Abstract
Purpose
Gaucher disease (GD) is a lysosomal storage disease caused by an autosomal recessive inherited deficiency of the lysosomal enzyme glucocerebrosidase. The aim of this study is to describe jaw bones’ involvement and dental radiographic features in paediatric Gaucher disease patients (type I and type III).
Methods
The study population of this case–control study included: 42 Gaucher patients (study group) and 84 medically free children (control group). The radiographic images of both groups were analysed for the following findings: generalised bone rarefaction, localised rarefaction and enlarged bone marrow spaces, thinning of cortex, pseudocystic radiolucent lesions, anodontia and dental anomalies. Dental age assessment of Gaucher patients using the Demirjian’s method was also performed.
Results
Generalised rarefaction showed almost similar percentages in both types of Gaucher disease cases. Localised rarefaction was noted in 30.77% and 18.75% of Gaucher disease type III and type I, respectively. Pseudocystic radiolucent lesions, thinning of cortex, anodontia and dental anomalies were more prevalent in type III Gaucher patients. The mean chronological and mean dental ages in both sexes of Gaucher patients were not statistically significant.
Conclusion
Thinning of cortex, localised rarefaction and generalised rarefaction are the most common jaw bone findings in Gaucher patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gaucher disease (GD) is the most common lysosomal storage disorder caused by an autosomal recessive inherited deficiency of the lysosomal enzyme glucocerebrosidase. This leads to the accumulation of the lipid glucocerebroside in the lysosome of the macrophages which become dilated Gaucher cells. Many of the disease manifestations result from infiltration of Gaucher cells into organs of the reticulo-endothelial system, particularly the spleen, liver and bone marrow (Saranjam et al. 2012).
Gaucher disease occurs in 1 in 50,000 to 100,000 people in the general population and increases in communities with consanguineous marriages, inbreeding, or geographic isolates (Lukina et al. 2010; Nalysnyk et al. 2017). GD manifests with broad phenotypic variation, ranging from neonatal lethality to asymptomatic octogenarians (Sidransky 2012). Three clinical forms of the disease are defined based on the absence (type I) or presence (types II and III) of neurological signs.
Type I is the most common type, with an ethnic predisposition in Ashkenazi Jews. Patients of this form are usually asymptomatic and show wide variation in the extent of signs with common presenting features of hepatosplenomegaly, hypersplenism, bleeding and bone pain. CNS involvement is usually rare, but patients may develop Parkinson disease. The onset of signs can occur at any age (Beutler and Gbowskira 2001; Zimran and Elstein 2010). Type II is more acute and shows high mortality rate by the age of 2 years (Lachmann et al. 2004). It has two common subtypes: neonatal, which has two null or recombinant mutations with common signs of hydrops fetalis; congenital ichthyosis, with lethal involvement of CNS. The infantile type presents one null and one severe mutation as common genotype with common presenting features of abnormal horizontal eye movements, strabismus, opisthotonos, trismus and severe CNS involvement (Zimran and Elstein 2010).
Type III is the chronic form where patients present some features in childhood with at least one neurological sign. (Schiffmann and Vellodi 2007). It encompasses three different subtypes, Subtype 3a usually presents with abnormal horizontal eye movements and myoclonic seizures. Subtype 3b shows common presenting features of abnormal horizontal eye movement, hepatosplenomegaly and growth retardation. Subtype 3c, which is common in Palestinian Arabs, Japanese and Spanish, presents with common signs of abnormal horizontal eye movements and calcification of cardiac valves (Zimran and Elstein 2010).
Bone association is one of the most common signs attributed to GD, mainly type I and type III, which is characterised by recurrent and sudden onset of painful episodes of “bone crisis”. The underlying pathology of bone disease is related to the accumulation of Gaucher cells that infiltrate the bone marrow compartment and lead directly or indirectly to localised bone defects, including cortical thinning, osteonecrosis and lytic lesions. Skeletal involvement follows the three basic processes: focal disease (irreversible lesions such as osteonecrosis and osteosclerosis), local disease (reversible abnormalities adjacent to heavily involved marrow such as cortical thinning and long bone deformity) and generalised osteopenia (Wenstrup et al. 2002).
