Abstract
Purpose of review
Dengue virus (DENV) infections are a global health emergency. Humans can experience multiple DENV infections, owing to the presence of four antigenically distinct DENV serotypes, DENV1–4. DENV infection results in a spectrum of disease, ranging from mild and self-limiting to severe and life-threatening illness. Currently, one dengue vaccine is approved, although with modest efficacy, so dengue-directed therapeutics are still needed to minimize the disease burden. The current standard-of-care treatment for dengue is only supportive but there is a pipeline of therapeutic treatments that are in development.
Recent findings
In the past decade, many promising anti-dengue drug targets have been identified. Antivirals directly influencing the virus replication cycle or immune modulatory drugs with host targets that affect inflammation have been explored. Recently, targeting mast cells (MCs) or MC products has also emerged as a promising treatment option for dengue-associated vascular pathologies. Several human clinical trials have been conducted or are ongoing to test the efficacy of various therapeutics with an end-goal of treating dengue. Special emphasis is given to safe and tolerable drugs, which could be re-purposed and introduced into clinical practice quickly.
Summary
This review highlights the current standard-of-care treatment for dengue and discusses various antiviral and host target drugs that are currently being pursued at various stages of preclinical and clinical development.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Dengue fever epidemiology
Dengue disease, known as dengue fever, is caused by infection with dengue virus (DENV), which is a member of the genus Flavivirus of the family Flaviviridae. Like other members of its family such as Zika virus and Japanese encephalitis virus (JEV), DENV is an enveloped single-stranded, positive-sense RNA virus [1]. The number of DENV cases remains difficult to quantify, with recent estimates putting the number at nearly 400 million annually, with a quarter of these being symptomatic [2•]. Dengue cases have been identified in over 100 countries; only a fraction of which were affected by DENV 50 years ago, highlighting the rapid emergence of the disease [3]. This has been attributed to several factors, including rapid urbanization, increased international travel, and obstacles to controlling the mosquito population [4, 5]. The main route of transmission to humans is through the bite of a DENV-infected mosquito, particularly the Aedes species, Aedes aegypti and Aedes albopictus [5]. This partially explains the locations where dengue disease has been identified, as these areas are where mosquitos can thrive.
Due to improvements in recognizing dengue disease and prompt treatment, the global mortality rate has decreased. One study examining dengue outbreaks from 1990 to 2015 noted that though mortality was 1.9% before 2010, it decreased to 0.7% thereafter [6], though the overall incidence of dengue has been increasing [7]. Of the one-quarter of infected individuals that are symptomatic, a smaller proportion will progress to severe dengue, known as dengue hemorrhagic fever (DHF) and dengue shock syndrome (DSS) [8]. Rates of DHF/severe dengue vary in different regions of the world and with different outbreaks but are around 5% of symptomatic cases [8]. Severe dengue, if appropriately treated, has low mortality rates [9]. However, due to a large number of infected patients per year, there are an estimated 10,000 dengue-related deaths annually [7]. It is important to note that the rates of DENV infection can fluctuate depending on seasonal and cyclical patterns. Seasonal patterns are thought to be due to the weather conditions permitting mosquito growth [10], while cyclical patterns can result from changes in the immunity of the population for DENV [11] as well as the emergence of different virus strains which are more pathogenic [12].
Clinical course of disease
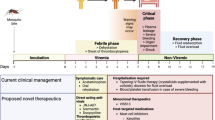
DENV is transmitted through the bite of an infected mosquito. Following an incubation period of 4–6 days, patients may start to develop common flu-like symptoms such as fever, myalgia, headache, and rash [8]. The level of viremia is highest during the febrile phase, with a resolution as the fever lyses [13] (Fig. 1). Thrombocytopenia, or a reduction in platelet counts, is a sign that appears early in the febrile phase and is highly suggestive of DENV infection, with more than 80% of patients experiencing thrombocytopenia [14]. Possibly due to the reduction in platelets, bleeding dyscrasias may occur, such as gum bleeding, petechiae, or more rarely and in the context of more moderate/severe disease, purpura, epistaxis, ecchymosis, gastrointestinal bleeding, or hypermenorrhea [15, 16]. Patients often feel lethargic and experience headache, classically retro-orbital, and arthralgia. Other laboratory findings include leukopenia and raised liver enzymes. The febrile phase lasts between 2 and 7 days, following which is the critical phase, where severe dengue may develop [8, 15].
The progression of clinical dengue disease. In humans, DENV infection begins with a mosquito bite, followed by an incubation period of 4–6 days before fever onset. Other common symptoms include rash and headache. The febrile phase lasts for 2–7 days during which peak viremia correlates with a high fever. Thrombocytopenia is a hallmark feature of clinical dengue disease and begins as fever develops. At the onset of fever and viremia, there is an opportunity for the diagnostic confirmation of DENV infection. As viremia is subsiding, some patients may develop a severe form of dengue disease, also known as DHF and DSS, resulting in the possibility of death. A secondary infection with a heterologous DENV strain results in viremia that is shorter, owing to the presence of a cross-reactive immune response generated against a previous DENV strain. The risk of severe dengue is enhanced during a secondary heterologous DENV infection
In 2009, the World Health Organization (WHO) released new guidelines classifying dengue disease into three broad categories: dengue with and without warnings signs and severe dengue. Some warning signs include enlargement of the liver, known as hepatomegaly, fluid accumulation, and mucosal bleeding. The significance of these warning signs is that they indicate the likelihood of progression to severe dengue, which can be lethal if untreated [17••]. “Severe dengue” replaced the older terminology of dengue hemorrhagic fever (DHF) and dengue shock syndrome (DSS), which were characterized by severe bleeding and circulatory compromise with fluid accumulation due to vascular leakage respectively. Due to this, depending on clinical and laboratory findings, patients may be admitted for monitoring and treatment, as death from dengue can be reduced with good supportive care [17••]. Death from dengue occurs mostly due to shock, attributed to leakage of the intravascular volume into the extravascular space following an increase in vascular permeability, and severe bleeding such from the gastrointestinal tract. The mortality rate can be as high as 10% to under 1%, depending on the location due to the access and quality of care available [6, 9, 18]. Additionally, other comorbidities such as age and risk factors such as hypertension and diabetes increase the likelihood of mortality [19]. This suggests that both host and viral factors play a role in determining disease severity and outcome [16]. Following the critical phase, the recovery phase marks the improvements in the patient’s symptoms. In cases of vascular leakage and thrombocytopenia, there is the resolution of extravasated fluid and recovery of platelet counts.
