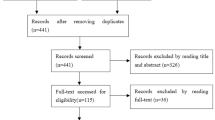Key summary points
This study is the first to explore the prevalence of oral frailty among older adults through systematic review and meta‑analysis.
AbstractSection FindingsThe overall pooled prevalence of oral frailty and oral pre-frailty in older adults was 24% and 57% respectively.
AbstractSection MessageOral frailty was common in older adults and various characteristics such as gender, source, country, study design and evaluation scales may affect its prevalence.
Abstract
Background
In recent years, oral frailty was proposed as a new concept regarding dental and oral health in older adults. Poor oral health is linked to preserving general health and has become a geriatric public health problem that deeply affects healthy aging. While in present, evidence on the prevalence associated with oral frailty in older adults remains unclear.
Objective
To systematically evaluate the prevalence of oral frailty among older adults, stratified by relevant factors such as gender, source, study design, region, and the evaluation scales for oral frailty and provide an evidence-based foundation for healthcare professionals and policymakers to formulate relevant measures.
Methods
Ten electronic databases were systematically searched from inception to September 2023, including PubMed, Web of Science, Embase, PsycINFO, The Cochrane Library, CINAHL, China National Knowledge Infrastructure Database (CNKI), Chinese Biomedical Database (Sinomed), Weipu Database, and Wanfang database. Based on the Stata 15.0 software package, a random effect model was used to calculate the pooled prevalence of oral frailty among older adults. In addition, sensitivity analysis, subgroup analysis, and meta-regression were conducted based on different study characteristics to detect heterogeneity sources. Funnel plots, Begg’s and Egger’s tests were used to evaluate the publication bias.
Results
Eighteen studies with a total of 12,932 older adults were included for meta-analysis. The pooled prevalence of oral frailty and oral pre-frailty was 24% (95% CI: 20–28%) and 57% (95% CI: 52–61%) respectively. Based on different assessment tools of oral frailty, the pooled prevalence of oral frailty was higher when using the OFI-8 scale (44.1%; 95% CI: 35.4–52.8%) than the OFI-6 scale (18.3%; 95% CI: 15.8–20.8%) or OF checklist (22.1%; 95% CI: 17.4–26.7%). The prevalence of oral frailty was higher among older adults in females (23.8%; 95% CI: 18.4–29.2%), hospital settings (31%, 95% CI: 16.6–45.5%), cross-sectional design (26.7%, 95% CI: 19.2–34.2%), and China (45.9%, 95% CI: 34.4–57.3%).
Conclusions
Our study showed that oral frailty was common among older adults and various characteristics may affect its prevalence. Thus, healthcare professionals and policymakers should take oral frailty seriously in clinical practice and program planning and develop more preventive measures for oral frailty among older adults.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Worldwide, countries are experiencing a rapid demographic transition and the proportion of older people will steadily grow. According to relevant data from the World Health Organization, the number of people over the age of 80 will reach nearly 434 million by 2050 [1]. For many older people, aging comes with more health problems such as chronic and multiple diseases [2, 3], and an elevated frailty risk [4]. Frailty is a biophysiological disorder defined as a dynamic state characterized by decreased physiological reserves and increased vulnerability to stressors which is associated with a higher susceptibility to negative health outcomes including falls, cognitive disorders, function impairment and death [5,6,7].
Due to the multidimensional and multisystematic nature of frailty, practitioners and scholars have extended the concept of frailty to different domains including physical [7], cognitive [8], psychological and social frailty phenotypes [9]. However, there is no universal consensus on emerging questions regarding prioritising domains in frailty contexts and some related issues are still being debated [10]. Therefore, the term oral frailty phenotype is a relatively novel construct proposed as a geriatric syndrome by Tanaka when considering the frail older people is becoming a scientific interest on this topic and has increasingly been adopted in recent years [11, 12].
