Abstract
Sewage water from an urban township in India has been used as a growth media for cultivation of a green alga Chlorella ellipsoidea in relation to phycoremediation and integrated approach for production of antioxidant and value added products. The concentration level of nitrate, phosphate, ammonia and total dissolved solid got significantly reduced in the sewage water media at post-stationary phase. An increased level of dissolved oxygen was observed on 30th day of incubation. The alga was efficient in the uptake of nutrients from sewage water. Most notably, complete removal of total dissolved solid was observed. Phaeopigment and physiological stress indices were virtually unaltered in most cases indicating facile adaptability. The stationary phase of the algal species was stable for 40 days as against only 22 days in BG11(N+) medium. Dry cell weight of the sewage water grown alga has been found to be about five times higher than that grown in BG11(N+) medium. The biomass accrued from sewage water as growth medium showed around 25% carbohydrate, 35% lipid and 56% protein and relatively higher amount of enzymatic and non-enzymatic antioxidants.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Algae are well recognised for their potential role in biotransformation of pollutants, nutrients and xenobiotic from waste water and carbon dioxide from waste air (Babaei et al. 2018). The release of wastewater to the receiving natural water bodies poses serious environmental challenges (De-Bashan and Bashan 2010). Microalgae by way of nutrient removal can help counter eutrophication in the aquatic ecosystem and are unique in sequestering carbon dioxide (Olguín 2003). The domestic sewage water being rich in organic carbon, nitrogen, phosphorus and other compounds supports the growth of microalgae (Liang et al. 2013).
Like fungi and bacteria, many algae are capable of sorbing heavy metals from polluted waters (Mehta and Gaur 2005; Yadav et al. 2017). Microalgae cultivation are now-a-days integrated with various wastewater as growth media to reduce the production cost of biomass. Apart from being cost-effective, recovering nutrients from waste water through microalgae assimilation is also considered a viable and sustainable option. Growing algae using sewage water (Renuka et al. 2013; Singh et al. 2017; Bansal et al. 2018) have been documented. The technique is considered efficient in producing high value added products such as proteins, carbohydrates, lipids, enzymatic, non-enzymatic antioxidants and biofuels as an alternative source of energy (Kumar et al. 2014; Marchão et al. 2017). Blend of organic and inorganic materials, sewage water generally contains carbohydrates, fats, proteins, amino acids, and volatile acids besides inorganic ions such as sodium, calcium, potassium, magnesium, chlorine, sulphur, phosphate, bicarbonate, ammonium salts and heavy metals (Tebbutt 1983). Silchar, a class II urban township in Assam state in India alone produces 8000 kilo litres of sewage water daily which is generally released into the river Barak (CPCB 1999; Datta Ray et al. 2000). Municipal wastewater is replete with pathogenic and non-pathogenic bacteria besides various toxin compounds (Shannon et al. 2007). Wastewater treatment by physico-chemical means are quite expensive, cumbersome and often not feasible. Thus low-cost effective biological waste treatment methods have drawn immense attention of researchers across the world (Yadav et al. 2017; Babaei et al. 2018). All algal species, however, cannot thrive in wastewater environment and selection of algal species is very crucial in the context of wastewater treatment (Pittman et al. 2011). The nutrient uptake potential of genus Chlorella from wastewater has been explored rather extensively (Megharaj et al. 1992; Marchão et al. 2017; Salgueiro et al. 2018).
Accordingly we report herein an assessment of growth of an algal species Chlorella ellipsoidea using raw sewage water of Silchar town located in the state of Assam, India in relation to phycoremediation, biomass production via an integrated approach of nutrient uptake process.
