Abstract
Background
To confirm the diagnosis of periprosthetic joint infection (PJI), the Infectious Diseases Society of America (IDSA) and the International Consensus Meeting (ICM) have defined criteria that include histology as a minor criterion and the sonication method only as an additional criterion. The aim of this monocentric, retrospective study was to investigate the value of histology and whether sonication leads to a more accurate diagnosis.
Materials and methods
All revision surgeries for knee and hip arthroplasty between 2017 and 2020 were included. With regard to microbiological diagnostic, conventional culture of periprosthetic biopsies and sonication of explant material were performed. In addition, histology and non-specific inflammatory markers (CRP, leukocytes) were recorded.
Results
A total of 78 patients with PJI and 62 aseptic controls were included. From both microbiological methods (conventional culture / sonication), Staphyloccus (S.) epidermidis and S. aureus were detected most frequently. However, compared to the conventional microbiology, a higher sensitivity was calculated for sonication, albeit with a lower specificity in relation to a PJI. In two logistic regression models for the significance of all diagnostic parameters in PJI, the AUC was 0.92 and 0.96 with histology in particular making the decisive contribution in both models (p < 0. 001, both models).
Conclusion
Since histology showed the highest accuracy in the current study, its importance in the PJI criteria should be reevaluated. Sonication shows a high sensitivity for germ detection with a lower specificity and should only be used in combination with the conventional culture for microbiolgical diagnostics.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Due to demographic changes and longer life expectancy, the number of joint replacements is increasing rapidly. Currently, more than 1 million arthroplasties per year are performed in the USA. A significant increase to up to four million operations per year is expected by 2030 [1, 2]. A corresponding increase in complications and revisions is to be expected.
Periprosthetic joint infection (PJI) is a major problem. Bozic et al. showed that the incidence of revision surgery due to infection after hip replacement is 14.8% [3] and after knee replacement 25.2% [4]. The diagnosis of PJI has far-reaching consequences for patients, as revision surgery, multiple operations and long-term antibiotic treatment are necessary. Long hospitalisation is the result. The economic burden on the healthcare system is correspondingly high. Vanhegan et al. used the example of total hip arthroplasty to show that septic revisions are almost twice as expensive as periprosthetic fractures and aseptic loosening [5]. Müller et al. point out that the costs of treating a PJI are generally not covered for hospitals in Germany [6].
There are various diagnostic criteria and algorithms for PJI [7,8,9,10]. The value of the respective criteria for diagnosing a PJI is the subject of current research due to the lack of a gold standard. However, the definitions of a PJI according to the Infectious Diseases Society of America (IDSA) [7] and the International Consensus Meeting (ICM) [8] are used worldwide. Major criteria for a PJI in both bodies are the detection of the same infectious agent in two tissue samples taken or the presence of a joint fistula (sinus tract communicating with the joint). In addition, there are minor criteria that can be used to diagnose a PJI; this is particularly important when the microbiological examinations have been carried out without detection of germs. Since the diagnosis of a PJI is not based on a single diagnostic marker, but several parameters must be considered (multi-parameter diagnostics) and misinterpretations may occur [11]. None of the commonly used criteria has 100 per cent sensitivity and specificity.
As already mentioned, microbiological diagnostics play an important role in the diagnosis of PJI. However, here too, caution is required when interpreting the results: For example, biofilm-forming microorganisms are not necessarily detectable in the periprosthetic tissue samples (high rate of negative findings). Furthermore, low-grade infections are often problematic in diagnostics, as the causative germs (e.g. coagulase-negative staphylococci, Cutibacterium propioni) are usually part of the skin flora and patients often experience few symptoms [10, 12]. In the case of microbiologically negative findings or in the differentiation of contamination from low-grade infections, histological examination could be of substantial importance [13]. In addition, sonication of explanted prosthesis components has been proposed to improve the sensitivity of microbiological diagnostics [14].
Against the background of the diagnostic problems, the aim of the present field study was to investigate the usefulness of histology in the multiorchestrated diagnosis of PJI including non-specific inflammatory markers, sonication method and conventional microbiological tissue culture from periprosthetic sample material.
Materials and methods
Study design
In this monocentric, retrospective cohort study, all cases of revision surgery for suspected PJI performed in our clinic were included when sonication method of the explant material was performed (e.g. suspicious infections with biofilm producing bacteria, low grade infections / chronic infections). The study period was from 02/2017 to 12/2020. Sonications of antibiotic-containing spacers and sonications of shoulder TEPs were excluded.
The PJI was based on the criteria of the International Consensus Meeting 2018 (ICM) and was used as a reference [8] (Table 1).
The sonication method and conventional microbiology of periprostetic tissue samples were performed independently at different institutes. For the evaluation of inflammatory markers, the results of C-reactive protein (CRP) and leukocytes in the peripheral blood were documented on the day of admission of the patients.