Poor oral health and decline in oral function are highly prevalent among older adults [13], which can result in poor nutrition status and chronic diseases [14, 15] and are strongly associated with different components of frailty. A systematic review suggested a positive longitudinal association between poor oral health, including fewer remaining teeth and an impaired oral function, and frailty among older adults [16].To recognize the multidimensional nature of poor oral health status, oral frailty was proposed and defined by Tanaka as an age-related gradual decrease in oral function (chewing, swallowing, oral motor skill, and tongue pressure) and they identified that oral frailty can be a significant predictor of physical frailty, sarcopenia, disability and mortality respectively [12]. Moreover, another systematic review proved that oral frailty indicators can significantly determine a series of major adverse health-related outcomes including death, functional disability, quality, hospitalization, and falls among older people [17]. In light of these findings, to promote global healthy aging, oral frailty deserves more attention and further exploration.
Although several studies have been published on oral frailty among older adults, there is no summary of existing studies to date to explore the prevalence of oral frailty in older adults. Therefore, the primary objective of our study is to conduct a systematic review and meta-analysis to quantitatively synthesize the overall prevalence of oral frailty among older people and identify associated factors that may affect the prevalence of oral frailty among older adults. We also aim to provide evidence-based support for healthcare professionals and develop more targeted strategies for oral frailty.
Methods
Design
This systematic review was performed based on the Preferred Reporting Items for Systematic Reviews and Meta-analyses Statement (PRISMA) guidelines and the detailed study protocol was registered on PROSPERO (CRD42023458721).
Selection criteria
Inclusion criteria: (1) study type was observational studies including cross-sectional study, case–control study or cohort study in English or Chinese language; (2) participants were older adults living in community, hospitals or nursing homes (aged 60 years or above); (3) specific assessment tools or methods of oral frailty were explicitly mentioned; (4) outcome measures were the prevalence of oral frailty in older adults.
Exclusion criteria: (1) reviews, case reports, and meta-analysis; (2) conference, commentary and animal experiments; (3) duplicate publication; (4) unable to obtain the full data or text.
Search strategy
A comprehensive search of ten electronic databases including PubMed, Web of Science, Embase, PsycINFO, The Cochrane Library, CINAHL, CNKI, Chinese Biomedical Database (Sinomed), Weipu Database, and Wanfang database was performed from database inception to September 2023. The MeSH words and free terms employed in our study encompassed (“aged” or “aging” or “aged” or “aging” or “elder*” or “senior*” or “old*” or “geriatric*” or “old people” or “old adults”) and (“oral frailty” or “oral function” or “oral vulnerability” or “oral weakness”). The detailed search strategies of each database were described in Appendix 1, Supplementary Material.
Methodological quality assessment
Two reviewers independently assessed the quality of the included studies. The appraisal of risk bias recommended by the Agency for Healthcare and Research and Quality tool (AHRQ) [18] was adopted to evaluate the quality of cross-sectional studies, in which 0–3 points were regarded as low quality, 4–7 points were regarded as medium quality, and 8–11 points were considered high quality respectively. The Newcastle–Ottawa Scale [19] was used to assess the methodological quality of each included study for case–control and cohort studies. The NOS scale had eight items and assessed each study from three domains with scores of 0–4 points, 5–6 points, and ≥7 points indicating low, medium, and high quality. Any disagreement in the quality of the rating process was resolved with a third researcher (J.Y.L. or Z.Y.) through discussion and consultation.
Selection of studies
Two authors (L.T. and L.L.) independently reviewed and cross-checked all eligible articles based on titles, abstracts, and full texts after deleting duplicates. Controversial parts between two researchers over the judgment of article inclusion were resolved by discussion or consultation with another researcher (J.Y.L.) until reaching a consensus.
Data extraction
The retrieved articles from each electronic database were imported and managed in Endnote version X9. The same two reviewers (L.T. and L.L.) independently extracted primary source data from each study and cross-checked them. A data extraction table was established and included the information extraction characteristics such as the first author, publication year, sample size, country, type of study design, average age of participant, source of the study participants, diagnostic criteria, and the prevalence of oral frailty and oral pre-frailty.
Data analysis
The statistical software package Stata 15.0 was used to analyze the pooled prevalence and 95% CI. Cochrane’s Q, I2, and p value statistics [20] were used to investigate the degree of heterogeneity among included studies. A fixed effect model was employed for meta-analysis if I2 ≤ 50% and the p value >0.05 which showed low or moderate heterogeneity; otherwise, a random effect was used to estimate the combined prevalence and 95% CI of oral frailty among older adults. Sensitivity analysis was employed to explore the stability and reproducibility of the results by eliminating each enrolled study. Subgroup analysis and meta-regression were conducted to investigate possible significant factors of heterogeneity. In addition, a funnel plot, Begg’s test, and Egger’s test were applied to investigate publication bias.
