Abstract
Plant growth promoting rhizomicroorganisms (PGPR) play an important role in improving plant growth, nutrition and yield of different crops. Chilly is one of the major commercial crops of India with large export potential. The first experiment was conducted with 10 PGPR to investigate their effects on growth and yield of chilly. Although most of the plant parameters studied were statistically not significant, 2 PGPR viz. Paenibacillus polymyxa and Pantoea dispersa showed agronomic improvement in plant growth as compared to control and other treatments. The second experiment was conducted with these 2 PGPR plus 8 more PGPR in order to select the best PGPR for inoculating chilly. Inoculation significantly improved the growth, nutrition and fruit yield as compared to uninoculated control. Considering plant dry biomass and fruit yield, Methylobacterium radiotolerans proved to be the best PGPR. Further screening with M. radiotolerans plus 2 more PGPR viz. Bacillus sonorensis and Paenibacillus elgii on 2 common varieties of chilly resulted in enhanced plant dry biomass, nutrition and fruit yield. The results clearly brought out that B. sonorensis is the most promising PGPR inoculant for chilly. The plant growth promoting traits revealed that B. sonorensis is a P-solubilizer and able to produce indole acetic acid, siderophore, chitinase, hydrogen cyanide and good in biofilm formation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Over the last few decades, the high increase in crop yield was achieved through high input of inorganic fertilizers and pesticides. However, currently emphasis is on sustainable agriculture, which uses less of chemical inputs like fertilizers/pesticides having adverse effect on soil and environment. Microbial inoculants can fulfill diverse beneficial interactions in plants leading to promising solutions for sustainable and environment-friendly agriculture. It is well known that PGPR play an important role in maintaining crop and soil health through nutrient cycling and uptake, suppression of plant pathogens, induction of resistance in plant host and direct stimulation of plant growth [1, 2]. Application of PGPR as biofertilizer reduces the cost of crop production. A large number of bacteria like Azospirillum, Azotobacter, Bacillus, Enterobacter, Pseudomonas, Klebsiella and Paenibacillus have been isolated from rhizosphere of various crops and used as PGPR [3, 4]. PGPR can be the best alternative to chemical fertilizers for sustainable and eco-friendly agriculture.
Chilly (Capsicum annuum L.) belonging to the family Solanaceae is one of the major commercial crop of India with large export potential. India is the largest producer of chilly in the world producing about 1.2 million tons every year [5]. However the production of chilly in India is considerably low when compared to the large area (930,000 hectares) of its production [6]. The application of chemical fertilizers and pesticides is increasing every year to attain the maximum production of chilly. The use of chemical fertilizers in India has increased up to 170 times in the last 50 years [7]. Different varieties are grown for vegetables, spices, condiments, sauces, pickles, etc. Chilly powder is the most important ground spice item exported from India. Since, chilly being a major spice with tremendous export potential, the emphasis needs to be given for sustainable cultivation of chilly. The main objective of the present study was to screen and select the efficient PGPR which can be used as an inoculant to improve growth, nutrition and yield of chilly.
Material and Methods
The investigations were carried out under pot culture conditions. Three different sets of pot culture experiments were conducted to screen and select the efficient PGPR for inoculating chilly. In the first pot culture trial 10 PGPR viz. Paenibacillus polymyxa, Pantoea dispersa, Pantoea agglomerans, Pseudomonas sp., Bacillus sp., Bacillus pumilus, Pseudomonos putida, Trichoderma viride, Bacillus sp. EB 02 and Bacillus sp. EB 04 were screened. In the second experiment 2 promising PGPR selected from the first trial (Paenibacillus polymyxa and Pantoea dispersa) plus 8 new PGPR (Paenibacillus sp., Pantoea agglomerans, Methylobacterium radiotolerans, Exiguobacterium acetylicum, Bacillus subtilis, Pseudomonas fluorescens, Azospirillum brasilense and Azotobacter chroococcum) were screened, and in the third experiment the best PGPR from the second experiment (Methylobacterium radiotolerans) plus 2 newer PGPR (Bacillus sonorensis and Paenibacillus elgii) were used. The PGPR used in the first and second experiments were obtained from culture collection centers and from scientists working with PGPR. The 2 new PGPR used in the third experiment were isolated by the last two authors. Bacillus sonorensis was isolated from the tomato rhizosphere and Paenibacillus elgii was isolated from chitin enriched soil [8].
