Abstract
Cardiovascular diseases (CVDs) are the main causes of mortality and disability worldwide. Although the initial therapeutic approach to improve the main cardiovascular (CV) risk factors is based on nonpharmacological measures, mainly lifestyle modifications, compliance to recommendations is often poor and inadequate. Therefore, in recent years the use of several nutraceuticals, i.e., nutrients and/or bioactive compounds of plant or microbial origin with potentially healthful effects, has become widespread. However, to date, the scientific data on the possible benefits of the use of nutraceuticals are still inconclusive, due to the absence of adequately controlled intervention studies at least for some of them. Against this background, the scientific evidence derived from controlled intervention studies in relation to the effects of some nutraceuticals (i.e. fiber, phytosterols, soy products, red yeast rice, polyphenols and berberine) on the main CV risk factors (body weight, blood pressure, blood glucose levels and plasma lipids) in humans will be reviewed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Cardiovascular diseases (CVDs), including coronary heart disease and stroke, are the main causes of mortality and disability both largely preventable through the control of risk factors via lifestyle modifications and preventive medication.
The initial therapeutic approach to the main risk factors for CVDs should always include nonpharmacological measures—such as lifestyle interventions, which are very similar for all CV risk factors [1]. In fact, current recommendations strongly encourage the reduction of total energy intake from foods and the increase in physical activity, especially in overweight/obese individuals. Moreover, saturated fat should be limited to less than 10% of calories, saturated and trans fatty acids should be replaced with polyunsaturated (PUFA) (up to 10% of calories) or monounsaturated (MUFA) fatty acids (10–15% of calories), and dietary fiber intake should be increased (30–45 g/day). Daily salt intake also should be reduced by at least one third and, if possible, to < 5 g per day. Moreover, dietary patterns that include a variety of fruits, vegetables, seeds and nuts (2–3 servings of each per day), pulses and whole grains are strongly encouraged, to promote health and lower CVD risk [2, 3].
However, adherence to lifestyle modification to prevent CVDs is often poor due to unsatisfactory patient compliance and poor adherence and maintenance in the medium- to long-term [4]. In addition, some dietary components with a potentially beneficial activity on CV risk factors are present only in scanty amounts in food. Therefore, the use of nutraceuticals has become widespread in recent years.
Nutraceuticals are nutrients and/or bioactive compounds of plant or microbial origin, with possible beneficial effects on human health when supplemented in adequate amounts (often above those present in foods). Nutraceuticals may be added to different foods and beverages (as in fortified foods), or taken in the form of dietary supplements (liquid preparations, tablets, capsules).
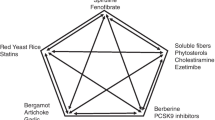
A growing number of nutraceuticals with variable activity on different CV risk factors have been proposed; unfortunately, scientific research has produced conflicting results for some of them. Hence, the aim of this report is to review and discuss the scientific evidence derived from controlled intervention studies regarding the effects of some nutraceuticals (i.e. fiber, phytosterols, soy products, red yeast rice, polyphenols and berberine) on the main CV risk factors (body weight, blood pressure, blood glucose levels and plasma lipids) in humans.
2 Effects on Body Weight
Overweight and obesity are currently recognized as the major risk factors for the development of a number of chronic diseases, including diabetes, dyslipidemia, CVDs and some types of cancer. Due to the dramatic increase in obesity prevalence, researchers have attempted to find effective complementary therapies for obesity and its complications, thus identifying in the supplementation of some nutraceuticals, mainly fibers and polyphenols, a potential tool and strategy to facilitate body weight management and influence obesity associated CV risk factors.
2.1 Fiber
The overall evidence from clinical intervention studies indicates soluble/viscous fibers as the most effective supplements for the management of body weight. Previous meta-analyses of twelve randomized controlled trials (RCTs) evaluated the effects of supplementation of different types of isolated soluble fiber (manno-oligosaccharides, galacto-oligosaccharides, fructo-oligosaccharides, β-glucan, flaxseed mucilage, mannans and dextrin) on outcomes related to weight management. Overall, the results showed that, independently of energy restriction or weight loss counseling, soluble fiber supplementation, compared to placebo, significantly reduced Body Mass Index (BMI) by 0.84 kg/m2, body weight by 2.52 kg and body fat by 0.41% in overweight and obese adults, at a mean dose of 18.5 g/day, over a follow-up of 2–17 weeks [5]. In line with this trend, findings from a three-arm, parallel, blind RCT on 83 overweight/obese individuals, showed that the supplementation of a commercial soluble viscous non-starch polysaccharide complex (PolyGlycoplex-PGX, as softgels or granules), containing konjac glucomannan, sodium alginate and xanthan gum, for 12 weeks, led to a significant reduction in waist circumference (− 2.5 cm) in the whole study population, as well as a to significant decrease in body weight (− 1.4 kg) in the subgroup of participants who consumed the recommended dose of fiber (about 12.2 g/day) [6].
Isolated glucomannan is also effective in the management of body weight [7, 8]. In fact, results from a glucomannan-focused meta-analysis of 14 RCTs showed a statistically significant, albeit small, reduction in body weight, of 0.79 kg in studies lasting a mean of 5.2 week [7]. However, the benefits observed with glucomannan supplementation on weight loss were not independent of the dietary modifications implemented in these RCTs. These findings have been confirmed by a meta-analyses of only 6 RCTs, in overweight/obese individuals, reporting a significant effect of glucomannan supplementation on body weight at different points during the intervention periods: at week 2 (− 0.21 kg), week 4 (− 2.04 kg) and week 5 (− 1.3 kg), at an average dose of glucomannan between 1.24 and 3.99 g/day [8].
The positive effect of glucomannan supplementation on the reduction of body weight was already confirmed in 2010 by the European Food Safety Authority (EFSA). The effect claimed is achieved when ≥ 3 g of glucomannan is supplemented daily in three 1-g doses, together with one to two glasses of water before the meal, in the context of an energy-restricted diet.
Other types of soluble fiber supplements, such as psyllium, dextrin and alginate, in combination with an energy restriction program, have proven capable to affect body weight and body composition and thus to be of some help in the management of obesity [9,10,11].
In conclusion, based on the available evidence in humans, it seems that soluble/viscous fiber supplements may be helpful in the management of body weight; it is nevertheless important to underline that the significant reduction in body weight observed in many studies is of small entity.
2.2 Polyphenols
Among polyphenols, flavonoids appear to be the most involved in obesity and weight management. In particular, epigallocatechin-3-gallate (EGCG), mainly abundant in green tea, represents the subclass of flavonoids best associated with weight loss.
Some years ago, Basu and colleagues [12], in a clinical trial in obese people, showed that an 8-week supplementation of encapsulated green tea extracts, containing 870 mg of catechins, resulted in a statistically significant reduction in body weight and BMI, by 1.9 kg and 0.6 kg/m2, respectively, compared to placebo. The results of two other randomized controlled trials performed with either encapsulated green tea extracts (800 mg catechins) for 8 weeks or decaffeinated green tea extracts (EGCG 856.8 mg) for 12 weeks confirms the results obtained by Basu [13, 14].