Materials and methods
Collection and analyses of domestic sewage water sample
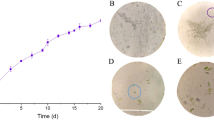
Domestic sewage water samples were collected in clean polythene bottles from the apartment’s outlets of Vivekananda road, Silchar town, Assam (Fig. 1a).The colour of the domestic sewage water was ‘greyish’ with BOD and COD values being 600 ± 0.18 mg/l and 1520 ± 0.18 mg/l. (Table 1), respectively. The samples were filtered to remove any suspended particles and stored at 4 °C until further use. Standard protocols were followed for analysis of domestic sewage water (APHA 2005).
Microalgae and growth condition
Chlorella ellipsoidea (AUS/EC/JR/PSGA294) was collected from submerged polythene surface in domestic sewage water of Silchar town, Assam state in India and pure culture developed. The algal species (Fig. 1b) was maintained in nitrogen enriched BG11(N+) media under continuous illumination (2000 lx) at 24 ± 1 °C (Rippka et al. 1979). The isolated microalga was identified using standard keys (Prescott 1952). The C. ellipsoidea was grown in varying concentration of domestic sewage water with five treatments (Fig. 1c) including BG11(N+) media (Table 2).The microalga grown in BG11(N+) medium served as a control. The flasks for phycoremediation series in different amount of domestic sewage water is shown in Fig. 1d. Flasks were shaken twice a day to avoid adherence of cells at bottom surface of conical flask.
The culture was thoroughly vortexed and cell count was performed using a Neubauer haemocytometer. The chlorophyll a (Chl a) was determined spectrophotometrically (Strickland and Parsons 1968).
Nutrient removal analysis
The nutrient removal capacity of C. ellipsoidea was assessed at the first and last day of the experiment by measuring physico-chemical properties of domestic sewage water. The removal rate of NO3− and PO43− were calculated as per equation: removal rate ( mg L−1 day−1) = Ci − Cf/t; where Ci and Cf are initial and final concentration of nutrients or DO and t is the time duration.
DCW of C. ellipsoidea
On 30th day of treatment, 10 ml of homogenised culture were filtered through pre-weighed Whatman filter (grade 42) papers and dried at 60 °C until constant weight of DCW was obtained.
Biochemical analysis of C. ellipsoidea
The total carbohydrate was determined according to anthrone method (Spiro 1966) and total protein was determined following modified folin’s method (Herbert et al.1971). Lipid estimation has been carried out as per Bligh and Dyer (1959).
Estimation of enzymatic and non-enzymatic antioxidants
Catalase activity was estimated according to Aebi (1984). Peroxidase activity was assayed by the method of Kar and Mishra (1976). Superoxide dismutase (SOD) activity was determined as per method of Van Rossum et al. (1997). Glutathione peroxidase activity was estimated according to standard procedure (Rotruck et al. 1973). Ascorbic acid contents were determined as per Roe and Kuether (1943). Glutathione reductase was assayed by the method of Schaedle and Bassham (1977).
Phaeopigment and physiological stress indices
Physiological stress indices (chlorophyll to phaeophytin ratio) of C. ellipsoidea grown in domestic sewage water and domestic sewage water + BG11(N+) treatments were determined according to the standard method (Megateli et al. 2009).
Conversion of phaeophytin from chlorophyll was effected by addition of 10µL HCl (35% GR, Merck) to 3 ml of extract. The ratios, D430/D410 (Phaeopigment Index), D430/D665 (Margalef Index I), D480/D665 (Margalef Index II) were evaluated (Martinez-Abaigar and Nùñez-Oliveira 1998).
Statistical analyses
Correlation study was performed using the Software Statistical Package for Social Sciences (SPSS Version 21.0). Data are average of three experiments ± S.D, n = 3.