The soil used in the present study was collected from an uncultivated field from a depth of 0–15 cm and has been classified as a fine, kaolinitic isohyperthermic kanhaplustulf. In the first and second experiments, variety NS-1701 was used only. In the third experiment, 2 varieties of chilly seeds commonly cultivated in this region viz, NS-1701 and Ujala F1 hybrid were used. Chilly seeds were sown in the pro-trays containing coco-peat enriched with vermicompost as substrate. After 30 days, uniform size seedlings were transplanted to polythene bags (21 × 14 cm) filled with soil, sand and vermicompost mix in the ratio 1:1:0.25 (v/v/v basis). The PGPR used in the present study were grown in liquid media and 10 ml of inoculum with 108 cfu/ml culture was added to the respective pot according to the treatment. Ten replications were maintained for each treatment and plants were watered when necessary.
Plant Parameters Studied
The height of the plant and stem girth were recorded at 60 days after transplanting (DAP). Biovolume index (BI) was calculated using the formula given below [9].
Chilly fruits were harvested 70 DAP and fresh weight of the fruits was taken for each plant separately. The shoot of the plants were cut and removed from soil surface and then roots of the plants were collected from soil, washed thoroughly and kept for drying at 60 °C in an oven. The dry weight of shoot and root was determined after drying to a constant weight. Dried shoot and root samples were then powdered using a blender. The major nutrients (N, P and K) in powdered plant samples were analyzed using the methods outlined by Jackson [10]. The data was analyzed using one way analysis of variance. The means were compared by Duncan’s multiple range test at 5% level [11].
PGP Traits of Bacillus Sonorensis
Bacillus sonorensis was found to be the best in increasing the growth, nutrition and yield of chilly and hence it was characterized for its PGPR traits. Phosphate solubilization was checked on Pikovskaya’s and National Botanical Research Institute’s phosphate (NBRIP) medium agar plates. Overnight culture was spotted on both plates, incubated at 30 °C for 72 h and checked for zone of solubilization [12]. Indole acetic acid production was detected as described by Brick et al. [13]. Siderophore production was checked on Chrome Azurol S agar plates as described by Schwyn and Neilands [14]. Chitinase assay was performed as described by Das et al. [15]. Qualitative hydrogen cyanide (HCN) production was checked using nutrient agar supplemented with 0.44% of glycine [16]. Biofilm formation by Bacillus sonorensis during growth was examined in borosilicate glass tubes, as described by Yousef et al. [17] and O’Toole and Kolter [18]. The ability of the bacterium to utilize 1-aminocyclopropane-1-carboxylate (ACC) as a sole source of nitrogen was assayed as described by Li et al. [19].
Results and Discussion
Under pot culture studies, chilly plants varied in their response to inoculation with different PGPR. Significant increases in growth and yield of agronomically important crops in response to inoculation with PGPR have been reported [20, 21]. Studies have also shown that the growth-promoting ability of some bacteria may be highly specific to certain plant species, cultivar and genotypes [22]. Poi and Kabi [23] reported that inoculation with Azotobacter strains isolated from the rhizosphere soils of squash, wheat and jute improved the yield but the strains were crop specific. The importance of screening of different PGPR and their potential features on plant growth and yield for different plants have been studied earlier [20, 24]. Main objective of this study was to screen and select the efficient PGPR to be used as an inoculant to increase the growth, yield and nutrition of chilly.