Diagnosis of dengue infection
Dengue disease may be suspected with signs and symptoms consistent with the initial presentation of DENV infection, including flu-like illness, fever, and myalgia [15], particularly during a DENV outbreak or in endemic areas. However, early diagnosis allows for clinical personnel to be aware of the potential for severe infection in the later course of the disease, permitting close monitoring. Several methods have been utilized to identify DENV infection. Two traditional virologic approaches, virus isolation through inoculation of either mosquitoes or cell lines and the plaque reduction neutralization test [20], are no longer routinely practiced due to the long and labor-intensive laboratory protocols they require. Currently, three methods are commonly utilized for diagnosis: nucleic acid-based techniques, antigen detection, and serological tests [21] (Fig. 1). While detection of viral products can be done early in the disease course, IgM and IgG against DENV usually appear only after 3–6 days of infection [8]. Commercial kits for each of these methods have been produced and are now commonly used for the diagnosis of dengue with high sensitivity and specificity, with results available within the day [22]. While nucleic acid-based techniques have the benefit of being highly sensitive and able to identify the serotype of DENV causing the infection [23], which is beneficial for surveillance, these molecular tests require the resources of sufficient laboratory equipment and skill. Currently, commercial kits for detection of DENV IgM and NS1 antigen are available and tests can be done rapidly in the outpatient setting [24•, 25]. Thus, the choice of the test to use will depend on multiple factors, such as duration of illness and cost and availability of a testing laboratory.
Treatment options
Current standard of care
There are very limited treatments available for dengue disease. The updated treatment guidelines proposed by the WHO [17••] focus on two aspects: symptomatic therapy and fluid administration. Symptomatic therapy is limited to acetaminophen as an analgesic and antipyretic. Use of aspirin and non-steroidal anti-inflammatory drugs is discouraged due to their antiplatelet activity, which could aggravate the bleeding risk due to thrombocytopenia. Fluid administration depends on the status of the patient. In mild cases and if the patient is able, oral administration is sufficient, though patients should be regularly reviewed [26]. In more severe cases, such as following the development of warning signs, hospitalization and administration of fluids by the intravenous route will be necessary. Both crystalloids and colloids have been evaluated for the treatment of DENV infection. Crystalloids are preferred due to the reduced risk of coagulopathy and allergy [27••]. However, colloids have been shown to be more efficacious in the setting of severe shock, particularly in terms of hematocrit and blood pressure [28]. Administration of albumin in crystalloid refractory shock has been shown in an observational study to be beneficial [29]. The added benefit of hospitalization is the ability to monitor the patient closely for progression to severe disease, particularly through serial laboratory investigations [30, 31•], since the onset of shock can occur rapidly within a few hours of warning signs. Various clinical scoring systems have also been proposed, based on a mix of vital signs, clinical findings, and laboratory investigations [30, 32], though none have been standardized and utilized globally.
In the case of severe dengue, manifested by severe bleeding or vascular leakage, more aggressive fluid administration will be required, potentially combined with blood transfusion. In this context, avoiding potential sources of bleeding is key, and patients should avoid unnecessary interventions and reduce the risk of trauma. It is important to ensure that despite shock during severe dengue secondary to intravascular depletion due to vascular leakage, fluid resuscitation still needs to be well-titrated. Patients can develop fluid overload manifested by pulmonary edema or ascites during the recovery phase if the excessive fluid was administered [33].
Development of therapeutics targeting the virus
The goal for therapeutics for DENV infection is to create an antiviral that could inhibit viral replication, reduce transmissibility, or reduce disease severity. Ideally, a drug for DENV should have a good safety profile and can be taken orally, as the majority of patients have mild symptoms. In the case of reducing disease severity, an issue lies with the short duration of illness, meaning that patients will have to be identified early in order to be able to receive this therapy. The same is true for an antiviral that targets viral replication and transmissibility, as symptomatic patients are already rapidly clearing the virus [13]. Moreover, warning signs appear later in the disease course when viremia has already subsided making it difficult to predict early which patient will suffer from severe dengue disease. These requirements have made finding treatment for DENV difficult. A number of drugs have been tested in humans, none with any success thus far, though there are still several drugs under investigation. These drugs in development include those targeting viral factors or and those targeting host factors. As viruses are obligate intracellular parasites and they rely on the host cellular machinery to replicate, making drugs that target host proteins involved directly in virus replication a promising strategy for therapy. While host protein-directed antiviral strategies have the limitation that the host proteins or pathways influenced by the drugs may be adversely impacted, resulting in the potential of side effects or even toxicity, there is also an advantage because of the limited ability of the virus to acquire resistance mutations. Antivirals with both DENV and human host targets as mechanisms of action are discussed below (Table 1).
Antiviral drugs
The genome of DENV measures around 11 kilobases with a single open reading frame. The viral genome encodes 10 proteins: 3 structural proteins (capsid protein C, membrane protein M, and envelope protein E) and 7 nonstructural proteins (NS1, NS2a, NS2b, NS3, NS4a, NS4b, and NS5) [1]. The viral proteins help in the assembly of a viral replication complex and, therefore, are prime targets of antiviral drug discovery. One potential strategy to treat dengue is to reduce viral replication. Due to levels of viremia correlating with severity [13], it was believed that reducing viremia would allow for treatment of disease. A number of viral proteins have been identified as therapeutic targets. As an RNA virus, DENV relies on its RNA-dependent RNA polymerase for replication [1]. The nucleoside analogue balapiravir was first designed to inhibit the RNA polymerase of hepatitis C virus [34]. Due to the similarity between the RNA polymerases of both hepatitis C and DENV [35], it was believed that balapiravir would be an effective antiviral. Though in vitro data supported its antiviral ability, a clinical trial failed to show any benefit in viremia and duration of illness [36]. A follow-up study suggested that this was due to poor conversion of balapiravir to its active form, suggesting under-dosing as the reason for treatment failure [37]. Similarly, NITD-008, a nucleoside inhibitor targeting RNA-dependent RNA polymerase, showed a strong inhibitory effect against all 4 DENV serotypes both in vitro and in vivo [38]. However, its development was halted due to the acute renal toxicity observed in preclinical studies. Other virus-specific drug targets that are currently being explored include NS2b, NS3, NS4b, and capsid proteins [39,40,41], affecting various stages of the virus replication cycle. Screening of large compound libraries has also enabled the identification of many potential inhibitors of viral proteins [42]. These are still in a nascent stage, with hopes that several could be beneficial in treating dengue.
Another target that has recently gained attention as a DENV antiviral is an enzyme α-glucosidase, which is necessary for N-linked glycosylation of viral prM and E proteins [43]. Absence of N-linked glycosylation reduced the infectivity and release of DENV from host cells [44]. Celgosivir, an alpha-glucosidase antagonist demonstrated impaired folding and trapping of viral NS1 in the endoplasmic reticulum and provided protection against lethal DENV challenge in mice [45]. Though DENV-infected mice treated with celgosivir had reduced viremia and a mortality benefit, a phase 1b randomized placebo-controlled trial failed to show a clear benefit in reducing viremia or disease course [46]. Subsequent studies on the drug pharmacokinetics and dosage suggested that increasing the drug dosage may improve the efficacy of this drug [47, 48].
Statins are a class of drugs that inhibit cholesterol synthesis by interfering with the enzyme HMG-CoA reductase. Apart from their lipid-lowering ability, statins possess anti-inflammatory properties [49]. Due to the importance of membrane lipids in the Flavivirus life cycle, it was hypothesized that statins could interfere with viral replication [50]. Supporting this, studies in a mouse model of dengue infection demonstrated that statins have both antiviral and anti-inflammatory properties [51]. However, a clinical trial assessing lovastatin in DENV infection failed to demonstrate any benefit in viremia or disease course, though the authors acknowledged that the study may not be adequately powered [52].