Results and discussion
Physico-chemical properties of domestic sewage water
The colour of domestic sewage water was greyish (Table 1). The temperature was 32 ± 0.56 °C. BOD, COD and DO of water was 600 ± 0.18, 1520 ± 0.18 and 2.2 ± 23 mg/L, respectively. The total alkalinity was 10 ± 1.4 mg/L, free CO2 was 36.98 ± 0.13 mg/L and TDS was found to be 500 ± 0.18 mg/L. The SS was 50 ± 0.54 mg/L. The chloride and calcium concentrations were found to be 60 ± 0.67 mg/L, 60 ± 0.13 mg/L, respectively. The sulphate, nitrate, magnesium and ammonia of sewage water was present at a concentration of 50 ± 1.6 mg/L, 12 ± 1.5 mg/L, 30 ± 0.23 mg/L and 30 ± 0.45 mg/L, respectively. The phosphate concentration was found to be 70 ± 0.45 mg/L. These results (Table 1) are in compliance with a recent study from this town (Sarmah and Rout 2017).
Effect of sewage water on cell number and Chl a
Growth study of C. ellipsoidea in different treatments (Fig. 2a) show the lag period and log period to be longest in raw domestic sewage water treatment. The treatment c showed a shorter lag period than that of raw sewage treatment. The treatment d, control BG11(N+) and the treatment e showed same log period. Stationary period was found to be the longest for the treatment b. Similar growth profiles were observed in the treatments d and e. Maximum cell number (1008 × 106cell/ml) was observed in the treatment b on 16th day of the treatment (Fig. 2). The cell number in domestic sewage water grown C. ellipsoidea were considerably higher than that of BG11(N+) grown culture. This suggests that C. ellipsoidea can grow well in sewage water without additional nitrogen source. This is in conformity with another species of C. vulgaris which showed higher growth in confectionery effluent water (Kumar et al. 2014).The C. ellipsoidea was found growing till 40 days, while the standard BG11(N+) medium registered growth till 22 days from inoculation. A wild-type Chlorella sp. isolated from freshwater was found to grow, adapt easily in diluted dairy manure samples in comparison to TAP media (Wang et al. 2010). In a related study, Chlorella sp. grown in municipal sewage water, 33% decline in growth rate was observed in comparison to standard media (Mutanda et al. 2011).
In this study, the peak point of growth of C. ellipsoidea was observed on day 16 of cultivation. The Chl a content was highest in the treatment b (raw sewage water), lowest in e (Fig. 2b). Due to high nutrient content, sewage water used in the present study may serve as a suitable media for cultivation of C. ellipsoidea. The pH was monitored daily for the different treatment series for a period of 30 days (Fig. 3). The alga reduced the dissolved CO2 concentrations of sewage water through photosynthesis which in turn raised the pH level. The growth of alga was affected by pH. High pH can lower the growth of the microalgae besides inhibiting the photosynthetic rate of the alga (Leavitt et al. 1999). The algal growth was measured in terms of Chl a content. A positive correlation was noted for the residual phosphate (r = 0.974**), nitrate (0.765**), ammonia (0.532**) and TDS (0.768**) and increased rate of DO (0.438*) in the medium with the Chl a concentration of the culture (algal biomass) (Table 3).
DCW of C. ellipsoidea
The DCW was quite higher (39.67 mg L−1) in sewage water compared to BG11(N+) medium (18.67 mg L−1) (Table 4), reflecting a significant increase in DCW (50%) in domestic sewage water in 4th day of the treatment. For 30th day of the treatments also, the dry cell weights in sewage water (998 mg L−1) were found to be higher compared to BG11(N+) medium (718 mg L−1). In a recent study, Parachlorella kessleri grown in municipal waste water has been shown to produce 50% more biomass as compared to TAP and ASW media (Singh et al. 2017).
Removal rate of nitrate, phosphate and ammonia
Nitrate, phosphate and ammonia removal rates were found to be maximum (4.79, 5.85, 4.05 mg L−1 day−1, respectively) in the treatment b (raw sewage water) as compared to BG11 (N+) media (Fig. 4). It is generally believed that there is direct correlation between chlorophyll production and nutrients removal efficiencies of alga in sewage water. In the present case, both parameters recorded highest in log period of C. ellipsoidea. The nutrient sequestration pattern suggests that removal efficiencies were associated with algal growth (Kim et al. 2013; Delgadillo-Mirquez et al. 2016; Babaei et al. 2018).This can be ascribed to nitrogen dependence of C. ellipsoidea from sewage water and large surface area and high binding affinity of algae. Cell wall of algae is considered as main binding site for nutrients and metals (Romero-Gonzalez et al. 2001). Phosphate removal rate was very high presumably owing to the utilization of phosphorus for their growth.