In the first experiment, 10 different PGPR were used. It was found that PGPR generally increased the plant height and stem girth as compared to uninoculated plants. Increased fruit yield was observed in six treatments viz, Paenibacillus polymyxa, Pantoea dispersa, Pseudomonas sp., Trichoderma viride and Bacillus sp. (EB 04) over control. The total plant (shoot + root) dry biomass was high in treatments inoculated with PGPR as compared to the uninoculated treatment. Though most of the plant parameters studied were statistically not significant, 2 PGPR viz., P. polymyxa and P. dispersa showed agronomic improvement in plant growth as compared to control; the increase in fruit yield in these two treatments being 33 and 17%, respectively.
The second experiment was conducted with 2 PGPR selected based on the results of the first experiment plus 8 more PGPR. Results showed that the plant height and stem girth of the chilly plants 60 DAP increased significantly with the PGPR, M. radiotolerans followed by P. fluorescens, B. subtilis and A. chroococcum. Other treatments did not differ significantly from the uninoculated control. The plants inoculated with M. radiotolerans and P. fluorescens had significantly greater biovolume-index and fruit yield as compared to other treatments and uninoculated control (Table 1). Methylobacterium sp., playing an important role in promoting the germination and plant growth, has been reported by many workers [25–27]. The dry weight of shoot was significantly more in 6 out of 10 PGPR inoculation treatments. The highest dry weight of the root was obtained in the treatment of M. radiotolerans which was statistically at par with A. chroococcum and A. brasilense treatments. The increase in total dry biomass was found in most of the PGPR inoculated plants as compared to uninoculated plants. However highest total dry biomass was recorded in plants treated with M. radiotolerans which was statistically at par with B. subtilis treatment (Table 2). Giving weightage to biovolume index, plant dry biomass and fruit yield, M. radiotolerans proved to be the best.
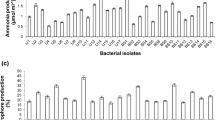
In the third pot experiment, further screening with M. radiotolerans plus 2 more new PGPR viz. Bacillus sonorensis and Paenibacillus elgii was done using 2 varieties of chilly viz. NS-1701 and Ujala F1 hybrid. Due to enhanced plant height and stem girth, the biovolume index was significantly higher in plants treated with B. sonorensis (Fig. 1) followed by M. radiotolerans in both the varieties of chilly. Inoculation of chilly plants (both varieties) with Bacillus sonorensis also showed significant increase in fruit yield. The increase in plant dry biomass and fruit yield because of inoculation with B. sonorensis was 41 and 57.74% in NS-1701, and 36 and 37.07% in Ujala F1 hybrid, respectively. The increase in plant dry biomass and fruit yield because of inoculation with B. sonorensis was 6.31 and 19.02% respectively as compared to that of M. radiotolerans treatment. B. sonorensis enhancing the growth and plant dry weight of tomato and sorghum has also been observed earlier by the authors [8]. The results of the major plant nutrient analysis brought out that N, P and K concentration of chilly samples was significantly high in plants inoculated with Bacillus sonorensis (Tables 3, 4). Hence, it can be concluded from the present study that B. sonorensis is the most promising PGPR for inoculating both the varieties of chilly. Since Bacillus sonorensis proved to be the best bacterium among 20 PGPR screened for inoculating chilly, its PGPR traits were studied and the results are given in Table 5. The traits such as P-solubilization, production of indole acetic acid (IAA), siderophore, chitinase, HCN, ACC deaminase, and biofilm formation confirmed that it is a PGPR.
