Abstract
Spodoptera frugiperda (J E Smith) (fall armyworm) (Lepidoptera: Noctuidae), is a key pest of maize that has recently entered in India causing damage and yield loss. A biocontrol-based integrated pest management (IPM) strategy was designed and evaluated in farmer’s field during rabi and kharif season (2018–2019). IPM strategy comprising installation of controlled release FAW pheromone traps, four releases of Trichogramma pretiosum Riley, two sprays of neem oil, one spray of each Bacillus thuringiensis (NBAIR-BT25) and Metarizium anisopliae (NBAIR Ma-35) resulted in 76 and 71.64% egg mass; 80 and 74.44% larval population reduction at 60 days after treatment during rabi and kharif season, respectively. Cob yield per acre in biocontrol-based IPM field was higher than the farmer’s practice (6–7 sprays of emamectin benzoate 5% SG) during both the seasons, and it resulted in 38.3 and 42.29% gain in yield per acre during rabi and kharif, respectively. Therefore, this module forms a base to manage the fall armyworm in an eco-friendly and farmer friendly manner. Future research with other alternatives has also been discussed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fall armyworm (FAW), Spodoptera frugiperda (JE Smith) (Lepidoptera: Noctuidae) is a lepidopteran and a key insect pest of maize. This pest is a native to neotropics in America and first reported as an invasive pest in Africa in the rainforest zones of Nigeria in 2016 (Akutse et al. 2019). Subsequently, it spread to different parts of Africa. Montezano et al. (2018) reported that FAW attacks 353 host plant species belonging to 76 plant families with preference to poaceae family. In India, this pest was reported on maize during May, 2018 in Karnataka. Since then, it has spread to many states of India causing havoc to maize production. In India during 2017, maize production was 28.7 million tons, but due to this insect pest, production fell by 3.2% to 27.8 million tons (Manupriya 2019). When an invasive like S. frugiperda enters a country, as an emergency response chemical insecticides are used to tackle the menace. Application of insecticides always poses a risk to environment, health and nontarget insects. In Latin America, insecticides have been used to manage this pest (Gutiérrez-Moreno et al. 2019). Their high cost and development of insecticide resistance in S. frugiperda make them unsuitable in long run. The use of synthetic insecticides was decreased by 47.8% after the introduction of Bt maize in America (Brookes and Barfoot 2017). Due to repeated use of insecticides in many countries where Bt maize was not available, it has developed resistance to several synthetic insecticides, viz., Carbamates, Organophosphates and Pyrethroids–Pyrethrins (Yu et al. 2003; Mota-Sanchez and Wise 2017). Furthermore, FAW has developed field evolved resistance to most of the Bt proteins (Cry1F, Cry1Ac, Cry1Ab, etc.) (Gutiérrez-Moreno et al. 2019) and has been reported in different regions of the USA (Sisay et al. 2018). This suggests the urgent need of an integrated management strategy for the sustainable and safe control of this invasive pest.
Many natural enemies have been found associated with this pest in different regions. Molina-Ochoa et al. (2003), documented ca. 150 species of parasitoids and parasites associated with FAW from Americas and the Caribbean basin. In India, Shylesha et al. (2018) also reported egg, larval, larval-pupal parasitoids and predators attacking different stages of this pest on maize. Biocontrol and biopesticides approaches are eco-friendly, sustainable and appropriate alternative to chemical insecticides. These approaches form a strong base, and they are the key component of any integrated pest management programme (IPM).
In India, most of the farmers are small holding farmers who cannot afford to bear the cost of expensive insecticides. Since FAW is a new invasive to India, there are no experimental data available for its management. Hence, it is crucial to determine the efficiency and potency of local natural enemies and native strain of biopesticides to deploy them further in IPM module.
In India, ICAR-National Bureau of Agricultural Insect Resources (NBAIR) holds bioagents and the compatible technologies available to tackle this pest in Indian conditions. These bioagents and compatible technologies have been tested in laboratory for their efficiency and potency. Therefore, a biocontrol-based IPM strategy was formulated by incorporating controlled release FAW pheromone traps, Trichogramma pretiosum Riley as an egg parasitoid, promising indigenous Bacillus thuringiensis isolate (NBAIR-BT25), Metarhizium anisopliae strain (NBAIR Ma-35) and neem oil to validate the module in Indian condition to manage this pest.