DO of sewage water
Dissolved oxygen, an important water quality parameter was found to have significantly high rate of enhancement (4.2 mg L−1 day−1) in the treatment b (raw sewage water) (Fig. 4). About 46% hike has earlier been observed in DO concentration in sewage water when incubated with Calothrix sp. (Renuka et al. 2013). In another recent study, 96% increase in DO rate was observed for Scenedesmus sp. in tannery waste water (Ajayan et al. 2015). It is evident that increased rate of DO associated with reduced dissolved CO2 concentrations through photosynthesis raises the pH level (Borowitzka 1998).
Removal rate of TDS
The maximum TDS removal rate (7.48 mg L−1 day−1) was observed in the treatment b, raw domestic sewage water, as compared to BG11(N+) media (Fig. 4). Notably, the species C. ellipsoidea in the present study, showed the potential to reduce the TDS below permissible limit. The TDS removal was found to be 41% for C. vulgaris in tannery wastewater (Das et al. 2017).
Phaeopigment and physiological stress
Stress condition in different plants such as algae (Marker et al. 1980), bryophytes (Spitale 2009), and vascular plants (Megateli et al. 2009) were calculated by measuring photosynthetic pigment composition. Photosynthetic pigment composition was calculated at the end day of the sewage water phycoremediation. Phaeopigment, Margalef I and Margalef II indices were calculated (Fig. 5). For the different treatments, phaeopigment and physiological stress indices revealed a trend of a > c > d > b > e. The Magalef index I and II also showed a similar trend indicating virtually no stress level for the alga. Similar low stress levels were found in phycoremediation of sewage water by Chlorella sp. (Renuka et al. 2013).
Biochemical contents
The carbohydrate content (35 ± 0.72%) of C. ellipsoidea grown in sewage water was higher compared to that of the BG11(N +) grown (30 ± 0.81%). Total protein content (56 ± 0.77%) of C. ellipsoidea grown in domestic sewage water was also higher than that of the BG11(N+) grown (43 ± 0.54%). Lipid content of C. ellipsoidea grown in sewage water (34 ± 0.67%) was relatively higher than that of the BG11 (N+) grown (24 ± 0.52%, Fig. 6). It is noteworthy that biochemical constituents of C. ellipsoidea were obtained 2–3 times higher as compared to those of BG11(N+) media maintained culture. The alga, Acutodesmus dimorphus was found to contain around 25% lipid and 30% carbohydrate (Chokshi et al. 2016). For C. sorokiniana, cultivated in municipal sewage water, an enhanced amount of biomass (77.14 mg L−1 day−1), lipid (24.91 mg L−1 day−1), carbohydrate (20.10 mg L−1 day−1) and protein (22.36 mg L−1 day−1) were observed (Ramsundar et al. 2017).
Enzymatic and non-enzymatic antioxidants
The sewage water grown C. ellipsoidea showed 56U/mg of protein of catalase, 54U/mg of protein of peroxidase, 63U/mg of protein of superoxide dismutase, 53U/mg of protein of glutathione peroxidase and the laboratory maintained C. ellipsoidea showed 53 U/mg of protein of catalase, 51 U/mg of protein of peroxidase, 60 U/mg of protein of superoxide dismutase and 47 U/mg of protein of glutathione peroxidase. Non-enzymatic antioxidants, 2.5 µg/mg of ascorbic acid, 69 U/mg of protein of glutathione reductase were recorded in C. ellipsoidea grown in sewage water. Ascorbic acid and glutathione reductase content in control were 1.18 µg/mg and 61 U/mg of protein, respectively. In a recent study, C. vulgaris cultivated in effluent of a confectionery waste water revealed similar results (Kumar et al. 2014). The non-enzymatic antioxidants were shown to be highly crucial in scavenging reactive oxygen species (ROS).
