Abstract
Introducion
Treatment strategies for patients with pre-hypertension and low–moderate cardiovascular (CV) risk may include nutraceutical compounds (NCs).
Aim
To investigate the efficacy and safety of a new-generation of NC in lowering BP values and improving metabolic profile, in a group of hyper-cholesterolemic subjects with pre-hypertension.
Methods
131 subjects with pre-hypertension (systolic BP 130–139 mmHg and/or diastolic BP 85–89 mmHg) without organ damage and history of CV diseases were enrolled. 66 subjects were treated with a once-daily oral formulation of a NC (red yeast rice, Berberine, Coenzyme Q10, folic acid and chrome) added to diet for 3 months, while 65 patients followed a diet only. Differences in serum total cholesterol (TC), low- and high-density lipoprotein cholesterol (LDLC and HDLC), triglycerides (TG), glycemia, creatine phosphokinase (CPK), aspartate aminotransferase (AST) alanine aminotransferase (ALT) and body mass index (BMI) were evaluated.
Results
At the end of treatment, significant reductions of TC, LDLC, TG glucose levels were observed in both treatment groups, while HDLC values increased in the active treatment group only. A greater reduction of TC, LDLC and glycemia was observed in the treatment group. TG levels were not different within the two groups. BP and BMI levels remained unchanged, as well AST, ALT; CPK slightly increased in both groups, but it remained in the normal range.
Conclusions
In patients with pre-hypertension, NC supplementation was safe, well tolerated and effective in improving lipid pattern and glucose levels and in preventing the progression to overt hypertension.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Pre-hypertension is defined as having a systolic blood pressure (BP) reading of between 120 and 139 and/or a diastolic BP reading of 80–89 values [1]. As observed in clinical studies and in the real-life population, pre-hypertension is frequently associated with different metabolic abnormalities that increase the cardiovascular (CV) risk regardless of BP. In this respect, the TECUMSEH BP study reported that people with pre-hypertension were significantly more likely to be overweight, and have higher total cholesterol (TC), triglycerides (TG), insulin, glucose levels, and lower high-density lipoprotein cholesterol (HDLC), than normotensive study participants [2]. Furthermore, patients with either pre-hypertension or the above-mentioned CV risk factors have a 2–3 times higher risk to develop a sustained hypertension, compared to the patients with normal BP values [1, 3, 4]. However, in these patients, pharmacological treatments failed to demonstrate a reduction in CV events as found in high-risk patients [1, 5, 6] and the potential side effects of the anti-hypertensive pharmacological therapy lead to the unfavorable risk benefit ratio [7].
For these reasons, the 2018 ESH/ESC hypertension guidelines recommend that patients with pre-hypertension and low–moderate CV risk should be treated with non-pharmacological interventions and should be offered lifestyle advice (i.e. low salt and alcohol intake, high consumption of vegetables and fruits, weight reduction and regular physical activity), because this approach reduces their risk of progressing to established hypertension [1, 8]. Unfortunately, lifestyle changes alone do not seem to be effective on long-term, as the reduction in low density lipoprotein cholesterol (LDLC) levels with diet is observed only in 3–6% of cases [9, 10]. In the last years, the non-pharmacological interventions have been combined to the use of nutraceutical compounds (NCs), that proved to be helpful in improving BP values and metabolic alterations in people with hypertension, dyslipidemia and at low CV risk [11, 12].
The aim of this study was to investigate the efficacy and safety of a new-generation of nutraceutical compounds (NC), added to diet and lifestyle management versus diet alone, in lowering BP values and improving lipid and glucose profile, in a group of hyper-lipidemic subjects with pre-hypertension.