In this paper, we have also estimated yield and cost–benefit of biocontrol-based IPM practices and farmer’s practice for management of FAW.
Materials and methods
Insect rearing, bioagents and semiochemicals
Laboratory culture of thelytokous T. pretiosum (National Accession number: NBAII-MP-tri-70) is being continuously mass multiplied on the eggs of rice grain moth, Corcyra cephalonica Stainton (National Accession number: NBAII-MP-PYR-01) in insectary and being supplied to other organizations and farmers. A liquid formulation of Bacillus thuringiensis (NBAIR-BT25) (GenBankMN327970) and talc formulation of entomopathogenic fungus Metarhizium anisopliae (NBAIR Ma-35) were developed in the bureau and evaluated to observe their efficacy to manage larval population of FAW in field after screening in the laboratory. A controlled release matrix developed by ICAR-NBAIR and Jawaharlal Nehru Centre for Advanced Scientific Research (JNCASR) Bengaluru, India for release of FAW pheromone was used in sleeve trap.
Liquid formulation of NBAIR-BT25
The culture was grown in T3 broth (Travers et al. 1987) and incubated for 7 days at 30 °C on an industrial shaker (Make Orbitek) with continuous shaking at 250 rpm. Samples were centrifuged at 5000 rpm for 15 min. Pellets (spores and parasporal crystals) were formulated directly with 2% glycerol, PVP-1%, trehalose 1%, proline 1%, yeast extract 1%, Tween 80-0.1% (sterile) and 10% of active ingredient. Molasses (1%) was added at the time of spraying as UV protectant.
Powder formulation of Metarhizium anisopliae (NBAIR Ma-35)
The fungus, M. anisopliae (NBAIR Ma-35) was grown in one litre conical flask containing 500 ml medium of Sabouraud’s dextrose yeast extract broth (SDYB) (dextrose 20 g, mycological peptone 10 g, yeast extract 5 g in 1L of distilled water) in an orbital shaker at 25 ± 2 °C temperature, 150 rpm for 8 days. The 8-day-old culture broth was mixed in sterilized talcum powder in the ratio of 1:2 (500 mL broth: 1 kg talc) in sterilized tray under laminar air flow and dried to 8% moisture. The talc formulation of M. anisopliae (NBAIR Ma-35) contains 2.0 × 108 cfu/g. The formulation was stored in milky white polypropylene pouches for further use in field evaluation trials.
Pheromone traps
The ICAR-NBAIR and JNCASR controlled release FAW pheromone lure with Z9-Tetradecenyl acetate as a major compound and Z7—dodecenyl acetate; Z11—hexadecenyl acetate blend as minor compound (altogether 3 mg per vial) was loaded in 10 mg of amorphous silica in polyethylene vial. These lures were housed in a sleeve trap that consisted of three parts, viz., canopy, funnel-shaped trap base and collection device. The canopy was 140 mm in diameter. The funnel top had an opening of 110 mm. The bottom opening of the funnel was 30 mm in diameter, and it was attached to a non-collapsible polyethylene bag 0.33 gauge of length 760 mm. This setup was installed in a wooden pole and placed in the field @ 10 traps per acre 20 days after sowing. The trap height was adjusted every week matching the crop canopy. The lures were changed once in 30 days. The number of insects trapped was collected at weekly intervals.
Neem oil
The commercial formulation of neem oil (EC formulation containing 0.03% azadirachtin) was used at a dose of 3 mL/L for managing FAW in field experiment and obtained from local market.
Farmer’s practice
In farmer’s practice field, emamectin benzoate 5% SG (trade name Volax, Indofil Industries Limited) (only insecticide used by farmers for managing FAW when FAW entered in India) was used to manage FAW.
Field Studies
Experimental design
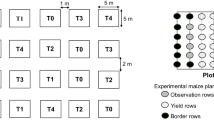
To assess the efficacy of IPM module in controlling S. frugiperda, two field experiments were conducted in FAW infested Maize field, at Manchenahalli, Gauribidanur, Karnataka (13° 31′ N, 77° 34′ 50.6′′ E, 918 m above sea level), India, during December 2018–March 2019 (rabi season) and June 2019–October 2019 (kharif season).The experiments were conducted in maize (cv. Pioneer) field of 4000 m2, with a plant spacing of (75 × 20 cm) where plants were sown following ridges and furrow method in red sandy loam soil. The experimental layout was a randomized complete block design with two treatments (T1 = Farmer’s practice; T2 = biocontrol-based IPM practices). There were 20 replicates (block) per treatment (4000 m2). The details of these treatments are given in Table 1. Both the treatments were separated by a buffer plot to reduce the probability of parasitoid moving to neighbouring plot.