Pathological fractures or fractures even after minor trauma are common complications of GD (Mistry et al. 2017). Osteopaenia is prevalent among type I GD patients and low bone density becomes evident by the age of 5 years with the highest prevalence rate in adolescence (Mistry et al. 2011). Long bone involvement is common in GD, whereas maxillofacial bone involvements are less commonly seen. These manifestations can be detected by routine dental X-rays, are often asymptomatic and include generalised bone rarefaction, enlarged marrow space, cortical thinning, osteosclerosis, radiolucent psuedocystic lesions and demineralisation of the mental region (Carter et al. 1998; Heasman 1991; Horwitz et al. 2007). The mandible has been reported to be a nidus of Gaucher cell infiltration and/or bone crisis in several case reports (Bender 1959; Sela et al. 1972; Lustmann et al. 1991; Schwartz et al. 1998; Baldini et al. 2018).
Studies reporting maxillofacial bone involvement among Gaucher disease patients are relatively few and include small numbers of patients, mainly case reports presenting 1 or 2 cases and case series presenting up to 28 cases (Horwitz et al. 2007; Ahmadieh et al. 2014; Carter et al. 1998). The purpose of this study was to describe jaw bones involvement and dental radiographic features in 42 paediatric Egyptian Gaucher disease patients (type I and type III) using panoramic radiographs and to compare the radiographic findings of the study group with an age- and sex-matched control group.
Subjects and methods
The population of this cross-sectional study included a total of 126 children divided into two groups. The study group included 42 patients diagnosed with GD through laboratory investigations and were recruited from Abu El Reesh children Hospital, Cairo, Egypt. The control group included 84 healthy children with matching age and sex, recruited from the outpatient clinic of the Oral and Maxillofacial Radiology department, Faculty of Dentistry, Cairo University; for screening and diagnostic purposes.
Visceral and haematological involvement data were recorded from the patient’s records as part of their routine follow-up. Short stature was reported by comparing the child’s height to the CDC growth curves. The date of birth as documented in hospital records determined the chronological age of each child.
Dental history, extra-oral and intra-oral examination were recorded by a single paediatric dentist at Abu-El Reesh Children Hospital.
Panoramic radiographs were carried out using Proline XC machine (Planmeca, Helsinki-Finland) with 60–68 kVp, 5–7 mA according to the child’s age and 18 s exposure time.
The radiographic assessment for digital panoramic images was performed by an oral and maxillofacial radiologist of 15 years’ experience for bony and dental abnormalities. Assessment was done twice with 2 weeks interval, and in case of presence of inconsistent radiographic findings in some cases, a third assessment was done 2 weeks later. Each assessment session was of half an hour duration with haphazard arrangement of digital images.
The radiographic images of both groups were analysed for the following findings which were commonly reported in previous studies: generalised bone rarefaction, localised rarefaction and enlarged bone marrow spaces, thinning of cortex, pseudocystic radiolucent lesions, anodontia and dental anomalies. (Nobre et al. 2012; Saranjam et al. 2012). Thinning of the inferior border of mandible in the anterior region was judged by comparison with other parts of the inferior border. Cyst-like radiolucent lesions were referred to as pseudocyst, because biopsy was not feasible to confirm diagnosis as all lesions were asymptomatic.
Dental age assessment of the Gaucher patients using the Demirjian’s method (Demirjian et al. 1973) was also performed. Determination of dental age was based on the rate of development and calcification of tooth buds using Demirjian’s method which includes eight developmental stages from A to H. The seven permanent left mandibular teeth from the central incisor to the second molar were scored, using the score tables, based on their developmental stage in the panoramic radiographs. The sum of seven individual scores gives maturity score, which was converted into dental age using Demirjian’s conversion charts.
Statistical package used for this study: R statistical package, version 3.3.1 (2016-06-21), copyright (C) 2016. All variables were described in terms of frequencies and percentages. Pearson’s Chi-squared test was used to determine whether any of the radiographic variable data were related to the presence of GD. Odds ratios were calculated using multinomial regression models where GD is the dependent variable and radiographic signs are the independent variables. Also, interactions between different radiographic signs were examined. The chronological age and dental age were compared using the unpaired Student’s t test. The results of all tests were considered to be statistically significant if p value was ≤ 0.05.