Sonication of removed endoprosthesis material
The explanted prosthesis material (prosthesis components including mobile parts) were packed in the operating theatre in sterile plastic boxes (reusable, disinfected and sterilised at 134° C, double-wrapped) and sent directly to the in-house laboratory. There, the implant boxes were opened under laminar flow and the explants were doused with 60 ml Ringer's solution. The implant box was then gently swirled for approx. 30 s and placed in an ultrasonic bath (Bandelin BactoSonioc 14.2, Germany) at 100% intensity (40 ± 5 kHz) for one minute. The implant boxes were shaken again under laminar flow for 30 s. Next, two Falcon tubes (polypropylene conical tubes, flat top, 50 ml, Falcon, USA) were each loaded with 20 ml sonicate and centrifuged at 2000 rpm for 5 min. The supernatant was carefully pipetted off until approx. 2 ml remained in the tip of the tube. Finally, 100 µL of the centrifugate was spread on semi-solid / liquid culture media (Chocolate Agar, Schaedler Agar (with 5% sheep blood), MacConkey Agar, Kochblut Agar, Thioglycollate Bouillon, Biomerieux, France) using a Drigalski spatula. The agar plates were incubated for 14 days at 37° C (Schaedler agar: anaerobic incubation). All plates exhibiting positive growth were quantified and identified according to the routine established by the laboratory.
Conventional microbiological culture of periprosthetic biopsies
Periprosthetic tissue biopsies (4—6 pieces) were sent immediately to the laboratory in sterile tubes and homogenised there under sterile conditions using tissue homogenisers (tissue homogenisers according to Potter–Elvehjem, VWR International, Germany). The homogenised mixture was then spread on semi-solid / liquid culture media (Chocolate Agar, Schaedler Agar (with 5% sheep blood), MacConkey Agar, Kochblut Agar, Thioglycollate Bouillon, Biomerieux, France). The agar plates were incubated for 14 days at 37° C (Schaedler agar: anaerobic incubation). All plates exhibiting positive growth were quantified and identified according to the routine established by the laboratory.
Routine germ identification and resistance testing
Germ identification was carried out using MALDI-Tof analysis (Matrix assisted laser desorbtion ionization time of flight analysis; Vitek MS, Biomerieux, France). In this technology, bacterial proteins are mixed with matrices, typically organic acids (α-Cyano-4-hydroxycinnamic acid (HCCA)), which can adsorb the laser energy to assist sample desorption and transfer protons for sample ionization. Mostly single charged ions or ions with low charge number are produced. The charged ions are accelerated in an electric field to gain kinetic energy, and then pass a field-free flight tube for separation, wherein the ions with larger mass-to-charge (m/z) ratio arrive at the end of the flight tube with a longer flight time whereas the ones with smaller m/z with a shorter flight time. The mass spectrum of the bacterial proteins is compared with a database. MALDI technique is a very precise and rapid technology to identify bacterial species. Resistance testing was performed using automated microdilution technology (Vitek 2, Biomerieux, France).
Histological examination (according to [15])
Three to seven tissue samples taken from the respective joint during the procedure were fixed in buffered formalin immediately after excision and sent to the histopathological laboratory. The bone tissue was decalcified with EDTA solution. The samples were embedded in paraffin, sectioned with a microtome and stained with haematoxylin & eosin and Prussian blue. Periprosthetic joint infection was considered to be present if either three neutrophils were found in a high power field (HPF) or five or more neutrophils were found in 5 HPF (magnification × 400). Cases with three and four neutrophils in 5 HPF were considered suspicious for periprosthetic joint infection. Neutrophils located within the superficial fibrin exudate or intravascularly were not considered.
Statistics
Analysis of possible differences between groups were performed using the Fishers exact test or the student’s t-test. Logistic regression was evaluated using leave-one-out cross-validation. For this purpose, one patient was removed from the data set, the coefficients for the logistic regression were determined with the remaining data set and the logistic regression model was then applied to the previously removed patient. The AUC analysis was performed on this basis. Logistic regression was calculated with forward selection on the one hand and with all variables (histology, sonicate or microbiology, CRP and leukocytes) on the other. The stated coefficients for the logistic regression models with all variables are based on the entire data set (without leave-one-out cross-validation). The difference in AUCs in the logistic regression models was calculated using a two-sided DeLong's test. Statistical analysis was performed with the statistics software R version 4.2.3 or with SPSS version 29.0.1.0.
Results
Patient characteristics
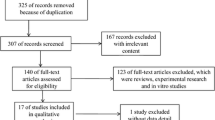
A total of 140 patients with suspected PJI were included in the study. Of these patients, 78 cases were defined as PJI / or possible PJI according to the consensus recommendation, with 42 patients fulfilling the major criteria (Table 2). Of these infections, 11 were defined as early infections (within four weeks after joint replacement surgery); 34 patients were receiving antibiotic therapy on admission. The remaining patients (n = 62) were defined as aseptic controls.
The age structure of patients with aseptic or septic revisions was comparable (p = 0.97), the mean age was 72.5 years (Table 2). There is no difference in the gender distribution (p = 0.62). The mean length of stay for PJI was almost twice as long (33.4 d) as for aseptic changes (17 d; p = 0.01).