Chloroquine is most well known for its role as an anti-malarial. In the context of DENV, it has been demonstrated to inhibit virus replication by preventing the proteolytic processing of viral proteins [53]. In addition, similar to statins, chloroquine has an added anti-inflammatory function that could theoretically also be of clinical benefit [54]. These two properties of chloroquine were demonstrated in vivo in a monkey model, demonstrating reduced inflammation and viremia [55]. However, one randomized placebo-controlled human trial failed to demonstrate any benefit of chloroquine treatment in viremia [56]. With regard to disease course, one study suggested an improvement in patient symptoms [57] while the other showed the opposite, with more adverse effects in the chloroquine-treated group [56]. Overall, neither study showed any significant improvement in disease progression to severe dengue.
Ivermectin, an anti-parasitic drug, commonly used to treat nematode infections such as river blindness, filariasis, scabies, and others, was shown to have anti-DENV antiviral activity [58]. The antiviral activity of ivermectin relies on its ability to inhibit host nuclear import receptors, importin α and β, which play a crucial part in DENV replication [59, 60]. A clinical trial is currently being conducted to assess the efficacy of ivermectin against dengue disease (NCT02045069).
Monoclonal therapeutics
Due to recent gains in our knowledge of virion and virion-antibody complexed structures, monoclonal antibodies are emerging as promising therapeutics against dengue. These antibodies either target viral surface glycoproteins, thus preventing either viral attachment [61] or fusion with the host membrane [62], or they target viral NS1, which has been implicated in disease pathogenesis [63]. NS1 targeting with monoclonal antibodies was shown to improve survival in an immune-compromised mouse model of DENV disease [64], but this has not yet been investigated in humans. There are examples of monoclonal human antibodies targeting both linear and quaternary DENV E protein structures that result in viral neutralization. Generally, these antibodies can neutralize specific DENV serotypes. For example, antibodies 14c10, 1F4, 4CAU, and 4C2I can neutralize DENV1 by binding to the hinge region of the E protein domains I and II [65•]. Similarly neutralizing only one serotype, antibodies 5J7 and 3J6U can neutralize DENV3 [65•]. Antibodies targeting the E protein dimer and domain III also can be neutralizing and can be either type-specific (e.g., 4UIF, 5A1Z, and 4UIH against DENV2) or group reactive (e.g., 4UTB and ab513 against DENV1–4) [65•]. The recent advances in our knowledge of monoclonal antibodies that could provide protection or potentially enhance the severity of dengue disease have been extensively discussed elsewhere [65•, 66, 67]. Several of these antibodies have shown in vivo efficacy in animal models; however, none have yet progressed to clinical stages of development.
Development of therapeutics targeting the host
As severe dengue disease is thought to be largely immune-mediated, additional therapeutic strategies have been proposed and tested that are designed to mitigate the pathological host response to DENV.
Corticosteroids are widely used in autoimmune diseases due to their ability to suppress host inflammation [68]. Increased levels of proinflammatory cytokines are correlated with disease severity [69], suggesting a role for inflammation in tissue damage and vascular leakage. Despite a large number of clinical trials being carried out, data regarding the efficacy of steroids has been unclear. Steroids are an efficacious treatment for immune thrombocytopenic purpura (ITP), wherein there is autoimmune destruction of platelets [70]. Thrombocytopenia in dengue was similarly thought to be due to an autoimmune mechanism in which autoantibodies against platelets result in their destruction [71, 72]. Two placebo-controlled studies looking at whether steroids could be beneficial in the treatment of thrombocytopenia failed to show any efficacy [73, 74]. Though there is now consensus that steroids are not useful for treating thrombocytopenia during dengue infection, there are some discordant data with regard to the utility of steroids for treating severe dengue, particularly in patients with shock. A retrospective review of patients receiving steroid treatment suggested some benefit [75]. Conversely, more recent studies showed no mortality benefit during severe dengue with steroid administration [76, 77]. Due to the lack of clear support for steroid efficacy and the theoretical risks of immunosuppression, they are currently not recommended for the treatment of dengue [17••]. However, these clinical studies have highlighted that there are minimal side effects with steroid administration during dengue [78].
As mentioned previously, thrombocytopenia was thought to be due to the autoimmune destruction of platelets [71, 79]. Hence, it was proposed that the immune modulating capability of intravenous immunoglobulin (IVIG) might be beneficial in the treatment of dengue disease. These antibodies are pooled from several healthy donors. A randomized placebo-controlled trial testing the efficacy of IVIG failed to demonstrate any benefit in platelet counts [80]. However, in some individual cases, IVIG has been beneficial in restoring platelet counts [81], which might reflect the potential of thrombocytopenia to be caused through varied mechanisms. A common trend in which IVIG has been beneficial has been in atypical presentations, particularly prolonged thrombocytopenia, beyond the typical duration of 7–10 days. Better patient stratification and understanding of the disease process might identify patients who would benefit from this treatment. While IVIG has shown limited benefit, another antibody therapy, anti-D immune globulin, has shown greater benefit in treating thrombocytopenia. This antibody targets the Rho(D) antigen present on red blood cells and was first used to prevent hemolytic disease of the newborn [82]. Randomized placebo-controlled trials have shown faster recovery in platelet counts in anti-D immune globulin treated patients [83, 84]. However, an issue with this treatment would be the risk of side effects such as hemolysis and its efficacy in only Rh+ patients, which would limit its widespread use. Furthermore, both IVIG and anti-D immune globulin represent expensive treatments, which would restrict their use in communities with limited resources.
In the event of hemorrhage, blood transfusion may be necessary. Since low platelet counts could contribute to hemorrhage, it was initially thought that platelet transfusions could be useful. However, multiple studies have highlighted that there is no benefit in platelet transfusions [85], with some showing an increased risk for adverse events such as allergy [85, 86]. One study has identified that platelet transfusion can also delay the normalization of platelet counts [87]. Thus, strong clinical necessity needs to be present before platelet transfusions are carried out in dengue patients [88].
Targeting mast cells to reduce vascular leakage and thrombocytopenia
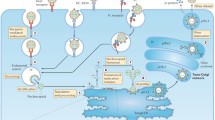
An increasing number of studies have implicated mast cells (MCs) and products released by them in worsening dengue disease. MCs are immune cells that reside in nearly all tissues and frequently surround blood vessels [89]. At the site of infection in the skin, MCs are poised to be able to react to DENV infection following a mosquito bite and respond by degranulation, which involves the release of pre-formed mediators including proteases and anti-coagulants, as well as the de novo synthesis of cytokines and lipid mediators [90, 91]. MCs also interact with T cells to promote viral clearance at the cutaneous infection site [92]. However, in contrast to this early beneficial role of MCs in virus clearance, when the infection becomes systemic, excessive release of MC mediators was shown to promote vascular leakage in mice [93•, 94]. In human dengue patients, an increase in serum levels of the MC-specific protease, chymase, was found to occur with dengue but to be highest in severe DENV cases (DHF/DSS) compared to the milder dengue fever [31•, 93•]. Since MC proteases have been shown to promote vascular leakage through their actions on the endothelium, it suggests that the release of MC products may directly promote the vascular signs of severe dengue [93•]. Consistent with this data, vascular leakage in a mouse model of DENV infection was absent in MC-deficient animals and when MC activation was blocked by drugs in the class of “mast cell stabilizers,” including ketotifen and cromolyn, which have been approved for use to treat allergic conditions including allergy and asthma [93•]. Currently, a clinical trial is underway to assess the efficacy of ketotifen in reducing vascular leakage during dengue disease (NCT02673840).
