mining has been reported to significantly decrease the terrestrial carbon pool (Ahirwal and Maiti 2018a), therefore, development of remediation technology which effectively sequesters carbon in the reclaimed coal mine spoil is necessary to combat global warming (Ahirwal et al. 2020). Biochar as an amendment during mine spoil reclamation not only enriches C pool in the derelict soil but also supports plant growth (Masto et al. 2013; Mohan et al. 2018). In an unfertilised soil, an increased CO2 flux after biochar application has been observed due to the soil microbial activity. Also, the substrate provides matrix for microbial decomposition resulting in the increase in the CO2 released. However, in reclaimed coal mine spoil, the labile fraction of carbon is much less compared to the recalcitrant carbon (Ghosh et al. 2020). Thus, CO2 flux in a barren soil has been reported to increase up to a certain period and then remain stable post biochar application. Carbon in biochars, originally removed from the atmosphere as CO2 during plant growth, remains in soils for hundreds of years (Camps-Arbestain et al. 2015; Lehmann et al. 2015). However, both positive and negative mineralization and CO2 emission have been reported. Biochar stability and its interaction with soil biota depend upon the biochar feedstock, the production method, the pyrolysis residence times and temperature (Ameloot et al. 2013). Biochar can act as an amendment to address problems related to remediation of degraded land (Ducey et al. 2021). However, a few studies have also reported the negative impact of biochar application on plant growth at high application rate (60 t ha−1) possibly due to increased soil alkalinity resulting in decreased nutrient availability and potentially Na toxicity (Gonzaga et al. 2018). However, the coal mine spoil is so impoverished that there will be probably no negative impact of biochar application.
Invasive weeds like Lantana camera naturally colonise coal mine overburden dumps and often cause allelopathy during plantation stage of reclamation. Lantana has very high biomass content and lignocellulosic composition (Ghosh et al. 2020). Invasive weeds like Lantana can be easily uprooted, converted to biochar in the site itself and used in the pit plantation technique during restoration of coal mine spoils (Kaur et al. 2018; Radhaboy et al. 2019). Thus, we propose that abundantly growing weeds in abandoned overburden dumps can be harnessed during the dry tropic summer months and converted to biochar. The biochar produced can be very easily applied in the pit plantation method. Biochar can be mixed with mine spoil and commercially important trees can be planted in such pits.
Influence of biochar in coal mine spoil enzymatic activities has not been reported widely, and experiments are essential for designing practical applicability in field scale for mine spoil quality and fertility enhancement. Thus, the influence of biochar application on enzyme activity in coal mine spoil and its correlation with the soil organic matter and mine spoil CO2 flux need attention. The study tested the hypothesis that biochar produced from invasive weeds will improve enzymatic activities, CO2 flux and fertility of coal mine spoil. The objectives of this study were to assess the impact of Lantana biochar (1) on the mine spoil physio-chemical properties after 6 months incubation in pilot scale application; (2) mine spoil enzymatic activity involved in C cycling and respiration, and (3) the economics involved for its application in a field scale.
2 Materials and methods
2.1 Biochar production and characterisation
Lantana camara growing in the coal mine overburden dumps was used as feedstock for biochar preparation. Feedstock was pyrolysed in a pre-heated muffle furnace at 450 ℃ for 60 min residence time. Biochar yield was calculated by the formula: yield (%) = weight of biochar (g)/weight of feedstock (g) × 100. pH and electrical conductivity of the filtrate (1:5; solid: deionised water; w/v) were measured by using multi-parameter probe (HI-2020, Hanna Instruments, India) (Singh et al. 2010). Organic carbon was determined by the potassium dichromate oxidation method (Walkley and Black 1934). The elemental analysis (C, H and N) was done using a CHNS-O Elemental Analyser-Euro vector EuroEA 3000, Italy. Bulk density and porosity of the biochar samples were estimated by using a column experiment (Ghosh et al. 2020). The surface morphology of the biochar was determined by field-emission scanning electron microscopy (FE-SEM Supra55, Carl Zeiss, Germany) and an FTIR (Fourier-transform infrared spectrophotometer) spectrum was used to analyse surface functional groups present in the biochar samples.
2.2 Collection of coal mine spoil
Coal mine spoil sampling was done from Tetulmari opencast project (OCP) (23° 48′ 210″ N and 86° 20′ 27″) Dhanbad (Fig. 1). Spoil samples were sieved in the field itself (< 2 mm) to get rid of the large boulders and rocks present in the mining site. Bulk spoil samples were brought back to the institute campus to conduct a pilot scale study on the pit application of biochar.
2.3 Coal mine spoil characterisation
pH and EC of the mine spoil were determined with a multiparameter probe (HI-2020, Hanna Instruments, India) in a spoil and water slurry (spoil: water, 1:2.5, w/v). Organic carbon (OC) was calculated by chromic acid wet oxidation method (Walkley and Black 1934). Cation exchange capacity (CEC) was determined by the ammonium acetate method (Jackson 1973). A Kjeldahl distillation unit (KJELODIST-EAS VA, Pelican equipment's Inc. India) was used for available nitrogen (Av-N) content in the mine spoil. Total C, H and N were measured by CHNS-O Elemental Analyser (Eurovector EuroEA 3000, Italy) by flash combustion technique (980 °C). Water holding capacity (WHC) was determined by placing the spoil samples in a Keen box (5.6 × 1.6 cm) in a water bath for 24 h (Maiti 2013). Bulk density (BD) was measured by soil core method (Maiti, 2013). Available potassium and sodium were determined by ammonium acetate method. Available phosphorus was extracted by alkaline sodium bicarbonate (pH 8.5) and measured by a UV–VIS Spectrophotometer, Shimadzu Corporation, Japan (Ahirwal et al. 2017).