Results
Search results
A total of 4137 articles were retrieved from ten electronic databases based on the search strategy, as shown in Fig. 1. After excluding 2173 duplicates, 1870 articles were removed by two authors who screened the titles and abstracts independently. 94 full-text articles were retrieved and evaluated to determine eligibility. Finally, 18 articles were eligible for inclusion.
Study characteristics
Of the 18 studies, 12 studies used a cross-sectional design, and 6 used a cohort design approach, involving 12,932 older adults (Table 1). The majority of studies (18/19) were published in the previous five years and were carried out in Japan (12), China (3), and Taiwan (3). The total sample size was 12,932 and the sample sizes ranged from 204 to 2011.
Risk of bias
Of the 12 cross-sectional studies evaluated by AHRQ, seven were rated as high quality, and another five were moderate quality (see Appendix 2, Supplementary Material); in the 6 cohort studies, three were high quality, and three studies were considered as moderate quality (see Appendix 3, Supplementary Material).
The prevalence of oral frailty in older adults
There were 18 included studies in total, and all of them reported the prevalence of oral frailty among older adults. The frequency of oral frailty in older adults ranged from 9.5% to 59.2%. Considering the high heterogeneity detected in 18 studies (Q = 683.08, I2 = 97.5%, p < 0.001), a random-effect model was used to assess the pooled prevalence of oral frailty in older adults, which was determined to be 24% (95% CI: 20–28%, p < 0.001; Fig. 2). Eight studies reported the prevalence of oral pre-frailty in older adults. Given the considerable heterogeneity observed among these studies (Q = 84.53, I2 = 91.7%, p < 0.001), a random-effect model was employed for effect size synthesis. The outcome revealed the pooled prevalence of oral pre-frailty among older adults was 57% (95% CI: 52–61%, p < 0.001; Fig. 3).
Subgroup analysis and meta-regression analysis
Subgroup analysis of the oral frailty in older adults is displayed in Table 2. The results of subgroup analysis by gender showed that the prevalence of oral frailty was higher in female adults (23.8%, 95% CI: 18.4–29.2%) than in male adults (21.9%, 95% CI: 17–26.8%). The prevalence of oral frailty was higher in hospital older adults (31%, 95% CI: 16.6–45.5%) than in community older adults (23.1%, 95% CI: 18.5–27.8%). Based on the oral frailty assessment tool, including the OF-6 scale, OF-8 scale, and OF checklist, the combined estimations of the prevalence of oral frailty were 18.3%, 44.1%, and 22.1% respectively. Studies using a cross-sectional methodology had a higher estimated pooled prevalence of oral frailty (26.7%, 95% CI: 19.2–34.2%) than those using a cohort design (19%, 95% CI: 16.3–21.7%). The overall pooled prevalence of oral frailty in Japan, China, and Taiwan was 20% (95% CI: 16.9–23.1%), 45.9% (95% CI: 34.4–57.3%), 18.3% (95% CI: 12.5–24.2%). We chose the country/region, study design, publication year, assessment scale, and mean age as covariates for meta-regression analysis. However, no significant statistical difference was detected in the regression coefficients of the above covariates between the designated reference subgroups (p > 0.05; Table 3).
Sensitivity analysis and publication bias
After individually removing each study, the pooled results of the sensitivity analysis had no significant change (see Fig. 4), indicating the robustness of the pooled prevalence of oral frailty among older adults. According to the funnel plot, Egger’s test (t = 0.63, p = 0.54), and Begg’s test (Z = 0.76, p = 0.449), there was a small possibility of publication bias in this analysis.