Conversely, other clinical studies have failed to show a beneficial effect of green tea catechin supplements on body weight and/or body composition. There may be different reasons for these discrepancies, one of which may be related to the possible confounding effect of caffeine. In fact, green tea extract naturally contains caffeine, a component known to influence per se body weight and body composition by enhancing sympathetic nervous system activity and regulating appetite [15]. Therefore, as a metabolic stimulant, the concentration of caffeine in green tea extract supplements should be carefully examined in relation to the independent effects of catechins on body weight. To this regard, a meta-analysis of 15 RCTs tried to characterize the relation between green tea catechins with and without caffeine, with changes in anthropometric parameters in overweight/obese people. This meta-analysis showed that the supplementation of catechins with caffeine, at a dose ranging from 583 to 714 mg/day over a median period of 12 week, decreased body weight, BMI and waist circumference by 1.38 kg, 0.55 kg/m2 and 1.93 cm, respectively, compared with a caffeine-matched control [16]. On the other hand, the few studies included in this meta-analysis on effects of catechins without concomitant caffeine administration did not show benefits on any of the anthropometric endpoints assessed.
In conclusion, results from RCTs evaluating the effect of tea catechin supplementation on outcomes related to obesity management are still discordant and inconclusive. Several confounding factors can influence the results of the available clinical trials, such as the concomitant presence of caffeine, but also the ethnicity of participants (Asians seems to be more responsive to caffeine) and the implementation or not of weight loss strategies.
2.3 Omega-3 Polyunsaturated Fatty Acids (n-3 PUFAs)
Based on studies performed in animals some years ago, it has been hypothesized that n-3 PUFA supplementation could influence body weight regulation to some extent, by increasing body fat oxidation or energy expenditure [17]. Instead, the evidence from RCTs in humans is limited and very inconsistent. Findings from a meta-analysis of 11 RCTs in overweight/obese adults, showed that n-3 PUFA supplementation, at a mean range dose of 1.5–6.0 g/day for 3 to 24 weeks, has no effects on body weight and/or BMI, compared to placebo. However, a subgroup analyses of only 7 studies included in this meta-analysis, revealed a significant reduction in waist circumference after n-3 PUFA supplementation compared to placebo [18]. In line with these findings, more recently Jacobo-Cejudo and colleagues [19], in a randomized, single-blind, placebo-controlled pilot study in type 2 diabetic patients, showed that the supplementation of 520 mg of docosahexaenoic acid (DHA) + eicosapentaenoic acid (EPA) fish-oil in the form of 2 soft gels per day for 24 weeks, resulted in a 3.3 cm reduction in waist circumference, compared to baseline values—but not vs. the placebo group—without affecting body weight, BMI and body fat. Furthermore, the supplementation of n-3 PUFA seems to be more effective in the reduction of body fat if associated with regular physical exercise and within the context of low calorie diets, even if the results are not univocal [20, 21].
In conclusion, the overall evidence from RCTs regarding the effect of n-3 PUFA supplementation on body weight reduction is very limited and, if anything, seems to suggest only a small effect on body fat reduction and distribution.
3 Effects on Blood Pressure
High blood pressure (BP) is one of the most relevant independent risk factors for CVDs and the most prevalent all over the world. The major international guidelines have stressed the preventive impact of dietary and life-style intervention in order to reach and maintain optimal BP levels [22, 23]. Given the difficulties in implementing dietary changes, nutraceuticals have been considered a useful option.
3.1 Fiber
The overall body of evidence on the effect of fiber supplements on BP is not very conclusive, at least for some types of fiber. A meta-analysis of 24 RCTs showed that fiber supplementation (11.5 g/day) significantly reduced BP, but did not analyze the effects according to type of fiber [24].
Instead, Evans and colleagues [25], performed a detailed analysis of RCTs focusing on the effects of fiber with different chemical structure, on BP in healthy populations. The authors found that diets rich in β-glucans, provided as oat bran, oat meals or oat beta-glucan-soluble powder—compared with wheat-based control products, reduced systolic blood pressure (SBP) by 2.9 mmHg (95% CI 0.9–4.9) and diastolic blood pressure (DBP) by 1.5 mmHg (95% CI 0.2–2.7), with a median difference in beta-glucans of 4 g, whereas little or no statistical evidence of impact on BP of the other types of dietary fiber was found.
More recently, another meta-analysis of RCTs evaluated the BP effects of different types of viscous fiber supplementation (β-glucan from oat and barley, guar gum, konjac, pectin and psyllium) in healthy and hypertensive individuals [26]. Overall, the results showed that viscous fiber reduced SBP (− 1.59 mmHg) and DBP (− 0.39 mmHg) at a median dose of 8.7 g/day (1.45–30 g/day) over a median follow-up of 7 weeks. Within the fiber types, SBP reductions were observed for psyllium fiber supplementation [− 2.39 mmHg (95% CI − 4.62, − 0.17)], while for beta-glucan, which also has highly viscous properties, only a trend to SBP and DBP reductions was observed. Moreover, hypertensive subjects seem to be more responsive to viscous fiber supplements.
The overall findings demonstrate a modest but significant reduction in SBP and DBP following viscous soluble fiber supplementation, in particular beta-glucans and psyllium.
3.2 Polyphenols
Different studies in humans support the hypothesis that some polyphenols are able to exert favorable effects on BP. A meta-analysis conducted by Zhang et al. [27] showed that grape seed extracts (100–2000 mg/day), rich in flavonoids, resveratrol and phenolic acids, supplemented for a mean period of 8 weeks can reduce SBP in obese subjects (WMD = − 4.469; 95% CI − 6.628, − 2.310; P < 0.001) and in patients with metabolic syndrome (WMD = − 8.487; 95% CI − 11.869, − 5.106; P < 0.001).
As for flavanol-rich cocoa products, two meta-analyses of RCTs revealed a significant reduction in SBP (− 4.5 or − 3.2 mmHg, respectively) and DBP (− 2.5 or − 2.0 mmHg, respectively) following cocoa flavanol intake ranging from 30 to 1000 mg/day in short-term trials of 2–18 week duration [28, 29]. Particularly, the meta-analysis from Ried et al. [29] indicated that the hypotensive effect is present in hypertensive and prehypertensive groups and not in normotensive groups.
A recent meta-analysis showed that in hypertensive patients soy isoflavone intake is associated with reduction in SBP by − 5.94 (95% CI − 10.55, − 1.34 mmHg, P = 0.01) and DBP by − 3.35 (95% CI − 6.52, − 0.19 mmHg, P = 0.04) [30]. As for healthy subjects, daily ingestion of 25–375 mg soy isoflavones (aglycone equivalents) for 2–24 weeks significantly decreased only SBP by 1.92 mmHg (95% CI − 3.45, − 0.39; P = 0.001) compared with placebo [31].