2 Methods
2.1 Study Design
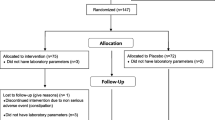
We conducted a prospective, single center, randomized open post-market trial to evaluate the effect of adding a NC to a diet regimen on BP values and metabolic parameters in patients with pre-hypertension and at low CV risk. The study protocol was approved by local Ethics Committee and institutional review boards and conducted in accordance with ICH Harmonized Tripartite Guidelines for Good Clinical Practice and the Declaration of Helsinki Principles, and was conducted from 2017 to 2018. All patients gave informed consent for the study participation.
2.2 Patients and Treatment
131 subjects, recruited in the Hypertension Centre, were enrolled in the study. Inclusion criteria were age ≥ 18 years, pre-hypertension, hyper-lipidemia defined as TC ≥ 200 mg/dl and LDLC between 130 and 190 mg/dl. Exclusion criteria were diabetes mellitus, positive history or clinical signs of ischemic heart disease, organ damage (left ventricular hypertrophy diagnosed by electrocardiogram and carotid plaque by ultrasonography), presence of neoplastic or hepatic diseases, chronic renal failure, disabilities that prevent the patient from collaborating, pregnancy or breast-feeding, use of lipid lowering drugs. All patients were prescribed a standardized Mediterranean diet, including a high intake of fish, fruits, vegetables, legumes, olive oil, unrefined whole grains and a moderate intake of lean meats and alcohol intake [13]. Patients were randomized into two groups: 66 (Group A) were treated for 3 months with a once-daily oral formulation of a NC that combines red yeast rice (equivalent of 3 mg of Monacolin K), 500 mg of Berberine, 30 mg of Coenzyme Q10, 200 mcg of folic acid and 40 mg of chrome (Liposcudil BBR, Piam Farmaceutici, GE, Italy). The compound was included in a gastro-resistant tablet, with an inner content of a Chitosan polymer and sulphurated N-acetylcysteine, an acidic molecule that in the enteric fluid causes cationization of Chitosan, and has a well-known mucolytic action. A second group of 65 patients followed a diet program only (Group B).
2.3 Assessments
Assessment visits were performed twice: at the enrollment (baseline visit) and at the end of the study (follow-up visit, after three month of treatment). At the baseline, demographic and lifestyle data were collected after obtaining informed written consent from all the recruited patients. Systolic BP, diastolic BP, heart rate (HR) were measured, and subjects were classified into current smokers (≥ 1 cigarette daily) and non-smokers. A blood sample was taken to assess the serum levels of TC, LDLC, HDLC, TG, glucose, creatinine (SCr), uricemia, aspartate aminotransferase (AST), alanine aminotransferase (ALT) and creatine phosphokinase (CPK). Fasting TC, TG, HDLC, serum glycaemia, creatinine, CPK and transaminases levels were measured by enzymatic method, while LDLC was calculated using Friedewald’s formula. Body mass index (BMI) was calculated as the ratio of weight (in kg) to squared height (in m). At the end of treatment, the assessment of PAS, PAD, TC, LDLC, HDLC, TG, glucose, BMI, AST, ALT and CPK was repeated, and the possible adverse events were collected.
2.4 Statistical Analysis
Continuous variables are indicated as mean ± standard deviation. The effect of the diet or the treatment with the NC on the different biomarkers was calculated with the T student two-tailed test with a significance level α = 0.05. As for HDLC, given the difference in the initial averages, even if not significant, the significance of the means of the difference between the basal and final measurements was calculated. Statistical analysis was performed using StatPlus software, Analyst Soft Version v6 package. The null hypothesis was always rejected for values of p < 0.05.
3 Results
The general characteristics of the study groups at baseline are showed in Table 1. All subjects had normal glycemia, uricemia, CPK, transaminases and SCr levels at baseline. No differences were found between the two groups.