Results
The study sample of the current study consisted of 42 patients with GD (study group): 26 males and 16 females with a mean age of 9.54 ± 4.25 years. The control group consisted of 84 healthy children as reported in medical history taken from their guardians: 45 males and 39 females with a mean age of 11.37 ± 1.83 years.
The study group was further subdivided into two subgroups according to GD type (type I and type III). The haematological results of the study group were as follows: mean haemoglobin count was 10.16 ± 1.43 and 10.78 ± 1.59 and platelet count was 158 ± 94.17 and 201.62 ± 142.99 in type I and type III, respectively. The normal range of haemoglobin level in children and adolescents is 12–15 gm/dL and normal platelet count ranges between 150,000 and 450,000 platelets per microlitre. Systemic findings and current medications for the study group are summarised in Table 1.
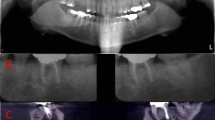
Analysis of the radiographic findings for GD cases versus control cases is presented in Table 2. Generalised rarefaction showed almost similar percentages in both type I and type III GD cases. Localised rarefaction was more prevalent in type III GD, and wide bone marrow spaces were more prevalent in type I GD (Fig. 1). Pseudocystic radiolucent lesions, thinning of cortex, anodontia and dental anomalies were more prevalent in type III GD (Fig. 2).
Chi-squared test results showed that there was a significant association between both types of GD and generalised rarefaction, wide bone marrow spaces, pseudocystic radiolucencies, thinning of cortex, dental anomalies and absence of abnormal radiographic features (p value < 0.05).
Regression model analysis revealed that patients with generalised rarefaction, wide bone marrow spaces and thinning of cortex are more likely to have type I GD. However, the presence of pseudocystic radiolucencies and dental anomalies are not associated with type I GD (Table 3).
Patients with no abnormal radiographic features have a lower risk for type I GD compared to those who have abnormal radiographic features with an odds ratio of 0.1 (95% CI 0.03–0.39, p value = 0.0009). On the other hand, patients with generalised rarefaction, pseudocystic radiolucent lesions, thinning of cortex and dental anomalies are more likely to have type III GD. However, wide bone marrow spaces are not associated with type III GD (p value = 0.3464). Patients with no abnormal radiographic features are less likely to have type III GD compared to those who have abnormal radiographic features with an odds ratio of 0.13 (95% CI 0.05–0.37, p value = 0.0001).
The results of multinomial regression models using interaction terms showed that there was no interaction between different radiographic signs among both groups of type I and type III GD (Table 3).
Intra-oral dental examination showed that five patients presented with delayed permanent teeth eruption. Regarding dental development determination using Demirjian’s method, GD patients were divided into two groups only, 26 males and 16 females due to the small number of patients in each age category. Dental age and chronological age were found to be 8.95 (± 4.01) and 8.42 (± 3.53), and 9.92 (± 4.47) and 8.93 (± 3.92) years for males and females, respectively. Although dental age was lower than chronological age, no statistically significant difference was found between them using unpaired Student’s t test (p value equals 0.4125 and 0.7038 for males and females, respectively).
Discussion
Gaucher disease is a rare genetic disease manifesting in three major types according to the presence or absence of neurological manifestations. Type I is the most common, while type III is seen in 5% of patients and is mainly seen in Northern Europe, Egypt and East Asia (Tylki-szumanska et al. 2010).
Systemic findings of the current study showed that organomegaly, short stature and bone pain were common among type I Gaucher patients (Grummer-Strawn et al. 2010). Low haemoglobin count was reported in 71% and low platelet count in 35.7% of Gaucher patients. Relatively similar results were reported in the case series of Sheth et al. (2011). Both haematological parameters increase the risk to bleeding and delayed healing; therefore, consultation with the haematologist before dental procedures that might induce bleeding is mandatory in GD patients.
Type III cases in the current study presented with organomegaly, bone pain and bone fracture which relatively agreed with Hill et al. (1996).
