Abstract
Fall armyworm (FAW), Spodoptera frugiperda (J. E. Smith) (Lepidoptera: Noctuidae), is a serious invasive insect pest affecting global maize production. Effective integrated management of FAW is essential to minimize the yield losses. The objective of this study is to determine a suitable package for the sustainable management of FAW by validating the potentiality of various integrated approaches through field trials. In the present study, five different integrated pest management treatments consisted of different components were synthesized and evaluated against fall armyworm in maize-based systems during winter (December 2022–April 2023) and rainy (July–November 2023) seasons. The data on the number of plants damaged, leaf damage rating (1–9 scale), and natural enemies such as spiders, coccinellids, and earwigs were recorded from 20 randomly selected plants at 7 and 14 days after the first and second sprays. Yield data (q ha−1) were recorded at the time of harvest. Treatment 1 consisting of pheromone traps at a rate of 4 per acre with ICAR-NBAIR lures, erection of bird perches at a rate of 10 per acre, seed treatment with Chlorantraniliprole 50 FS at a rate of 5.6 ml per kilogram of seed, and spray application of azadirachtin 1500 ppm at the rate of 5 ml per litre, and Metarrhizium anisopliae with spore count of 1 × 108 cfu/g (1 kg per acre) at a rate of 5 g per litre significantly reduced the per cent plant infestation (12.7) and leaf damage rating (2.1) by FAW larvae compared to untreated control (39.7, 4.1), respectively. The higher natural enemy population (spiders, coccinellids, and earwigs) was also observed in Treatment 1 (8.8) compared to Treatment 5 (4.1) (chemical control). Furthermore, higher grain yield of 51.5 q ha−1 was obtained in Treatment 1 with a cost–benefit ratio of 1:2.1, whereas in untreated control, the yield obtained was 29.0 q ha−1 with a cost–benefit ratio of 1:1.3. The highest per cent of avoidable yield losses of 43.6 was observed in Treatment 1 in comparison with other treatments. Integration of sustainable management approaches reduces the application of chemical insecticides and enhances the population of natural enemies which would be beneficial to maize farmers.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fall armyworm (FAW), Spodoptera frugiperda (J. E. Smith), is an invasive insect pest of maize posing a serious threat to global food security (Stokstad 2017a, b). The pest migrated from neotropical areas of America to West Africa in early 2016 (Georgen et al. 2016) and quickly spread to most of the countries in sub-Saharan Africa. From African countries, it has invaded Yemen, India, China, Thailand, Myanmar, Sri Lanka, Bangladesh, Nepal, Cambodia, the Philippines, Vietnam, Indonesia, China, South Korea, Japan, Australia, Papua New Guinea, Jordan, and Syria (Prasanna et al. 2021; CABI 2022). In India, Karnataka experienced the first outbreak of FAW in 2018, which caused significant economic yield losses in maize (Sharanabasappa et al. 2018; Shylesha et al. 2018), and within 1 year, it spreads to major maize-growing ecologies (Rakshit et al. 2019; Suby et al. 2020). It is a highly polyphagous insect pest that attacks more than 350 host plants including maize (Zea mays L.), rice (Oryza sativa), sorghum (Sorghum bicolor), cotton (Gossypium spp. L), and soybean (Glycine max) (Barros et al. 2010). FAW is highly eurytopic and could complete 12 generations in a year in tropical climates (Busato et al. 2005). The larvae feed on vegetative and reproductive parts of the plant including leaf whorls, tassels, and cobs, and cause significant yield losses. Adhikari et al. (2020) reported that the effect of fall armyworm damage on maize yield is about 50–80% resulting in loss of millions of dollars. In Africa, without effective FAW management options, this pest causes significant maize yield losses ranging from 8.3 to 20.6 million tonnes per year, resulting in losses of $2.5–6.2 million (Mendesil et al. 2023). In 2017, maize production in India reached 28.7 million tonnes, but a decline of 3.2% occurred due to infestation by insect pests, resulting in a production of 27.8 million tonnes (Manupriya 2019). Presently, FAW is established in over 42 countries, with an estimated annual impact of $13 billion.
In India, FAW has strongly established due to favourable climatic conditions, high reproductive ability, and capacity to migrate over long distances (Rose et al. 1975). The sudden invasion of the FAW in India has prompted numerous recommendations advocating for the use of chemical insecticides applied as foliar sprays. However, the FAW tendency to feed within the whorl portion of maize poses a challenge as it makes the insecticides ineffective in reaching their target site. Additionally, the use of these toxic pesticides has led to the development of resistance in pests (Gutierrez-Moreno et al. 2019), toxicity to beneficial fauna (Abudulai et al. 2018), pest resurgence, and poses environmental and human health risks (Roubos et al. 2014). The reliance on chemical pesticides to mitigate yield losses caused by FAW has increased production costs, rendering maize farming less profitable. In India, where a significant number of farmers are smallholders facing financial constraints, the emergence of FAW as a new invasive pest underscores the dearth of experimental data on integrated pest management (IPM). Recently, various biorational management strategies have gained prominence for FAW control. Hence, it is imperative to assess the effectiveness and potential of diverse biorational management strategies and indigenous natural enemies for potential integration into comprehensive IPM strategies.
Pheromones have proven to be valuable in various aspects of insect pest management, including monitoring, mass-trapping, and disrupting mating patterns, and have been particularly effective in tracking male populations, as documented by Mitchell et al. (1985, 1989). Employing pheromone traps for the surveillance of FAW adults emerges as the most effective method for determining the requisite number of pesticide applications needed to manage the pest in maize, as indicated by Cruz et al. (2010). Monitoring through pheromone traps is considered a critical activity and a fundamental tool in the proactive control of insect pests, as highlighted by Stokstad (2017a, b) and Hendrichs et al. (2021). Bird perches play a crucial role in supporting insectivorous birds that actively prey on insect pests throughout the entire crop growth period. The incorporation of bird perches is a fundamental component of IPM, proving highly effective in reducing populations of lepidopteran larvae. Rao et al. (1998) underscored the constructive contribution of birds in significantly diminishing larval populations of Spodoptera litura and Helicoverpa armigera in groundnut crops. Seed treatment is one of the most important IPM technologies which has the greatest potential to tackle FAW infestation especially during early growth stages of maize (Chinwada et al. 2023). Furthermore, treating seeds with chemicals requires less insecticide, reduces environmental contamination, and exposure to natural enemies (Nault et al. 2004).