3.3 Omega-3 Polyunsaturated Fatty Acids (n-3 PUFAs)
There is a wealth of data on the BP-lowering effect of n-3 PUFAs. A recent meta-analysis of 70 RCTs showed that the consumption of omega-3 PUFAs (0.3–15 g/day) for 4–26 weeks reduced SBP by 4.5 mmHg (95% CI − 6.1, − 2.8) and DBP by 3.0 mmHg (95% CI − 4.3, − 1.7), in untreated hypertensive individuals [32]. The hypotensive effect is significant also in normotensive individuals, albeit to a more modest extent (SBP − 1.25 mmHg; DBP − 0.62 mmHg). These results are in line with those from two previous meta-analyses of RCTs showing that omega-3 PUFA supplementation (≥ 3 g/day) for over 2 weeks was associated with an improvement in SBP and DBP [33, 34], again more evident in hypertensive patients. On the other hand, type 2 diabetic patients are not responsive to omega-3 supplementation for what concerns blood pressure [35].
Finally, based on current knowledge, EFSA has established that an intake of about 3 g/day of EPA and DHA are required to obtain the claimed effect on blood pressure.
3.4 Red Yeast Rice, Berberine and Coenzyme Q10
There is very limited evidence in the literature on the effect of red yeast rice (RYR), berberine and coenzyme Q10 on BP. As for RYR, a meta-analysis of RCTs—most of which with low methodological quality—has shown that RYR + conventional therapy has no significant BP-lowering effect compared to placebo + conventional therapy in hypertensive subjects [36].
On the other hand, a meta-analysis of RCTs with berberine suggests that the addition of the latter to lifestyle intervention can be effective in reducing BP (SBP: MD = − 5.97 mmHg, 95% CI (− 9.19, − 2.74), P = 0.0003; DBP: MD = − 2.69 mmHg, 95% CI (− 5.06, − 0.31), P = 0.03) compared to lifestyle intervention alone or placebo [37]. The same positive effect on BP has been observed with berberine + hypotensive drug vs the same hypotensive drug (SBP: MD = − 4.91 mmHg, 95% CI (− 8.72, − 1.1), P = 0.01; DBP: MD = − 2 mmHg, 95% CI (− 3.76, − 0.24), P = 0.03) [37].
Data on the possible effect of coenzyme Q10 on BP are very discordant. In fact, while a previous meta-analysis concluded that coenzyme Q10 has the potential to lower SBP by up to 17 mmHg and DBP by up to 10 mmHg in hypertensive patients [38], a more recent one clearly showed no significant effect either on SBP or DBP when only controlled studies vs placebo (only two studies) were considered [39].
4 Effects on Blood Glucose Levels
Fasting and postprandial hyperglycemia is associated with increasing risk of CVDs and mortality among patients with abnormal glucose metabolism and in healthy population [40,41,42]. Therefore, treatment strategies to reduce blood glucose either at fasting or in the postprandial state become crucial to reduce the risk of CVDs.
A growing number of nutraceuticals with variable glucose lowering activity have been proposed as a quite novel approach to treat hyperglycemia, although the scientific research on their use has produced conflicting results and the reliability of a number of trials has been flawed by methodological limitations.
4.1 Fiber
Data from clinical trials seem to suggest that soluble fiber supplementation significantly reduces blood glucose levels, especially in the postprandial state, in subjects with pre-diabetes and in patients with diabetes. The glucose-lowering effect of this type of fiber ranges from 7 mg/dl (for glucomannan) to 45 mg/dl (for psyllium, in the postprandial state), with possible variations in relation to the doses used in the different trials [5, 43,44,45].
Although most intervention trials with fiber supplements are of short duration and performed on small groups of subjects, the results are quite consistent, especially for β-glucans and pectin. In fact, based on current knowledge, these types of fiber have been the object of a specific claim by EFSA for the maintenance of optimal postprandial glucose levels at a dosage of 4 g for each 30 g of available carbohydrates for β-glucans, and 10 g for meal for pectin.
4.2 Polyphenols
Epidemiological and clinical studies support the favorable effects of polyphenol-rich diets, characterized by a higher amount of polyphenols from cereals, fruit, vegetables, coffee, tea, and chocolate, in preventing diabetes and in improving blood glucose levels [46,47,48,49].
Against this background, in recent years, the possible glucose-lowering effect of different types of polyphenols added to the habitual diet is raising increasing interest. Two metanalysis of randomized controlled trials indicate that flavan-3-ols, present in green tea, green tea extracts, and chocolate, improve glucose metabolism (significant decrease in fasting blood glucose and glycosylated hemoglobin), and that this effect is caffeine-independent and more evident with higher doses of epigallocatechin (from 230 to 1200 mg/day) [50, 51]. Similar effects appear to be present for soy isoflavones. In fact, along the same line, a metanalysis of few intervention studies has shown that soy isoflavones—in particular genistein at 40–160 mg/day—significantly reduce fasting blood glucose and serum insulin although this effect is very limited from clinical point of view and present only in postmenopausal women [52].
A small number of intervention studies evaluated the supplementation of phenolic compounds derived from extra-virgin olive oil and olive leaves, i.e., oleuropein (at a mean dose of 51.1 mg/day) and hydroxytyrosol (at a mean dose of 9.7 mg/day); the results are quite promising, as they show a significant decrease in fasting blood glucose and glycosylate hemoglobin, but the evidence is still too scant for any conclusion to be drawn [53, 54].
Finally, some studies on resveratrol—one of the most studied phenolic compounds, largely present in grape peels—seem to support its hypoglycemic action, showing a decrease in glycemia, hemoglobin and insulin, but only in people with diabetes [55].
In conclusion, although the results of some intervention studies seem to suggest a possible hypoglycemic effect related in particular to some types of polyphenols used as supplements, they must be confirmed by longer-term trials on larger population before recommendations may be given. Actually, up to now no claims have been released for polyphenols regarding the modulation of glucose metabolism.
4.3 Berberine
Berberine is an isoquinoline plant alkaloid present in several plants, used since many years in China for its different therapeutic properties.
To date, studies in type 2 diabetic patients, and metanalysis of these studies show that a dose of berberine between 0.5 and 1.5 g/day for 8–12 weeks reduces fasting and postprandial blood glucose as well as HbA1c either when used alone or in combination with oral hypoglycemic agents [56]. Similar effects have been observed in subjects with metabolic syndrome, in whom berberine administration induced a 36% remission of the presence of metabolic syndrome, decreased waist circumference, systolic blood pressure, TG, glucose levels and insulin secretion, and increased insulin sensitivity [57].
While berberine seems to be effective in improving blood glucose levels, some limitations need to be considered: (1) the evidence comes mainly from studies carried out in Asians, at least for the use of berberine alone, and it is not transferable to other ethnic groups, (2) no data is as yet available on the long-term safety of its use.