Conclusion
Cultivation of C. ellipsoidea algae in untreated municipal sewage water without any additional nutrients afforded remarkable growth and biomass production. The lipid rich green alga was able to effectively sequester nitrate and phosphate, increase the DO level, and lower the TDS well below the permissible limit. The study also demonstrate that the microalga is capable of efficiently remediate domestic sewage water, mitigate carbon dioxide as it grow proficiently in polluted water. High lipid production in sewage water relative to the control medium may be exploited for its feasibility in biofuel generation. The antioxidant produced by the alga during the remediation of domestic sewage water is anticipated to be of significance in pharmaceutical, food and cosmetic applications. Employing domestic sewage water to harvest algae for production of value added chemicals could thus serve as an integrated approach for manifold applications.
Data availability
All data generated or analyzed during this study are included in this published article.
Abbreviations
- DO:
-
Dissolved oxygen
- BOD:
-
Biological oxygen demand
- COD:
-
Chemical oxygen demand
- TDS:
-
Total dissolved solid
- SS:
-
Suspended solid
- DCW:
-
Dry cell weight
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Ajayan KV, Selvaraju M, Unnikannan P, Sruthi P (2015) Phycoremediation of tannery wastewater using microalgae Scenedesmus species. Int J Phytoremediation 17:907–916
APHA (2005) Standard methods for examination of water and wastewater, 21st edn. APHA, AWAA, WPCF, Washington
Babaei A, Mehrnia MR, Shayegan J, Sarrafzadeh M-H, Amini E (2018) Evaluation of nutrient removal and biomass production through mixotrophic, heterotrophic, and photoautotrophic cultivation of Chlorella in nitrate and ammonium wastewater. Int J Environ Res 12:167. https://doi.org/10.1007/s41742-018-0077-z
Bansal A, Shinde O, Sarkar S (2018) Industrial wastewater treatment using phycoremediation technologies and co-production of value-added products. J Bioremediat Biodegrad 9:1. https://doi.org/10.4172/21556199.1000428
Bligh EG, Dyer WJ (1959) A rapid method for total Lipid extraction and purification. Can J Biochem. physiol. 37:911–917
Borowitzka MA (1998) Limits to growth. In: Wong YS, Tam NFY (eds) Wastewater treatment with algae. Springer, Berlin, pp 203–226
Chokshi K, Pancha I, Ghosh A, Mishra S (2016) Microalgal biomass generation by phycoremediation of dairy industry wastewater: an integrated approach towards sustainable biofuel production. Bioresour Technol 221:455–460
CPCB (1999) Status of water supply and Wastewater Collection Treatment & Disposal in Class I Cities- 1999, Control of Urban Pollution Series:CUPS/44/1999-2000
Das C, Naseera K, Ram A, Meena RM, Ramaiah N (2017) Bioremediation of tannery wastewater by a salt-tolerant strain of Chlorella vulgaris. J Appl Phycol 29:235–243
Datta Ray B, Mazhari HK, Passah PM, Pandey MC (2000) Population, poverty, and environment in North-East India. Concept publishing company. ISBN 10:8170227704/ISBN 13:9788170227700
de-Bashan LE, Bashan Y (2010) Immobilized microalgae for removing pollutants: review of practical aspects. Bioresour Technol 101:1611–1627. https://doi.org/10.1016/j.biortech.2009.09.043
Delgadillo-Mirquez L, Lopes F, Taidi B, Pareau D (2016) Nitrogen and phosphate removal from wastewater with a mixed microalgae and bacteria culture. Biotechnol Rep 11:18–26