The mean age was 55.7 ± 9.6 years; 53% of subjects were males. Mean BP levels were 136/85 mmHg (group A) and 136/84 mmHg (group B). At the follow up visit, a significant decrease was observed in glycemia, TC, TG, LDLC, PAS and PAD in both treatment groups, but the difference was significantly greater in the active treatment groups for TC (203 ± 16 vs 234 ± 15 vs, − 13.6%, p < 0.001), LDLC (128 ± 17 vs 155 ± 15, − 17.7%, p < 0.001) and serum glucose 87.5 ± 7.3 vs 94.9 ± 9.5, − 7.7%, p < 0.001). HDLC significantly increased only in NC treated group (54.1 ± 9.1 vs 51.5 ± 9.3, + 4.8%, p < 0.001) (Table 2 and Fig. 1).
TG levels were significantly different from baseline to the end of treatment in both group (106 ± 34 vs 139 ± 54 vs, p < 0.001 for the treatment group and 109 ± 45 vs 134 ± 65, p < 0.001 for the control group, respectively), but no difference was found at the end of treatment between groups.
No significant differences between groups and between baseline and the end of treatment were found in BMI, AST and ALT in both groups. Either in Group A or in group B a slight increase was observed in CPK, but it remained in the normal range. Gender difference was not found in either group. 1 subject in the active and 1 in the control group withdrew from the study due to dyspepsia; other two subjects (1 in the active treatment and 1 in the control group) refused to continue the study.
4 Discussion
Many studies have shown that in hypertensive and hyper-cholesterolemic subjects, NCs are able to improve BP levels and metabolic parameters [8, 10, 14], endothelial dysfunction [15] and aortic stiffness [16]. The present post-market study has evidenced that the NC compound tested containing a combination of red yeast rice, berberine, coenzyme Q10 and chrome, resulted able in improving lipid serum profile and glucose pattern when added to the diet. These changes were higher than the improvement obtained with diet alone.
The role of NCs in the primary prevention of CV diseases is not necessarily due to a single compound but to an additive and synergistic effect of several combined components [17]. Monacolin K, an active component of red yeast rice, is a lovastatin analogue that inhibits 3-Hydroxy-3-methyl-glutaryl-coenzyme A (HMG-CoA) reductases, showing a lipid-lowering effect in different conditions, like metabolic syndrome [18] and dyslipidemia [19], even in patients intolerant to statins [20] and in elderly hypercholesterolemic patients [21]. Its efficacy in reducing total and LDLC has been well documented in randomized clinical trials [12, 22]. Berberine is an alkaloid extracted from several plants [23, 24], that stabilizes the mRNA of LDL-receptors and counteracts their degradation [25], inhibits lipolysis and synthesis of cholesterol and triglycerides, and increases the liver uptake of LDL, with a consequent reduction of plasmatic levels of total and LDLC [24]. In a meta-analysis of randomized controlled trials, Dong et al. reported that the administration of berberine induced a significant reduction of total cholesterol, triglycerides and LDL, and a remarkable increase of HDL, without causing serious adverse effects [26]. Kong et al. reported that the administration of berberine to hyper-cholesterolemic patients allowed a reduction of TC by 29%, TG by 35%, and LDLC by 25% [25]. Recent reviews report that berberine seems to reduce insulin resistance and to improve glycemic control, increasing the expression of insulin receptors, promoting the regeneration and the activity of pancreatic β cells, and enhancing the muscle uptake of glucose [24, 28, 29]. The effects of berberine are synergistic with the action of monacolin K, due to inhibition of Proprotein convertase subtilsin-kexin type 9 (PCSK9) transcription [25, 27]. Treatment with a combination of monacolin K, berberine and policosanols reduces cholesterol levels and improves aortic stiffness in low-moderate risk hypercholesterolemic patients [16, 28,29,30,31,32]. In a randomized trial in hyper-cholesterolemic patients intolerant to statins, the combination of berberine and red yeast rice resulted more effective than ezetimibe in reducing TC, LDLC and non-HDLC [33]. Coenzyme Q10 (CoQ10) is a free radical scavenger with a potent antioxidant activity and a significant antihypertensive effect [34]; in this respect, a Cochrane meta-analysis reported that high doses of Coenzyme Q10 (i.e. 100 mg) reduces either systolic or diastolic BP [35]. In addition, CoQ10 improves also the lipid profile, insulin sensitivity and myocardial left ventricular function, decreases heart rate, and reduces plasma catecholamine levels [34]. Finally, chrome supplementation has shown to significantly improve glycaemia among patients with diabetes [36], and in a randomized, controlled clinical trial a combination of chrome and amino acids decreased the glucose peak by almost 30% compared to placebo [37].