Bisphosphonate (BPs) therapy was used by 9.5% of the total sample of Gaucher patients in the current study, and 95% were on enzyme replacement therapy (ERT). Various bisphosphonates have been used by patients with GD as monotherapy for osteopaenia/osteonecrosis as they promote proliferation and differentiation of osteoblast-like cells. They are also used as an adjunct to ERT which has proven to be beneficial in alleviating organomegaly, anaemia and thrombocytopenia in patients with GD (Serratrice et al. 2016). Bisphosphonates are known to inhibit osteoclastic bone resorption; this hypothesis suggests a value for these agents as adjuvant therapy for the treatment of bone complications in GD. Bender and Bender (1996) also reported that ERT improved the rarefaction of the mandible bilaterally with no evidence of osteolysis.
Although BPs are anti-resorptive drugs that act on osteoclasts and maintain bone density; osteonecrosis of the jaw is a rare reported complication of long-term intravenous bisphosphonate treatment and seems to be triggered by exposed bone and subsequent bacterial contamination, typically after dental extraction (Rasmusson and Abtahi 2014).
Most jaw manifestations in the current study were present in the mandible, not in maxilla. This may be because the mandible is considered a potential reservoir for Gaucher cell infiltration (Wenstrup et al. 2002). Comparing Gaucher patients type III and type I with the control group by means of panoramic images, four manifestations had statistically significant differences: generalised rarefaction, wide bone marrow spaces, thinning of cortex and dental anomalies, which implies that those manifestations are specific features for GD.
Generalised rarefaction showed almost similar percentages in both type III (19.23%) and type I (18.75%) GD cases which agreed with Bender and Bender (1996), who observed generalised rarefactions in only 5 out of 28 Gaucher type I patients. However, it disagrees with Heasman (1991), Nobre et al. (2012) and Karabulut et al. (1997), who found that rarefactions were detected in all Gaucher cases. Those differences can be explained by that in the current study, 95% of Gaucher cases were on ERT which might have improved bone rarefaction and reversed the necrotic and lytic changes in bone.
Wide bone marrow spaces were noted in 31.25% of the cases with type I GD, which contradicts the results of Nobre et al. (2012) who recognised enlarged marrow spaces in nine out of ten cases, and also disagreed with the findings of Bender and Bender (1996) and Karabulut et al. (1997).
Thinning of cortex was more prevalent in type III Gaucher patients (38.46%) than type I patients (25%), which disagrees with others studies that showed that this manifestation was one of the most prevalent findings (Heasman 1991; Nobre et al. 2012).
Prevalence of bony manifestations as thinning of cortex is crucial in management of patients with GD as they might have increased risk of bone fractures even from minor trauma (Mistry et al. 2017); therefore, tooth extraction and minor oral surgeries must be performed as atraumatically as possible in those patients.
Pseudocystic radiolucent lesions were more prevalent in type III Gaucher patients (15.38%) versus (6.25%) in type I patients, which partially agrees with Nobre et al. (2012) who observed pseudocystic lesions in one panoramic radiograph in his study. However, the results of this study disagree with other studies where the most common radiographic observation in an affected mandible is the presence of pseudocystic or honeycombed radiolucent lesions mainly in the premolar–molar regions (Weigler et al. 1967; Michanowicz et al. 1967; Bildman et al. 1972; Lustmann et al. 1991; Horwitz et al. 2007). Biopsy may be required for more definitive diagnosis of cyst lesions, which was not performed in the current study as all the lesions were asymptomatic and the current study is an observational study and as per patients consent only clinical and radiographic examination were included. Formation of pseudocystic lesions might be attributed to the presence of thin bone trabeculae and multiple wide bone spaces which may coalesce together giving a cystic appearance.
Anodontia (26.92%) and dental anomalies (19.23%) were more prevalent in type III Gaucher patients that may be explained by the accumulation of Gaucher cells, which increase intraosseous pressure and compromise the vascularity of the area by extrinsic compression or occlusion of these vessels (Bender and Bender 1996; Horwitz et al. 2007).
Differences in prevalence of dental and jaw manifestations in the current study can be attributed to the increased number of subjects in this study compared to other studies.