The use of biopesticides, including entomopathogenic formulations and botanicals, represents an exciting alternative to synthetic pesticides. This approach is advantageous due to its cost-effectiveness, rapid degradation, lower likelihood of harming natural enemies, and overall environmentally friendly nature (Sisay et al. 2019). The FAW has been identified as highly susceptible to various entomopathogens, including fungi, viruses, bacteria, and nematodes (Molina-Ochoa et al. 2003). The use of entomopathogens has proven to be an effective management strategy, particularly during severe outbreaks (De Faria and Wraight 2007). Entomopathogenic fungi such as Beauveria bassiana, Metarrhizium anisopliae, and nematodes, i.e. Heterorhabditis indica and Steinernema carpocapsae NBAIRS 59, were effective in the management of FAW (Haase et al. 2015; Akutse et al. 2019; Patil et al. 2022; Sayed et al. 2022). Fakeer et al. (2023) found that entomopathogenic fungi isolates B. bassiana AUMC3563 and M. anisopliae AUMC2605 were effective against S. frugiperda, resulting in mortality rates ranging from 10.0 to 80.33%. EPNs have demonstrated efficacy in controlling various lepidopteran species, making them a promising option for pest management (Andalo et al. 2010; De Oliveira Giannasi et al. 2018; Viteri et al. 2018). Various natural enemies have been identified in different geographical areas in association with this pest. In India, Shylesha et al. (2018) documented the presence of egg, larval, larval–pupal parasitoids, and predators actively preying on various developmental stages of the FAW on maize. Veena et al. (2023) investigated the impact of parasitoid female age and host egg age on the parasitization rates of S. frugiperda eggs by Trichogramma pretiosum and Telenomus remus. Results revealed that increasing age in T. pretiosum led to a decrease in parasitism and adult emergence. In contrast, T. remus females showed no significant effect on parasitization with age, but younger females produced a more male-biased progeny, and optimal parasitism occurred in 24- and 48-h-old eggs. Several botanicals such as azadirachtin were also found effective against larvae of FAW (Tulashie et al. 2021). Firake et al. (2023) found that bamboo-leaf prickly ash (Zanthoxylum armatum) extract has strong insecticidal and oviposition inhibition activity against FAW at a rate of 22.0 mL/L. GCMS analysis revealed that the most abundant constituents of the composition are monoterpenes, sesquiterpenes and their derivatives (55%), followed by fatty acids and their derivatives (34%), and aromatic compounds (10%).
Keeping in view of the above aspects, it is hypothesized that the integration of different strategies including slow-release pheromone lures, bird perches, botanicals, and biopesticides would reduce infestation and damage by FAW in maize. Therefore, in the present study, five different treatments including slow-release pheromone lures, inanimate bird perches, botanicals, biopesticides, and newer generation chemical molecules as seed treatment were evaluated to determine a suitable package for the sustainable management of FAW through field trials.
Materials and methods
Description of the study area
The study was conducted during winter (December 2022–April 2023) and rainy (July–November 2023) seasons at Winter Nursery Centre, (17.325429" N latitude and 78.397010" E longitude) ICAR-Indian Institute of Maize Research, Rajendranagar, Hyderabad, Telangana.
Experimental design and trial management
The maize single cross hybrid DHM 121 was sown at a spacing of 75 × 20 cm in 5-m row length with a plot size of 60 m2 for each module. The experiments were laid out in randomized block design with four replications. Five different treatments were evaluated and compared the effectiveness with control. Treatment 1 consisted of five components: (i) Seed treatment utilizing Chlorantraniliprole 50 FS at a rate of 5.6 ml per kilogram of seed. (ii) Pheromone traps were strategically installed at a density of 4 traps per acre after germination. (iii) Bird perches were erected at a rate of 10 per acre after germination. (iv) At 20 days after germination (DAG), a spray application of azadirachtin 1500 ppm was administered at a concentration of 5 ml per litre. (v) At 35 DAG, M. anisopliae formulation with a spore count of 1 × 108 cfu/g (1 kg per acre) was applied at a rate of 5 g per litre. Treatment 2 consisted of three components: (i) The installation of pheromone traps at 4 traps per acre. (ii) At 15 DAG, a spray application of azadirachtin 1500 ppm was administered at a concentration of 5 ml per litre. (iii) At 30 DAG, S. carpocapsae NBAIRS 59 formulation was applied at a rate of 20 g per litre. Treatment 3 comprised of four components: (i) The installation of pheromone traps at 4 traps per acre. (ii) The erection of bird perches at a rate of 10 per acre. (iii) A spray application of azadirachtin 1500 ppm at 15 DAG with a concentration of 5 ml per litre. (iv) At 30 DAG, B. bassiana formulation was applied at a rate of 5 g per litre. Treatment 4 included three components: (i) Controlled release FAW pheromone traps at a density of 16 traps per acre. (ii) Spray of B. bassiana at 5 g per litre at 15 DAG. (iii) Spray of Pseudomonas fluorescens at 10 g per litre at 30 DAG. Treatment 5 consisted of two components: (i) Chlorantraniliprole 18.5 SC at a rate of 80 ml per acre, applied at a concentration of 0.4 ml per litre at 20 DAG. (ii) Emamectin benzoate 5 SG at a rate of 80 g per acre was applied at 0.4 g per litre at 30 DAG. The untreated control group did not receive any plant protection measures throughout the experiment. The crop was raised by following the recommended agronomic practices, viz. the cultivation process commenced with thorough land preparation, involving deep ploughing followed by harrowing and levelling. Urea (46%N)-125 kg ha−1 was applied in three equal splits at the time of sowing, 30–35 and 50–55 days after sowing. Diammonium phosphate (18%N, 46%P2O5)-125 kg ha−1 was applied at the time of sowing. Muriate of potash (60%K2O)-50 kg ha−1was applied at the time of sowing and at 50–55 days after sowing. The crop was sown with a spacing of 75 × 20 cm. As part of the pre-emergence strategy, atrazine was applied at a rate of 2.5 kg ha−1, mixed in 500 L of water. Throughout the growth stages, irrigation was administered immediately after sowing, at the knee-high stage, flowering, and during grain filling stage. The careful timing of these nutrient applications aimed to optimize crop development and yield. Harvesting was carried out when cobs reached a moisture content of 20–25%. This meticulous approach to the maize cultivation from land preparation to harvest was designed to enhance overall crop productivity and quality. In each treatment, the interventions were made as per the schedule for two consecutive seasons.