In fact, so far, neither EFSA nor FDA has yet released specific claims on the efficacy and safety of berberine.
4.4 Combination of Nutraceuticals
Nutraceutical combinations have been used as therapeutic strategies, especially for patients whose lipid and glucose levels were marginally high, but not to the extent as to warrant the prescription of either lipid- or glucose-lowering medications. A combination of nutraceuticals containing red yeast rice, berberine, policosanol, astaxanthin, Coenzyme Q10 and folic acid has been reported to be effective in reducing glucose levels, although the magnitude of the reduction varied across trials, the difference partly ascribable to different durations and sample sizes of trials exploring lipid- and glucose-lowering effects of such combination [58, 59].
5 Effects on Plasma Lipids
Dyslipidemia, in particular high LDL-cholesterol (LDL-C) concentrations, are considered one of the main CV risk factors [60]. Lifestyle and different drugs are very effective in optimizing lipid profile. However, in some conditions and in some individuals, nutraceuticals as “pharmaceutical-grade nutrients” could represent a potentially useful tools to reduce LDL-C and optimize the overall plasma lipid profile in the general population as well as in people at high CV risk—in the latter cases in addition to the pharmacological therapy.
5.1 Fiber
Pirro et al. recently summarized the evidence from randomized controlled trials, analyzing the results of relevant meta-analyses [61]. The LDL-C-lowering effect of fiber ranges from − 6 mg/dl (β-glucan and chitosan) to − 22 mg/dl (guar gum) in both healthy subjects and in patients with either hypercholesterolemia or diabetes. The effect of fiber supplementation on triglycerides (TG) and HDL-cholesterol (HDL-C) is less clear, even if a reduction of TG concentrations (− 11 mg/dl) has been observed for chitosan and glucomannan [61].
Based on current evidence, some fibers obtained a claim by FDA (β-glucan and psyllium) and by EFSA (β-glucan, chitosan, glucomannan, guar gum, hydroxypropylmethylcellulose and pectin) for the maintenance of optimal LDL-C concentrations. EFSA has stated the effectual amount for the specific fibers as follow: β-glucan ≥ 3 g/day, chitosan 3 g/day, glucomannan 4 g/day, guar gum 10 g/day, hydroxypropylmethylcellulose 5 g/day and pectin 6 g/day [61].
5.2 Polyphenol
During the last decades, there has been substantial interest in the potential role of polyphenols in the modulation of lipid metabolism.
The main evidence comes from studies exploring the hypolipidemic effect of cocoa polyphenols (flavan-3-ols). Overall, the results of meta-analyses of randomized controlled trials show that cocoa polyphenols (from 166 to 2110 mg/day) significantly affect LDL-C, HDL-C and TG concentrations although with very limited effects from a clinical point of view [51, 62, 63]. As for tea polyphenols (flavan-3-ols), the current evidence is not conclusive due to the small number of high quality trials and the variability in dosage, exposure and study population [64,65,66,67,68]. However, the overall findings suggest that green tea may have greater effect than black tea, at least on LDL-C concentrations, due to the higher amount of catechins. As a matter of fact, the consumption of green tea beverages or extracts (from 145 to 3000 mg/day of catechins) significantly reduced LDL-C by 2.19 mg/dl [64, 65]. Conversely, similar amounts of black tea failed to show a hypocholesterolemic activity in any of the meta-analyses performed [66, 67]. None of the meta-analyses showed any effect on HDL-C [64,65,66,67,68] or TG concentrations [68].
Looking at other specific phenolic compounds, a recent meta-analyses on quercetin supplementation has shown to significantly reduce LDL-C and TG (− 5 mg/dl and − 7 mg/dl, respectively), and to increase HDL-C concentrations (2 mg/dl) even if these effects might not be clinically relevant [69]. However, a previous meta-analysis showed a meaningful reduction of triglycerides concentrations (− 24.54 mg/dl) when more than 500 mg/day of quercetin were supplemented. Greater effect on LDL-C were observed for anthocyanin supplementation in both healthy [70] and dyslipidemic subjects [71] (− 14 mg/dl and − 22 mg/dl, respectively). Moreover, anthocyanins reduced TG (26 mg/dl) and increased HDL-C (6 mg/dl) in dyslipidemic subjects [71], whereas no effect on these parameters was detected in healthy subjects [70]. For resveratrol, one of the phenolic compounds mostly studied, the current evidence does not support its hypolipidemic action (with resveratrol doses ranging from 8 mg/day to 1500 mg/day) [72, 73].
Finally, the results of a meta-analyses have shown that isoflavone extracts (on average 15 mg/day) seem to effectively reduce TG concentrations (− 41 mg/dl) in patients with hypercholesterolemia, whereas no variations in LDL-C and HDL-C were observed [74].
Due to the limited clinical evidence, no claims have so far been released for polyphenols for the modulation of lipid concentrations. However, it should be pointed out that hydroxytyrosol and its derivative (oleuropein and tyrosol) have received a claim for the protection of LDL particles from oxidative damage (effective dose 5 mg/day), likely suggesting that polyphenol may exert cardioprotective effects through mechanisms other than modulation of lipoprotein metabolism.
5.3 Omega-3 Polyunsaturated Fatty Acids (n-3 PUFAs)
There is growing evidence supporting the beneficial effects of omega 3 supplementation (2–3 g/day) on TG (− 20 mg/dl on average), particularly among people with more elevated baseline concentrations [75,76,77,78]. In some of these intervention studies, the decrease in TG was accompanied by a slight increase in LDL-C [79], while, on the whole, no effect on HDL-C has been consistently reported.
Based on current evidence, omega 3 supplementation (2–4 g/day) is recommended by the main international guidelines for the treatment of hypertriglyceridemia, and in addition to lifestyle changes and statin use in people with TG concentrations ranging from 200 to 500 mg/dl [80]. EFSA released a claim for 2 g/day of EPA and DHA to obtain the maintenance of normal fasting blood concentrations of triglycerides.
5.4 Phytosterols
Clinical studies and meta-analyses have demonstrated that phytosterols consumption (2–3 g/day) induces a mean 10%-reduction of LDL-C in healthy subjects and in patients with hypercholesterolemia and/or diabetes, with little or no effects on HDL-C and TG [61].
Due to the consistent evidence, FDA and EFSA endorsed a claim related to the use of phytosterols for LDL-C reduction. However, EFSA recommended not to exceed a dose of 3 g/day to prevent sitosterolemia (a rare autosomal recessive disease characterized by phytosterol accumulation), and suggests that patients receiving lipid-lowering medications should use phytosterols under medical supervision [61].
5.5 Red Yeast Rice
The hypolipidemic action of monacolin K, a natural analogue of lovastatin produced by the Monascus purpureus-fermentation of red rice, has been proven by meta-analyses of clinical trials carried out in healthy subjects and in patients with dyslipidemia, diabetes, coronary heart disease or hypertension [61]. The reduction of LDL-C was − 30 mg/dl on average for a mean monacolin k supplementation of 10 mg/day. With the same amount of monacolin K, an hypotriglyceridemic effect was observed (− 26 mg/dl on average), whereas no influence on HDL-C was detected.