Herbert D, Phipps PJ, Strange RE (1971) Chemical analysis of microbial cells. In: Norris JR, Ribbons DW (eds) Methods in microbiology, vol 5B. Academic Press, London, pp 209–344
Kar M, Mishra D (1976) Catalase, peroxidase and polyphenoloxidase activities during rice leaf senescence. Plant Physiol 57:315–319
Kim S, Park JE, Cho YB, Hwang SJ (2013) Growth rate, organic carbon and nutrient removal rates of Chlorella sorokiniana in autotrophic, heterotrophic and mixotrophic conditions. Bioresour Technol 144:8–13
Kumar R, Rao RP, Subramanian VV, Sivasubramanian V (2014) Enzymatic and non-enzymatic antioxidant potentials of Chlorella vulgaris grown in effluent of a confectionery industry. J Food Sci Technol 51:322–328
Leavitt PR, Findlay DL, Hall RI, Smol JP (1999) Algal responses to dissolved organic carbon loss and pH decline during whole-lake acidification: evidence from paleolimnology. Limnol Oceanogr 44:757–773. https://doi.org/10.4319/lo.1999.44.3_part_2.0757
Liang S, Xu M, Zhang T (2013) Life cycle assessment of biodiesel production in China. Bioresour Technol 129:72–77. https://doi.org/10.1016/j.biortech.2012.11.037
Marchão L, Lopes da Silva T, Gouveia L, Reis A (2017) Microalgae-mediated brewery wastewater treatment: effect of dilution rate on nutrient removal rates, biomass biochemical composition, and cell physiology. J Appl Phycol. https://doi.org/10.1007/s10811-017-1374-1
Marker AFH, Nusch EA, Rai H, Riemann B (1980) The measurement of photosynthetic pigments in freshwaters and standardization of methods: conclusions and recommendations. Arch Hydrobiol Beih Ergebn Limnol 14:91–106
Martinez-Abaigar J, Nùñez-Oliveira E (1998) Ecophysiology of photosynthetic pigments in aquatic bryophytes. In: Bates JW, Ashton NW, Duckett JG (eds) Bryology for the Twenty-first century. Maney Publishing and the British Bryological Society, Cambridgeshire, pp 277–292
Megateli S, Semsari S, Couderchet M (2009) Toxicity and removal of heavy metals (cadmium, copper and zinc) by Lemna gibba. Ecotoxicol Environ Saf 72:1774–1780
Megharaj M, Pearson HW, Vekateswarlu K (1992) Removal of nitrogen and phosphorus by immobilized cells of Chlorella vulgaris and Scenesdesmus bijugatus isolated from soil. Enzyme Microbi Technol 14:656–658
Mehta SK, Gaur JP (2005) Use of algae for removing heavy metal ions from wastewater. Crit Rev Biotechnol 25:11352
Mutanda T, Karthikeyan S, Bux F (2011) The Utilization of Post-chlorinated Municipal Domestic Wastewater for Biomass and Lipid Production by Chlorella spp. under Batch Conditions. Appl Biochem Biotechnol 164:1126–1138
Olguín EJ (2003) Phycoremediation: key issues for cost-effective nutrient removal processes. Biotech Adv 22:81–91
Pittman JK, Dean AP, Osundeko O (2011) The potential of sustainable algal biofuel production using wastewater resources. Bioresour Technol 102(1):17–25. https://doi.org/10.1016/j.biortech.2010.06.035
Prescott GW (1952) Algae of Western Great Lakes Area. Sci Publisher West Germany, Ottokoeltz
Ramsundar P, Guldhe A, Singh P, Bux F (2017) Assessment of municipal wastewaters at various stages of treatment process as potential growth media for Chlorella sorokiniana under different modes of cultivation. Bioresour Technol 227:82–92
Renuka N, Sood A, Ratha SK, Prasanna R, Ahluwalia AS (2013) Nutrient sequestration, biomass production by microalgae and phycoremediation of sewage water. Int J Phytoremediation 15:789–800