Regarding dental development determination; the dental age using Demirjian’s method was lower than the chronological age for most of the study sample, yet the mean chronological and mean dental ages in both sexes were not statistically significant, which agrees with Carter et al. (1998). They linked the delayed eruption of teeth to the delayed achievement of peak bone mass, and, often, of full adult height in some patients with GD.
Conclusion
Localised rarefaction and generalised rarefaction are the most common jaw bone findings in both types of GD.
The presence of pseudocystic radiolucencies and dental anomalies is more prevalent in type III GD, while wide bone marrow spaces are more prevalent in type I GD.
Dental development results can be considered preliminary and further studies comparing larger number of Gaucher patients versus controls are recommended.
Clinical significance
The current results offer paediatric dentists an overview of the possible systemic and dental manifestations accompanying GD, which is crucial information when planning for dental management of those cases.
References
Ahmadieh A, Farnad F, Sedghizadeh P. Gaucher disease with jawbone involvement: a case report. J Med Case Rep. 2014;8:360.
Baldini M, Cairati G, Uliviei FM, Cassineria E, Chalouhi KK, Poggiali E, Borin L, Burghignoli V, Cesana BM, Cappellini MD. Skeletal involvement in type 1 Gaucher disease: not just bone mineral density. Blood Cells Mol Dis. 2018;68:148–52.
Bender IB. Dental observations in Gaucher’s disease; a twenty-year follow-up. Oral Surg Oral Med Oral Pathol. 1959;12(5):546–61.
Bender IB, Bender AL. Dental observations in Gaucher disease: review of the literature and two case reports with 13- and 60-year follow-ups. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1996;82:650–9.
Beutler E, Gbowskira GA. Gaucher disease. In: Scriver CR, Beaudet SL, Sly WS, Valle D, editors. The metabolic and molecular bases of inherited disease. New York: McGraw-Hill; 2001. p. 3635–8.
Bildman B, Martinez MJR, Robinson LH. Gaucher disease discovered by mandibular biopsy: report of case. J Oral Surg. 1972;30(7):510–2.
Carter LC, Fischman SL, Mann J, Elsein D, Stabholz A, Zimran A. The nature and extent of jaw involvement in Gaucher disease: observations in a series of 28 patients. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1998;85(2):233–9.
Demirjian A, Goldestein H, Tanner JM. A new system of dental age assessment. Hum Biol. 1973;45(2):211–27.
Heasman PA. Mandibular lesions in Gaucher disease. Oral Surg Oral Med Oral Pathol. 1991;72(4):506.
Hill S, Damaska B, Tsokos M, Kreps C, Brady R, Barton N. Radiographic findings in type 3 b Gaucher disease. Pediatr Radiol. 1996;26(12):852–60.
Horwitz J, Hirsh I, Machtei EE. Oral aspects of Gaucher’s disease: a literature review and case report. J Periodontol. 2007;78(4):783–8.
Grummer-Strawn LM, Reinold C, Krebs NF, Centers for Disease Control and Prevention. Use of World Health Organization and CDC growth charts for children aged 0–59 months in the United States. MMWR Recomm Rep. 2010;59(9):1–15.
Karabulut N, Ahmetoglu A, Ariyurek M, Erol C. Obliteration of maxillary and sphenoid sinuses in Gaucher’s disease. Br J Radiol. 1997;70:533–5.
Lachmann R, Grant I, Halsall D, Cox T. Twinpairs showing discordance of phenotype in adult Gaucher’s disease. QJM. 2004;97:199–204.
Lukina E, Watman N, Arregun EA, et al. Improvement in hematological, visceral, and skeletal manifestations of Gaucher disease type 1 with oral eliglustat tartrate (Genz-112638) treatment: 2-year results of a phase 2 study. Blood. 2010;116:4095–8.
Lustmann J, Ben-Yehuda D, Somer M, Ulmansky M. Gaucher disease affecting the mandible and maxilla. Int J Oral Maxillofac Surg. 1991;20(1):7–8.
Michanowicz AE, Michanowicz JP, Stein GM. Gaucher disease: report of a case. Oral Surg Oral Med Oral Pathol. 1967;23(1):36–42.