Data Collection
Data were collected from 20 randomly selected plants by observing foliar damage on maize before as well as 7 and 14 days after each spray imposition. Maize leaves/whorls with pin holes, ragged edges, large irregular holes on leaves/whorls, and skeletonized leaves giving a window pane appearance are used as basis for plant damage. Leaf damage rating score was recorded on maize from 20 randomly selected plants before as well as 7 and 14 days after each spray imposition based on rating scale for leaf damage caused by FAW. The rating scale included categories such as 1—healthy plant/no damage/visible symptoms (Resistant); 2—few short/pin size holes/scraping on few leaves (1–2) (Resistant); 3—short/pin size holes/scraping on several leaves (3–4) (Resistant); 4—short/pin size holes/scraping on several leaves (5–6) and a few long elongated lesions (1–3 Nos) up to 2.0-cm length present on whorl and/or adjacent fully opened leaves (Resistant); 5—several holes with elongated lesions (4–5 Nos) up to 4.0-cm length and uniform/irregular shaped holes present on whorl and or adjacent fully opened leaves (Moderately Resistant); 6—several leaves with elongated lesions (6–7 Nos) up to 6.0-cm length and uniform/irregular shaped holes present on whorl and adjacent fully opened leaves (Moderately Resistant); 7—several long lesions (> 7 Nos) up to 10-cm length and uniform/irregular shaped holes common on one-half of the leaves present on whorl and adjacent fully opened leaves (Susceptible); 8—several long lesions > 10-cm length and uniform/irregular shaped holes common on one half to two-thirds of leaves present on whorl and adjacent fully opened leaves (Susceptible); and 9—complete defoliation of whorl of the plant (Susceptible) (Lakshmi Soujanya et al. 2022). Similarly, the natural enemies including spiders, coccinellids, and earwigs were monitored from the same 20 randomly selected plants before, as well as 7 and 14 days after each spray application. Grain yield was recorded on each treatment at 12% moisture content at the time of harvest and computed on a hectare basis. The benefit–cost ratio was calculated based on the minimum support price of maize. The sequential details of the different interventions implemented in each module are furnished in Table 1.
Statistical analysis
Data of per cent plant infestation, leaf damage rating score, number of natural enemies, and yield were subjected to analysis of variance (ANOVA) in a randomized complete block design using SAS version 9.3. The significance of differences between treatment means was judged by the least significant difference (LSD) at P ≤ 0.05.
Results
Per cent plant infestation and leaf damage rating score
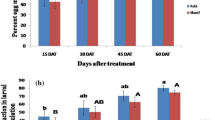
The pooled data of two seasons were presented here. At 7 days (F5, 15 = 59.46; P < 0.0001) after the first spray, the per cent plant infestation was significantly lowest in Treatment 1 (18.1) followed by Treatment 5 (24.3), Treatment 4 (35.6), Treatment 3 (42.5), and Treatment 2 (45.0) compared to untreated control (45.0) (Table 2). At 14 days (F5,15 = 27.62; P < 0.0001) after the first spray, the per cent plant infestation was lowest in Treatment 1 (13.1) followed by Treatment 5 (14.3), Treatment 4 (18.7), Treatment 3 (23.1), and Treatment 2 (25.0) in comparison with control (38.7). At 7 days (F5, 15 = 51.48; P < 0.0001) after the second spray, the per cent plant infestation was significantly lowest in Treatment 1 (7.5) and Treatment 5 (9.3), Treatment 4 (11.2), Treatment 3 (13.7), and Treatment 2 (15.6) when compared to untreated control (33.1). At 14 days (F5, 15 = 134.64; P < 0.0001) after the second spray, the per cent plant infestation was significantly lowest in the Treatment 1 (3.1) followed by the Treatment 5 (4.3), Treatment 4 (5.6), Treatment 3 (6.8), and Treatment 2 (8.7) compared to untreated control (30.6).
At 7 and 14 days after the first spray, the pooled mean leaf damage rating of the two seasons was in the range of 2.4–2.8 among all the IPM treatments, whereas in untreated control, it was 3.8–4.1 (Table 3). The minimum leaf damage rating was observed in Treatment 1 (2.4, 2.3) followed by Treatment 5 (2.5, 2.3), Treatment 4 (2.6, 2.5), Treatment 3 (2.7, 2.6), and Treatment 2 (2.8, 2.7) at 7 (F5, 15 = 65.40; P < 0.0001) and 14 days (F5, 15 = 85.93; P < 0.0001) after the first spray, respectively. The pooled mean leaf damage rating was in the range of 1.3–2.6 at 7 (F5, 15 = 161.98; P < 0.0001) and 14 days (F5, 15 = 49.70; P < 0.0001) after the second spray, whereas in untreated control, it was 4.7–5.0. The minimum leaf damage rating was observed in Treatment 1 (2.1, 1.3) followed by Treatment 5 (2.2, 1.7), Treatment 4 (2.3, 2.1), Treatment 3 (2.4, 2.3), and Treatment 2 (2.6, 2.5) at 7 and 14 days after the second spray, respectively.
Occurrence of natural enemies
Due to the low number of natural enemies such as coccinellids, spiders, and earwigs, the count was aggregated collectively rather than being recorded individually. Natural enemies such as spiders, coccinellids, and earwigs were recorded at 7 and 14 days after the first and second sprays. The pooled data of two seasons indicated that maximum number of natural enemies were observed in untreated control (10.3, 9.8) followed by Treatment 1 (8.6, 9.0), Treatment 4 (8.2, 8.7), Treatment 3 (7.8, 8.2), and Treatment 2 (7.6, 7.8) compared to Treatment 5 (2.7, 2.8) at 7 (F5, 15 = 16.22; P < 0.0001) and 14 days (F5, 15 = 24.7; P < 0.0001) after the first spray, respectively (Table 4). The same trend was followed at 7 (F5, 15 = 21.77; P < 0.0001) and 14 days (F5, 15 = 14.1; P < 0.0001) after the second spray. The population of natural enemies was high in untreated control (11.0, 10.3) followed by Treatment 1 (8.8, 9.1), Treatment 4 (8.3, 8.6), Treatment 3 (7.8, 8.3), and Treatment 2 (7.7, 7.8) compared to Treatment 5 (2.1, 3.6). The population of natural enemies was quite high in untreated control compared to remaining modules as there were no chemical interventions in control plot.
Grain yield and economics
The effectiveness of Treatment 1 was also reflected in the grain yield (F5, 15 = 127.84; P < 0.0001) (Table 5). Treatment 1 recorded the highest grain yield of 51.5 q.ha−1 followed by Treatment 5 (49.8 q.ha−1), Treatment 4 (47.2 q.ha−1), Treatment 3 (46.5 q.ha−1), and Treatment 2 (43.9 q.ha−1) whereas the lowest grain yield was observed in untreated control (29.0 q.ha−1). The per cent increase in grain yield obtained over control were 77.4, 71.5, 62.5, 60.1, and 51.3 in Treatment 1, Treatment 5, Treatment 4, Treatment 3, and Treatment 2, respectively. The highest per cent avoidable grain yield loss of 43.6 was observed in Treatment 1 followed by Treatment 5 (41.7) and Treatment 4 (38.4), Treatment 3 (37.5), and Treatment 2 (33.9). Similarly, the cost–benefit ratio was maximum in Treatment 1 (1:2.1) followed by Treatment 5 (1:2.0) and Treatment 3 (1:2.0) whereas Treatment 4 and Treatment 2 registered the lowest BCR of 1:1.9 and 1:1.8, respectively.