Discussion
To the best of our knowledge, this is the first comprehensive review of the existing evidence to examine the prevalence of oral frailty in older adults based on large sample sizes. Owing to the different baseline characteristics and study or region limitations among the included articles, the prevalence of oral frailty ranged from 9.5% to 59.2%. Our current systematic and meta-analysis included 18 studies, comprising 12,932 older adults and discovered the pooled prevalence of oral frailty and oral pre-frailty was 24% (95% CI: 20–28%), 57% (95% CI: 52% ~ 61%) respectively. The estimated prevalence in this meta-analysis was similar to the previous review [34]. Compared to a survey result conducted in the context of rural communities in China [35], our findings presented a lower prevalence of oral frailty among older adults. While comparing the prevalence of oral frailty among older adults conducted in Taiwan and Japan [31, 37], our results present a higher prevalence level of oral frailty. It is also noteworthy that the prevalence of oral frailty notably exceeds the detection rates of physical frailty [38] (4.3%), cognitive frailty [39] (9%), and social frailty [40] (18.8%) among older people. The potential reasons for the disparity in these findings include differences in healthcare systems across countries and regions, disparities in lifestyle and hygiene practices, and an insufficient level of concern among older adults about their oral health conditions. Consequently, these factors may contribute to the higher prevalence of oral frailty among older people. Nonetheless, our findings revealed that oral frailty was common among older adults and many older persons were already influenced by oral frailty. Therefore, public health personnel and policymakers should take the high prevalence of oral frailty among older persons seriously and implement some targeted and preventative strategies to help them deal with oral frailty timely.
The subgroup analysis based on gender found that the prevalence of oral frailty was 23.8% in females and 21.9% in males, consistent with previous research. Relevant studies [41, 42] indicated that due to the earlier development of permanent teeth in females compared to males, they were consequently subjected to extended periods of masticatory wear and oral macrobiotic erosion, potentially leading to the premature manifestation of deleterious oral health conditions in female older adults. Furthermore, in older postmenopausal women, the sustained decline in estrogen levels within the body made them susceptible to disturbances in bone metabolism and calcium loss. The gingiva, serving as a target organ for estrogen, is particularly prone to causing oral issues such as alveolar bone osteoporosis, reduced salivary secretion, and diminished salivary flow velocity. Consequently, these factors can incite the onset of periodontal diseases, xerostomia, dental caries, and ultimately culminate in oral frailty [43]. However, according to some studies conducted in Japan [26, 44], their findings showed that oral frailty in older adults was not impacted by gender factors. The association between sex and oral frailty could not be confirmed based on current evidence and more relevant high-quality studies were expected to fully explore this topic.
The subgroup analysis by source revealed that the prevalence of oral frailty was higher among older adults in the hospital than in the community (31% vs 23.1%). This disparity may be explained by the community’s higher proportion of elderly citizens, as well as its strong focus on improving the quality of life and relevant healthcare and dental information for its older citizens. Furthermore, the community has demonstrated a considerable degree of sophistication in its health promotion endeavors and health service initiatives [45], which may help older adults living in the community attach great importance to their oral condition. Meanwhile, older adults in hospitals often presented with a multitude of comorbidities, diminished physical resilience, and during their hospitalization, they often tended to prioritize therapeutic interventions for their diseases, inadvertently neglecting their oral hygiene status. A cross-sectional research in Japan revealed that medical providers pay insufficient attention to clinical patients and infrequent referral by medical providers to dental providers [46]. Therefore, establishing a standard routine screen program for oral frailty is critical to maintaining general health among older adults, especially in non-gerodontologic settings. In light of these discoveries, healthcare practitioners in hospital settings should enhance the health consciousness and health literacy of senior citizens through the consistent implementation of diverse health education initiatives, thereby ameliorating the extant condition of oral frailty among older people. However, we could not conduct meta-analysis to pool the prevalence of oral frailty among older adults living in nursing homes due to insufficient studies on this topic. Future studies are recommended to conduct in nursing home settings and further explore the association between the prevalence of oral frailty and study source.
The subgroup analysis by study design showed that the prevalence of oral frailty in cross-sectional studies was 26.7% (95% CI: 19.2–34.2%) and 19% (95% CI: 16.3–21.7%) for cohort studies. Methodology design could be attributed to explain this discrepancy in which cross-sectional studies and cohort studies refer to different inclusion and exclusion criteria. In addition, some studies [23, 31, 47] in this meta-analysis excluded several older individuals who did not fulfill the follow-up requirements. Thus these missing data arising from nonadherence to the follow-up protocol may have contributed to the difference in the prevalence of oral frailty.