Interventions in farmer’s field (T1)
During rabi and kharif season, maize plants received six and seven sprays of emamectin benzoate 5% SG, respectively at a dose of 0.4 g/L at weekly interval started at 20 DAS.
Interventions in biocontrol-based IPM practices (T2)
Pheromone traps and egg parasitoid to target egg mass
Pheromone traps were installed @ 10 traps/acre when the crop was 20 days old to monitor and mass trap adult moth population. The release of T. pretiosum was initiated when crop was twenty-five days old (next day after first catch of adult moths). A total of four releases were made at weekly intervals at the rate of 50,000 parasitized eggs per hectare. The parasitoids were released in field when at least 5% adult emergence (pharate) was observed. During each release, trichocards containing ca.16,000 parasitized eggs were cut into 16 bits of 4 × 1.5 cm size and were stapled to the lower side of upper part of maize leaf and were uniformly dispersed in the field.
Biopesticides and neem oil to manage FAW larvae
Maize plants received two sprays of neem oil (0.03%) at a dose of 3 mL/L with the help of knapsack sprayer at 30 and 54 days after sowing (DAS). The liquid formulation of B. thuringiensis (NBAIR BT25) was thoroughly mixed, using a sticker (gum acacia 1%) and sprayed once @ 20 mL/L on maize plants at 5.00 pm using knapsack sprayer at 38 DAS. One spray of M. anisopliae (NBAIR Ma-35) (1 × 108 cfu/g) was applied at the dose of 5 g/L at 45 DAS. Neem oil and NBAIR BT25 were used to target young larvae and were sprayed during early infestation.
Observations: To estimate the egg mass and larval population of FAW, five plants were randomly selected per replicate and the number of egg mass of FAW and FAW larvae/plant was counted after thorough inspection in both the treatments. For parasitism, observations were made for presence of dark black eggs (characteristic of parasitism) after 2nd and 4th release of parasitoid. During experiment, FAW eggs were not removed from the plants except after 4th release of egg parasitoid where black eggs were removed and per cent parasitism was computed. Percentage reduction in larval population and egg mass was calculated at 15, 30, 45 and 60 days after treatment (DAT). The number of larval population and egg mass was compared between farmer’s practice and biocontrol-based IPM. Maize yield and cost–benefit of biocontrol-based IPM and farmer’s practice were also calculated for both the seasons.
Statistical analysis
All analyses were undertaken using SPSS Statistics version 25.0 (IBM). The data obtained from the experiments were analysed for normality and homoscedasticity. Wherever required to meet normality, square root transformation and arcsine transformation were used. Per cent reduction in larval population and egg mass was compared at 15, 30, 45 and 60 days after first treatment using one-way ANOVA. Independent t test was carried out to compare the mean number of larvae/plant and mean number of egg mass/plant between biocontrol-based IPM and farmer’s practice field. When ANOVA was significant, comparison of all the relevant means was made using Tukey’s post hoc significance test at a significance level of 5%. The cost of both the treatments was estimated on the basis of cost of production of bioagents, biopesticides, labour cost and prevailing market price of neem oil and insecticide. Yield was also estimated for both the treatments. Net profit was estimated based on the income through the yield and the cost per acre of all the inputs from both the treatments.
Results
Effect on egg mass
Results exhibited 76.25% reduction in S. frugiperda egg mass at 60 days after treatment (DAT) (first release of parasitoid) which was on par with 30 and 45 DAT and significantly higher than 15 DAT (46.76%) (F3,76 = 3.963; P = 0.01) in biocontrol-based IPM field (Fig. 1a) during rabi season. A number of egg mass were significantly low in biocontrol-based IPM field compared to farmer’s practice at 15 (t = 2.9, df = 38, P = 0.006), 30 (t = 3.9, df = 38, P < 0.0001, 45 (t = 5.9, df = 38, P < 0.0001) and 60 (t = 7.2, df = 38, P < 0.0001) DAT (Fig. 2a). Egg reduction was observed after second release of egg parasitoid and continued till the end of crop.