Discussion
Studies reported by various researchers revealed that the application of integrated approaches was successful in the management of the FAW population in maize (Thilagam et al. 2020; Varshney et al. 2021; Warkad et al. 2021; Keerthi et al. 2023; Mendesil et al. 2023). In the present study, different interventions were implemented simultaneously to substitute chemical pesticides with newer generation seed treatment molecules, slow-release pheromone lures, bird perches, and biopesticides for sustainable management of FAW based on action thresholds. In general, the recommended spray interventions start at 2–3 weeks after seedling emergence (Van den Berg et al. 2021). However, the timings of intervention depend upon action thresholds based on genotype and environmental conditions. In the present study, action thresholds were determined based on pheromone trap catches and per cent plant infestation depending on the phenological stage of the crop. Similarly, Prasanna et al. (2018) stated that the action thresholds are to be used at the early stages of maize development as the first generation of FAW is completed by that time. Recently, information on action thresholds based on economic injury levels have been developed in Colombia for two maize hybrids, again at early and late whorls stages in terms of FAW density per plant (Jaramillo-Barrios et al. 2020).
Among the different treatment, Treatment 1 (seed treatment with Chlorantraniliprole 50 FS at the rate of 5.6 ml per kg seed + installation of pheromone traps with controlled release NBAIR lures at the rate of 4 per acre + erection of bird perches at the rate of 10 per acre + spray with azadirachtin 1500 ppm at the rate of 5 ml per litre at 20 DAG + application of M. anisopliae formulation with spore count of 1 × 108 cfu.g−1 (1 kg per acre) at the rate of 5 g per litre at 35 DAG) was found effective in reducing per cent plant infestation and leaf damage rating score followed by Treatment 5 (Chemical control) and Treatment 4 (installation of pheromone traps with controlled release NBAIR lures (16 per acre) + first spray of B. bassiana at the rate of 5 g per litre at 15 DAG + second spray P. fluorescens at the rate of 10 g per litre at 30 DAG). The effect of Treatment 3 (installation of pheromone trap at the rate of 4 per acre + erection of bird perches at the rate of 10 per acre + spray with azadirachtin 1500 ppm at the rate of 5 ml per litre at 15 DAG + application of B. bassiana formulation at the rate of 5 g per litre at 30 DAG) and Treatment 2 (installation of pheromone traps at the rate of 4 per acre + spray with azadirachtin 1500 ppm at the rate of 5 ml per litre at 15 DAG + application of S. carpocapsae NBAIRS 59 formulation at the rate of 20 g per litre at 30 DAG) was intermediate when compared to untreated control. The interventions followed in Treatments 1, 2, 3, and 4 are sustainable and safe to non-target organisms and the environment. Kavyashree et al. (2022) also reported that the adoption of IPM technologies including installation of pheromone traps (10/ ha) at the time of sowing, removal of egg masses and neonates, application of sugar solution 10% (two sprays at fortnightly intervals, 1st at 15 and 2nd at 30 DAS), and spraying of M. rileyi @ 3 g. L−1 was found effective against FAW.
In Treatment 1, seed treatment protected up to 20 DAG as it is a promising technology for managing FAW in the early stages of maize plants. The present result was in agreement with Oliveira et al. (2022) who reported that seed treatment is effective and is more advantageous if incorporated with other pest management strategies. Similarly, Suganthi et al. (2022) observed that seed treatment in maize with diamide insecticides would be useful for up to 15 days in preventing FAW foliar damage. Chlorantraniliprole when applied through seed treatment is absorbed by maize plants and translocated into the whole plant from the seed through roots to stem and leaves (Pes et al. 2020). It is an anthranilic diamide (IRAC Group 28) that binds to the ryanodine receptor with selective potency against insect versus mammalian forms of the receptor (Selby et al. 2017). Seye et al. (2023) reported that seed treatment with cyantraniliprole reduced the severity of leaf damage (67%) by FAW in maize.
As pheromones produced by insects are crucial for the sexual communication of males and females (Raina 1997), exploiting this behaviour for monitoring is helpful for the successful management of FAW. Pheromone traps are considered as the best means to monitor FAW adults and also to decide the time of insecticide application to control the pest in maize (Cruz et al. 2010, 2012; Abang et al. 2022). Similar results were obtained in the present study, in which NBAIR slow-release pheromone lures were found effective in monitoring and reducing FAW populations in Treatments 1, 2, 3, and 4. Erection of bird perches also resulted in a reduction of pest populations in Treatments 1 and 3 as these made insect-feeding birds such as Black Drango convenient to sit and prey on FAW larvae. Similar observations were made in Australian soybean fields, where the installation of T-shaped perches expanded the habitat for birds, leading to a decline in pest numbers (Lindell et al. 2018). Modern farmers embracing IPM strategies, such as ecological pest control involving insectivorous birds, have adopted these practices (Seni and Halder 2022). Kumar and Cheema (2020) employed T-shaped bird perches to leverage the services of insectivorous birds in feeding on lepidopteran larvae. The lowest population of H. armigera larvae (2.29 individuals/m row length) was observed in plots featuring trap crops such as marigold and bird perches, along with neem insecticidal sprays in Egyptian clover. Mehta et al. (2010) also addressed H. armigera management in tomato fields using neem biopesticide and T-shaped bird perches, and the lower larval survival was observed in plots with T-shaped perches compared to control.