The amount of 10 mg/dl of monacolin K from red yeast rice has received the claim for the maintenance of normal LDL-C concentrations from EFSA and FDA [61].
However, it must be taken into account that monacolin K should have the same restrictions as lovastatin, according to an FDA warning. Moreover, the Joint Commission of Experts of the Federal Office of Consumer Protection and Food Safety and the Federal Institute for Drugs and Medical Devices in Germany have stated that products providing 5 mg/day of monacolin K should be classified as drugs since they have a significant pharmacological action. In this regard, very recently, EFSA has concluded that “the available information on the adverse effects reported in humans was judged to be sufficient to conclude that monacolins from red yeast rice when used as food supplements were of significant safety concern at the use level of 10 mg/day” [81].
5.6 Berberine
To date, evidence of a lipid-lowering effect of berberine comes mainly from studies carried out in Asian populations and cannot be easily extrapolated to other ethnic groups. Nevertheless, the main findings considered in three meta-analyses [61] show that berberine (0.5–1.5 g/day) lowers LDL-C and TG concentrations (on average − 22 mg/dl and 37 mg/dl, respectively), while beneficially increases HDL-C (on average 2 mg/dl).
So far, neither EFSA nor FDA have released specific claims on the efficacy of berberine.
5.7 Soy Protein
Soy protein has been largely investigated, even if it is not easy to evaluate its hypolipidemic effect independently of the isoflavone content. Overall, the evidence suggested that soy protein may improve lipid metabolism, affecting particularly LDL-C, but the extent of these effects is not at all conclusive [61]. Furthermore, soy protein seems to act only in individuals with high baseline LDL-C concentrations for whom other pharmacological therapies are now more indicated.
No claims for soy protein and lipid concentrations have been authorized by EFSA, whereas the claim released by FDA in 1999 is under debate as a result of more recent evidence.
5.8 Coenzyme Q10
In recent years, the coenzyme Q10 (CoQ10) has attracted scientific interest for its possible role on lipid profile.
A meta-analysis of RCTs showed that the consumption of CoQ10 at low doses (< 200 mg/day) significantly reduced serum triglycerides levels by 0.17 mmol/l, without affecting other plasma lipids, in overweight and obese individuals with type 2 diabetes [82]. On the other hand, Sahebkar and colleagues, in a meta-analyses of seven RCTs, suggested a significant lowering effect of CoQ10 supplementation on plasma Lipoprotein-a (Lp(a)) levels, particularly in patients with baseline Lp(a) values ≥ 30 mg/dl. No change in the plasma concentrations of total cholesterol, LDL-cholesterol, HDL-cholesterol and triglycerides was reported in this meta-analysis [83]. Findings from a more recent meta-analysis including eight RCTs showed that taking CoQ10 significantly decreased total cholesterol and increased HDL-cholesterol levels in patients with coronary artery disease (CAD), without any effect on LDL-cholesterol, triglycerides and Lp(a) levels [84].
In conclusion, due to the limited clinical evidence, no conclusive indication can be given about the possible efficacy of coenzyme Q10 supplementation on plasma lipids.
5.9 Combination of Nutraceuticals
The available evidence on the effect of fiber and phytosterol combination has shown a slightly higher cholesterol-lowering action compared to the single components. Conversely, the combination of phytosterols and monacolin K did not provide any additional effect compared to the individual nutraceuticals [61].
A recent meta-analyses focusing on the effect of a nutraceutical comprising monacolin K (3 mg), berberine (500 mg), policosanols (10 mg), astaxanthin (0.5 mg), coenzyme Q10 (2 mg), and folic acid (200 μg) has shown that this combination reduced LDL-C (24 mg/dl) and TG (14 mg/dl), and increase HDL-C concentrations (2 mg/dl) [59].
6 Conclusions
The scientific evidence indicates a possible role of some nutraceuticals in the development of different CV risk factors (Table 1). This is also supported by evidence of the potential beneficial effects of single nutraceuticals and few red yeast rice based combinations of nutraceuticals on surrogate markers of CV risk, as vascular function [85].
Fiber, mainly the soluble/viscous type, seems to have beneficial effects on different CV risk factors even if the entity of these effects is not very high. Furthermore, LDL-cholesterol levels are beneficially influenced by phytosterol, red yeast rice, and berberine, which seems to act positively also on blood glucose levels, plasma triglycerides, HDL-cholesterol and blood pressure.
The effects of some types of polyphenols on different CV risk factors seem quite suggestive but there is urgent need for very well controlled intervention studies on large populations and for long periods of time, to be sure also of the safety aspect. Indeed, this is true for all nutraceuticals and applies also to the use of nutraceutical combinations. The clinical use of the combination of different nutraceuticals has become widespread, with the aim to act simultaneously on different CV risk factors and on the same risk factor through different mechanisms. However, also for this specific aspect we need to have more scientific evidence deriving from well-controlled intervention studies, as required for drugs.
While awaiting such evidence, we can say that some nutraceuticals may be useful in particular circumstances and in well-identified individuals, i.e. those at low-moderate CV risk who do not yet need drugs, or in addition to drug therapy—in line with what suggested by scientific societies. In any case, nutraceuticals should be taken under medical/nutritionist supervision. This last point is crucial since nutraceuticals, to be effective, should be taken regularly and at the recommended dosage.
References
Prabhakaran D, Anand S, Watkins D, Gaziano T, Wu Y, Mbanya JC, et al. Cardiovascular, respiratory, and related disorders: key messages from Disease Control Priorities, 3rd edition. Lancet. 2018;391(10126):1224–36.
Reynolds A, Mann J, Cummings J, Winter N, Mete E, Te Morenga L. Carbohydrate quality and human health: a series of systematic reviews and meta-analyses. Lancet. 2019;393(10170):434–45.
Piepoli MF, Hoes AW, Agewall S, Albus C, Brotons C, Catapano AL, et al. 2016 European Guidelines on cardiovascular disease prevention in clinical practice. The Sixth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of 10 societies and by invited experts). Eur Heart J. 2016;37:2315–81.
Chapman K. Can people make healthy changes to their diet and maintain them in the long term? A review of the evidence. Appetite. 2010;54(3):433–41.
Thompson SV, Hannon BA, An R, Holscher HD. Effects of isolated soluble fiber supplementation on body weight, glycemia, and insulinemia in adults with overweight and obesity: a systematic review and meta-analysis of randomized controlled trials. Am J Clin Nutr. 2017;106(6):1514–28.
Solah VA, Kerr DA, Hunt WJ, Johnson SK, Boushey CJ, Delp EJ, et al. Effect of fibre supplementation on body weight and composition, frequency of eating and dietary choice in overweight individuals. Nutrients. 2017;9(2):149.