Bacillus sp. are known to be P solubilizers and IAA producers and inoculation with Bacillus spp. improving growth of maize [28] and wheat [29] have been reported. Husen [30] reported that soil bacterial isolate Bacillus cereus produces siderophores and hence it can be used as efficient PGPR to increase crop yield. Shobha and Kumudini [31] isolated a Bacillus sp. which produced IAA, ammonia, HCN and siderophore, and inhibited the pathogen Fusarium oxysporum and suggested that it can be used as a PGPR. Zahir et al. [32] reported that the inoculation with rhizobacterial strains containing ACC-deaminase activity significantly promoted root, shoot and other growth contributing parameters of wheat at all salinity levels both under axenic and pot conditions. ACC deaminase is a master regulator of ethylene synthesis in plants. Bacteria possessing ACC deaminase activity reduce the level of stress hormone ethylene resulting in plant growth promotion under various abiotic stresses such as salt, drought, flooding, and heavy metal [33]. Thus, it is believed that the strain of B. sonorensis used in the present study will also protect plants against abiotic stresses. This aspect needs further investigation.
Conclusions
In conclusion, the present study demonstrates that PGPR are not host specific, however they exhibit host preference, thus emphasizing the need for screening different PGPR in order to select the best to be used as an inoculant for a particular crop. Out of the 3 pot experiments conducted using 20 PGPR, the results clearly brought out that B. sonorensis is the most promising PGPR inoculant for chilly. Studies also confirmed that B. sonorensis possessed all the PGPR traits.
References
Haas D, Defago G (2005) Biological control of soil-borne pathogens by fluorescent pseudomonads. Nat Rev Microbiol 3:307–319
Kloepper JW, Ryum M, Zhang S (2004) Induced systemic resistance and promotion of plant growth by Bacillus spp. Phytopathol 94:1259–1266
Saharan B, Nehra V (2011) Plant growth promoting rhizobacteria: a critical review. Life Sci Med Res 21:1–30
Vessey JK (2003) Plant growth promoting rhizobacteria as biofertilizers. Plant Soil 255:571–586
Khan MS, Raj SK (2006) First report of molecular detection of an Aster yellows phytoplasma (Candidatus Phytoplasma asteris) isolate infecting chilli (Capsicum annuum) in India. Plant Pathol 55:822
Bharathi R, Vivekananthan R, Harish S, Ramanathan A, Samiyappan R (2004) Rhizobacteria-based bioformulations for the management of fruit rot infection in chillies. Crop Prot 23:835–843
FAO Data (2010) Greening agriculture in India: an overview of opportunities and constraints. http://www.fao.org/docrep/article/agrippa/658_en00.htm#TopOfPage
Vaikuntapu PR, Dutta S, Samudrala RB, Rao VR, Kalam S, Podile AR (2014) Preferential promotion of Lycopersicon esculentum (tomato) growth by plant growth promoting bacteria associated with Tomato. Indian J Microbiol 54(4):403–412
Hatchell GE (1985) Production of bare root seedlings. In: Proceedings of 3rd Biennial Southern Silviculture Research Conference, pp 395–402
Jackson ML (1973) Soil chemical analysis. Prentice Hall Pvt. Ltd., New Delhi
Little TM, Hill FJ (1978) Agricultural experimentation design and analysis. Wiley, NewYork
Nautiyal CS (1999) An efficient microbiological growth medium for screening phosphate solubilizing microorganisms. FEMS Microbiol Lett 170:265–270
Brick JM, Bostock RM, Silversone SE (1991) Rapid in situ assay for indole acetic acid production by bacteria immobilized on nitrocellulose membrane. Appl Environ Microbiol 57:535–538
Schwyn B, Neilands JB (1987) Universal chemical assay for the detection and determination of siderophore. Anal Biochem 160:47–56
Das SN, Dutta S, Anil K, Neeraja C, Sarma PVSRN, Srinivas V, Podile AR (2010) Plant growth promoting chitinolytic Paenibacillus elgii responds positively to tobacco root exudates. J Plant Growth Regul 29:409–418