Several researchers reported that biopesticides as potential tool for integrated pest management which are environmentally friendly and can replace chemical pesticides for insect pest control (Peters 1996; Bateman et al. 2018Rioba et al. 2020). The use of biopesticides such as azadirachtin, entomofungal formulation—M. anisopliae, and entomopathogenic nematode formulation—S. carpocapsae NBAIRS 59 in conjunction with other management strategies which were included in Treatments 1, 2, 3, and 4 resulted in a reduction of synthetic insecticides and were found successful in managing FAW. The previous studies reported that botanicals such as neem extracts have shown 70% mortality of FAW (Maredia et al. 1992; Silva et al. 2015); Tulashie et al. (2021) observed azadirachtin as a potential bioinsecticide against larvae of FAW. Sisay et al. (2019) examined an extract from Azadirachta indica against FAW and observed a mortality rate exceeding 95% within 72 hours of application. Akutse et al. (2019) reported that certain isolates of M. anisopliae demonstrated an ovicidal effect, resulting in egg mortality ranging from 79.5 to 87%. Specifically, isolates ICIPE41 and ICIPE7 of M. anisopliae caused mortality rates of 96.5% and 93.7% in neonate larvae of FAW, respectively. Garcia and Bautista (2011) documented that B. bassiana (strain Bb42), isolated from FAW larvae collected in the field, exhibited a high virulence with a mortality rate of 96.6% in the 2nd instar larvae at a concentration of 1 × 109 conidia ml-1. Ramirez-Rodriguez and Sanchez-Pena (2016) evaluated the pathogenicity of a B. bassiana strain, initially isolated from soil but later introduced as an endophyte in maize. It was confirmed that the endophytic strain caused 75% larval mortality by the 14th day of the experiment. Additionally, Cruz-Avalos et al. (2019) observed that three strains of M. anisopliae, Ma22, Ma41, and Mr8, induced 100% mortality in both eggs and neonate larval stages of FAW. The previous studies also suggested that EPNs have potential as biological control agents against FAW (Caccia et al. 2014; Viteri et al. 2018). Molina Ochoa et al. (1999) found that S. carpocapsae and S. riobravis were effective in controlling FAW during the prepupal stage. Researchers have suggested that the use of entomopathogenic nematodes (EPNs) on maize silk could enhance mortality of FAW during the prepupal stage (Negrisoli et al. 2010). Garcia et al. (2008) reported that around 280 infective juveniles of Steinernema sp. can kill 100% of the third-instar FAW, while 400 infective juveniles of H. indica cause 75% mortality of FAW. Similarly, Acharya et al. (2020) reported that the FAW larvae were most susceptible to S. carpocapsae. Patil et al. (2022) reported that both H. indica and S. carpocapsae caused 100% mortality in the third- and fourth-instar larvae of FAW, whereas 82.5 and 75.0% mortality were observed in pupae, respectively. Similarly, a study conducted by Ratnakala et al. (2023), reported that H. indica and S. carpocapsae exhibited notable ovicidal, larvicidal, and pupicidal effects on S. frugiperda. Additionally, these EPNs had a significant impact on the adult stage, leading to deformities and mortality.
In the present study, Treatment 1 also recorded a higher number of natural enemies, viz. spiders, ladybird beetles, and earwigs followed by Treatments 4, 3, and 2 compared to Treatment 5. The minimum population of beneficial fauna was observed in Treatment 5 due to the toxic nature of chemical pesticides. However, a maximum number of natural enemies were observed in untreated control due to no spray intervention.
The present result is in agreement with Geetha (2021) who observed the maximum population of natural enemies in integrated pest management modules compared to non-IPM fields. Kavyashree et al. (2022) also reported higher populations of ants and coccinellids in the biointensive integrated module against FAW in maize. Molina-Ochoa et al. (2003) documented the presence of 150 species of parasitoids and parasites associated with FAW in America and the Caribbean basin. Shylesha et al. (2018) reported the existence of egg, larval, and larval–pupal parasitoids, as well as predators actively preying on different developmental stages of the FAW on maize. In the present study, Treatment 1 recorded the highest grain yield, maximum avoidable yield losses and also contributed a higher cost–benefit ratio in comparison with other modules. The present result is in line with Varshney et al. (2021) who reported that IPM strategy comprising installation of controlled release FAW pheromone traps, four releases of T. pretiosum Riley, two sprays of neem oil, and one spray of each Bacillus thuringiensis (NBAIR-BT25) and M. anisopliae (NBAIR Ma-35) resulted in 38.3 and 42.2% gain in yield per acre during rabi and kharif (2018–19), respectively. Similarly, Srinivasan et al. (2022) reported 50.2% avoidable yield losses in complete protection, i.e. application of recommended insecticides by Tamil Nadu Agricultural University when the incidence was observed while 45.36% was noticed in window-based application (azadirachtin 1% EC @ 2 ml.L−1 or emamectin benzoate 5% SG @ 200 g ai/ ha for first window (15–20 days) and spinetoram 11.7% w/w SC @ 30 g a.i/ha or Chlorantraniliprole 18.5% SC @ 40 g a.i/ha or Novaluron 10EC @ 100 g a.i./ha) against FAW in maize.
Conclusions
The findings of this study revealed that Treatment 1, comprised of various integrated pest management (IPM) approaches such as seed treatment with Chlorantraniliprole 50 FS at the rate of 5.6 ml per kg seed, setting up of pheromone traps at the rate of 4 per acre, installation of bird perches at the rate of 10 per acre, spray application of azadirachtin 1500 ppm at the rate of 5 ml per litre, and foliar spray of M. anisopliae with spore count of 1 × 108 cfu/g (1 kg per acre) at the rate of 5 g per litre effectively controlled FAW infestation in maize. Additionally, Treatment 1 enhanced natural enemy population, contributing to a more sustainable pest management strategy and also resulted in the highest grain yield among all the treatments, highlighting its economic viability with a high cost–benefit ratio. This emphasizes the importance of adopting IPM strategies, which combine cultural, biological, and adopting chemical control measures as last resort, for successful and sustainable FAW management in maize. Moreover, Treatment 1 not only addresses immediate FAW infestation concerns but also helps minimize the likelihood of resistance development, providing a long-term solution for small holder maize farmers. The integrated nature of this approach aligns with effective pest management strategies while considering the ecological balance within the maize ecosystem.
Data availability
All data generated and analysed during this study are indicated in the manuscript.