a Percentage egg mass reduction over farmer’s practice at 15, 30, 45 and 60 days after treatment during rabi and kharif (2018–2019). Data represent mean ± SE. Bars with different small letters on the top of error bars indicate significant differences among different DAT during rabi, and bars with different capital letters indicate significant differences among different DAT during kharif (P < 0.05, Tukey’s post hoc test). b Percentage larval population reduction over farmer’s practice at 15, 30, 45 and 60 days after treatment during rabi and kharif (2018–2019). Data represent mean ± SE. Bars with different small letters on the top of error bars indicate significant differences among different DAT during rabi and bars with different capital letters indicate significant differences among different DAT during kharif (P < 0.05, Tukey’s post hoc test)
a Effect of biocontrol-based IPM practices and farmer’s practice on average number of egg mass per plant in both the treatments at 15, 30, 45 and 60 DAT during rabi (2018–2019). Data represent mean ± SE; statistical differences are based on independent t test (P < 0.05; n = 20). b Effect of biocontrol-based IPM practices and farmer’s practice on average number of egg mass per plant in both the treatments at 15, 30, 45 and 60 DAT during kharif (2019). Data represent mean ± SE; statistical differences are based on independent t test (P < 0.05; n = 20)
During kharif season, per cent egg reduction of FAW was significantly higher in biocontrol-based IPM field at 60 DAT (71.64%) (F3,76 = 4.723; P = 0.004) compared to 15 DAT (42.75%) (Fig. 1a). When both the treatments were compared in terms of egg mass, significantly low number of FAW eggs were observed in biocontrol-based IPM field at 30 (t = 2.1, df = 38, P = 0.03), 45 (t = 5.12, df = 38, P < 0.0001) and 60 DAT (t = 7.05, df = 38, P < 0.0001) except at 15 DAT (P = 0.21) (Fig. 2b).
Effect on larval population
During rabi season, in biocontrol-based IPM field, 80.28% reduction in larval population was observed at 60 days after treatment (DAT) which was significantly higher than 15 DAT (44.62%) (F3,76 = 3.871; P = 0.01) (Fig. 1b). However, it was on par with per cent larval reduction at 30 and 45 DAT. Results of “t test” also exhibited that in biocontrol-based IPM field average number of larvae/plant were significantly lower (0.62 ± 0.09, 0.27 ± 0.05, 0.25 ± 0.05, 0.2 ± 0.03) compared to farmer’s field (1.38 ± 0.19, 0.79 ± 0.14, 0.9 ± 0.11, 0.97 ± 0.96) at 15 (P = 0.002), 30 (P = 0.004), 45 (P < 0.0001) and 60 (P < 0.0001) DAT (Fig. 3a).
a Effect of biocontrol-based IPM practices and farmer’s practice on average number of larvae per plant in both the treatments at 15, 30, 45 and 60 DAT during rabi (2018–2019). Data represent mean ± SE; statistical differences are based on independent t test (P < 0.05; n = 20). b Effect of biocontrol-based IPM practices and farmer’s practice on average number of larvae per plant in both the treatments at 15, 30, 45 and 60 DAT during kharif (2019). Data represent mean ± SE; statistical differences are based on independent t test (P < 0.05; n = 20)
During kharif season, significant difference was observed in per cent reduction in larval population at 60 DAT in biocontrol-based IPM field and it was significantly higher (F3,79 = 5.646; P = 0.002) than 15 DAT (38.05%) (Fig. 1b). As observed in rabi season, also average number of larvae/plant were significantly lower in biocontrol-based IPM field compared to farmer’s field at 15 (t = 9.7, df = 38, P < 0.0001), 30 (t = 7.36, df = 38, P < 0.0001), 45 (t = 14.2, df = 38, P < 0.0001) and 60 DAT (t = 11.4, df = 27.2, P < 0.0001) (Fig. 3b).
Yield
During both the seasons, the cob yield in biocontrol-based IPM field (35 and 32.3 q/acre during rabi and kharif, respectively) was higher than farmer’s practice (25.3 and 22.7 q/acre during rabi and kharif, respectively). Though the cost for all the interventions made in biocontrol-based IPM for the management of FAW was high compared to farmers’ practice, it resulted in high net profit during rabi and kharif compared to farmer’s practice (Table 2).