2.4 Experimental design of biochar amendment study
The experimental design was a completely randomised block design with mine spoil as control and biochar amendment at 2% and 3% (w/w) with five replicates for each dose (Fig. 2a). The mine spoil samples collected from the field were air dried at room temperature in laboratory condition (25–25 °C), and divided into three equal parts. One part was mixed with biochar at 2% (w/w) and another with 3% (w/w). The control and amended mine spoil samples were placed in PVC lined pits each of 45 cm × 45 cm × 30 cm dimension (Fig. 2b). A total of 15 (3 amendments × 5 replicates) such pits were prepared in the ESE department of Indian Institute of Technology (Indian School of Mines) Dhanbad campus. A PVC collar (11 cm in height and 21 cm in diameter) was placed up to a depth of 5 cm at each plot having overburden dump as shown in Fig. 2b, c. Initially, distilled water was added to obtain a field capacity of 50%. The coal mine spoil was left naturally to incubate for 6 months in mid-June. The spoil enzymatic activity was measured on the beginning in June, mid-way in August (3 months) and the end of incubation in November (6 months). After the incubation period, mine spoil respiration was measured for 4 months (November–February).
2.5 Enzyme activities
The mine spoil samples were analysed for four enzymatic activities and the changes that occurred after 3 and 6 months of incubation were measured. The soil enzymatic activities such as dehydrogenase (DHA), invertase, amylase and cellulase were measured. Glasswares were previously treated by 2 M nitric acid for 24 h then further cleaned with distilled water. All chemical and reagents used were of analytical grade. DHA was determined by reducing triphenyl tetrazolium chloride (TCC) to triphenyl formazane (TPF) (Casida Jr 1977). The results were expressed as µg TPF g−1 soil 24 h−1. For invertase (EC 3.2.1.26), amylase (EC 3.2.1.1) and cellulase (EC 3.2.1.4) activities, soil samples were incubated in sucrose, starch and carboxy methyl cellulose substrate, respectively, with Sorensen’s buffer (pH 5.5, 0.06 M) for 24 h at 30 °C. The produced sugar was measured colometrically by 3, 5–dinitrosalicylic acid (Arey, 2010). The activities of invertase, amylase and cellulase were finally represented by µg glucose per g soil after 24 h.
2.6 Geometric mean of enzyme activities
The geometric mean of enzyme activities (GMea) can be used as an index to quantify the all enzymatic activity values in a single numerical value (García-Ruiz et al. 2008; Paz-Ferreiro et al. 2012). Literature review suggested that the reported GMea are of agricultural or garden soil, its value in coal mine degraded soil has not been reported yet (Zhang et al. 2015). Thus, GMea of the assayed enzymes activities of the biochar amended soil was calculated by Eq. 1:
where, DHA, I, A and C are dehydrogenase, invertase, amylase and cellulase activities, respectively.
2.7 Measurement of CO2 flux
CO2 flux was monitored in control plot (only coal mine spoil) and biochar amended mine spoil (2–3%) which was incubated for a period of 6 months. Spoil CO2 flux was monitored by LICOR LI-8100 (LICOR Inc. Lincoln, USA). A PVC collar (11 cm in height and 21 cm in diameter) was inserted up to a depth of 5 cm at each plot (Fig. 2b). The observations for CO2 flux were taken for a period of four months (120 days) from November 2018 to February 2019. 12 h data from 8:00 to 20:00 (+ 5:30 GMT) was measured at 20 min interval, each lasting for 90 s. LI-8100 PC Client v3.0.1 windows interface software was used for the operation of the CO2 flux system using Lenovo laptop.
Spoil temperature was recorded by inserting a temperature probe into the spoil (type E thermocouple, p/n 8100–201; 6.4 mm in diameter, 25 cm in length). Spoil moisture was recorded by placing a soil moisture probe up to a depth of 5 cm (ECHO Model EC-5, p/n 8100–202; 5 cm in length). Relative humidity was generated by the instrument itself. The moisture probe generated the output in mV (500–1300 mV) from which volumetric water content (VWC) was measured by Eq. 2:
Millivolts (mV) vs. moisture content (%, w/w) was in the range of 640 mV to 902 mV and corresponding moisture content ranged from 1.2% to 16.6%. The regression equation used is as follow (Eq. 3):
2.8 Statistics
One-way ANOVA (analysis of variance) was used to compare the means of biochar and mine spoil characteristics. Differences between individual means were evaluated using Duncan's multiple range test at p < 0.05. Pearson’s correlation analysis with a significance level of p < 0.01 and p < 0.05 was performed to identify the correlation between variables. For all statistical analysis MS-Excel and IBM-SPSS 2019 software were used.
3 Results and discussions
3.1 Biochar properties
The general properties of biochar are given in Table 1. A 54.9% yield of biochar was obtained at this pyrolysis condition from Lantana feedstock. Biochar obtained had an alkaline pH (9.34) and EC of 4.76 mS cm−1. The total elemental carbon, nitrogen, hydrogen and oxygen were 51.64%, 6.08%, 5.88% and 7.64%, respectively. The low H/C ratio (0.11) of Lantana biochar represents its high degree of aromatisation while the low O/C ratio (0.14) represents the polarity of the biochar. The C/N ratio of 9.18 indicates that the pyrolysis improves organic matter which enhances the labile carbon, thus making it available for microbial activity in the spoil. The ratios are indicative of the prolonged stability of carbon in the soil.