In our study, Liposcudil™ BBR showed a positive trend in lowering BP values, without reaching statistical significance. In this respect, hypertension is a disease characterized by inflammation, oxidative stress and immune dysfunction in the vascular system [1]. The increase in BP levels is a bidirectional issue with vascular endothelial dysfunction, leading to hypertension. In turn, hypertension increases the endothelial dysfunction [15], and as consequence, a logical approach to the prevention of hypertension is to improve vascular function. In this respect, the literature supports a positive interaction between the use of NCs and the BP levels [10]. In our study, after 3 months of treatment with NCs, the BP values remained unchanged, thus preventing the progression to overt hypertension that is frequently observed in the subject with pre-hypertension over the years. This is probably due to the effective treatment of CV risk factors (i.e. dyslipidemia and hyperglycemia) that often co-exist in subjects with pre-hypertension. In addition, this non-pharmacological approach offers potential savings to patients and reduces the need for anti-hypertensive drugs.
4.1 Study Limitations
The study does not include a double-blind control treatment and assessments at intermediate timelines. Furthermore, we included a limited population, and a long-term treatment and follow up were not planned.
In conclusion, the addition of NCs to a dietetic regimen, appears to be safe and able to improve lipid serum profile and glucose levels, and to prevent the progression to overt hypertension. These results need to be confirmed by further studies, involving a larger sample of subjects with a longer follow up.
References
Williams B, Mancia G, Spiering W, Agabiti Rosei E, Azizi M, Burnier M et al. ESC/ESH guidelines for the management of arterial hypertension. Eur Heart J. 2018;39:3021–104.
Julius S, Jamerson K, Mejia A, Krause L, Schork N, Jones K. The association of borderline hypertension with target organ changes and higher coronary risk. Tecumseh Blood Pressure Study. JAMA. 1990;264(3):354–8.
Leitschuh M, Cupples LA, Kannel W, Gagnon D, Chobanian A. High-normal blood pressure progression to hypertension in the Framingham Heart Study. Hypertension. 1991;17(1):22–7.
Luders S, Schrader J, Berger J, Ungerj T, Walter Zidekk W, Bohm M, et al. The PHARAO study: prevention of hypertension with the angiotensin-converting enzyme inhibitor ramipril in patients with pre-hypertension—a prospective, randomized, controlled prevention trial of the German Hypertension League. J Hypertens. 2008;26:1487–96.
Thomopoulos C, Parati G, Zanchetti A. Effects of blood-pressure-lowering treatment on outcome incidence. 12. Effects in individuals with high-normal and normal blood pressure: overview and meta-analyses of randomized trials. J Hypertens. 2017;35:2150–60.
Yusuf S, Bosch G, Dagenais G, Zhu J, Xavier D, Liu L, Pais P, et al. Cholesterol lowering in intermediate-risk persons without cardiovascular disease. N Engl J Med. 2016;374:2021–31.
Nieuwlaat R, Schwalm JD, Khatib R, Yusuf S. Why are we failing to implement effective therapies in cardiovascular disease? Eur Heart J. 2013;34:1262–9.
Borghi C, Cicero AFG. Nutraceuticals with a clinically detectable blood pressure-lowering effect: a review of available randomized clinical trials and their meta-analyses. Br J Clin Pharmacol. 2017;83:163–71.