Discussion
The combination of tactics, viz., installation of pheromone traps, four releases of T. pretiosum at weekly interval, two sprays of neem oil (30 and 54 DAS) and one spray of each BT25 (38 DAS) and Ma-35 (45 DAS) used in the biocontrol-based IPM field reduced egg mass as well as larval population compared to farmer’s practice during both the seasons. In Africa, IPM of FAW is mainly based on cultural practices, push–pull strategies and intercropping (Hruska 2019). He further stated that among all the parasitoids and predators, in African countries, tachinids and braconids are the important parasitoids responsible for 60 and 30% parasitism of the FAW larvae, respectively. Shylesha et al. (2018) also reported many parasitoids and predators attacking FAW in India. Though there are many reports of association of natural enemies with FAW from different regions, augmentation of bioagents is essential because native population of parasitoids and predators associated with this pest cannot control it. Therefore, in present study augmentation of T. pretiosum was carried out along with pheromone traps, biopesticides and neem oil.
Pheromone traps serve as an important tool to monitor adult population in field and help to decide time of egg parasitoid release and also spray schedule of biopesticides/chemicals. In the present study, first catch of moth was observed at 23 days after sowing (DAS), and then, release of egg parasitoid and other interventions were started. Subsequently, moth catch started declining and again reached to peak at 40DAS followed by decrease in moth catch. We also observed maximum number of egg laying during initial 30–45 DAS. Thus, egg parasitoid was released from 25 to 47 DAS and two sprays of neem oil (30 and 54 DAS) were taken up to target egg and early instars. Egg parasitoid might play an important role in reduction of initial population of fall armyworm and decrease the subsequent load of population along with other factors, viz., crop phenology and other abiotic factors. Therefore, it is advisable to release trichocards during initial stage of crop (after first catch of moths till 45–50 days).
In Latin America, T. pretiosum and Telenomus remus Nixon are the common species used in FAW management (Van Lenteren and Bueno 2003). In Mexico, T pretiosum is also used in augmentative biological control programme to manage S. frugiperda (Jaraleño-Teniente et al. 2020). Figueiredo et al. (2015) documented efficiency of T. pretiosum against FAW causing 79.2% egg mass and 19.4% gain of productivity. Besides that, T. pretiosum was found in 93.79% of the parasitized eggs of fall armyworm as an effective and frequent parasitoid (Beserra et al. 2002). We observed during rabi, after second release, seven egg masses of FAW were parasitized of the twelve egg masses. Similarly, after fourth release, total 7 egg masses were observed and 4 egg masses of them were containing parasitized eggs. In biocontrol-based IPM field, the average egg mass parasitized by egg parasitoid was 20.75%. During kharif, the average egg mass parasitized by egg parasitoid was 22.25%.
In India, recently we have also observed natural parasitization of FAW eggs by Trichogramma chilonis Ishii. This is a dominant species and is being used in many ecosystems to target lepidopteran pests in India. We also observed natural parasitization of FAW eggs by T. remus which is a very potent parasitoid of FAW eggs and is being used in augmentative biocontrol targeting fall armyworm and other species of Spodoptera Guenée (Lepidoptera: Noctuidae). In India, this species was imported from New Guinea to control Achaea janata (Linneus) (Sankaran 1974). Natural parasitization of FAW egg by this species shows its establishment in Indian ecosystems. This species has immense potential to target FAW eggs compared to T. pretiosum because it can parasitized the deepest layer of egg mass and provide maximum parasitization (Bueno et al. 2008).Thus, these two parasitoids could be evaluated along with biopesticides (Bt, entomopathogenic fungus, nuclear polyhedrosis virus) to manage FAW.
In the present study, reduction in larval population was observed due to use of EPF, Bt and neem oil, though neem oil and EPF might have helped in egg reduction to some extent. Our results are corroborated with Akutse et al. (2019) who observed ovicidal effect of some of the isolates of M. anisopliae causing egg mortality of FAW ranged from 79.5 to 87%. Isolates of M. anisopliae (ICIPE 41 and ICIPE 7) inflicted 96.5% and 93.7% mortality to the neonate larvae, respectively. However, they were less effective on second instar FAW larvae. On the contrary of that, 72.5% mortality to third instar FAW larvae was observed by M. anisopliae isolate CP-MA1 with high doses of 5.3 × 105 conidia mL−1 in Mexico (Romero-Arenas et al. 2014). In India, work has been initiated to screen potential isolates of EPF effective for FAW larvae. Most of the studies are confined to laboratory. However, Mallapur et al. (2018) observed 58.91–62.87% reduction of FAW infestation by using Nomuraea rileyi. Emphasis should be given to isolate fungus strain naturally causing infection to FAW larvae and assess its efficacy. García et al. (2011) isolated a strain of B. bassiana from FAW larvae which was able to cause 96% mortality to second instar larvae (@ 1 × 109 conidia mL−1. Further ovicidal effect of both B. bassiana and M. anisopliae was observed on other species of Spodoptera Guenée, viz., S. litura and Spodoptera exigua (Hubner) by different workers (Anand and Tiwary 2009; Al-Kherb 2014) and other lepidopteran insect pests such as Maruca vitrata (Fabricius) (Ekesi et al. 2002), and Potato tuber moth, Phthorimaea operculella Zeller (63% mortality) (Khorrami et al. 2018).