Many vasoactive lipid mediators are released by activated MCs, including leukotrienes and platelet activating factor (PAF) and, in addition to MCs, these can also be released by activated endothelium and other immune cell types. In mice, a leukotriene inhibitor, montelukast was effective in reducing DENV-induced vascular leakage [93•], as was a PAF-inhibitor. PAF had previously been implicated in DENV-induced vascular leakage in animal models [95]. Promisingly, a recent preliminary clinical trial tested the efficacy of the drug rupatadine, an orally available drug that has dual PAF and histamine-1-receptor blocking activities [96•]. Although the primary study endpoint of reduced development of pleural effusions or ascites was not met, post hoc analyses revealed significantly higher platelet counts, lower levels of liver enzymes that could indicate lesser tissue damage, and smaller volume effusions on certain days of the study protocol, suggesting that further studies are needed to fully assess the utility of the drug against dengue vasculopathy [96•].
Aside from PAF, other inflammatory mediators that regulate the MC-platelet axis and coagulation are of interest as possible therapeutic targets. One such MC product, the monoamine serotonin, was recently shown to be required in vivo for DENV-induced thrombocytopenia [97•]. The reduction in platelets was reversed when mice were treated with 5HT2 receptor inhibitory drugs ketanserin or sarpogrelate, which target the receptor for serotonin on platelets. Inhibition of serotonin reuptake by platelets using fluoxetine also prevented thrombocytopenia [97•]. Although further testing in humans is needed, these studies suggest that MCs or their mediators are important drug targets in treating severe dengue pathologies, including vascular permeability, plasma leakage, and thrombocytopenia.
Other potential therapies
Papaya leaf extracts (PLE) have been suggested to have a diverse range of beneficial activities during dengue disease. Although shown to have no direct antiviral activity [98], it possesses anti-inflammatory and platelet augmenting activities in vitro and in vivo [99, 100]. Treatment with PLE in humans showed increased platelet counts during dengue disease [101, 102]. However, the exact mechanism through which platelets are restored is unknown and further studies are needed to identify compounds present in the PLE that may have therapeutic potential against dengue disease. Recent studies also highlight the potential of vitamin E supplementation in dengue disease. Vitamin E supplementation on top of standard therapy has been shown to be beneficial in clinical trials, reducing derangements in liver function [103] and promoting recovery in platelet counts [104] during dengue infection.
Conclusions
The current mainstay of treatment for DENV infection remains supportive care. It is difficult to predict dengue severity since warning signs appear late in the disease. Therefore, hospitalization can improve the disease outcome due to the ability to monitor disease progression and using appropriate interventions. Currently, appropriate fluid management is the best way to reduce mortality. Ideally, a drug that can reduce the spread of infection and/or limit the risk of severe disease could dramatically improve health outcomes. However, a barrier to developing targeted therapeutics remains our limited understanding of dengue disease pathogenesis, which is likely to be multi-factorial and involve both virus-intrinsic factors and the host response. Further understanding of the disease process will allow for the identification of better targets for therapy. Though there have been recent advances in vaccines and vector control, effective targeted dengue therapeutics are still urgently needed and would reduce the health and economic burdens of disease.
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Lindenbach BD, Rice CM. Flaviviridae: the viruses and their replication. In: Knipe DM, Howley PM, editors. Fields Virology. Philadelphia: Lippincott Williams & Wilkins; 2001. p. 991–1041.
• Bhatt S, Gething PW, Brady OJ, Messina JP, Farlow AW, Moyes CL, et al. The global distribution and burden of dengue. Nature. 2013;496(7446):504–7. https://doi.org/10.1038/nature12060 This paper estimates that the global disease burden of dengue virus is much higher than previously thought.
Brady OJ, Gething PW, Bhatt S, Messina JP, Brownstein JS, Hoen AG, et al. Refining the global spatial limits of dengue virus transmission by evidence-based consensus. PLoS Negl Trop Dis. 2012;6(8):e1760. https://doi.org/10.1371/journal.pntd.0001760.
Gubler DJ. Dengue, Urbanization and globalization: the unholy trinity of the 21(st) century. Trop Med Health. 2011;39(4 Suppl):3–11. https://doi.org/10.2149/tmh.2011-S05.
Wilder-Smith A, Ooi EE, Horstick O, Wills B. Dengue. Lancet. 2019;393(10169):350–63. https://doi.org/10.1016/S0140-6736(18)32560-1.
Guo C, Zhou Z, Wen Z, Liu Y, Zeng C, Xiao D, et al. Global epidemiology of dengue outbreaks in 1990-2015: a systematic review and meta-analysis. Front Cell Infect Microbiol. 2017;7:317. https://doi.org/10.3389/fcimb.2017.00317.
Stanaway JD, Shepard DS, Undurraga EA, Halasa YA, Coffeng LE, Brady OJ, et al. The global burden of dengue: an analysis from the global burden of disease study 2013. Lancet Infect Dis. 2016;16(6):712–23. https://doi.org/10.1016/S1473-3099(16)00026-8.
St John AL, Rathore APS. Adaptive immune responses to primary and secondary dengue virus infections. Nat Rev Immunol. 2019;19:218–30. https://doi.org/10.1038/s41577-019-0123-x.
Md-Sani SS, Md-Noor J, Han WH, Gan SP, Rani NS, Tan HL, et al. Prediction of mortality in severe dengue cases. BMC Infect Dis. 2018;18(1):232. https://doi.org/10.1186/s12879-018-3141-6.
Liu-Helmersson J, Stenlund H, Wilder-Smith A, Rocklov J. Vectorial capacity of Aedes aegypti: effects of temperature and implications for global dengue epidemic potential. PLoS One. 2014;9(3):e89783. https://doi.org/10.1371/journal.pone.0089783.
Ooi EE, Hart TJ, Tan HC, Chan SH. Dengue seroepidemiology in Singapore. Lancet. 2001;357(9257):685–6. https://doi.org/10.1016/S0140-6736(00)04137-4.
Bennett SN, Holmes EC, Chirivella M, Rodriguez DM, Beltran M, Vorndam V, et al. Molecular evolution of dengue 2 virus in Puerto Rico: positive selection in the viral envelope accompanies clade reintroduction. J Gen Virol. 2006;87(Pt 4:885–93. https://doi.org/10.1099/vir.0.81309-0.
Vaughn DW, Green S, Kalayanarooj S, Innis BL, Nimmannitya S, Suntayakorn S, et al. Dengue in the early febrile phase: viremia and antibody responses. J Infect Dis. 1997;176(2):322–30.