Joseph B, Patra RR, Lawrence R (2007) Characterization of plant growth promoting rhizobacteria associated with chickpea (Cicer arietinum L.). Int J Plant Prod 1:141–152
Yousef CF, Travieso ML, Espinosa UM (2008) Different, overlapping mechanisms for colonization of abiotic and plant surfaces by Pseudomonas putida. FEMS Microbiol Lett 288:118–124
OˈToole GA, Kolter R (1998) Initiation of biofilm formation in Pseudomonas fluorescens WCS365 proceeds via multiple, convergent signalling pathways: a genetic analysis. Mol Microbiol 28:449–461
Li Z, Chang S, Lin L, Li Y, An Q (2011) A colorimetric assay of 1-aminocyclopropane-1-carboxylate (ACC) based on ninhydrin reaction for rapid screening of bacteria containing ACC deaminase. Lett Appl Microbiol 53(2):178–185
Adriana A, Anelise B, Thais S, Felipe GP, Luciano KV, Luciane MPP (2012) Screening of plant growth promoting rhizobacteria isolated from sunflower (Helianthus annuus L.). Plant Soil 356:245–264
Hemlata C, Bagyaraj DJ, Anita S (2013) Plant growth-promoting bacterial endophytes from sugarcane and their potential in promoting growth of the host under field conditions. Exp Agric 49:43–52
Nowak J (1998) Benefits of in vitro ‘‘biotization’’ of plant tissue cultures with microbial inoculants. In vitro Cell Dev Biol Plant 34:122–130
Poi SC, Kabi MC (1979) Effect of Azotobacter inoculation on growth and yield of jute and wheat. Indian J Agri Sci 49:478–480
Hussain MI, Asghar HN, Arshad M, Shahbaz M (2013) Screening of multi-traits rhizobacteria to improve maize growth under axenic conditions. J Anim Plant Sci 23:514–520
Holland MA (1997) Occams razor applied to hormonology are cytokinins produced by plants? Plant Physiol 115:865–868
Ivanova EG, Doronina NV, Trossenko YA (2001) Acrobic methylobacteira are capable of synthesizing auxins. Mikrobiolgiya 70:345–347
Omer ZS, Tombolini R, Broberg A, Gerhardson B (2004) Indole-3-acetic acid production by pink-pigmented facultative methylotrophic bacteria. Plant Growth Regul 43:93–96
Beneduzi A, Peres D, da Costa PB, Bodanese Zanettini MH, Passaglia LM (2008) Genetic and phenotypic diversity of plant-growth-promoting bacilli isolated from wheat fields in southern Brazil. Res Microbiol 159:244–250
Trivedi P, Pandey A (2008) Plant growth promotion abilities and formulation of Bacillus megaterium strain B 388 (MTCC6521) isolated from a temperate Himalayan location. Indian J Microbiol 48:342–347
Husen E (2003) Screening of soil bacteria for plant growth promotion activities in vitro. Indones J Agric Sci 4:27–31
Shobha G, Kumudini BS (2012) Antagonistic effect of the newly isolated PGPR Bacillus spp. on Fusarium oxysporum. Int J Appl Sci Eng Res 1:463–474
Zahir ZA, Ghani U, Naveed M, Nadeem SM, Asghar HN (2009) Comparative effectiveness of Pseudomonas and Serratia sp. containing ACC-deaminase for improving growth and yield of wheat (Triticum aestivum L.) under salt-stressed conditions. Arch Microbiol 191:415–424
Glick BR, Cheng Z, Czarny J, Duan J (2007) Promotion of plant growth by ACC deaminase-producing soil bacteria. Eur J Plant Pathol 119:329–339
Acknowledegments
The first two authors are grateful to The National Academy of Sciences, India (NASI), Allahabad for the financial support under the Senior Scientist Jubilee Fellowship for carrying out this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Thilagar, G., Bagyaraj, D.J., Podile, A.R. et al. Bacillus sonorensis, a Novel Plant Growth Promoting Rhizobacterium in Improving Growth, Nutrition and Yield of Chilly (Capsicum annuum L.). Proc. Natl. Acad. Sci., India, Sect. B Biol. Sci. 88, 813–818 (2018). https://doi.org/10.1007/s40011-016-0822-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40011-016-0822-z