According to our study, the prevalence of oral frailty in older adults ranged from 18.3 to 44.1% depending on the different oral frailty assessment tools. With the growing body of research on oral frailty, there has been a concurrent proliferation of assessment tools dedicated to its scrutiny. Nonetheless, a unified standard for the assessment of oral frailty remained conspicuously absent. The OF-6 scale was introduced by Tanaka in 2018 [12] which is based on a cohort study. It comprised a total of six items, with the first four being objective indicators and the latter two being subjective assessments. The total score of 0 points was defined as non-OF, 1–2 points were oral pre-frailty, and 3 or more points were OF. This assessment method presently enjoys widespread usage in research. However, it was imperative to underscore that the examination elements within the scale necessitate the expertise of dental professionals and the utilization of specialized equipment, rendering it less amenable to swift and routine screening. In 2021, Tanaka introduced the OFI-8 scale [48] to identify oral frailty in older adults residing in the community. This measure consists of eight subjective questions with a scoring range of 0–11 points, with higher scores indicating a higher risk of oral frailty. For each incremental point in the OFI-8 score, the risk of disability increases by a factor of 1.1, and the risk of oral frailty rises by a factor of 1.3. Notably, this scale is characterized by its simplicity of operation, requiring no specialized equipment, rendering it suitable for swift screening of individuals at a high risk of oral frailty within community and outpatient settings. The OF checklist was proposed by the Japan Dental Association and validated for screening of oral frailty in Japanese people [49]. Consisting of 8 items, the OF checklist assessed oral frailty in older adults from tooth loss, poor oral function, oral health-related behaviors, and declining social participation [44]. However, there still exists some challenges in evaluating oral frailty across countries and cultures, future studies could validate the efficacy of various assessment tools for oral frailty and establish a standard of practice for oral frailty screening.
The subgroup analysis by countries/regions found that the pooled prevalence of oral frailty among older adults in China was higher than in Chinese Taiwan and Japan. In high-income countries or regions, older individuals often demonstrate a greater consciousness of oral health [12, 27], making them more inclined to comply with a regular routine of oral hygiene practices, including frequent dental cleanings and check-ups, as well as the purchase of high-quality oral hygiene products. However, there exists no additional evidence to substantiate the correlation between country/region and oral frailty. Hence, it is imperative that future studies undertake a more comprehensive investigation into the impact of countries/regions on oral frailty among older adults.
Strength and limitations
This systematic review and meta-analysis conducted a comprehensive literature retrieval of 10 databases, ensuring the stability and authenticity of the search results. Furthermore, a comprehensive quality evaluation of the included studies was performed rigidly by two independent researchers using relevant assessment tools. Finally, the results of this meta-analysis highlighted the importance of early screening and early intervention to reduce the prevalence of oral frailty among older adults. However, this study had some limitations. To begin with, limited by the characteristics of the single-rate meta-analysis [50], a high level of heterogeneity was detected inevitably among the included studies. Second, despite we conducted sufficient measures including subgroup analysis and meta-regression to explore potential sources of heterogeneity, considerable heterogeneity still existed in our study. Third, most of the included studies were from Asia countries or regions, which may affect the representation of the results in our meta-analysis and limit the global generalizability of the findings. More studies in other countries or regions are needed to further investigate the prevalence of oral frailty in different areas. Finally, unpublished documents and grey literature were not included in this meta-analysis though the databases had been searched deeply, which may miss some potentially relevant studies.