The FE-SEM of the biochar revealed a highly porous morphology (Fig. 3). This provided a large surface area and substrate for microbial activity and nutrient accumulation. The FTIR spectra showed five strong peaks representing various functional groups. The O–H bond at 3391 cm−1 was prominent due to the breaking of hydrogen bonded hydroxyl groups at higher temperatures (Fig. 4). Other bonds such as –CH3 (2924 cm−1), –CH2 (2870 cm−1), C=O (1600–1700 cm−1) due to cellulose of the feedstock were also present.
3.2 Changes in mine spoil physio–chemical properties
The change in the physio–chemical properties due to Lantana biochar amendment in a coal mine spoil has been shown in Table 2. Electrical conductivity showed a steep rise from 90.06 mS cm−1 in spoil to 721.66 mS cm−1 at 3%, w/w application rate (p < 0.05). The increase in cation exchange capacity from control to 3% (w/w) amendment (7.24 vs. 15.85 Cmol kg−1) might be responsible for the steep rise in electrical conductivity. Mohan et al. (2018) reported the similar results by rice husk and corn cover biochar application. The organic carbon increased with increasing biochar application rate in the mine spoil, and significant increase (p < 0.05) of 192% was observed at 3% (w/w) biochar amendment compared to the control (Table 2). Gonzaga et al. (2018) reported similar results by the application of coconut husks and pine wood chips biochar. Total carbon increased from 3% in mine spoil to 21% at 3% (w/w) biochar application rate. Total N and H did not show any significant changes (p < 0.05) by biochar application. Water holding capacity (27%) and bulk density (decreased by 13%) were also effected significantly by its application at 3% (w/w). Fellet et al. (2011) reported an increase in water-holding capacity by 5% at 10% biochar application in a mine spoil. In another mine spoil, Kelly et al. (2014) reported a decrease in bulk density decreased by 16.4% and 19.7% for soil sampled from different sites.
3.3 Effect of biochar amendment on enzymatic activities of coal mine spoil
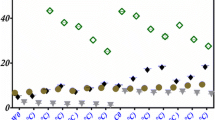
3.3.1 Dehydrogenase activity
Effect of biochar application on the spoil enzymatic activities at the end of 6-month incubation study has been shown in Fig. 5a. A significant increase (p < 0.05) of 83% was observed in dehydrogenase (DHA) value at 3% (w/w) compared to control. DHA values at 2 and 3% (w/w) application rates were significantly different during the middle of the study. DHA is considered to be the index of microbial metabolic activity and most importantly the indicator of respiration of the soil) (Maiti 2013; Gascó et al. 2016). Mukhopadhyay et al. (2020) reported that yard waste biochar addition at 10% (w/w) increased the coal mine spoil DHA activity from 34.02 µg TPF g−1 soil 24 h−1 in control to 59.71 µg TPF g−1 soil 24 h−1. Wang et al. (2015a, b) reported similar findings in DHA activity by addition of biochar produced from maize at 300–600 °C. Liu et al. (2018) also reported a positive effect of coconut shell @5% (w/w) on the DHA activity in a soil incubation experiment.
3.3.2 Invertase activity
Invertase activity was also found to increase significantly (p < 0.05) up to 2% (w/w) biochar application while a decreased was observed at 3% biochar amendment (Fig. 5b). At 2% (w/w) invertase activity increased from 3 µg glucose g−1 soil 24 h−1 in the control mine spoil in the beginning of the experiment to 14 µg glucose g−1 soil 24 h−1 after 3 and 6 months of the experiment. However, at 3% (w/w) application rate, there were no significant changes in the invertase activity. Invertase enzyme in microorganisms catalyses the hydrolysis of sucrose to glucose and fructose. Thus, the changes in the invertase activity in the spoil act as an indicator of microbial activity. Zhang et al. (2015) reported an increase in invertase activity by using rice straw and peanut hull biochars in an acidic spoil. Similarly, Khadem and Raiesi, (2017) found a 1.3- to 5.8-fold increase in invertase activity by corn stalk biochar in calcareous soil. A decrease in enzymatic activity beyond 2% (w/w) biochar amendment can be due to its ability to adsorb organic and inorganic molecules. Nie et al. (2018) reported that sugarcane and bagasse biochar at different doses (1.5, 2.25 and 3.0 t ha−1), increased the invertase activity 1.2, 1.5 and 1.7 times, respectively, compared to the control. At higher level of application, biochar particles might inhibit soil enzymes or their corresponding substrates by blocking their reaction sites (Bailey et al. 2011).