Many of the strains of B. thuringiensis were screened and provided excellent results in laboratory for FAW (Loto et al. 2019; dos Santos et al. 2009; Álvarez et al. 2009). Polanczyk et al. (2000) reported 100 and 80.4% mortality of FAW larvae using Bt aizawai HD 68 and Bt thuringiensis 4412, respectively. de Souza et al. (2009) identified B. thuringiensis (israelensis type) showing toxicity to S. frugiperda (LC50 of 69.07 µg cm−2). Capalbo et al. (2001) devised solid state fermentation of B. thuringiensis subsp. tolworthi for field use against S. frugiperda that showed 100 % mortality of neonate larvae. In present study, the cumulative effect of Bt, EPF and neem oil has provided more than 77% larval population reduction. Many workers have proven the efficacy of neem products to many of the lepidopteran pests. Further, Silva et al. (2015) observed the seed cake extract of Azadirachta indica, causing high larval mortality of fall armyworm. Sisay et al. (2019) tested extract of Azadirachta indica, against FAW and observed more than 95% larval mortality 72 h after application.
Our study indicates that the present strategy of IPM including installation of pheromone traps, four releases of T. pretiosum at weekly interval, two sprays of neem oil and one spray of each BT25 and Ma-35 provided the effective control of fall armyworm which was superior to chemical insecticides. This is only a limited trial and first module to test against FAW on emergency basis. Further, trials are being done at hot spot locations with more elements and different seasons in larger plots. However, availability of these biopesticides and biocontrol agent is a serious concern. Therefore, the next challenge is to make biopesticides available to the farmers after registration.
There are many other egg-larval, larval parasitoids which showed high parasitism. Further research should be carried out to multiply and evaluate these parasitoids along with T. remus, T. chilonis and other promising isolates of EPF, Bt and NPV to provide more elements in IPM for better and timely management of fall armyworm.
References
Akutse KS, Kimemia JW, Ekesi S, Khamis FM, Ombura OL, Subramanian S (2019) Ovicidal effects of entomopathogenic fungal isolates on the invasive fall armyworm Spodoptera frugiperda (Lepidoptera: Noctuidae). J Appl Entomol 143:626–663
Al-Kherb WA (2014) Virulence bio-assay efficiency of Beauveria bassianaand Metarhizium anisopliaefor the biological control of Spodoptera exigua(Hübner) (Lepidoptera: Noctuidae) eggs and the 1st instars larvae. Aust J Basic Appl Sci 8(3):313–323
Álvarez A, Pera LM, Loto F, Virla EG, Baigorí MD (2009) Insecticidal crystal proteins from native Bacillus thuringiensis: numerical analysis and biological activity against Spodoptera frugiperda. Biotechnol Lett 31:77–82
Anand R, Tiwary BN (2009) Pathogenicity of entomopathogenic fungi to eggs and larvae of Spodoptera litura, the common cutworm. Biocontrol Sci Technol 19(9):919–929
Beserra EB, Dias CTDS, Parra JRP (2002) Distribution and natural parasitism of Spodoptera frugiperda (Lepidoptera: Noctuidae) eggs at different phenological stages of corn. Fla Entomol 85:588–593
Brookes G, Barfoot P (2017) GM crops: global socio-economic and environmental impacts (1996–2015). PG Economics LTD, Dorcester. www.pgeconomics.co.uk/pdf/2017globalimpactstudy
Bueno RCODF, Carneiro TR, Pratissoli D, Bueno ADF, Fernandes OA (2008) Biology and thermal requirements of Telenomusremus reared on fall armyworm Spodoptera frugiperda eggs. Ciênc Rural 38:1–6
Capalbo DMF, Valicenti FH, Moraes IO, De Oliveira Iracema, Pelizer LH (2001) Solid-state fermentation of Bacillus thuringiensis tolworthi to control fall armyworm in maize. Electron J Biotechnol 4(2):9–10