Sood N, Baker WL, Coleman CI. Effect of glucomannan on plasma lipid and glucose concentrations, body weight, and blood pressure: systematic review and meta-analysis. Am J Clin Nutr. 2008;88(4):1167–75.
Zalewski BM, Chmielewska A, Szajewska H, Keithley JK, Li P, Goldsby TU, et al. Correction of data errors and reanalysis of “The effect of glucomannan on body weight in overweight or obese children and adults: a systematic review of randomized controlled trials”. Nutrition. 2015;31(7–8):1056–7.
Abutair AS, Naser IA, Hamed AT. Soluble fibers from psyllium improve glycemic response and body weight among diabetes type 2 patients (randomized control trial). Nutr J. 2016;15(1):86.
Jane M, McKay J, Pal S. Effects of daily consumption of psyllium, oat bran and polyGlycopleX on obesity-related disease risk factors: a critical review. Nutrition. 2018;57:84–91.
Georg Jensen M, Kristensen M, Astrup A. Effect of alginate supplementation on weight loss in obese subjects completing a 12-wk energy-restricted diet: a randomized controlled trial. Am J Clin Nutr. 2012;96(1):5–13.
Basu A, Sanchez K, Leyva MJ, Wu M, Betts NM, Aston CE, et al. Green tea supplementation affects body weight, lipids, and lipid peroxidation in obese subjects with metabolic syndrome. J Am Coll Nutr. 2010;29(1):31–40.
Brown AL, Lane J, Holyoak C, Nicol B, Mayes AE, Dadd T. Health effects of green tea catechins in overweight and obese men: a randomised controlled cross-over trial. Br J Nutr. 2011;106(12):1880–9.
Chen IJ, Liu CY, Chiu JP, Hsu CH. Therapeutic effect of high-dose green tea extract on weight reduction: a randomized, double-blind, placebo-controlled clinical trial. Clin Nutr. 2016;35(3):592–9.
Belza A, Frandsen E, Kondrup J. Body fat loss achieved by stimulation of thermogenesis by a combination of bioactive food ingredients: a placebo-controlled, double-blind 8-week intervention in obese subjects. Int J Obes (Lond). 2007;31(1):121–30.
Phung OJ, Baker WL, Matthews LJ, Lanosa M, Thorne A, Coleman CI. Effect of green tea catechins with or without caffeine on anthropometric measures: a systematic review and meta-analysis. Am J Clin Nutr. 2010;91(1):73–81.
Flachs P, Horakova O, Brauner P, Rossmeisl M, Pecina P, Franssen-van Hal N, et al. Polyunsaturated fatty acids of marine origin upregulate mitochondrial biogenesis and induce beta-oxidation in white fat. Diabetologia. 2005;48(11):2365–75.
Zhang YY, Liu W, Zhao TY, Tian HM. Efficacy of omega-3 polyunsaturated fatty acids supplementation in managing overweight and obesity: a meta-analysis of randomized clinical trials. J Nutr Health Aging. 2017;21(2):187–92.
Jacobo-Cejudo MG, Valdés-Ramos R, Guadarrama-López AL, Pardo-Morales RV, Martínez-Carrillo BE, Harbige LS. Effect of n-3 polyunsaturated fatty acid supplementation on metabolic and inflammatory biomarkers in type 2 diabetes mellitus patients. Nutrients. 2017;9(6):E573.
Hill AM, Buckley JD, Murphy KJ, Howe PR. Combining fish-oil supplements with regular aerobic exercise improves body composition and cardiovascular disease risk factors. Am J Clin Nutr. 2007;85(5):1267–74.
Munro IA, Garg ML. Dietary supplementation with n-3 PUFA does not promote weight loss when combined with a very-low-energy diet. Br J Nutr. 2012;108(8):1466–74.
Williams B, Mancia G, Spiering W, Agabiti Rosei E, Azizi M, Burnier M, et al. 2018 Practice Guidelines for the management of arterial hypertension of the European Society of Hypertension and the European Society of Cardiology: ESH/ESC Task Force for the Management of Arterial Hypertension. J Hypertens. 2018;36(12):2284–309.
Appel LJ, Giles TD, Black HR, Izzo JL, Materson BJ, Oparil S, et al. ASH position paper: dietary approaches to lower blood pressure. J Clin Hypertens (Greenwich). 2009;11(7):358–68.
Streppel MT, Arends LR, van ‘t Veer P, Grobbee DE, Geleijnse JM. Dietary fiber and blood pressure: a meta-analysis of randomized placebo-controlled trials. Arch Intern Med. 2005;165(2):150–6.
Evans CE, Greenwood DC, Threapleton DE, Cleghorn CL, Nykjaer C, Woodhead CE, et al. Effects of dietary fibre type on blood pressure: a systematic review and meta-analysis of randomized controlled trials of healthy individuals. J Hypertens. 2015;33(5):897–911.
Khan K, Jovanovski E, Ho HVT, Marques ACR, Zurbau A, Mejia SB, et al. The effect of viscous soluble fiber on blood pressure: a systematic review and meta-analysis of randomized controlled trials. Nutr Metab Cardiovasc Dis. 2018;28(1):3–13.
Zhang H, Liu S, Li L, Mi J, Tian G. The impact of grape seed extract treatment on blood pressure changes: a meta-analysis of 16 randomized controlled trials. Medicine (Baltimore). 2016;95(33):e4247.
Desch S, Schmidt J, Kobler D, Sonnabend M, Eitel I, Sareban M, et al. Effect of cocoa products on blood pressure: systematic review and meta-analysis. Am J Hypertens. 2010;23(1):97–103.
Ried K, Sullivan T, Fakler P, Frank OR, Stocks NP. Does chocolate reduce blood pressure? A meta-analysis. BMC Med. 2010;8:39.
Liu XX, Li SH, Chen JZ, Sun K, Wang XJ, Wang XG, et al. Effect of soy isoflavones on blood pressure: a meta-analysis of randomized controlled trials. Nutr Metab Cardiovasc Dis. 2012;22(6):463–70.
Taku K, Lin N, Cai D, Hu J, Zhao X, Zhang Y, et al. Effects of soy isoflavone extract supplements on blood pressure in adult humans: systematic review and meta-analysis of randomized placebo-controlled trials. J Hypertens. 2010;28(10):1971–82.
Miller PE, Van Elswyk M, Alexander DD. Long-chain omega-3 fatty acids eicosapentaenoic acid and docosahexaenoic acid and blood pressure: a meta-analysis of randomized controlled trials. Am J Hypertens. 2014;27(7):885–96.
Appel LJ, Miller ER, Seidler AJ, Whelton PK. Does supplementation of diet with ‘fish oil’ reduce blood pressure? A meta-analysis of controlled clinical trials. Arch Intern Med. 1993;153(12):1429–38.
Geleijnse JM, Giltay EJ, Grobbee DE, Donders AR, Kok FJ. Blood pressure response to fish oil supplementation: metaregression analysis of randomized trials. J Hypertens. 2002;20(8):1493–9.