3.3.3 Amylase activity
Amylase activity increased significantly (p < 0.05) up to 2% (w/w) application rate; beyond this threshold a steep fall was observed at 3% biochar amendment (Fig. 5c). At 2% (w/w), the amylase activity increased form 11 µg glucose g−1 soil 24 h−1 in control to 38 µg glucose g−1 soil 24 h−1 after 3 months to 44 µg glucose g−1 soil 24 h−1 at the end of the incubation study. However, at 3% (w/w), a decrease in amylase activity was observed compared to the control. Awasthi et al. (2016) reported that biochar produced from wheat straw significantly improved amylase activity in a substrate. Amylase plays an important role of catalysing the hydrolytic depolymerisation of polysaccharides such as starch in soil to simple glucose molecule. Its presence is significantly correlated with microbial populations and moisture content. A decrease in the amylase activity beyond 2% (w/w) indicates that an application rate beyond this negatively affected microbial population responsible for amylase secretion in the spoil. At higher application rate, enzymes may be inactivated in biochar amended soils, by blocking or absorption of the substrate (Bailey et al. 2011).
3.3.4 Cellulase activity
In the current study, cellulase activity had a positive relation with biochar application (Fig. 5d). At 3% (w/w) of biochar amendment, the cellulase activity increased from 0.06 µg glucose g−1 soil 24 h−1 in control to 0.4 µg glucose g−1 soil 24 h−1 in the middle of the study period and 0.6 µg glucose g−1 soil 24 h−1 at the end of the incubation study. An average increase in 70% and 78% was observed at 2% and 3% (w/w) biochar application, respectively. A number of biochar-based studies reported that biochar application significantly stimulated the cellulase activity in the soil (Awasthi et al. 2016; Khadem and Raiesi 2017). Cellulase is another important enzyme involved in C-metabolism and produced by soil microbes that catalyse the decomposition of cellulose and some other related polysaccharides. The presence of cellulase indicates a nutrient-rich soil with enriched microbial activity.
3.4 Effect of biochar in geometric mean of enzyme activities
The geometric mean of enzyme activities (GMea) is an index to summarise the soil enzymes activities having various values and units (Paz-Ferreiro et al. 2012). GMea of the unamended mine spoil was 1.87 while it increased to 4.51 at 2% (w/w) and 3.25 at 3% (w/w) biochar amendments, clearly stating the fact that Lantana biochar at 2% application is best suited for improving the spoil biochemistry (Fig. 5e). The improvement in the GMea is related to soil organic carbon and microbial activity due to biochar application (Lehmann 2007) or by direct interaction between the enzymes and the biochar surfaces (Lehmann 2007).
3.5 Effect of biochar amendment on CO2 flux
The effect of biochar amendments at 2% (w/w) and 3% (w/w) on CO2 flux, surface CO2 concentrations, moisture, temperature and relative humidity at 5 cm depth was recorded for a 4-month period (Table 3). The control had much higher average CO2 flux value of 4.92 μ mol CO2 m−2 s−1 compared to 2.85 and 2.60 μ mol CO2 m−2 s−1 in 2% and 3% (w/w) biochar amendment. The CO2 flux value showed a significant (p < 0.05) 50% decrease by Lantana biochar application. It was found to be highly correlated with organic carbon, GMea, cellulase and DHA activity (Table 2). Studies report that changes in CO2 flux by biochar application is influenced by feedstock, pyrolysis temperature, substrate type and also the age of biochar (Gascó et al. 2019). Biochar amendment significantly increased the CO2 flux due to the increase in the organic matter in the substrate. On the other hand, wood biochar has been reported to reduce the CO2 emission by about 42.8%, and a mixture of saw dust and coffee husk biochar reduced the CO2 emissions by about 50% (Awasthi et al. 2016). Biochar prepared at higher temperature are generally known to be highly aromatic with high recalcitrant carbon (Al-Wabel et al. 2013). Majumder et al. (2019) reported that biochar had dual effects on soil carbon mineralization, depending upon the source material and pyrolysis temperature. In the present study, the mine spoil was incubated in natural conditions for 6 months, during this period the labile fraction of the biochar was acted upon by microbial activity leaving behind the high recalcitrant carbon. Thus, the 6-months aged biochar on the mine spoil had a lower CO2 flux than the unamended substrate.
3.6 Volumetric water content, relative humidity, spoil moisture and temperature
Incubated spoil moisture content was highest at 3% (w/w) amendment and least in control (Table 3). Mean relative humidity was also found to be highest at 3% (w/w) amendment (69%) and lowest in the control (45%) (Table 3). Biochar application in spoil affects its water holding capacity by modifying soil texture and structural properties. In addition, internal porosity also stores plant available water which in turn affects soil water retention properties (Rasa et al. 2018). The daily mean spoil temperatures at a 5-cm depth were 22.5, 20 and 19 °C for control, 2%, and 3%, respectively, with clear diurnal differences. The possible changes in spoil temperature resulting from biochar addition might affect other biophysical–chemical properties of the spoil.
3.7 Monthly variation
Spoil respiration was recorded for the months of November to February. In the control mine spoil, CO2 flux initially decreased from November to January, and started increasing after January. However, in the biochar amended mine spoil, a decrease was observed after November, which eventually remained stable up to the month of February (Fig. 6a). Similarly, the cumulative CO2 flux in the control mine spoil increased over the period of time while the biochar amended spoil had a lower cumulative CO2 flux value (Fig. 6b). A review conducted by Liu et al. (2016) reported that a positive response of soil CO2 flux was observed in the soil without vegetation and the unfertilised soil treated with manure and crop residue biochars. An increase in CO2 emission followed by stabilisation by poultry litter biochar was reported by Van Zwieten et al. (2013). The average surface CO2 showed an opposite trend, and the highest value was observed at 3% (w/w) biochar amendment and the least in unamended (Fig. 7c). However, the control and 2% (w/w) values did not vary significantly. Spoil moisture content was highest in biochar amended mine spoil during the period of study (Fig. 4d). Luo and Gu (2016) reported that peanut hull biochar at 3% (w/w) application rate increased the cumulative CO2 emission due to the enhanced organic carbon mineralisation caused by biochar addition. Similarly, Deng et al. (2020) reported that biochar addition increased carbon substrate for microbial activity which stimulated mineralisation and oxidation of biochar carbon and hence CO2 emission.