Conclusion
The overall prevalence of oral frailty and oral pre-frailty among older adults were revealed to be 24% and 57% respectively in our systematic review and meta-analysis which can provide an evidence-based foundation for future relevant research. Meanwhile, our study showed that the prevalence of oral frailty was impacted by gender, source, study design, region, and the evaluation scales for oral frailty. Therefore, healthcare professionals and policymakers should figure out targeted and effective interventions for oral frailty among older adults. Future cohort studies or clinical studies are expected to further verify the results found in this study and develop the best strategies and interventions to prevent older adults from oral frailty.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
World Health Organization (2018) Ageing and health. https://www.who.int/news-room/fact-sheets/detail/ageing-and-health
Vetrano DL, Palmer K, Marengoni A et al (2019) Frailty and multimorbidity: a systematic review and meta-analysis. J Gerontol A Biol Sci Med Sci 74(5):659–666. https://doi.org/10.1093/gerona/gly110
Lin T, Zhao Y, Xia X et al (2020) Association between frailty and chronic pain among older adults: a systematic review and meta-analysis. Eur Geriatr Med 11(6):945–959. https://doi.org/10.1007/s41999-020-00382-3
Suzuki T (2018) Health status of older adults living in the community in Japan: recent changes and significance in the super-aged society. Geriatr Gerontol Int 18(5):667–677. https://doi.org/10.1111/ggi.13266
Gobbens RJ, Luijkx KG, Wijnen-Sponselee MT et al (2010) Towards an integral conceptual model of frailty. J Nutr Health Aging 14(3):175–181. https://doi.org/10.1007/s12603-010-0045-6
Hoogendijk EO, Afilalo J, Ensrud KE et al (2019) Frailty: implications for clinical practice and public health. Lancet 394(10206):1365–1375. https://doi.org/10.1016/s0140-6736(19)31786-6
Fried LP, Tangen CM, Walston J et al (2001) Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci 56(3):146–156. https://doi.org/10.1093/gerona/56.3.m146
Sardone R, Castellana F, Bortone I et al (2021) Association between central and peripheral age-related hearing loss and different frailty phenotypes in an older population in Southern Italy. JAMA Otolaryngol Head Neck Surg 147(6):561–571. https://doi.org/10.1001/jamaoto.2020.5334
Pek K, Chew J, Lim JP et al (2020) Social frailty is independently associated with mood, nutrition, physical performance, and physical activity: insights from a theory-guided approach. Int J Environ Res Public Health 17(12):4239. https://doi.org/10.3390/ijerph17124239
Dibello V, Zupo R, Sardone R et al (2021) Oral frailty and its determinants in older age: a systematic review. Lancet Healthy Longev 2(8):e507–e520. https://doi.org/10.1016/s2666-7568(21)00143-4
Choi JH, Kang JH, Koh SB et al (2020) Development of an oral and maxillofacial frailty index: a preliminary study. J Oral Rehabil 47(2):187–195. https://doi.org/10.1111/joor.12890
Tanaka T, Takahashi K, Hirano H et al (2018) Oral frailty as a risk factor for physical frailty and mortality in community-dwelling elderly. J Gerontol A Biol Sci Med Sci 73(12):1661–1667. https://doi.org/10.1093/gerona/glx225
Murray Thomson W (2014) Epidemiology of oral health conditions in older people. Gerodontology 31(Suppl 1):9–16. https://doi.org/10.1111/ger.12085
Wakai K, Naito M, Naito T et al (2010) Tooth loss and intakes of nutrients and foods: a nationwide survey of Japanese dentists. Community Dent Oral Epidemiol 38(1):43–49. https://doi.org/10.1111/j.1600-0528.2009.00512.x
Lockhart PB, Bolger AF, Papapanou PN et al (2012) Periodontal disease and atherosclerotic vascular disease: does the evidence support an independent association?: a scientific statement from the American Heart Association. Circulation 125(20):2520–2544. https://doi.org/10.1161/CIR.0b013e31825719f3
Hakeem FF, Bernabé E, Sabbah W (2019) Association between oral health and frailty: a systematic review of longitudinal studies. Gerodontology 36(3):205–215. https://doi.org/10.1111/ger.12406