Abbreviations
- a.i/ha:
-
Active ingredient per hectare
- CBR:
-
Cost–benefit ratio
- cfu:
-
Colony-forming units
- DAG:
-
Days after germination
- EC:
-
Emulsifiable concentrate
- FAW:
-
Fall armyworm
- g.L− 1 :
-
Gram per litre
- ICAR:
-
Indian Council of Agricultural Research
- IPM:
-
Integrated pest management
- IRAC:
-
Insecticide Resistance Action Committee
- NBAIR:
-
National Bureau of Agricultural Insect Resources
- ppm:
-
Parts per million
- ml.kg− 1 :
-
Millilitre per kilogram
- q.ha− 1 :
-
Quintal per hectare
- SC:
-
Suspension concentrate
- SG:
-
Water-soluble granules
References
Abang AF, Nanga SN, Esi Ndanda RM, Doumtsop Fotio AR, Gonder MK, Kouebou C, Suh C, Fotso Kuate A, Fiaboe KK, Hanna R (2022) Reliability of pheromone trap catches and maize plant damage as criteria for timing fall armyworm control interventions in humid forest agroecology of central Africa. J Econ Entomol 115(6):1806–1816
Abudulai M, Seini SS, Nboyine J, Seidu A, Ibrahim YJ (2018) Field efficacy of some insecticides for control of bollworms and impact on non-target beneficial arthropods in cotton. Exp Agric 54(2):315–322
Acharya R, Hwang HS, Mostafiz MM, Yu YS, Lee KY (2020) Susceptibility of various developmental stages of the fall armyworm, Spodoptera frugiperda, to entomopathogenic nematodes. InSects 11(12):868
Adhikari K, Bhandari S, Dhakal L, Shrestha J (2020) Fall armyworm (Spodoptera frugiperda): a threat in crop production in Africa and Asia. Peruv J Agron 4(3):121–133
Akutse KS, Kimemia JW, Ekesi S, Khamis FM, Ombura OL, Subramanian S (2019) Ovicidal effects of entomopathogenic fungal isolates on the invasive fall armyworm Spodoptera frugiperda (Lepidoptera: Noctuidae). J Appl Entomol 143(6):626–634
Andaló V, Cavalcanti RS, Molina JP, Moino A Jr (2010) Substrates for storing entomopathogenic nematodes (Rhabditida: Steinernematidae, Heterorhabditidae). Sci Agric 67:342–347
Barros EM, Torres JB, Bueno AF (2010) Oviposition, development, and reproduction of Spodoptera frugiperda (JE Smith) (Lepidoptera: Noctuidae) fed on different hosts of economic importance. Neo Trop Entomol 39:996–1001
Bateman ML, Day RK, Luke B, Edgington S, Kuhlmann U, Cock MJ (2018) Assessment of potential biopesticide options for managing fall armyworm (Spodoptera frugiperda) in Africa. J Appl Entomol 142(9):805–819
Busato GR, Grützmacher AD, Garcia MS, Giolo FP, Zotti MJ, Bandeira JDM (2005) Exigênciastérmicas e estimativa do número de gerações dos biótipos" milho" e" arroz" de Spodoptera frugiperda. Pesqui Agro Pecu Bras 40:329–335
CABI (2022) Spodoptera frugiperdaIn: invasive species compendium. CAB International, Wallingford, UK. https://www.cabi.org/ISC/fallarmyworm
Caccia MG, Del Valle E, Doucet ME, Lax P (2014) Susceptibility of Spodoptera frugiperda and Helicoverpa gelotopoeon (Lepidoptera: Noctuidae) to the entomopathogenic nematode Steinernema diaprepesi (Rhabditida: Steinernematidae) under laboratory conditions. Chil J Agric Res 74:123–126
Chinwada P, Fiaboe KKM, Akem C, Dixon A, Chikoye D (2023) Assessment of effectiveness of maize seed treated with cyantraniliprole and thiamethoxam for management of fall armyworm, Spodoptera frugiperda (JE Smith). Crop Prot 174:106418
Cruz I, Figueiredo MDLC, Silva RBD, Silva IFD, Paula CD, Foster JE (2012) Using sex pheromone traps in the decision-making process for pesticide application against fall armyworm (Spodoptera frugiperda [Smith][Lepidoptera: Noctuidae]) larvae in maize. Int J Pest Manag 58(1):83–90
Cruz I, Figueiredo M, Da Silva RB (2010) Monitoramento de adultos de Spodoptera frugiperda (JE Smith) (Lepidoptera: Noctuidae) e Diatraea saccharalis (Fabricius) (Lepidoptera: Pyralidae) em algumas regiões produtoras de milho no Brasil.
Cruz-Avalos AM, Bivián-Hernández MDLÁ, Ibarra JE, Del Rincón-Castro MC (2019) High virulence of Mexican entomopathogenic fungi against fall armyworm,(Lepidoptera: Noctuidae). J Econ Entomol 112:99–107
De Faria MR, Wraight SP (2007) Mycoinsecticides and mycoacaricides: a comprehensive list with worldwide coverage and international classification of formulation types. Biol Control 43(3):237–256
De Oliveira Giannasi A, Roque Brambila C, Zart M, Guide BA, Alves VS (2018) Assessment of entomopathogenic nematodes in Agrotis ipsilon (Lepidoptera: Noctuidae) under laboratory and greenhouse conditions. Rev Colomb Entomol 44(1):25–31
Fakeer M, Hammam GH, Joo JH, Hussein KA (2023) Applicability of entomopathogenic fungi and essential oils against the fall armyworm Spodoptera frugiperda (Lepidoptera: Noctuidae). Int J Trop Insect Sci 44:1–9
Firake DM, Ghosh R, Kumar M, Milton AAP, Sanjukta RK, Behere GT, Pandit S (2023) Bioactivity of Zanthoxylum armatum fruit extract against Spodoptera frugiperda and Tuta absoluta. J Plant Dis Prot 130(2):383–392
Garcia LC, Raetano CG, Leite LG (2008) Application technology for the entomopathogenic nematodes Heterorhabditis indica and Steinernema sp. (Rhabditida: Heterorhabditidae and Steinernematidae) to control Spodoptera frugiperda (J.E. Smith) (Lepidoptera: Noctuidae) in corn. Neotrop Entomol 37:305–311
García C, Bautista N (2011) Pathogenicity of isolates of entomopathogenic fungi against Spodoptera frugiperda (Lepidoptera: Noctuidae) and Epilachna varivestis (Coleoptera: Coccinellidae). Colombian J Entomol 37:217–222
Geetha B (2021) Evaluation of IPM technologies for the management of fall armyworm, Spodoptera frugiperda (J E Smith) on maize in Tamil Nadu. Indian J plant prot 49(1)
Goergen G, Kumar PL, Sankung SB, Togola A, Tamò M (2016) First report of outbreaks of the fall armyworm Spodoptera frugiperda (JE Smith) (Lepidoptera, Noctuidae), a new alien invasive pest in West and Central Africa. PLoS ONE 11(10):0165632
Gutiérrez-Moreno R, Mota-Sanchez D, Blanco CA, Whalon ME, Terán-Santofimio H, Rodriguez-Maciel JC, DiFonzo C (2019) Field-evolved resistance of the fall armyworm (Lepidoptera: Noctuidae) to synthetic insecticides in Puerto Rico and Mexico. J Econ Entomol 112(2):792–802
Haase S, Sciocco-Cap A, Romanowski V (2015) Baculovirus insecticides in Latin America: historical overview, current status and future perspectives. Viruses 7(5):2230–2267