4 Correlation of GMea, CO2 flux and coal mine spoil properties
Pearson’s correlation showed all the enzymatic activities and GMea were significantly correlated with organic carbon (Fig. 7). Many studies have reported a positive correlation between the soil enzymatic activities and organic matter contents (Khadem and Raiesi 2017). Soil enzymes catalyse a large number of important reactions such as decomposition of soil organic carbon. A positive correlation between extracellular enzymes such as DHA (r = 0.95), amylase (r = 0.75), invertase (r = 0.86) and cellulase (r = 0.88) and organic carbon was observed.
Correlation analysis showed that enzymatic activities were positively correlated with biochar application and mine spoil organic carbon (Fig. 8a). A similar trend was also observed for CO2 flux (Fig. 8b). Biochar amendment and GMea and CO2 flux were related with R2 of 0.964 and 0.81, respectively (p < 0.05). Similarly, GMea (R2 = 0.84, p < 0.05) and mine spoil organic carbon (R2 = 0.96, p < 0.05) also showed positive correlation by biochar amendment. The results indicate that biochar addition to mine spoil might generally increase the activities of enzymes associated with carbon mineralization.
5 Implication of the study
The main objective of the current study was to test the feasibility of biochar application in a pit plantation method of mine reclamation. The pit size is generally 45 cm × 45 cm × 30 cm and filled with OB/topsoil along with tree sapling (Maiti 2013). Pit plantation with fruit bearing/ timber trees saplings along with biochar will promote plant growth and immobilise potentially toxic metals (Ghosh and Maiti 2020a, b). Thus, from the current study it can be concluded that biochar at 2% (w/w) application rate in a pit can ameliorate mine spoil properties for supporting plant growth.
Thus, from the findings of the present pilot scale study, it can be estimated that in 1 ha of mine degraded land, if the pits are at distance of 2.5 m, there will be a total of 1600 pits (100/2.5 in each row × 100/2.5 in each column). The diagrammatic representation of a 1 ha mine spoil is shown in Fig. 9. Each pit has a volume of 47,006 cm3 (45 cm × 45 cm × 30 cm) and the bulk density of the mine spoil is 1.2 g cm−3 (Table 1). Thus, 56 kg of mine spoil would be required in each pit and considering biochar application at 2% (w/w), 1.12 kg of biochar would be required per pit. Thus, the estimated amount of biochar required for 1 ha mine spoil remediation would be 1792 kg or 1.7 t ha−1. This method of pit plantation would reduce biochar requirement many folds, as the conventional spreading method has been reported to use 5–60 t ha−1 biochar application for amendment purpose (Ghosh and Maiti, 2020a, b).
In a mining area, before the eco-restoration practices start, the extracted overburden materials are often left for 1–2 years. During this period, the invasive weeds like Lantana grow abundantly in these areas which cause problems due to allelopathy. These weeds can be uprooted during the dry tropical months before monsoon, sun-dried and converted to biochar by charcoal-pit production method which is labour extensive. The estimated unit cost (CE, USD t−1) for biochar production and application can be calculated by Eq. 2 (Dickinson et al. 2015; Ghosh and Maiti 2020a, b):
where, UA = unit labour input for feedstock harvest and biochar production (d t−1); UB = unit labour input for biochar transport (d t−1); UC = unit labour input for biochar application to plantation pit (d t−1); Ci = labour cost (USD d−1).
Based on the above equation, the cost estimations have been shown in Table 4. The cost of biochar application is 120 USD t ha−1 by pit plantation method.
For upliftment of livelihood of stakeholders and employment generation, plantation of timber and fruit bearing trees having economical significance are often suggested. Also, plantation of native species will promote species richness and will promote better growth (Maiti and Ghosh 2020). Fruit bearing trees such as Psidium guajava (Guava), Mangifera indica (Mango), Artocarpus heterophyllus (Jackfruit), Syzygium cumini (Black plum) and Citrus spp. can provide economic benefits in the long run. Apart from those multipurpose trees such as Albizia lebbeck (Siris), Tectona grandis (Teak), Dalbergia sissoo (Shisham) and Gmelina arborea, Beechwood has high economic significance. Thus, the cost of fruit bearing tree sapling 50% (@₹25) and timber saplings 50% (@₹35) will cost 640 USD ha−1 (Table 4). Therefore, the total cost of biochar production, preparation of plantation pit and plantation will cost 844 USD ha−1.
6 Conclusions
Lantana biochar significantly improved GMea up to 2% (w/w) application rate. At 3% (w/w) mine spoil physio-chemical properties such as organic carbon (65%), WHC (19%), CEC (54%), BD (25%) were improved significantly compared to the unamended mine spoil. In general, DHA and invertase activity were increased up to 3% biochar amendment while amylase and cellulase activities increased up to 2% amendment and eventually decreased beyond this level. Decreases in mine spoil CO2 flux by 42% and 47% by 2 and 3% biochar amendment respectively, compared to the control were observed. Thus, Lantana biochar effectively remediates the physio–chemical and biological aspects of degraded and can be easily applied during ecological restoration by pit plantation method. The application rate with this method will be 1.7 t ha−1, which is lower than the reported rates of application. The cost of biochar preparation, application and plantation in this pit plantation technique at 2% is estimated to be 844 USD t ha−1. If fruit orchards and timber bearing plants are used for plantation in mine spoil, it will provide livelihood to stakeholders.