de Souza JDA, Jain S, de Oliveira CMF, Lucena WA (2009) Toxicity of a Bacillus thuringiensis israelensis-like strain against Spodoptera frugiperda. Biocontrol 54:467–473. https://doi.org/10.1007/s10526-008-9191-8
dos Santos KB, Neves P, Meneguim AM, dos Santos RB et al (2009) Selection and characterization of the Bacillus thuringiensis strains toxic to Spodoptera eridania (Cramer), Spodoptera cosmioides (Walker) and Spodoptera frugiperda (Smith) (Lepidoptera: Noctuidae). Biol Control 50:157–163
Ekesi S, Adamu RS, Maniania NK (2002) Ovicidal activity of entomopathogenic hyphomycetes to the legume pod borer, Marucavitrataand the pod sucking bug Clavigralla Tomentosicollis. Crop Prot 21(7):589–595. https://doi.org/10.1016/S0261-2194(02)00015-7
Figueiredo MDLC, Cruz I, da Silva RB, Foster JE (2015) Biological control with Trichogramma pretiosum increases organic maize productivity by 19.4%. Agron Sustain Dev 35:1175–1183
García C, González MB, Bautista MN (2011) Patogenicidad de aislamientos de hongosentomopatógenos contra Spodoptera fru- giperda(Lepidoptera: Noctuidae) Epilachnavarivestis (Coleoptera: Coccinellidae). Rev Colomb Entomol 37(2):217–222
Gutiérrez-Moreno R, Mota-Sanchez D, Blanco CA, Whalon ME, Terán-Santofimio H, Rodriguez-Maciel JC, DiFonzo C, (2019) Field-evolved resistance of the fall armyworm (Lepidoptera: Noctuidae) to synthetic insecticides in puerto rico and mexico. J Econ Entomol 112(2):792–802. https://doi.org/10.1093/jee/toy372
Hruska AJ (2019) Fall armyworm (Spodoptera frugiperda) management by smallholders. CAB Rev 14(043):1–11. https://doi.org/10.1079/pavsnnr201914043
Jaraleño-Teniente J, Lomeli-Flores JR, Rodríguez-Leyva E, Bujanos-Muñiz R, Rodríguez-Rodríguez SE (2020) Egg parasitoids survey of Spodoptera frugiperda (Smith) (Lepidoptera: Noctuidae) in maize and sorghum in Central Mexico. Insects 11:157. https://doi.org/10.3390/insects11030157
Khorrami F, Mehrkhou F, Mahmoudian M, Ghosta Y (2018) Pathogenicity of three different entomopathogenic fungi, Metarhizium anisopliaeIRAN 2252, Nomuraea rileyiIRAN 1020C and PaecilomycestenuipesIRAN 1026C against the Potato tuber moth, Phthorimaea operculella Zeller (Lepidoptera: Gelechiidae). Potato Res 61(4):297–308. https://doi.org/10.1007/s11540-018-9378-z
Loto FDV, Carrizo AE, Romero CM, Baigorí MD, Pera LM (2019) Spodoptera frugiperda(Lepidoptera: Noctuidae) strains from northern Argentina: esterases, profiles, and susceptibility to Bacillus thuringiensis (Bacillales: Bacillaceae). Fla Entomol 102:347–352
Mallapur CP, Naik AK, Hagari S, Praveen T, Patil RK, Lingappa S (2018) Potentiality of Nomuraearileyi (Farlow) Samson against the fall armyworm, Spodoptera frugiperda (J E Smith) infesting maize. J EntomolZool Stud 6(6):1062–1067
Manupriya (2019) Fall armyworm, destroyer of maize farms, causes concern in India. Published in Mongaway series on 2 September 2019. https://india.mongabay.com/2019/09/fall-armyworm-destroyer-of-maize-farms-causes-concern-in-india/
Molina-Ochoa J, Carpenter JE, Heinrichs EA, Foster JE (2003) Parasitoids and parasites of Spodoptera frugiperda(Lepidoptera: Noctuidae) in the Americas and Caribbean basin: an inventory. Fla Entomol 86:254–289
Montezano DG, Specht A, Sosa-Gómez DR, Roque-Specht VF, Sousa-Silva JC, Paula-Moraes SV, Peterson JA, Hunt TE (2018) Host plants of Spodoptera frugiperda (Lepidoptera: Noctuidae) in the Americas. Afr Entomol 26:286–300
Mota-Sanchez D, Wise J (2017) Arthropod pesticide resistance database. Michigan State University. https://www.pesticideresistance.org/
Polanczyk RA, da Silva RFP, Fiuza LM (2000) Effectiveness of Bacillus thuringiensis strains against Spodoptera frugiperda (Lepidoptera: Noctuidae). Braz J Microbiol 31:165–167