Hendrichs J, Pereira R, Vreysen MJ (2021) Area-wide integrated pest management: development and field application. Taylor & Francis, p 1028
Jaramillo-Barrios CI, Varón-Devia EH, Monje-Andrade B (2020) Economic injury level and action thresholds for Spodoptera frugiperda (JE Smith) (Lepidoptera: Noctuidae) in maize crops. Rev Fac Nac Agron Medellin 73(1):9065–9076
Kavyashree BA, Deshmukh SS, Kalleshwaraswamy CM, Shivanna BK, Sridhar S (2022) Impact of various modules on ants and coccinellids of fall armyworm, Spodoptera frugiperda (J. E Smith) in Maize ecosystem. Biol Forum 414(2):1555–1558
Keerthi MC, Suroshe SS, Doddachowdappa S, Shivakumara KT, Mahesha HS, Rana VS, Gupta A, Murukesan A, Casini R, Elansary HO, Shakil NA (2023) Bio-intensive tactics for the management of invasive fall armyworm for organic Maize production. Plants 12(3):685
Kumar R, Cheema HK (2020) Restricting lepidopteran herbivory through trap cropping and bird perches in Egyptian clover (Trifolium alexandrinum L.). Egypt J Biol Pest Control 30(1):1–8
Lakshmi Soujanya PL, Sekhar JC, Yathish KR, Karjagi CG, Rao KS, Suby SB, Jat SL, Kumar B, Kumar K, Vadessery J, Subaharan K (2022) Leaf damage based phenotyping technique and its validation against fall armyworm, Spodoptera frugiperda (JE Smith), in Maize. Front Plant Sci 13:906207
Lindell C, Eaton RA, Howard PH, Roels SM, Shave ME (2018) Enhancing agricultural landscapes to increase crop pest reduction by vertebrates. Agric Ecosyst Environ 257:1–11
Manupriya (2019) Fall armyworm, destroyer of maize farms, causes concern in India. Published in Mongaway series on 2 September 2019. https://india.mongabay.com/2019/09/fall-armyworm-destroyer-of-maize-farms-causes-concern-in-india/
Maredia KM, Segura OL, Mihm JA (1992) Effects of neem, Azadirachta indica on six species of maize insect pests. Trop Pest Manag 38:190–195
Mehta KS, Patyal SK, Rana RS, Sharma RC (2010) Eco-friendly techniques for the management of Helicoverpa armigera in tomato. J Biopest 3:296–303
Mendesil E, Tefera T, Blanco CA, Paula-Moraes SV, Huang F, Viteri DM, Hutchison WD (2023) The invasive fall armyworm, Spodoptera frugiperda, in Africa and Asia: responding to the food security challenge, with priorities for integrated pest management research. J Plant Dis Prot 130(6):1175–1206
Mitchell ER, Tumlinson JH, McNeil JN (1985) Field evaluation of commercial pheromone formulations and traps using a more effective sex pheromone blend for the fall armyworm (Lepidoptera: Noctuidae). J Econ Entomol 78(6):1364–1369
Mitchell ER, Agee HR, Heath RR (1989) Influence of pheromone trap colour and design on capture of male velvet bean caterpillar and fall armyworm moths (Lepidoptera: Noctuidae). J Econ Entomol 15:1775–1784
Molina-Ochoa J, Lezama-Gutierrez R, Hamm JJ, Wiseman BR, Lopez-Edwards M (1999) Integrated control of fall armyworm (Lepidoptera: Noctuidae) using resistant plants and entomopathogenic nematodes (Rhabditida: Steinernematidae). Fla Entomol 82:263–271
Molina-Ochoa J, Lezama-Gutierrez R, Gonzalez-Ramirez M, Lopez-Edwards M, Rodriguez-Vega MA, Arceo-Palacios F (2003) Pathogens and parasitic nematodes associated with populations of fall armyworm (Lepidoptera: Noctuidae) larvae in Mexico. Fla Entomol 86(3):244–253
Nault BA, Taylor AG, Urwiler M, Rabaey T, Hutchison WD (2004) Neonicotinoid seed treatments for managing potato leafhopper infestations in snap bean. Crop Prot 23(2):147–154
Negrisoli AS, Garcia MS, Negrisoli CRCB (2010) Compatibility of entomopathogenic nematodes (Nematoda: Rhabditida) with registered insecticides for Spodoptera frugiperda (Smith, 1797) (Lepidoptera: Noctuidae) under laboratory conditions. Crop Prot 29:545–549
Oliveira C, Orozco-Restrepo SM, Alves AC, Pinto BS, Miranda MS, Barbosa MH, Pereira EJ (2022) Seed treatment for managing fall armyworm as a defoliator and cutworm on maize: plant protection, residuality, and the insect life history. Pest Manag Sci 78(3):1240–1250
Patil J, VaddarLinga V, Vijayakumar R, Kesavan Subaharan K, Om N, Bakthavatsalam N, Priyank HM, Sekhar JC (2022) Biocontrol potential of entomopathogenic nematodes for the sustainable management of Spodoptera frugiperda (Lepidoptera: Noctuidae) in maize. Pest Manag Sci 78(7):2883–2895
Pes MP, Melo AA, Stacke RS, Zanella R, Perini CR, Silva FM, CarúsGuedes JV (2020) Translocation of chlorantraniliprole and cyantraniliprole applied to corn as seed treatment and foliar spraying to control Spodoptera frugiperda (Lepidoptera: Noctuidae). PLoS ONE 15(4):0229151
Peters A (1996) The natural host range of Steinernema and Heterorhabditis spp. and their impact on insect populations. Biocontrol Sci Technol 6(3):389–402
Prasanna BM, Huesing JE, Eddy R, Peschke VM (2018) fall armyworm in Africa: A guide for integrated pest management. USAID; CIMMYT vii, 109
Prasanna BM, Huesing JE, Peschke VM, Nagoshi RN, Jia X, Wu K, Trisyono YA, Tay T, Watson A, Day R, Eddy R (2021) Fall armyworm in Asia: invasion, impacts, and strategies for sustainable management. CIMMYT 1–10
Raina AK (1997) Control of pheromone production in moths. In: Cardé RT, Minks AK (eds) Insect pheromone research. Springer, pp 21–30. https://doi.org/10.1007/978-1-4615-6371-6_2
Rakshit S, Ballal CR, Prasad YG, Sekhar JC, Lakshmi Soujanya P, Suby SB, Jat SL, Siva Kumar G, Prasad JV (2019) Fight against fall armyworm Spodoptera frugiperda (J. E. Smith). ICAR-Indian Institute of Maize Research, Ludhiana, Punjab, -52
Ramirez-Rodriguez D, Sánchez-Peña SR (2016) Endophytic Beauveria bassiana 1 in Zea mays: pathogenicity against larvae of fall armyworm. Spodoptera Frugiperda Southwest Entomol 41(3):875–878
Rao GVR, Wightman JA, Rao VR (1998) Birds as insect control agents in the groundnut crop. In: Dhindsa M, Rao S, Syamsundar P, Parasharya BM (eds) Birds in agricultural ecosystem. Society for Applied Ornithology, New Delhi, pp 79–83
Ratnakala B, Kalleshwaraswamy CM, Rajkumar M, Mallikarjuna HB (2023) Biocontrol potential of entomopathogenic nematodes against invasive fall armyworm. Spodoptera Frugiperda in India Biol Control 185:105304