Availability of data and materials
The datasets generated and/or analysed during the current study are not publicly available due as this is a part of an on-going Ph.D. thesis, but are available from the corresponding author on reasonable request.
References
Ahirwal J, Maiti SK (2018a) Assessment of soil carbon pool, carbon sequestration and soil CO2 flux in unreclaimed and reclaimed coal mine spoils. Environ Earth Sci. https://doi.org/10.1007/s12665-017-7185-5
Ahirwal J, Maiti SK (2018b) Development of Technosol properties and recovery of carbon stock after 16 years of revegetation on coal mine degraded lands, India. Catena 166:114–123. https://doi.org/10.1016/j.catena.2018.03.026
Ahirwal J, Maiti SK, Singh AK (2017) Changes in ecosystem carbon pool and soil CO2 flux following post-mine reclamation in dry tropical environment. India Sci Total Environ 583:153–162. https://doi.org/10.1016/j.scitotenv.2017.01.043
Ahirwal J, Kumar A, Maiti SK (2020) Effect of fast-growing trees on soil properties and carbon storage in an afforested coal mine land (India). Minerals 10(10):840. https://doi.org/10.3390/min10100840
Al-Wabel MI, Al-Omran A, El-Naggar AH, Nadeem M, Usman ARA (2013) Pyrolysis temperature induced changes in characteristics and chemical composition of biochar produced from conocarpus wastes. Bioresour Technol 131:374–379. https://doi.org/10.1016/j.biortech.2012.12.165
Ameloot N, De Neve S, Jegajeevagan K, Yildiz G, Buchan D, Funkuin YN, Prins W, Bouckaert L, Sleutel S (2013) Short-term CO2 and N2O emissions and microbial properties of biochar amended sandy loam soils. Soil Biol Biochem 57:401–410. https://doi.org/10.1016/j.soilbio.2012.10.025
Arey NC (2010) Manual of environmental analysis. Ane Books Pvt Ltd, New Delhi
Awasthi MK, Wang Q, Ren X, Zhao J, Huang H, Awasthi SK, Lahori AH, Li R, Zhou L, Zhang Z (2016) Role of biochar amendment in mitigation of nitrogen loss and greenhouse gas emission during sewage sludge composting. Bioresour Technol 219:270–280. https://doi.org/10.1016/j.biortech.2016.07.128
Bailey VL, Fansler SJ, Smith JL, Bolton H (2011) Reconciling apparent variability in effects of biochar amendment on soil enzyme activities by assay optimization. Soil Biol Biochem 43:296–301. https://doi.org/10.1016/j.soilbio.2010.10.014
Camps-Arbestain M, Amonette JE, Singh B, Wang T, Schmidt HP (2015) A biochar classification system and associated test methods. In: Lehmann J, Joseph S (eds) Biochar for environmental management: science, technology and implementation, 2nd edn. Routledge, London, p 165
Casida LE Jr (1977) Microbial metabolic activity in soil as measured by dehydrogenase determinations. Appl Environ Microbiol 34:630–636
Deng B, Shi Y, Zhang L, Fang H, Gao Y, Luo L, Feng W, Hu X, Wan S, Huang W, Guo X, Siemann E (2020) Effects of spent mushroom substrate-derived biochar on soil CO2 and N2O emissions depend on pyrolysis temperature. Chemosphere 246:125608. https://doi.org/10.1016/j.chemosphere.2019.125608
Dickinson D, Balduccio L, Buysse J, Ronsse F, van Huylenbroeck G, Prins W (2015) Cost-benefit analysis of using biochar to improve cereals agriculture. GCB Bioenergy 7:850–864. https://doi.org/10.1111/gcbb.12180
Ducey TF, Novak JM, Sigua GC, Ippolito JA, Rushmiller HC, Watts DW, Trippe KM, Spokas KA, Stone KC, Johnson MG (2021) Microbial response to designer biochar and compost treatments for mining impacted soils. Biochar. https://doi.org/10.1007/s42773-021-00093-3
Fellet G, Marchiol L, Delle Vedove G, Peressotti A (2011) Application of biochar on mine tailings: effects and perspectives for land reclamation. Chemosphere 83:1262–1267. https://doi.org/10.1016/j.chemosphere.2011.03.053
Frouz J, Prach K, Pižl V, Háněl L, Starý J, Tajovský K, Materna J, Balík V, Kalčík J, Řehounková K (2008) Interactions between soil development, vegetation and soil fauna during spontaneous succession in post mining sites. Eur J Soil Biol 44:109–121. https://doi.org/10.1016/j.ejsobi.2007.09.002
Futa B, Oleszczuk P, Andruszczak S, Kwiecińska-Poppe E, Kraska P (2020) Effect of natural aging of biochar on soil enzymatic activity and physicochemical properties in long-term field experiment. Agronomy 10:449. https://doi.org/10.3390/agronomy10030449
García-Ruiz R, Ochoa V, Hinojosa MB, Carreira JA (2008) Suitability of enzyme activities for the monitoring of soil quality improvement in organic agricultural systems. Soil Biol Biochem 40:2137–2145. https://doi.org/10.1016/j.soilbio.2008.03.023
Gascó G, Paz-Ferreiro J, Cely P, Plaza C, Méndez A (2016) Influence of pig manure and its biochar on soil CO2 emissions and soil enzymes. Ecol Eng 95:19–24. https://doi.org/10.1016/j.ecoleng.2016.06.039