Dibello V, Lobbezoo F, Lozupone M et al (2023) Oral frailty indicators to target major adverse health-related outcomes in older age: a systematic review. Geroscience 45(2):663–706. https://doi.org/10.1007/s11357-022-00663-8
Zeng X, Zhang Y, Kwong JS et al (2015) The methodological quality assessment tools for preclinical and clinical studies, systematic review and meta-analysis, and clinical practice guideline: a systematic review. J Evid Based Med 8(1):2–10. https://doi.org/10.1111/jebm.12141
Stang A (2010) Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol 25(9):603–605. https://doi.org/10.1007/s10654-010-9491-z
Higgins JP, Thompson SG, Deeks JJ et al (2003) Measuring inconsistency in meta-analyses. BMJ 327(7414):557–560. https://doi.org/10.1136/bmj.327.7414.557
Hoshino D, Hirano H, Edahiro A et al (2021) Association between oral frailty and dietary variety among community-dwelling older persons: a cross-sectional study. J Nutr Health Aging 25(3):361–368. https://doi.org/10.1007/s12603-020-1538-6
Iwasaki M, Motokawa K, Watanabe Y et al (2020) A two-year longitudinal study of the association between oral frailty and deteriorating nutritional status among community-dwelling older adults. Int J Environ Res Public Health 18(1):213. https://doi.org/10.3390/ijerph18010213
Iwasaki M, Motokawa K, Watanabe Y et al (2020) Association between oral frailty and nutritional status among community-dwelling older adults: the takashimadaira study. J Nutr Health Aging 24(9):1003–1010. https://doi.org/10.1007/s12603-020-1433-1
Iwasaki M, Watanabe Y, Motokawa K et al (2021) Oral frailty and gait performance in community-dwelling older adults: findings from the Takashimadaira study. J Prosthodont Res 65(4):467–473. https://doi.org/10.2186/jpr.JPR_D_20_00129
Komatsu R, Nagai K, Hasegawa Y et al (2021) Association between physical frailty subdomains and oral frailty in community-dwelling older adults. Int J Environ Res Public Health 18(6):29–31. https://doi.org/10.3390/ijerph18062931
Kugimiya Y, Watanabe Y, Ueda T et al (2020) Rate of oral frailty and oral hypofunction in rural community-dwelling older Japanese individuals. Gerodontology 37(4):342–352. https://doi.org/10.1111/ger.12468
Kuo YW, Lee JD (2022) Association between oral frailty and physical frailty among rural middle-old community-dwelling people with cognitive decline in Taiwan: a cross-sectional study. Int J Environ Res Public Health 19(5):2884. https://doi.org/10.3390/ijerph19052884
Kusunoki H, Ekawa K, Kato N et al (2023) Association between oral frailty and cystatin C-related indices-A questionnaire (OFI-8) study in general internal medicine practice. PLoS ONE 18(4):e0283803. https://doi.org/10.1371/journal.pone.0283803
Lin YC, Huang SS, Yen CW et al (2022) Physical frailty and oral frailty associated with late-Life depression in community-dwelling older adults. J Pers Med 12(3):459. https://doi.org/10.3390/jpm12030459
Hironaka S, Kugimiya Y, Watanabe Y et al (2020) Association between oral, social, and physical frailty in community-dwelling older adults. Arch Gerontol Geriatr 89:104105. https://doi.org/10.1016/j.archger.2020.104105
Nishimoto M, Tanaka T, Hirano H et al (2023) Severe periodontitis increases the risk of oral frailty: a six-year follow-up study from kashiwa cohort study. Geriatrics (Basel) 8(1):25–30. https://doi.org/10.3390/geriatrics8010025
Ohara Y, Motokawa K, Watanabe Y et al (2020) Association of eating alone with oral frailty among community-dwelling older adults in Japan. Arch Gerontol Geriatr 87:104014. https://doi.org/10.1016/j.archger.2020.104014
Wang L, Ju M, Wang T et al (2023) Oral frailty and its influencing factors in community-dwelling elderly population. J Nurs Sci 38(18):112–116. https://doi.org/10.3870/j.issn.1001-4152.2023.18.112
Yamamoto T, Tanaka T, Hirano H et al (2022) Model to predict oral frailty based on a questionnaire: a cross-sectional study. Int J Environ Res Public Health 19(20):13244. https://doi.org/10.3390/ijerph192013244