Tang JL, Armitage JM, Lancaster T, Silagy CA, Fowler GH, Neil HA. Systematic review of dietary intervention trials to lower blood total cholesterol in free-living subjects. BMJ. 1998;316:1213–20.
Mazza A, Lenti S, Schiavon L, D’Avino M, Ramazzina E, Casiglia E. Nutraceuticals for serum lipid and blood pressure control in hypertensive and hypercholesterolemic subjects at low cardiovascular risk. Adv Ther. 2015;32(7):680–90.
Sirtori CR, Galli C, Anderson JW, Arnoldi A. Nutritional and nutraceutical approaches to dyslipidemia and atherosclerosis prevention: focus on dietary proteins. Atherosclerosis. 2009;203:8–17.
Li Y, Jiang L, Jia Z, Xin W, Yang S, Yang Q, Wang L. A meta-analysis of red yeast rice: an effective and relatively safe alternative approach for dyslipidemia. PLoS One. 2014;9:e98611.
Estruch R, Ros E, Martinez-Gonzalez MA. Mediterranean diet for primary prevention of cardiovascular disease. N Engl J Med. 2013;69:676–7.
Rivellese AA, Ciciola P, Costabile G, Vetrani C, Vitale M. The possible role of nutraceuticals in the prevention of cardiovascular disease. High Blood Press Cardiovasc Prev. 2019;26(2):101–11.
Affuso F, Ruvolo A, Micillo F, Saccà L, Fazio S. Effects of a nutraceutical combination (berberine, red yeast rice and policosanols) on lipid levels and endothelial function randomized, double-blind, placebo-controlled study. Nutr Metab Cardiovasc Dis. 2010;20(9):656–61.
Pirro M, Lupattelli G, Del Giorno R, Schillaci G, Berisha S, Mannarino MR, et al. Nutraceutical combination (red yeast rice, berberine and policosanols) improves aortic stiffness in low moderate risk hypercholesterolemic patients. Pharma-Nutrition. 2013;1:73–7.
Alissa EM, Ferns GA. Functional foods and nutraceuticals in the primary prevention of cardiovascular diseases. J Nutr Metab. 2012;2012:569486.
Izzo R, de Simone G, Giudice R, Chinali M, Trimarco V, De Luca N, et al. Effects of nutraceuticals on prevalence of metabolic syndrome and on calculated Framingham Risk Score in individuals with dyslipidemia. J Hypertens. 2010;28:1482–7.
Trimarco B, Benvenuti C, Rozza F, Pagnano G, Manzi MV, Piglia A, et al. Clinical evidence of efficacy of red yeast rice and berberine in a large controlled study versus diet. Med J Nutr Metab. 2011;4:133–9.
Cicero AFG, Derosa G, Bove M, Imola F, Borghi C, Gaddi AV. Long term effectiveness and safety of a nutraceutical-based approach to reduce cholesterolemia in statin intolerant subjects with and without metabolic syndrome. Curr Top Nutrac Res. 2009;7(3–4):121–6.
Marazzi G, Cacciotti L, Pelliccia F, Iaia L, Volterrani M, Caminiti G, et al. Long-term effects of nutraceuticals (berberine, red yeast rice, policosanol) in elderly hypercholesterolemic patients. Adv Ther. 2011;28:1105–13.
Cicero AFG, Colletti A, Bajraktari G, Olivier D, Djuric DM, Ezhov M, et al. Lipid-lowering nutraceuticals in clinical practice: position paper from an International Lipid Expert Panel. Nutr Rev. 2017;75(9):731–67.
Barbagallo CM, Cefalù AB, Noto D, Averna MR. Role of nutraceuticals in hypolipidemic therapy. Front Cardiovasc Med. 2015;2:22. https://doi.org/10.3389/fcvm.2015.00022.
Chander V, Asval JS, Dobhal R, Uniyal DP. A review on Pharmacological potential of Berberine; an active component of Himalayan Berberis aristata. J Phytopharmacol. 2017;6(1):53–8.