Gascó G, Álvarez ML, Paz-Ferreiro J, Méndez A (2019) Combining phytoextraction by Brassica napus and biochar amendment for the remediation of a mining soil in Riotinto (Spain). Chemosphere 231:562–570. https://doi.org/10.1016/j.chemosphere.2019.05.168
Ghosh D, Maiti SK (2020a) Can biochar reclaim coal mine spoil? J Environ Manage 272:111097. https://doi.org/10.1016/j.jenvman.2020.111097
Ghosh D, Maiti SK (2020b) Biochar assisted phytoremediation and biomass disposal in heavy metal contaminated mine soils: a review. Int J Phytoremediat 10:1–8. https://doi.org/10.1080/15226514.2020.1840510
Ghosh D, Masto RE, Maiti SK (2020) Ameliorative effect of Lantana biochar on coal mine spoil and growth of maize (Zea mays). Soil Use Manag 36(4):726–739. https://doi.org/10.1111/sum.12626
Gonzaga MIS, Mackowiak C, de Almeida AQ, de Carvalho Jr JIT, Andrade KR (2018) Positive and negative effects of biochar from coconut husks, orange bagasse and pine wood chips on maize (Zea mays L.) growth and nutrition. Catena 162:414–420. https://doi.org/10.1016/j.catena.2017.10.018
Jackson ML (1973) Soil Chemical Analysis. PHI Pvt, Ltd, New Delhi
Kaur M, Kumar M, Sachdeva S, Puri SK (2018) Aquatic weeds as the next generation feedstock for sustainable bioenergy production. Bioresour Technol 251:390–402. https://doi.org/10.1016/j.biortech.2017.11.082
Kelly CN, Peltz CD, Stanton M, Rutherford DW, Rostad CE (2014) Biochar application to hardrock mine tailings: soil quality, microbial activity, and toxic element sorption. Appl Geochem 43:35–48. https://doi.org/10.1016/j.apgeochem.2014.02.003
Khadem A, Raiesi F (2017) Influence of biochar on potential enzyme activities in two calcareous soils of contrasting texture. Geoderma 308:149–158. https://doi.org/10.1016/j.geoderma.2017.08.004
Lehmann J (2007) A handful of carbon. Nature 447(7141):143–144. https://doi.org/10.1038/447143a
Lehmann J, Kuzyakov Y, Pan G, Ok YS (2015) Biochars and the plant-soil interface. Plant Soil 395:1–5. https://doi.org/10.1007/s11104-015-2658-3
Liu S, Zhang Y, Zong Y, Hu Z, Wu S, Zhou J, Jin Y, Zou J (2016) Response of soil carbon dioxide fluxes, soil organic carbon and microbial biomass carbon to biochar amendment: a meta-analysis. GCB Bioenergy 8:392–406. https://doi.org/10.1111/gcbb.12265
Liu H, Xu F, Xie Y, Wang C, Zhang A, Li L, Xu H (2018) Effect of modified coconut shell biochar on availability of heavy metals and biochemical characteristics of soil in multiple heavy metals contaminated soil. Sci Total Environ 645:702–709. https://doi.org/10.1016/j.scitotenv.2018.07.115
Luo L, Gu J (2016) Alteration of extracellular enzyme activity and microbial abundance by biochar addition: implication for carbon sequestration in subtropical mangrove sediment. J Environ Manage 182:29–36. https://doi.org/10.1016/j.jenvman.2016.07.040
Maiti SK (2013) Ecorestoration of the coalmine degraded lands. Springer, India. https://doi.org/10.1007/978-81-322-0851-8
Maiti SK, Ghosh D (2020) Plant–soil interactions as a restoration tool. In: Prasad MNV, Pietrzykowski M (eds) Climate change and soil interactions. Elsevier, pp 689–730, https://doi.org/10.1016/B978-0-12-818032-7.00024-2
Majumder S, Neogi S, Dutta T, Powel MA, Banik P (2019) The impact of biochar on soil carbon sequestration: meta-analytical approach to evaluating environmental and economic advantages. J Environ Manage 250:109466. https://doi.org/10.1016/j.jenvman.2019.109466
Masto RE, Kumar S, Rout TK, Sarkar P, George J, Ram LC (2013) Biochar from water hyacinth (Eichornia crassipes) and its impact on soil biological activity. Catena 111:64–71. https://doi.org/10.1016/j.catena.2013.06.025
Munir MA, Yousaf B, Ali MU, Dan C, Abbas Q, Arif M, Yang X (2021) In situ synthesis of micro-plastics embedded sewage-sludge co-pyrolyzed biochar: implications for the remediation of Cr and Pb availability and enzymatic activities from the contaminated soil. J Clean Prod 302:127005. https://doi.org/10.1016/j.jclepro.2021.127005
Mohan D, Abhishek K, Sarswat A, Patel M, Singh P, Pittman CU (2018) Biochar production and applications in soil fertility and carbon sequestration-a sustainable solution to crop-residue burning in India. RSC Adv 8:508–520. https://doi.org/10.1039/c7ra10353k
Mukhopadhyay S, Masto RE, Singh AK, Singh PK (2020) Impact of the combined application of biochar and compost on mine soil quality and growth of lady’s finger (Abelmoschus esculentus). Bull Environ Contam Toxicol. https://doi.org/10.1007/s00128-020-03011-8