Sahana KS, Sujatha R. Clinical profile of dengue among children according to revised WHO classification: analysis of a 2012 outbreak from Southern India. Indian J Pediatr. 2015;82(2):109–13. https://doi.org/10.1007/s12098-014-1523-3.
Gubler DJ. Dengue and dengue hemorrhagic fever. Clin Microbiol Rev. 1998;11(3):480–96.
St John AL, Abraham SN, Gubler DJ. Barriers to preclinical investigations of anti-dengue immunity and dengue pathogenesis. Nat Rev Microbiol. 2013;11(6):420–6. https://doi.org/10.1038/nrmicro3030.
•• Dengue: Guidelines for Diagnosis, Treatment, Prevention and control: new edition. WHO guidelines approved by the guidelines review committee. Geneva .2009. Updated clinical guidelines for the characterization of dengue disease, to dengue with and without warning signs and severe dengue.
Moraes GH, de Fatima Duarte E, Duarte EC. Determinants of mortality from severe dengue in Brazil: a population-based case-control study. Am J Trop Med Hyg. 2013;88(4):670–6. https://doi.org/10.4269/ajtmh.11-0774.
Karunakaran A, Ilyas WM, Sheen SF, Jose NK, Nujum ZT. Risk factors of mortality among dengue patients admitted to a tertiary care setting in Kerala, India. J Infect Public Health. 2014;7(2):114–20. https://doi.org/10.1016/j.jiph.2013.09.006.
Guzman MG, Kouri G. Advances in dengue diagnosis. Clin Diagn Lab Immunol. 1996;3(6):621–7.
Guzman MG, Gubler DJ, Izquierdo A, Martinez E, Halstead SB. Dengue infection. Nat Rev Dis Primers. 2016;2:16055. https://doi.org/10.1038/nrdp.2016.55.
Hunsperger EA, Yoksan S, Buchy P, Nguyen VC, Sekaran SD, Enria DA, et al. Evaluation of commercially available diagnostic tests for the detection of dengue virus NS1 antigen and anti-dengue virus IgM antibody. PLoS Negl Trop Dis. 2014;8(10):e3171. https://doi.org/10.1371/journal.pntd.0003171.
Johnson BW, Russell BJ, Lanciotti RS. Serotype-specific detection of dengue viruses in a fourplex real-time reverse transcriptase PCR assay. J Clin Microbiol. 2005;43(10):4977–83. https://doi.org/10.1128/JCM.43.10.4977-4983.2005.
• Wang SM, Sekaran SD. Early diagnosis of dengue infection using a commercial dengue duo rapid test kit for the detection of NS1, IGM, and IGG. Am J Trop Med Hyg. 2010;83(3):690–5. https://doi.org/10.4269/ajtmh.2010.10-0117 This study describes the current laboratory standards for dengue diagnosis.
Vickers IE, Harvey KM, Brown MG, Nelson K, DuCasse MB, Lindo JF. The performance of the SD BIOLINE Dengue DUO(R) rapid immunochromatographic test kit for the detection of NS1 antigen, IgM and IgG antibodies during a dengue type 1 epidemic in Jamaica. J Biomed Sci. 2015;22:55. https://doi.org/10.1186/s12929-015-0164-9.
Lee IK, Lee WH, Yang KD, Liu JW. Comparison of the effects of oral hydration and intravenous fluid replacement in adult patients with non-shock dengue hemorrhagic fever in Taiwan. Trans R Soc Trop Med Hyg. 2010;104(8):541–5. https://doi.org/10.1016/j.trstmh.2010.05.003.
•• Wills BA, Nguyen MD, Ha TL, Dong TH, Tran TN, Le TT, et al. Comparison of three fluid solutions for resuscitation in dengue shock syndrome. N Engl J Med. 2005;353(9):877–89. https://doi.org/10.1056/NEJMoa044057.This study evaluates various fluid solutions as standard of care treatment for dengue shock syndrome
Dung NM, Day NP, Tam DT, Loan HT, Chau HT, Minh LN, et al. Fluid replacement in dengue shock syndrome: a randomized, double-blind comparison of four intravenous-fluid regimens. Clin Infect Dis. 1999;29(4):787–94. https://doi.org/10.1086/520435.
Ranjit S, Ramanathan G, Ramakrishnan B, Kissoon N. Targeted interventions in critically ill children with severe dengue. Indian J Crit Care Med. 2018;22(3):154–61. https://doi.org/10.4103/ijccm.IJCCM_413_17.
Lam PK, Ngoc TV, Thu Thuy TT, Hong Van NT, Nhu Thuy TT, Hoai Tam DT, et al. The value of daily platelet counts for predicting dengue shock syndrome: results from a prospective observational study of 2301 Vietnamese children with dengue. PLoS Negl Trop Dis. 2017;11(4):e0005498. https://doi.org/10.1371/journal.pntd.0005498.
• Tissera H, Rathore APS, Leong WY, Pike BL, Warkentien TE, Farouk FS, et al. Chymase level is a predictive biomarker of dengue hemorrhagic fever in pediatric and adult patients. J Infect Dis. 2017;216(9):1112–21. https://doi.org/10.1093/infdis/jix447 This study describes a mast cell product, chymase, as a prognostic biomarker for severe dengue disease.
Thanachartwet V, Wattanathum A, Oer-areemitr N, Jittmittraphap A, Sahassananda D, Monpassorn C, et al. Diagnostic accuracy of peripheral venous lactate and the 2009 WHO warning signs for identifying severe dengue in Thai adults: a prospective observational study. BMC Infect Dis. 2016;16:46. https://doi.org/10.1186/s12879-016-1386-5.
Premaratna R, Liyanaarachchi E, Weerasinghe M, de Silva HJ. Should colloid boluses be prioritized over crystalloid boluses for the management of dengue shock syndrome in the presence of ascites and pleural effusions? BMC Infect Dis. 2011;11:52. https://doi.org/10.1186/1471-2334-11-52.
Klumpp K, Leveque V, Le Pogam S, Ma H, Jiang WR, Kang H, et al. The novel nucleoside analog R1479 (4′-azidocytidine) is a potent inhibitor of NS5B-dependent RNA synthesis and hepatitis C virus replication in cell culture. J Biol Chem. 2006;281(7):3793–9. https://doi.org/10.1074/jbc.M510195200.
Yap TL, Xu T, Chen YL, Malet H, Egloff MP, Canard B, et al. Crystal structure of the dengue virus RNA-dependent RNA polymerase catalytic domain at 1.85-angstrom resolution. J Virol. 2007;81(9):4753–65. https://doi.org/10.1128/JVI.02283-06.
Nguyen NM, Tran CN, Phung LK, Duong KT, Huynh Hle A, Farrar J, et al. A randomized, double-blind placebo controlled trial of balapiravir, a polymerase inhibitor, in adult dengue patients. J Infect Dis. 2013;207(9):1442–50. https://doi.org/10.1093/infdis/jis470.