Cameron J, Ranheim T, Kulseth MA, Leren TP, Berge KE. Berberine decreases PCSK9 expression in HepG2 cells. Atherosclerosis. 2008;201(2):266–73.
Dong H, Zhao Y, Zhao L, Lu F. The effects of berberine on blood lipids: a systemic review and meta-analysis of randomized controlled trials. Planta Med. 2013;79:437–46.
Kong W, Wei J, Abidi P, Lin M, Inaba S, Li C, et al. Berberine is a novel cholesterol-lowering drug working through a unique mechanism distinct from statins. Nat Med. 2004;10:1344–51.
Derosa G, Maffioli P, Cicero AFG. Berberine on metabolic and cardiovascular risk factors: an analysis from preclinical evidences to clinical trials. Expert Opin Biol Ther. 2012;12(8):1113–24.
Tabeshpour J, Imenshahidi M, Hosseinzadeh H. A review of the effects of Berberis vulgaris and its major component, berberine, in metabolic syndrome. Iran J Basic Med Sci. 2017;20:557–68.
Poli A, Barbagallo CM, Cicero AFG, Corsini A, Manzato E, Trimarco B, et al. Nutraceuticals and functional foods for the control of plasma cholesterol levels. An intersociety position paper. Pharmacol Res. 2018;134:51–60.
Poli A, Visioli F. Pharmacology of nutraceuticals with lipid lowering properties. High Blood Press Cardiovasc Prev. 2019;26(2):113–8.
Cicero AFG, Fogacci F, Colletti A. Food and plant bioactives for reducing cardiometabolic disease risk: an evidence based approach. Food Funct. 2017;8(6):2076–88.
Pisciotta L, Bellocchio A, Bertolini S. Nutraceutical pill containing berberine versus ezetimibe on plasma lipid pattern in hypercholesterolemic subjects and its additive effect in patients with familial hypercholesterolemia on stable cholesterol-lowering treatment. Lipids Health Dis. 2012;11:123.
Houston MC. The role of nutrition, nutraceuticals, vitamins, antioxidants, and minerals in the prevention and treatment of hypertension. Altern Ther Health Med. 2013;19(S1):32–49.
Ho MJ, Bellusci A, Wright JM. Blood pressure lowering efficacy of coenzyme Q10 for primary hypertension. Cochrane Database Syst Rev. 2009;(4):CD007435. https://doi.org/10.1002/14651858.cd007435.pub2.
Balk EM, Tatsioni A, Lichtenstein A, Lau J, Pittas AG. Effect of chromium supplementation on glucose metabolism and lipids. A systematic review of randomized controlled trials. Diabetes Care. 2007;30(8):2154–63.
Ostman E, Forslund A, Oste R, Bjorck I. A drink containing amino acids and chromium picolinate improves postprandial glycemia at breakfast in healthy, overweight subjects. Funct Foods Health Dis. 2017;7(2):88–97.
Acknowledgements
The authors thank Dr. Maria Carla Marrè Brunenghi for the technical assistance during the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of interest
The authors’ declares that they have no conflict of interest.
Ethical approval
The study protocol was approved by local Ethics Committee and institutional review boards and conducted in accordance with ICH Harmonized Tripartite Guidelines for Good Clinical Practice and the Declaration of Helsinki Principles.
Informed consent
All patients gave informed consent for the study participation.
Additional information
This article is part of the topical collection on Nutraceuticals in Hypertension & Cardiovascular Prevention.
Rights and permissions
About this article
Cite this article
Mazza, A., Schiavon, L., Rigatelli, G. et al. The Effects of a New Generation of Nutraceutical Compounds on Lipid Profile and Glycaemia in Subjects with Pre-hypertension. High Blood Press Cardiovasc Prev 26, 345–350 (2019). https://doi.org/10.1007/s40292-019-00332-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40292-019-00332-6