Romero-Arenas O, Rivera A, Aragon A, Parraguirre C, Cabrera E, López F (2014) Mortality evaluation of armyworm (Spodoptera frugiperda JE Smith) by using Metarhizium anisopliae in vitro. J Pure Appl Microbiol 8:59–67
Sankaran T (1974) Natural enemies introduced in recent years for biological control of agricultural pests in India. Indian J Agric Sci 44:425–433
Shylesha AN, Jalali SK, Gupta A, Varshney R, Venkatesan T, Shetty P, Ojha R, Ganiger PC, Navik O, Subaharan K, Bakthavatsalam N, Ballal CR (2018) Studies on new invasive pest Spodoptera frugiperda (J. E. Smith) (Lepidoptera: Noctuidae) and its natural enemies. J Biol Control 32(3):1–7. https://doi.org/10.18311/jbc/2018/21707
Silva MS, Broglio SMF, Trindade RCP, Ferrreira ES, Gomes IB, Micheletti LB (2015) Toxicity and application of neem in fall armyworm. Commun Sci 6(3):359–364. https://doi.org/10.14295/cs.v6i3.808
Sisay B, Simiyu J, Malusi P, Likhayo P, Mendesil E, Elibariki N, Wakgiri M, Ayalew G, Tefera T (2018) First report of fall armyworm, Spodoptera frugiperda (Lepidoptera:Noctuidae), natural enemies from Africa. J Appl Entomol 142(8):800–804. https://doi.org/10.1111/jen.12534
Sisay B, Tefera T, Wakgari M, Ayalew G, Mendesil E (2019) The efficacy of selected synthetic insecticides and botanicals against fall armyworm, Spodoptera frugiperda, in maize. Insects. https://doi.org/10.3390/insects10020045
Travers RS, Martin PA, Reichelderfer CF (1987) Selective process for efficient isolation of soil Bacillus spp. Appl Environ Microbiol 53:1263–1266
Van Lenteren JC, Bueno VH (2003) Augmentative biological control of arthropods in Latin America. Biocontrol 48:123–139
Yu SJ, Nguyen SN, Abo-Elghar GE (2003) Biochemical characteristics of insecticide resistance in the fall armyworm, Spodoptera frugiperda (JE Smith). Pest Biochem Physiol 77:1–11
Acknowledgements
The authors would like to thank Indian Council of Agricultural Research and to Dr. Chandish R. Ballal, Director ICAR-NBAIR for providing research facilities. Authors are also thankful to the farmer friend, Mr. Srinivas for making his field available for the study and support. The financial grant received from CABI under a collaborative project is gratefully acknowledged.
Funding
This study was funded by CABI UK collaborative project “Emergency response to address Fall Armyworm (Spodoptera frugiperda) in India through deployment of proven IPM Technologies for its management”.
Author information
Authors and Affiliations
Contributions
NB and RV were involved in conceptualization; ANS and NB helped in funding acquisition; RR and BR contributed to biopesticides isolates; RV and YL were involved in egg parasitoid multiplication; NB and KS contributed to pheromone traps; RV, BP, AR, VA were involved in farmer's field experiment and data recording; RV helped in data analysis and preparation of manuscript; BR, NB, KS, RR, MC and VP contributed to review and editing manuscript; and all authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have not found any potential conflicts of interest, and all ethical aspects are considered.
Ethics approval/consent to participate
This manuscript does not contain any studies with human participants or animals performed by any of the authors.
Availability of data and material
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Varshney, R., Poornesha, B., Raghavendra, A. et al. Biocontrol-based management of fall armyworm, Spodoptera frugiperda (J E Smith) (Lepidoptera: Noctuidae) on Indian Maize. J Plant Dis Prot 128, 87–95 (2021). https://doi.org/10.1007/s41348-020-00357-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41348-020-00357-3