Chen YL, Abdul Ghafar N, Karuna R, Fu Y, Lim SP, Schul W, et al. Activation of peripheral blood mononuclear cells by dengue virus infection depotentiates balapiravir. J Virol. 2014;88(3):1740–7. https://doi.org/10.1128/JVI.02841-13.
Yin Z, Chen YL, Schul W, Wang QY, Gu F, Duraiswamy J, et al. An adenosine nucleoside inhibitor of dengue virus. Proc Natl Acad Sci U S A. 2009;106(48):20435–9. https://doi.org/10.1073/pnas.0907010106.
Luo D, Vasudevan SG, Lescar J. The flavivirus NS2B-NS3 protease-helicase as a target for antiviral drug development. Antivir Res. 2015;118:148–58. https://doi.org/10.1016/j.antiviral.2015.03.014.
Byrd CM, Dai D, Grosenbach DW, Berhanu A, Jones KF, Cardwell KB, et al. A novel inhibitor of dengue virus replication that targets the capsid protein. Antimicrob Agents Chemother. 2013;57(1):15–25. https://doi.org/10.1128/AAC.01429-12.
Byrd CM, Grosenbach DW, Berhanu A, Dai D, Jones KF, Cardwell KB, et al. Novel benzoxazole inhibitor of dengue virus replication that targets the NS3 helicase. Antimicrob Agents Chemother. 2013;57(4):1902–12. https://doi.org/10.1128/AAC.02251-12.
Hernandez-Morales I, Geluykens P, Clynhens M, Strijbos R, Goethals O, Megens S, et al. Characterization of a dengue NS4B inhibitor originating from an HCV small molecule library. Antivir Res. 2017;147:149–58. https://doi.org/10.1016/j.antiviral.2017.10.011.
Courageot MP, Frenkiel MP, Dos Santos CD, Deubel V, Despres P. Alpha-glucosidase inhibitors reduce dengue virus production by affecting the initial steps of virion morphogenesis in the endoplasmic reticulum. J Virol. 2000;74(1):564–72.
Mondotte JA, Lozach PY, Amara A, Gamarnik AV. Essential role of dengue virus envelope protein N glycosylation at asparagine-67 during viral propagation. J Virol. 2007;81(13):7136–48. https://doi.org/10.1128/JVI.00116-07.
Rathore AP, Paradkar PN, Watanabe S, Tan KH, Sung C, Connolly JE, et al. Celgosivir treatment misfolds dengue virus NS1 protein, induces cellular pro-survival genes and protects against lethal challenge mouse model. Antivir Res. 2011;92(3):453–60. https://doi.org/10.1016/j.antiviral.2011.10.002.
Low JG, Sung C, Wijaya L, Wei Y, Rathore APS, Watanabe S, et al. Efficacy and safety of celgosivir in patients with dengue fever (CELADEN): a phase 1b, randomised, double-blind, placebo-controlled, proof-of-concept trial. Lancet Infect Dis. 2014;14(8):706–15. https://doi.org/10.1016/S1473-3099(14)70730-3.
Sung C, Wei Y, Watanabe S, Lee HS, Khoo YM, Fan L, et al. Extended evaluation of virological, immunological and pharmacokinetic endpoints of CELADEN: a randomized, placebo-controlled trial of celgosivir in dengue fever patients. PLoS Negl Trop Dis. 2016;10(8):e0004851. https://doi.org/10.1371/journal.pntd.0004851.
Watanabe S, Chan KW, Dow G, Ooi EE, Low JG, Vasudevan SG. Optimizing celgosivir therapy in mouse models of dengue virus infection of serotypes 1 and 2: the search for a window for potential therapeutic efficacy. Antivir Res. 2016;127:10–9. https://doi.org/10.1016/j.antiviral.2015.12.008.
Jain MK, Ridker PM. Anti-inflammatory effects of statins: clinical evidence and basic mechanisms. Nat Rev Drug Discov. 2005;4(12):977–87. https://doi.org/10.1038/nrd1901.
Neufeldt CJ, Cortese M, Acosta EG, Bartenschlager R. Rewiring cellular networks by members of the Flaviviridae family. Nat Rev Microbiol. 2018;16(3):125–42. https://doi.org/10.1038/nrmicro.2017.170.
Martinez-Gutierrez M, Correa-Londono LA, Castellanos JE, Gallego-Gomez JC, Osorio JE. Lovastatin delays infection and increases survival rates in AG129 mice infected with dengue virus serotype 2. PLoS One. 2014;9(2):e87412. https://doi.org/10.1371/journal.pone.0087412.
Whitehorn J, Nguyen CVV, Khanh LP, Kien DTH, Quyen NTH, Tran NTT, et al. Lovastatin for the treatment of adult patients with dengue: a randomized, double-blind, placebo-controlled trial. Clin Infect Dis. 2016;62(4):468–76. https://doi.org/10.1093/cid/civ949.
Randolph VB, Winkler G, Stollar V. Acidotropic amines inhibit proteolytic processing of flavivirus prM protein. Virology. 1990;174(2):450–8.
van den Borne BE, Dijkmans BA, de Rooij HH, le Cessie S, Verweij CL. Chloroquine and hydroxychloroquine equally affect tumor necrosis factor-alpha, interleukin 6, and interferon-gamma production by peripheral blood mononuclear cells. J Rheumatol. 1997;24(1):55–60.
Farias KJ, Machado PR, Muniz JA, Imbeloni AA, da Fonseca BA. Antiviral activity of chloroquine against dengue virus type 2 replication in Aotus monkeys. Viral Immunol. 2015;28(3):161–9. https://doi.org/10.1089/vim.2014.0090.
Tricou V, Minh NN, Van TP, Lee SJ, Farrar J, Wills B, et al. A randomized controlled trial of chloroquine for the treatment of dengue in Vietnamese adults. PLoS Negl Trop Dis. 2010;4(8):e785. https://doi.org/10.1371/journal.pntd.0000785.
Borges MC, Castro LA, Fonseca BA. Chloroquine use improves dengue-related symptoms. Mem Inst Oswaldo Cruz. 2013;108(5):596–9.
Tay MY, Fraser JE, Chan WK, Moreland NJ, Rathore AP, Wang C, et al. Nuclear localization of dengue virus (DENV) 1-4 non-structural protein 5; protection against all 4 DENV serotypes by the inhibitor Ivermectin. Antivir Res. 2013;99(3):301–6. https://doi.org/10.1016/j.antiviral.2013.06.002.
Tay MY, Smith K, Ng IH, Chan KW, Zhao Y, Ooi EE, et al. The C-terminal 18 amino acid region of dengue virus NS5 regulates its subcellular localization and contains a conserved arginine residue essential for infectious virus production. PLoS Pathog. 2016;12(9):e1005886. https://doi.org/10.1371/journal.ppat.1005886.
Wagstaff KM, Sivakumaran H, Heaton SM, Harrich D, Jans DA. Ivermectin is a specific inhibitor of importin alpha/beta-mediated nuclear import able to inhibit replication of HIV-1 and dengue virus. Biochem J. 2012;443(3):851–6. https://doi.org/10.1042/BJ20120150.