Hartweg J, Farmer AJ, Holman RR, Neil A. Potential impact of omega-3 treatment on cardiovascular disease in type 2 diabetes. Curr Opin Lipidol. 2009;20(1):30–8.
Xiong X, Wang P, Li X, Zhang Y, Li S. The effects of red yeast rice dietary supplement on blood pressure, lipid profile, and C-reactive protein in hypertension: a systematic review. Crit Rev Food Sci Nutr. 2017;57(9):1831–51.
Lan J, Zhao Y, Dong F, Yan Z, Zheng W, Fan J, et al. Meta-analysis of the effect and safety of berberine in the treatment of type 2 diabetes mellitus, hyperlipemia and hypertension. J Ethnopharmacol. 2015;161:69–81.
Rosenfeldt FL, Haas SJ, Krum H, Hadj A, Ng K, Leong JY, et al. Coenzyme Q10 in the treatment of hypertension: a meta-analysis of the clinical trials. J Hum Hypertens. 2007;21(4):297–306.
Ho MJ, Li EC, Wright JM. Blood pressure lowering efficacy of coenzyme Q10 for primary hypertension. Cochrane Database Syst Rev. 2016;3:CD007435.
Coutinho M, Gerstein HC, Wang Y, Yusuf S. The relationship between glucose and incident cardiovascular events. A metaregression analysis of published data from 20 studies of 95,783 individuals followed for 12.4 years. Diabetes Care. 1999;22(2):233–40.
Stamler J, Vaccaro O, Neaton JD, Wentworth D. Diabetes, other risk factors, and 12-yr cardiovascular mortality for men screened in the Multiple Risk Factor Intervention Trial. Diabetes Care. 1993;16(2):434–44.
Lawes CM, Parag V, Bennett DA, Suh I, Lam TH, Whitlock G, et al. Blood glucose and risk of cardiovascular disease in the Asia Pacific region. Diabetes Care. 2004;27(12):2836–42.
Abutair AS, Naser IA, Hamed AT. The effect of soluble fiber supplementation on metabolic syndrome profile among newly diagnosed type 2 diabetes patients. Clin Nutr Res. 2018;7(1):31–9.
Gibb RD, McRorie JW, Russell DA, Hasselblad V, D’Alessio DA. Psyllium fiber improves glycemic control proportional to loss of glycemic control: a meta-analysis of data in euglycemic subjects, patients at risk of type 2 diabetes mellitus, and patients being treated for type 2 diabetes mellitus. Am J Clin Nutr. 2015;102(6):1604–14.
de Carvalho CM, de Paula TP, Viana LV, Machado VM, de Almeida JC, Azevedo MJ. Plasma glucose and insulin responses after consumption of breakfasts with different sources of soluble fiber in type 2 diabetes patients: a randomized crossover clinical trial. Am J Clin Nutr. 2017;106(5):1238–45.
Zhang YB, Chen WH, Guo JJ, Fu ZH, Yi C, Zhang M, et al. Soy isoflavone supplementation could reduce body weight and improve glucose metabolism in non-Asian postmenopausal women—a meta-analysis. Nutrition. 2013;29(1):8–14.
Bozzetto L, Annuzzi G, Pacini G, Costabile G, Vetrani C, Vitale M, et al. Polyphenol-rich diets improve glucose metabolism in people at high cardiometabolic risk: a controlled randomised intervention trial. Diabetologia. 2015;58(7):1551–60.
Annuzzi G, Bozzetto L, Costabile G, Giacco R, Mangione A, Anniballi G, et al. Diets naturally rich in polyphenols improve fasting and postprandial dyslipidemia and reduce oxidative stress: a randomized controlled trial. Am J Clin Nutr. 2014;99(3):463–71.
Vitale M, Vaccaro O, Masulli M, Bonora E, Del Prato S, Giorda CB, et al. Polyphenol intake and cardiovascular risk factors in a population with type 2 diabetes: the TOSCA.IT study. Clin Nutr. 2017;36(6):1686–92.
Zheng XX, Xu YL, Li SH, Hui R, Wu YJ, Huang XH. Effects of green tea catechins with or without caffeine on glycemic control in adults: a meta-analysis of randomized controlled trials. Am J Clin Nutr. 2013;97(4):750–62.
Hooper L, Kay C, Abdelhamid A, Kroon PA, Cohn JS, Rimm EB, et al. Effects of chocolate, cocoa, and flavan-3-ols on cardiovascular health: a systematic review and meta-analysis of randomized trials. Am J Clin Nutr. 2012;95(3):740–51.
Fang K, Dong H, Wang D, Gong J, Huang W, Lu F. Soy isoflavones and glucose metabolism in menopausal women: a systematic review and meta-analysis of randomized controlled trials. Mol Nutr Food Res. 2016;60(7):1602–14.
de Bock M, Derraik JG, Brennan CM, Biggs JB, Morgan PE, Hodgkinson SC, et al. Olive (Olea europaea L.) leaf polyphenols improve insulin sensitivity in middle-aged overweight men: a randomized, placebo-controlled, crossover trial. PLoS One. 2013;8(3):e57622.
Wainstein J, Ganz T, Boaz M, Bar Dayan Y, Dolev E, Kerem Z, et al. Olive leaf extract as a hypoglycemic agent in both human diabetic subjects and in rats. J Med Food. 2012;15(7):605–10.
Liu K, Zhou R, Wang B, Mi MT. Effect of resveratrol on glucose control and insulin sensitivity: a meta-analysis of 11 randomized controlled trials. Am J Clin Nutr. 2014;99(6):1510–9.
Pirillo A, Catapano AL. Berberine, a plant alkaloid with lipid- and glucose-lowering properties: from in vitro evidence to clinical studies. Atherosclerosis. 2015;243(2):449–61.
Pérez-Rubio KG, González-Ortiz M, Martínez-Abundis E, Robles-Cervantes JA, Espinel-Bermúdez MC. Effect of berberine administration on metabolic syndrome, insulin sensitivity, and insulin secretion. Metab Syndr Relat Disord. 2013;11(5):366–9.
Trimarco V, Battistoni A, Tocci G, Coluccia R, Manzi MV, Izzo R, et al. Single blind, multicentre, randomized, controlled trial testing the effects of a novel nutraceutical compound on plasma lipid and cardiovascular risk factors: results of the interim analysis. Nutr Metab Cardiovasc Dis. 2017;27(10):850–7.
Pirro M, Mannarino MR, Bianconi V, Simental-Mendía LE, Bagaglia F, Mannarino E, et al. The effects of a nutraceutical combination on plasma lipids and glucose: a systematic review and meta-analysis of randomized controlled trials. Pharmacol Res. 2016;08(110):76–88.
Jain KS, Kathiravan MK, Somani RS, Shishoo CJ. The biology and chemistry of hyperlipidemia. Bioorg Med Chem. 2007;15(14):4674–99.