Mistry PK, Lopez G, Schiffmann R, Barton NW, Weinreb NJ, Sidransky E. Gaucher disease: progress and ongoing challenges. Mol Genet Metab. 2017;120(1–2):8–21.
Mistry PK, Weinreb NJ, Kaplan P, Cole JA, Gwosdow AR, Hangartner T. Osteopenia in Gaucher disease develops early in life: response to imiglucerase enzyme therapy in children, adolescents and adults. Blood Cells Mol Dis. 2011;46(1):66–72.
Nalysnyk L, Rotella P, Simeone JC, Hamed A, Weinreb N. Gaucher disease epidemiology and natural history: a comprehensive review of the literature. Hematology. 2017;22:65–73.
Nobre RM, Ribeiro ALR, Alves-Junior SM, Tuji FM, Rodrigues Pinheiro MDG, Pinheiro LR, Pinheiro JJV. Dentomaxillofacial manifestations of Gaucher’s disease: preliminary clinical and radiographic finding. Dentomaxillofac Radiol. 2012;41(7):541–54.
Rasmusson L, Abtahi J. Bisphosphonate associated osteonecrosis of the jaw: an update on pathophysiology, risk factors and treatment. Int J Dent. 2014. https://doi.org/10.1155/2014/471035.
Saranjam HR, Sidransky E, Levine WZ, Zimran A, Elstein D. Mandibular and dental manifestations of Gaucher disease. Oral Dis. 2012;18(5):421–9.
Schiffmann R, Vellodi A. Neuronopathic Gaucher disease. In: Futerman AH, Zimran A, editors. Gaucher disease. Boca Raton: CRC Press, Taylor & Francis; 2007. p. 175–96.
Schwartz M, Weycer J, Mcgavran M. Gaucher’s disease involving the maxillary sinuses. Arch Otolaryngol Head Neck Surg. 1998;114(2):203–6.
Sela J, Polliack A, Ulmansky M. Involvement of the mandible in Gaucher disease. Report of a case with post-mortem findings. Br J Oral Surg. 1972;9(3):246–50.
Serratrice C, Carballo S, Serratrice C, Stirnrmann J. Imiglucerase in the management of Gaucher disease type 1: an evidence-based review of its place in therapy. Core Evid. 2016;11:37–47.
Sheth JJ, Ankleshwaria CM, Mistri MA, Nanavaty N, Methta SJ. Splenomegaly, cardiomegaly, and osteoporosis in a child with Gaucher disease. Case Rep Pediatr. 2011;2011:564868.
Sidransky E. Gaucher disease: insights from a rare Mendelian disorder. Discov Med. 2012;14:273–81.
Tylki-szumanska A, Vellodi A, El-Beshlawy A, Cole JA, Kolodny E. Neuronopathic Gaucher disease: demographic and clinical features of 131 patients enrolled in the International Collaborative Gaucher Group Neurological Outcomes Subregistry. J Inherit Metab Dis. 2010;33(4):339–46.
Weigler JM, Seldin R, Minkowitz S. Gaucher disease involving the mandible: report of case. J Oral Surg. 1967;25(2):158–63.
Wenstrup RJ, Roca-Espiau M, Wenreb NJ, Bembi B. Skeletal aspects of Gaucher disease: a review. Br J Radiol. 2002;75(Suppl 1):A2–12.
Zimran A, Elstein D. Lipid storage diseases. In: Lichtman MA, Kipps T, Seligsohn U, Kaushansky K, Prchal JT, editors. Williams hematology, vol. 8. New York: McGraw-Hill; 2010. p. 1065–71.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflicts of interest.
Ethical approval
This study was approved by the Research Ethics Committee, Faculty of Medicine, Cairo University, approval number R-7-2016. The current study followed the ethical standards of Helsinki declaration.
Informed consent
The purpose of the study was explained to the guardians of children and they all signed an informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mohamed, Y.S.A., Zayet, M.K., Omar, O.M. et al. Jaw bones’ involvement and dental features of type I and type III Gaucher disease: a radiographic study of 42 paediatric patients. Eur Arch Paediatr Dent 21, 241–247 (2020). https://doi.org/10.1007/s40368-019-00471-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40368-019-00471-3