Rioba NB, Stevenson PC (2020) Opportunities and scope for botanical extracts and products for the management of fall armyworm (Spodoptera frugiperda) for smallholders in Africa. Plants 9(2):207
Rose AH, Silversides RH, Lindquist OH (1975) Migration flight by an aphid, Rhopalosiphum maidis (Hemiptera: Aphididae), and a noctuid, Spodoptera frugiperda (Lepidoptera: Noctuidae). Can Entomol 107(6):567–576
Roubos CR, Rodriguez-Saona C, Isaacs R (2014) Mitigating the effects of insecticides on arthropod biological control at field and landscape scales. Biol Control 75:28–38
Sayed RM, Ibrahim SS, El-Gepaly HMKH (2022) Susceptibility of the fall armyworm, Spodoptera frugiperda (JE Smith) (Lepidoptera: Noctuidae), larvae to un-irradiated and gamma-irradiated entomopathogenic nematodes. Egypt J Biol Pest Control 32(1):119
Selby TP, Lahm GP, Stevenson TM (2017) A retrospective look at anthranilicdiamide insecticides: discovery and lead optimization to chlorantraniliprole and cyantraniliprole. Pest Manag Sci 73(4):658–665
Seni A, Halder J (2022) Role of predatory birds in insect pest management in agriculture. Agric Lett 3(2):2582–6522
Seye D, Silvie P, Brévault T (2023) Effect of maize seed treatment on oviposition preference, larval performance and foliar damage of the fall armyworm. J Appl Entomol 147(5):299–306
Sharanabasappa KCM, Asokan R, MahadevaSwamy HM, Maruthi MS, Pavithra HB, Hedge K, ShivarayNavi PST, Goergen G (2018) First report of the fall armyworm, Spodoptera frugiperda (J E Smith) (Lepidoptera: Noctuidae), an alien invasive pest on maize in India. Pest Manag Hortic Ecsyst 24(1):23–29
Shylesha AN, Jalali SK, Ankita G, Richa V, Venkatesan T, Pradeeksha S, Rakshit O, Ganiger PC, Omprakash N, Subaharan K, Bakthavatsalam N (2018) Studies on new invasive pest Spodoptera frugiperda (JE Smith) (Lepidoptera: Noctuidae) and its natural enemies. Biol Control 32(3):1–7
Silva MS, Maria S, Broglio F, Cristina R, Trindade P, Ferrreira ES, Gomes IB, Micheletti LB (2015) Toxicity and application of neem in fall armyworm. Comun Sci 6:359–364
Sisay B, Tefera T, Wakgari M, Ayalew G, Mendesil E (2019) The efficacy of selected synthetic insecticides and botanicals against Fall Armyworm, Spodoptera frugiperda. Maize Insects 10(2):45
Srinivasan T, Shanmugam PS, Baskaran V, Kavitha Z, Yasodha P, Ravi M, Sathiah N, Rabindra RJ, Krishnamoorthy SV, Muthukrishnan N, Vinothkumar B (2022) Estimation of avoidable yield loss in Maize Zea mays L. caused by the fall armyworm Spodoptera frugiperda (JE Smith) (Noctuidae: Lepidoptera). Ecol Environ Conserv 28(4):1946–1957
Stokstad E (2017a) New crop pest takes Africa at lightning speed. Science 356(6337):473–474
Stokstad E (2017b) New crop pest takes Africa at lightning speed. Science 356(6337):473–474. https://doi.org/10.1126/science.356.6337.473
Suby SB, Soujanya PL, Yadava P, Patil J, Subaharan K, Prasad GS, Babu KS, Jat SL, Yathish KR, Vadassery J, Kalia VK (2020) Invasion of fall armyworm (Spodoptera frugiperda) in India: nature, distribution, management and potential impact. Curr Sci 119:44–51
Suganthi A, Krishnamoorthy SV, Sathiah N, Rabindra RJ, Muthukrishnan N, Jeyarani S, Karthik P, Selvi C, Kumar GA, Srinivasan T, Harishankar K (2022) Bioefficacy, persistent toxicity, and persistence of translocated residues of seed treatment insecticides in maize against fall armyworm, Spodoptera frugiperda (JE Smith, 1797). Crop Prot 154:105892
Thilagam P, Dinakaran D, Nanthakumar S, Gopikrishnan A (2020) Reduction of maize fall armyworm (Spodoptera frugiperda, JE smith) population with the use of TNAU technology capsule. Int J Rec Res Life Sci 9(09):3322–3325
Tulashie SK, Adjei F, Abraham J, Addo E (2021) Potential of neem extracts as natural insecticide against fall armyworm (Spodoptera frugiperda (JE Smith)(Lepidoptera: Noctuidae). Case Stud Chem Environ Eng 4:100130
Van den Berg J, Britz C, du Plessis H (2021) Maize yield response to chemical control of Spodoptera frugiperda at different plant growth stages in South Africa. Agriculture 11(9):826
Varshney R, Poornesha B, Raghavendra A, Lalitha Y, Apoorva V, Ramanujam B, Rangeshwaran R, Subaharan K, Shylesha AN, Bakthavatsalam N, Chaudhary M (2021) Biocontrol-based management of fall armyworm, Spodoptera frugiperda (JE Smith) (Lepidoptera: Noctuidae) on Indian Maize. J Plant Dis Prot 128(1):87–95
Veena K, Hosamani A, Prabhuraj A, Hanchinal SG, Kenganal M, Deshmukh SS (2023) Efficiency of female age of egg parasitoids on parasitism of Spodoptera frugiperda (JE Smith)(Lepidoptera: Noctuidae) eggs of various ages. J Plant Dis Prot 131:1–8
Viteri DM, Linares AM, Flores L (2018) Use of the entomopathogenic nematode Steinernema carpocapsae in combination with low-toxicity insecticides to control fall armyworm (Lepidoptera: Noctuidae) Larvae. Florida Entomol 101:327–329
Warkad TP, Bhede BV, Shinde GS (2021) Development of integrated pest management module for fall armyworm Spodoptera frugiperda (JE Smith) on maize. Zool Entomol Lett 1(2):22–25
Acknowledgements
The authors are thankful to the Director, ICAR-Indian Institute of Maize Research, Ludhiana, and Acharya NG Ranga Agricultural University, Guntur, for supporting the research work. The authors gratefully acknowledge Director, ICAR-National Bureau of Agricultural Insect Resources, Bengaluru, for providing pheromone lures and biopesticides in the present study.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
PLS conceived and designed research. GAK conducted experiments and analysed the data. PLS and GAK wrote the manuscript. GAK and KRY maintained field. DVSRK, VMK, KRY, JCS, and HSJ reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Anilkumar, G., LakshmiSoujanya, P., Kumar, D.V.S.R. et al. Integrated approaches for the management of invasive fall armyworm, Spodoptera frugiperda (J. E. Smith) in maize. J Plant Dis Prot 131, 793–803 (2024). https://doi.org/10.1007/s41348-024-00914-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41348-024-00914-0