Pirro M, Vetrani C, Bianchi C, Mannarino MR, Bernini F, Rivellese AA. Joint position statement on “Nutraceuticals for the treatment of hypercholesterolemia” of the Italian Society of Diabetology (SID) and of the Italian Society for the Study of Arteriosclerosis (SISA). Nutr Metab Cardiovasc Dis. 2017;27(1):2–17.
Tokede OA, Gaziano JM, Djoussé L. Effects of cocoa products/dark chocolate on serum lipids: a meta-analysis. Eur J Clin Nutr. 2011;65(8):879–86.
Lin X, Zhang I, Li A, Manson JE, Sesso HD, Wang L, et al. Cocoa flavanol intake and biomarkers for cardiometabolic health: a systematic review and meta-analysis of randomized controlled trials. J Nutr. 2016;146(11):2325–33.
Zheng XX, Xu YL, Li SH, Liu XX, Hui R, Huang XH. Green tea intake lowers fasting serum total and LDL cholesterol in adults: a meta-analysis of 14 randomized controlled trials. Am J Clin Nutr. 2011;94(2):601–10.
Kim A, Chiu A, Barone MK, Avino D, Wang F, Coleman CI, et al. Green tea catechins decrease total and low-density lipoprotein cholesterol: a systematic review and meta-analysis. J Am Diet Assoc. 2011;111(11):1720–9.
Wang D, Chen C, Wang Y, Liu J, Lin R. Effect of black tea consumption on blood cholesterol: a meta-analysis of 15 randomized controlled trials. PLoS One. 2014;9(9):e107711.
Zhao Y, Asimi S, Wu K, Zheng J, Li D. Black tea consumption and serum cholesterol concentration: systematic review and meta-analysis of randomized controlled trials. Clin Nutr. 2015;34(4):612–9.
Hartley L, Flowers N, Holmes J, Clarke A, Stranges S, Hooper L, et al. Green and black tea for the primary prevention of cardiovascular disease. Cochrane Database Syst Rev. 2013;6:CD009934.
Menezes R, Rodriguez-Mateos A, Kaltsatou A, González-Sarrías A, Greyling A, Giannaki C, et al. Impact of flavonols on cardiometabolic biomarkers: a metaanalysis of randomized controlled human trials to explore the role of inter-individual variability. Nutrients. 2017;9(2):E117.
Yang L, Ling W, Du Z, Chen Y, Li D, Deng S, et al. Effects of anthocyanins on cardiometabolic health: a systematic review and meta-analysis of randomized controlled trials. Adv Nutr. 2017;8(5):684–93.
Liu C, Sun J, Lu Y, Bo Y. Effects of anthocyanin on serum lipids in dyslipidemia patients: a systematic review and meta-analysis. PLoS One. 2016;11(9):e0162089.
Sahebkar A, Serban C, Ursoniu S, Wong ND, Muntner P, Graham IM, et al. Lack of efficacy of resveratrol on C-reactive protein and selected cardiovascular risk factors—results from a systematic review and meta-analysis of randomized controlled trials. Int J Cardiol. 2015;189:47–55.
Sahebkar A. Effects of resveratrol supplementation on plasma lipids: a systematic review and meta-analysis of randomized controlled trials. Nutr Rev. 2013;71(12):822–35.
Qin Y, Niu K, Zeng Y, Liu P, Yi L, Zhang T, et al. Isoflavones for hypercholesterolaemia in adults. Cochrane Database Syst Rev. 2013;6:CD009518.
Bernstein AM, Ding EL, Willett WC, Rimm EB. A meta-analysis shows that docosahexaenoic acid from algal oil reduces serum triglycerides and increases HDL-cholesterol and LDL-cholesterol in persons without coronary heart disease. J Nutr. 2012;142(1):99–104.
Chen C, Yu X, Shao S. Effects of omega-3 fatty acid supplementation on glucose control and lipid levels in type 2 diabetes: a meta-analysis. PLoS One. 2015;10(10):e0139565.
Abdelhamid AS, Brown TJ, Brainard JS, Biswas P, Thorpe GC, Moore HJ, et al. Omega-3 fatty acids for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst Rev. 2018;7:CD003177.
Yan JH, Guan BJ, Gao HY, Peng XE. Omega-3 polyunsaturated fatty acid supplementation and non-alcoholic fatty liver disease: a meta-analysis of randomized controlled trials. Medicine (Baltimore). 2018;97(37):e12271.
Rivellese AA, Maffettone A, Vessby B, Uusitupa M, Hermansen K, Berglund L, et al. Effects of dietary saturated, monounsaturated and n-3 fatty acids on fasting lipoproteins, LDL size and post-prandial lipid metabolism in healthy subjects. Atherosclerosis. 2003;167(1):149–58.
Karalis DG. A review of clinical practice guidelines for the management of hypertriglyceridemia: a focus on high dose omega-3 fatty acids. Adv Ther. 2017;34(2):300–23.
EFSA Panel on Food Additives and Nutrient Sources added to Food (ANS), Younes M, Aggett P, Aguilar F, Crebelli R, Dusemund B, Filipi M, et al. Scientific opinion on the safety of monacolins in red yeast rice. EFSA J. 2018;16(8):5368.
Huang H, Chi H, Liao D, Zou Y. Effects of coenzyme Q10 on cardiovascular and netabolic biomarkers in overweight and obese patients with type 2 diabetes mellitus: a pooled analysis. Diabetes Metab Syndr Obes Targets Ther. 2018;11:875–86.
Sahebkar A, Simental-Mendia LE, Stefanutti C, Pirro M. Supplementation with coenzyme Q10 reduces plasma lipoprotein(a) concentrations but not other lipid indices: a systematic review and meta-analysis. Pharmacol Res. 2016;105:198–209.
Jorat MV, Tabrizi R, Mirhosseini N, Lankarani KB, Akbari M, Heydari ST, et al. The effects of coenzyme q10 supplementation on lipid profiles among patients with coronary artery disease: a systematic review and meta-analysis of randomized controlled trials. Lipids Helath Dis. 2018;17:230.
Bianconi V, Mannarino MR, Sahebkar A, Cosentino T, Pirro M. Cholesterol-lowering nutraceuticals affecting vascular function and cardiovascular disease risk. Curr Cardiol Rep. 2018;20:53.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Angela Albarosa Rivellese declares that she has no conflict of interest; Paola Ciciola declares that she has no conflict of interest; Giuseppina Costabile declares that she has no conflict of interest; Claudia Vetrani declares that she has no conflict of interest; Marilena Vitale declares that she has no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the studies.
Additional information
This article is part of the topical collection on Nutraceuticals in Hypertension & Cardiovascular Prevention.
Rights and permissions
About this article
Cite this article
Rivellese, A.A., Ciciola, P., Costabile, G. et al. The Possible Role of Nutraceuticals in the Prevention of Cardiovascular Disease. High Blood Press Cardiovasc Prev 26, 101–111 (2019). https://doi.org/10.1007/s40292-019-00309-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40292-019-00309-5