Lok SM, Kostyuchenko V, Nybakken GE, Holdaway HA, Battisti AJ, Sukupolvi-Petty S, et al. Binding of a neutralizing antibody to dengue virus alters the arrangement of surface glycoproteins. Nat Struct Mol Biol. 2008;15(3):312–7. https://doi.org/10.1038/nsmb.1382.
Fibriansah G, Ibarra KD, Ng TS, Smith SA, Tan JL, Lim XN, et al. DENGUE VIRUS. Cryo-EM structure of an antibody that neutralizes dengue virus type 2 by locking E protein dimers. Science. 2015;349(6243):88–91. https://doi.org/10.1126/science.aaa8651.
Avirutnan P, Punyadee N, Noisakran S, Komoltri C, Thiemmeca S, Auethavornanan K, et al. Vascular leakage in severe dengue virus infections: a potential role for the nonstructural viral protein NS1 and complement. J Infect Dis. 2006;193(8):1078–88. https://doi.org/10.1086/500949.
Beatty PR, Puerta-Guardo H, Killingbeck SS, Glasner DR, Hopkins K, Harris E. Dengue virus NS1 triggers endothelial permeability and vascular leak that is prevented by NS1 vaccination. Sci Transl Med. 2015;7(304):304ra141. https://doi.org/10.1126/scitranslmed.aaa3787.
• Fibriansah G, Lok SM. The development of therapeutic antibodies against dengue virus. Antivir Res. 2016;128:7–19. https://doi.org/10.1016/j.antiviral.2016.01.002This review discusses the monoclonal antibodies that are currently being evaluated preclinically for the treatment of dengue.
Rey FA, Stiasny K, Vaney MC, Dellarole M, Heinz FX. The bright and the dark side of human antibody responses to flaviviruses: lessons for vaccine design. EMBO Rep. 2018;19(2):206–24. https://doi.org/10.15252/embr.201745302.
Slon Campos JL, Mongkolsapaya J, Screaton GR. The immune response against flaviviruses. Nat Immunol. 2018;19(11):1189–98. https://doi.org/10.1038/s41590-018-0210-3.
Cupps TR, Fauci AS. Corticosteroid-mediated immunoregulation in man. Immunol Rev. 1982;65:133–55.
Yacoub S, Wills B. Predicting outcome from dengue. BMC Med. 2014;12:147. https://doi.org/10.1186/s12916-014-0147-9.
Wei Y, Ji XB, Wang YW, Wang JX, Yang EQ, Wang ZC, et al. High-dose dexamethasone vs prednisone for treatment of adult immune thrombocytopenia: a prospective multicenter randomized trial. Blood. 2016;127(3):296–302; quiz 70. https://doi.org/10.1182/blood-2015-07-659656.
Sun DS, King CC, Huang HS, Shih YL, Lee CC, Tsai WJ, et al. Antiplatelet autoantibodies elicited by dengue virus non-structural protein 1 cause thrombocytopenia and mortality in mice. J Thromb Haemost. 2007;5(11):2291–9. https://doi.org/10.1111/j.1538-7836.2007.02754.x.
Falconar AK. The dengue virus nonstructural-1 protein (NS1) generates antibodies to common epitopes on human blood clotting, integrin/adhesin proteins and binds to human endothelial cells: potential implications in haemorrhagic fever pathogenesis. Arch Virol. 1997;142(5):897–916.
Kularatne SA, Walathara C, Mahindawansa SI, Wijesinghe S, Pathirage MM, Kumarasiri PV, et al. Efficacy of low dose dexamethasone in severe thrombocytopenia caused by dengue fever: a placebo controlled study. Postgrad Med J. 2009;85(1008):525–9. https://doi.org/10.1136/pgmj.2008.078444.
Shashidhara KC, Murthy KA, Gowdappa HB, Bhograj A. Effect of high dose of steroid on plateletcount in acute stage of dengue fever with thrombocytopenia. J Clin Diagn Res. 2013;7(7):1397–400. https://doi.org/10.7860/JCDR/2013/6135.3143.
Premaratna R, Jayasinghe KG, Liyanaarachchi EW, Weerasinghe OM, Pathmeswaran A, de Silva HJ. Effect of a single dose of methyl prednisolone as rescue medication for patients who develop hypotensive dengue shock syndrome during the febrile phase: a retrospective observational study. Int J Infect Dis. 2011;15(6):e433–4. https://doi.org/10.1016/j.ijid.2011.03.006.
Tassniyom S, Vasanawathana S, Chirawatkul A, Rojanasuphot S. Failure of high-dose methylprednisolone in established dengue shock syndrome: a placebo-controlled, double-blind study. Pediatrics. 1993;92(1):111–5.
Sumarmo TW, Asrin A, Isnuhandojo B, Sahudi A. Failure of hydrocortisone to affect outcome in dengue shock syndrome. Pediatrics. 1982;69(1):45–9.
Tam DT, Ngoc TV, Tien NT, Kieu NT, Thuy TT, Thanh LT, et al. Effects of short-course oral corticosteroid therapy in early dengue infection in Vietnamese patients: a randomized, placebo-controlled trial. Clin Infect Dis. 2012;55(9):1216–24. https://doi.org/10.1093/cid/cis655.
Saito M, Oishi K, Inoue S, Dimaano EM, Alera MT, Robles AM, et al. Association of increased platelet-associated immunoglobulins with thrombocytopenia and the severity of disease in secondary dengue virus infections. Clin Exp Immunol. 2004;138(2):299–303. https://doi.org/10.1111/j.1365-2249.2004.02626.x.
Dimaano EM, Saito M, Honda S, Miranda EA, Alonzo MT, Valerio MD, et al. Lack of efficacy of high-dose intravenous immunoglobulin treatment of severe thrombocytopenia in patients with secondary dengue virus infection. Am J Trop Med Hyg. 2007;77(6):1135–8.
Kumar V, Gupta K, Soneja M, Biswas A. Intravenous immunoglobulin for severe thrombocytopenia in secondary dengue. BMJ Case Rep. 2018;2018. https://doi.org/10.1136/bcr-2018-224542.
Lazarus AH, Freedman J, Semple JW. Intravenous immunoglobulin and anti-D in idiopathic thrombocytopenic purpura (ITP): mechanisms of action. Transfus Sci. 1998;19(3):289–94.
de Castro RA, de Castro JA, Barez MY, Frias MV, Dixit J, Genereux M. Thrombocytopenia associated with dengue hemorrhagic fever responds to intravenous administration of anti-D (Rh(0)-D) immune globulin. Am J Trop Med Hyg. 2007;76(4):737–42.
Pannu AK, Bhalla A, Singhal M, Suri V, Shafiq N, Varma S. Safety and efficacy of a single dose of anti-D (WinRho((R))) in severe thrombocytopenia secondary to dengue virus infection. Indian J Crit Care Med. 2017;21(2):80–4. https://doi.org/10.4103/ijccm.IJCCM_386_16.
Lye DC, Archuleta S, Syed-Omar SF, Low JG, Oh HM, Wei Y, et al. Prophylactic platelet transfusion plus supportive care versus supportive care alone in adults with dengue and thrombocytopenia: a multicentre, open-label, randomised, superiority trial. Lancet. 2017;389(10079):1611–8. https://doi.org/10.1016/S0140-6736(17)30269-6.