Nie C, Yang X, Niazi NK, Xu X, Wen Y, Rinklebe J, Ok YS, Xu S, Wang H (2018) Impact of sugarcane bagasse-derived biochar on heavy metal availability and microbial activity: a field study. Chemosphere 200:274–282. https://doi.org/10.1016/j.chemosphere.2018.02.134
Novak JM, Ippolito JA, Ducey TF, Watts DW, Spokas KA, Trippe KM, Sigua GC, Johnson MG (2018) Remediation of an acidic mine spoil: Miscanthus biochar and lime amendment affects metal availability, plant growth, and soil enzyme activity. Chemosphere 205:709–718. https://doi.org/10.1016/j.chemosphere.2018.04.107
Palansooriya KN, Wong JTF, Hashimoto Y, Huang L, Rinklebe J, Chang SX, Bolan N, Ok WH, YS, (2019) Response of microbial communities to biochar-amended soils: a critical review. Biochar 1(1):3–22
Pandey D, Daverey A, Arunachalam K (2020) Biochar: production, properties and emerging role as a support for enzyme immobilization. J Clean Prod 255:120267. https://doi.org/10.1016/j.jclepro.2020.120267
Paz-Ferreiro J, Gascó G, Gutiérrez B, Méndez A (2012) Soil biochemical activities and the geometric mean of enzyme activities after application of sewage sludge and sewage sludge biochar to soil. Biol Fertil Soils 48:511–517. https://doi.org/10.1007/s00374-011-0644-3
Radhaboy G, Pugazhvadivu M, Ganeshan P, Ramshankar P (2019) Analysis of thermo chemical behaviour of Calotropis procera parts for their potentiality. Int J Ambient Energy. https://doi.org/10.1080/01430750.2019.1630309
Raiesi F, Khadem A (2019) Short-term effects of maize residue biochar on kinetic and thermodynamic parameters of soil β-glucosidase. Biochar 1(2):213–227
Rasa K, Heikkinen J, Hannula M, Arstila K, Kulju S, Hyväluoma J (2018) How and why does willow biochar increase a clay soil water retention capacity? Biomass Bioenerg 119:346–353. https://doi.org/10.1016/j.biombioe.2018.10.004
Sandhu S, Sekaran U, Ozlu E, Hoilett NO, Kumar S (2019) Short-term impacts of biochar and manure application on soil labile carbon fractions, enzyme activity, and microbial community structure. Biochar 1(3):271–282. https://doi.org/10.1007/s42773-019-00025-2
Singh B, Singh BP, Cowie AL (2010) Characterisation and evaluation of biochars for their application as a soil amendment. Soil Res 48(7):516–525. https://doi.org/10.1071/SR10058
Ussiri DAN, Lal R (2008) Method for determining coal carbon in the reclaimed minesoils contaminated with coal. Soil Sci Soc Am J 72:231–237. https://doi.org/10.2136/sssaj2007.0047
Van Zwieten L, Kimber SWL, Morris SG, Singh BP, Grace PR, Scheer C, Rust J, Downie AE, Cowie AL (2013) Pyrolysing poultry litter reduces N2O and CO2 fluxes. Sci Total Environ 465:279–287. https://doi.org/10.1016/j.scitotenv.2013.02.054
Walkley A, Black IA (1934) An examination of the Degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Sci 37(1):29–38
Wang X, Song D, Liang G, Zhang Q, Ai C, Zhou W (2015a) Maize biochar addition rate influences soil enzyme activity and microbial community composition in a fluvo-aquic soil. Appl Soil Ecol 96:265–272. https://doi.org/10.1016/j.apsoil.2015.08.018
Wang X, Zhou W, Liang G, Song D, Zhang X (2015b) Characteristics of maize biochar with different pyrolysis temperatures and its effects on organic carbon, nitrogen and enzymatic activities after addition to fluvo-aquic soil. Sci Total Environ 538:137–144. https://doi.org/10.1016/j.scitotenv.2015.08.026
Zhang Y, Tan Q, Hu C, Zheng C, Gui H, Zeng W, Sun X, Zhao X (2015) Differences in responses of soil microbial properties and trifoliate orange seedling to biochar derived from three feedstocks. J Soils Sediments 15:541–551. https://doi.org/10.1007/s11368-014-1032-z
Acknowledgements
The authors would like to acknowledge IIT (ISM), Dhanbad and MHRD, Government of India for giving contingency funds for research and scholarship to the first author (17DR000426).
Funding
The authors would like to acknowledge IIT (ISM), Dhanbad and MHRD, Government of India for giving contingency funds for research and scholarship to the first author (17DR000426). The authors would also like to thank the reviewers and editors for their insightful comments during the review process.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Dipita Ghosh. Prof. Subodh Kumar Maiti was the overall supervisor of the work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Ghosh, D., Maiti, S.K. Effect of invasive weed biochar amendment on soil enzymatic activity and respiration of coal mine spoil: a laboratory experiment study. Biochar 3, 519–533 (2021). https://doi.org/10.1007/s42773-021-00109-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42773-021-00109-y