Tang J, Tang XY, Zeng L et al (2023) Prevalence and influencing factors of oral frailty in the elderly of rural areas in Guizhou Province. Chin J Prev Contr Chron Dis 31(05):327–331. https://doi.org/10.16386/j.cjpccd.issn.1004-6194.2023.05.002
Tu HJ, Zhang SY, Fang YY et al (2023) Current situation and influencing factors of oral frailty in the community elderly. Chin J Nurs 58(11):1351–1356. https://doi.org/10.3761/j.issn.0254-1769.2023.11.011
Wang XY (2021) Oral frailty of the community elderly: the association between sarcopenia and nutrition and depression [D]. Taipei University of Nursing and Health Sciences
Ofori-Asenso R, Chin KL, Mazidi M et al (2019) Global incidence of frailty and prefrailty among community-dwelling older adults: a systematic review and meta-analysis. JAMA Netw Open 2(8):e198398–e198398. https://doi.org/10.1001/jamanetworkopen.2019.8398
Qiu Y, Li G, Wang X et al (2022) Prevalence of cognitive frailty among community-dwelling older adults: a systematic review and meta-analysis. Int J Nurs Stud 125(03):104112. https://doi.org/10.1016/j.ijnurstu.2021.104112
Zhang XM, Cao S, Gao M et al (2023) The prevalence of social frailty among older adults: a systematic review and meta-analysis. J Am Med Dir Assoc 24(1):29–37. https://doi.org/10.1016/j.jamda.2022.10.007
Nyström M, Haataja J, Kataja M et al (1986) Dental maturity in Finnish children, estimated from the development of seven permanent mandibular teeth. Acta Odontol Scand 44(4):193–198. https://doi.org/10.3109/00016358608997720
Ye X, Jiang F, Sheng X et al (2014) Dental age assessment in 7–14-year-old Chinese children: comparison of Demirjian and Willems methods. Forensic Sci Int 244:36–41. https://doi.org/10.1016/j.forsciint.2014.07.027
Adam M, Wooton J (2022) Menopause and oral health. Br Dent J 233(3):170. https://doi.org/10.1038/s41415-022-4568-0
Nomura Y, Ishii Y, Suzuki S et al (2020) Nutritional status and oral frailty: a community based study. Nutrients 12(9):2886. https://doi.org/10.3390/nu12092886
Li HW, Lee WJ, Lin MH et al (2021) Quality of life among community-dwelling middle-aged and older adults: function matters more than multimorbidity. Arch Gerontol Geriatr 95:104423. https://doi.org/10.1016/j.archger.2021.104423
Shimpi N, Schroeder D, Kilsdonk J et al (2016) Medical providers’ oral health knowledgeability, attitudes, and practice behaviors: an opportunity for interprofessional collaboration. J Evid Based Dent Pract 16(1):19–29. https://doi.org/10.1016/j.jebdp.2016.01.002
Tanaka T, Hirano H, Ikebe K et al (2023) Oral frailty five-item checklist to predict adverse health outcomes in community-dwelling older adults: a Kashiwa cohort study. Geriatr Gerontol Int 15(8):651–659. https://doi.org/10.1111/ggi.14634
Tanaka T, Hirano H, Ohara Y et al (2021) Oral Frailty Index-8 in the risk assessment of new-onset oral frailty and functional disability among community-dwelling older adults. Arch Gerontol Geriatr 94:104340. https://doi.org/10.1016/j.archger.2021.104340
Nomura Y, Ishii Y, Chiba Y et al (2021) Structure and validity of questionnaire for oral frail screening. Healthcare 9(1):45. https://doi.org/10.3390/healthcare9010045
Zhang T, Ren Y, Shen P et al (2021) Prevalence and associated risk factors of cognitive frailty: a systematic review and meta-analysis. Front Aging Neurosci 13:755926. https://doi.org/10.3389/fnagi.2021.755926
Funding
No funding was received for this study.
Author information
Authors and Affiliations
Contributions
Study design: LT, SYL, and YZJ. Data extraction and collection: LT, JYL, and ZY. Data analysis and interpretation: LT, WSY, and LYJ. Manuscript writing: LT and LL.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethics approval
None.
Informed consent
No informed consent is required for this review.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Systematic review registration: CRD42023458721.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, T., Shen, Y., Leng, Y. et al. The prevalence of oral frailty among older adults: a systematic review and meta‑analysis. Eur Geriatr Med 15, 645–655 (2024). https://doi.org/10.1007/s41999-023-00930-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41999-023-00930-7