Rippka R, Deruelles J, Waterbury JB, Herdman MR, Stenier Y (1979) Generic assignments, strain histories and properties of pure cultures of cyanobacteria. J Gen Microbiol 111:1–61. https://doi.org/10.1099/00221287-111-1-1
Roe JH, Kuether CA (1943) The determination of ascorbic acid in whole blood and urine through 2, 4-dinitrophenyl hydrazine derivative of Dehydroascorbic acid. J Biol Chem 147:399–407
Romero-Gonzalez ME, Williams CJ, Gardiner PH (2001) Study of the mechanisms of cadmium biosorption by dealginated seaweed waste. Environ Sci Technol 35:3025–3030
Rotruck JT, Pope AL, Ganther HE, Swanson AB, Hafeman DG, Hoeckstra WG (1973) Selenium: biochemical role as a component of glutathione peroxidase. Science 179:588–590
Salgueiro JL, Pérez L, Maceiras R, Sánchez A, Cancela A (2018) Semicontinuous culture of chlorella vulgaris microalgae for wastewater treatment. Int J Environ Res. https://doi.org/10.1007/s41742-018-0129-4
Sarmah P, Rout J (2017) Colonisation of Oscillatoria on submerged polythenes in domestic sewage water of Silchar town, Assam (India). J Algal Biomass Utln 8:135–144
Schaedle M, Bassham JA (1977) Chloroplast glutathione reductase. Plant Physiol 59(5):1011–1012
Shannon KE, Lee DY, Trevors JT, Beaudette LA (2007) Application of real time quantitative PCR for the detection of selected bacterial pathogens during municipal wastewater treatment. Sci Total Environ 382:121–129
Singh AK, Sharma N, Farooqi H, Abdin MZ, Mock T, Kumar S (2017) Phycoremediation of municipal wastewater by microalgae to produce biofuel. Int J Phytoremed 19:805–812
Spiro RG (1966) Analysis of sugars found in glycoproteins. Methods Enzymol 8:3–26
Spitale D (2009) Spatial distribution of bryophytes along a moisture gradient: an approach using photosynthetic pigments as indicators of stress. Ecol Res 24:1279–1286
Strickland JDH, Parsons TR (1968) A practical handbook of seawater analyses. Pigment Analysis. Bull Fish Res Bd Canada 167.
Tebbutt THY (1983) Principles of water quality control. Pergammon Press, Oxford
Van Rossum MWPC, Alberda M, Van der Plas LHW (1997) Role of oxidative damage in tulip bulb scale micropropagation. Plant Sci 130:207–216
Wang L, Min M, Li Y, Chen P, Chen Y, Liu Y, Wang Y, Ruan R (2010) Cultivation of green algae Chlorella sp. in different wastewaters from municipal wastewater treatment plant. Appl Biochem Biotechnol 162:1174–1186
Yadav KK, Gupta N, Kumar V, Singh J (2017) Bioremediation of heavy metals from contaminated sites using potential species: a review. Indian J Environ Prot 37:65–84
Acknowledgements
The authors acknowledges the Department of Science and Technology (DST/IS-STAC/CO2-SR-164/13 (G), Government of India, New Delhi for support. PS acknowledges the University Grants Commission, New Delhi for fellowship.
Funding
This research was partially supported by the project “Evaluation of indigenous freshwater algal species from Southern Assam (North East India) for carbon sequestration” from Department of Science and Technology (DST/IS-STAC/CO2-SR-164/13 (G), Government of India, New Delhi.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animal rights
There is no research involving human participants and or animals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sarmah, P., Das, S., Sharma, H. et al. Microalgal biomass generation by phycoremediation of sewage water: an integrated approach for production of antioxidant and value added products. Vegetos 32, 556–563 (2019). https://doi.org/10.1007/s42535-019-00056-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42535-019-00056-x