Khan Assir MZ, Kamran U, Ahmad HI, Bashir S, Mansoor H, Anees SB, et al. Effectiveness of platelet transfusion in dengue fever: a randomized controlled trial. Transfus Med Hemother. 2013;40(5):362–8. https://doi.org/10.1159/000354837.
Lee TH, Wong JG, Leo YS, Thein TL, Ng EL, Lee LK, et al. Potential harm of prophylactic platelet transfusion in adult dengue patients. PLoS Negl Trop Dis. 2016;10(3):e0004576. https://doi.org/10.1371/journal.pntd.0004576.
Thomas L, Kaidomar S, Kerob-Bauchet B, Moravie V, Brouste Y, King JP, et al. Prospective observational study of low thresholds for platelet transfusion in adult dengue patients. Transfusion. 2009;49(7):1400–11. https://doi.org/10.1111/j.1537-2995.2009.02132.x.
Kunder CA, St John AL, Abraham SN. Mast cell modulation of the vascular and lymphatic endothelium. Blood. 2011;118(20):5383–93. https://doi.org/10.1182/blood-2011-07-358432.
St John AL, Rathore AP, Yap H, Ng ML, Metcalfe DD, Vasudevan SG, et al. Immune surveillance by mast cells during dengue infection promotes natural killer (NK) and NKT-cell recruitment and viral clearance. Proc Natl Acad Sci U S A. 2011;108(22):9190–5. https://doi.org/10.1073/pnas.1105079108.
Rathore APS, St John AL. Immune responses to dengue virus in the skin. Open Biol. 2018;8(8). https://doi.org/10.1098/rsob.180087.
Mantri CK, St John AL. Immune synapses between mast cells and gammadelta T cells limit viral infection. J Clin Invest. 2019;129:1094–108. https://doi.org/10.1172/JCI122530.
• St John AL, Rathore AP, Raghavan B, Ng ML, Abraham SN. Contributions of mast cells and vasoactive products, leukotrienes and chymase, to dengue virus-induced vascular leakage. Elife. 2013;2:e00481. https://doi.org/10.7554/eLife.00481 This study demonstrates the potential of treating dengue vascular leakage with mast cell targeting drugs in pre-clinical animal models.
Syenina A, Jagaraj CJ, Aman SA, Sridharan A, St John AL. Dengue vascular leakage is augmented by mast cell degranulation mediated by immunoglobulin Fcgamma receptors. Elife. 2015;4. https://doi.org/10.7554/eLife.05291.
Souza DG, Fagundes CT, Sousa LP, Amaral FA, Souza RS, Souza AL, et al. Essential role of platelet-activating factor receptor in the pathogenesis of dengue virus infection. Proc Natl Acad Sci U S A. 2009;106(33):14138–43. https://doi.org/10.1073/pnas.0906467106.
• Malavige GN, Wijewickrama A, Fernando S, Jeewandara C, Ginneliya A, Samarasekara S, et al. A preliminary study on efficacy of rupatadine for the treatment of acute dengue infection. Sci Rep. 2018;8(1):3857. https://doi.org/10.1038/s41598-018-22285-x This preliminary clinical study highlights to potential of targeting platelet activating factor to improve dengue clinical outcomes.
• Masri MFB, Mantri CK, Rathore APS, St John AL. Peripheral serotonin causes dengue-induced thrombocytopenia through 5HT2 receptors. Blood. 2019;133:2325–37. https://doi.org/10.1182/blood-2018-08-869156 .This preclinical study shows that drugs targeting serotonin receptor 5HT2 may be useful for the treatment of dengue-induced thrombocytopenia.
Mohd Abd Razak MR, Mohmad Misnan N, Md Jelas NH, Norahmad NA, Muhammad A, Ho TCD, et al. The effect of freeze-dried Carica papaya leaf juice treatment on NS1 and viremia levels in dengue fever mice model. BMC Complement Altern Med. 2018;18(1):320. https://doi.org/10.1186/s12906-018-2390-7.
Norahmad NA, Mohd Abd Razak MR, Mohmad Misnan N, Md Jelas NH, Sastu UR, Muhammad A, et al. Effect of freeze-dried Carica papaya leaf juice on inflammatory cytokines production during dengue virus infection in AG129 mice. BMC Complement Altern Med. 2019;19(1):44. https://doi.org/10.1186/s12906-019-2438-3.
Sharma N, Mishra KP, Chanda S, Bhardwaj V, Tanwar H, Ganju L, et al. Evaluation of anti-dengue activity of Carica papaya aqueous leaf extract and its role in platelet augmentation. Arch Virol. 2019;164:1095–110. https://doi.org/10.1007/s00705-019-04179-z.
Srikanth BK, Reddy L, Biradar S, Shamanna M, Mariguddi DD, Krishnakumar M. An open-label, randomized prospective study to evaluate the efficacy and safety of Carica papaya leaf extract for thrombocytopenia associated with dengue fever in pediatric subjects. Pediatric Health Med Ther. 2019;10:5–11. https://doi.org/10.2147/PHMT.S176712.
Kasture PN, Nagabhushan KH, Kumar A. A multi-centric, double-blind, placebo-controlled, randomized, prospective study to evaluate the efficacy and safety of Carica papaya leaf extract, as empirical therapy for thrombocytopenia associated with dengue fever. J Assoc Physicians India. 2016;64(6):15–20.
Chathurangana PW, Samaranayake D, Quienters VG, Wickramasinghe P. Effects of vitamin E supplementation on the clinical outcome of dengue fever and dengue haemorrhagic fever in children. Asian Pacific Journal of Tropical Disease. 2017;7(11):645–9. https://doi.org/10.12980/apjtd.7.2017D7-176.
Vaish A, Verma S, Agarwal A, Gupta L, Gutch M. Effect of vitamin E on thrombocytopenia in dengue fever. Annals of Tropical Medicine and Public Health. 2012;5:282–5. https://doi.org/10.4103/1755-6783.102004.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Dr. Rathore has a pending patent application: PCT/SG2018/050552, Use of Serotonergic Drugs to Treat Virus-Induced Thrombocytopenia and an issued patent: PCT/US9763921B2: Novel dosing regimens of celgosivir for the treatment of dengue.
Dr. St. John has a pending patent application: PCT/SG2018/050552, Use of Serotonergic Drugs to Treat Virus-Induced Thrombocytopenia, and an issued patent: PCT/US9730921B2, Compositions and methods for treating mast cell-induced vascular leakage.
Dr. Masri discloses a pending patent application PCT/SG2018/050552, Use of SerotonergicDrugs to Treat Virus-Induced Thrombocytopenia.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Viral Infections
Rights and permissions
About this article
Cite this article
Masri, M.F.B., Rathore, A.P.S. & St. John, A.L. Therapeutics for Dengue. Curr Treat Options Infect Dis 11, 199–214 (2019). https://doi.org/10.1007/s40506-019-00193-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40506-019-00193-6