Influence of risk factors on the detection of a PJI
When analysing the risk factors for a PJI, diabetes (p = 0.72, 95%CI 0.53—2.57), obesity (defined with a body mass index > 25) (p = 0.45, 95%CI 0.61—3.14), renal insufficiency (p = 0.07, 95%CI 0.85—7.40) and nicotine abuse (p = 0.68, 95%CI 0.26—21.39) showed no significant influence. In the PJI group, at least one risk factor was found in 59% of the patients compared to 42% in the control group (p = 0.07).
Microbiological identification and microbes spectrum after using sonication of removed endoprosthetic material and conventional culture from tissue samples (Fig. 1)
In the PJI group, the most common microorganisms detected by sonication were Staphylococcus (S.) epidermidis (n = 20 (25.6%)), followed by S. aureus (14 (17.9)) and Streptococci (Str.) (11 (14.1)). The most common germs in the conventional culture of tissue samples were S. aureus (15 (19%)) and S. epidermidis (14 (18), followed by streptococci (7 (8.9)). Overall, 21.8% of the cases remained undetected after sonication and 32% after conventional culture of biopsy material.
In the aseptic control group, 75.8% of the sonicates and 91.9% of cultured biopsies have shown no growth after cultivation. So, most common germ particular found after sonication was Cutibacterium cutis (n = 7 (11.3%)).
Results of non-specific inflammatory markers (CRP and leukocytes) in PJI and controls
The concentrations of CRP and the leukocyte count in the peripheral blood of patients with PJI were significantly higher than in the controls (CRP: p < 0.001, leukocytes p = 0.010) (Table 2).
Significance of histology, sonication method, conventional culture from tissue samples and non-specific inflammatory markers (CRP, leukocytes) in PJI
To clarify the question of the importance of histology compared to sonication and culture from tissue samples, logistic regression (leave-one-out method) was calculated (Fig. 2): in one model, the diagnostic criteria histology, sonication, leukocytes, CRP were used for the calculation; in the other model, the results of conventional culture from biopsies were included instead of sonication. The first model resulted in an Area Under the Curve (AUC) of 0.92, 95% CI 0.87—0.97, whereby only histology (p < 0.001) was a significant parameter in the diagnosis of PJI (sonication p = 0.78, CRP p = 0.35, leukocytes p = 0.21). The model with conventional culture instead of sonication showed an AUC of 0.96, 95% CI 0.92—0.99. Here, histology (p < 0.001) and the conventional culture method (p = 0.01) were significantly associated with the diagnosis of PJI (for CRP p = 0.25, leukocytes p = 0.12 were calculated). The difference between the AUCs was significant (p = 0.0544).
Performance of two logistic regression models and calculation of the AUC to evaluate the significance of the individual diagnostic parameters in the context of multi-parameter diagnostics of PJI: in the first model (A), the parameters histology, conventional cultures of biopsies, leucocytes and CRP were taken into account, in the second model (B), sonication was used in place of the conventional culture. In both models, a high AUC of 0.96 (A) and 0.92 (B) was achieved, which differed significantly (p = 0.054). In principle, both multiparameter models are suitable for the diagnosis of PJI. The highly significant importance of histology in the diagnosis compared to all other parameters is also evident here.
The statistical indices sensitivity / specificity, negative predictive value (NPV) and positive predictive value (PPV) for the presence of a PJI for the parameters histology, conventional culture from biopsies, sonication method, CRP (calculated for the cut off concentrations 5 / 100 mg/L) and leukocytes (cut off 10 / nL) can be found in Table 3. Histology method defined as minor parameter has been found the most sensitive and specific parameter for diagnosing PJI. Sonication of explanted endoprothetic material compared to the conventional culture of biopsies has a higher sensitivity albeit with a lower specificity in diagnosing PJI.
Discussion
The diagnosis of PJI is a multiparameter diagnosis in everyday clinical practice. The present study on the value of the individual diagnostic criteria for PJI showed that histology was superior to conventional culture of biopsies and sonication method in terms of significance, sensitivity and specificity; the inflammatory parameters CRP and leukocytes in peripheral blood were only diagnostic parameters of second choice, depending on the cut-off. The ICM and IDSA criteria used to diagnose PJI did not show any differences in the present study, nor did the evaluation of individual risk factors such as diabetes mellitus, obesity, renal insufficiency and nicotine abuse reveal any particular risk constellations for PJI. The bacterial spectrum we determined (11 early, 67 late infections) largely corresponded to the bacterial spectrum known from the literature with frequent detection of coagulase-negative staphylococci and Staphylococcus aureus [16,17,18,19,20].
The identification of germs from periprosthetic biopsies or from sonicates with consecutive resistance testing is the basis for the optimal treatment of a PJI. Whether one of the two methods is superior in terms of sensitivity and specificity for the diagnosis of a PJI is the subject of a number of studies with controversial results [14, 16, 21,22,23,24,25,26,27: the studies by Dudareva et al. [28] and Ribeiro et al. [24] with a design comparable to our study showed comparable sensitivities of sonication and microbiology; however, the authors recommended sonication only as a supplement to microbiology due to its significantly poorer specificity. In the authors' opinion, sonication could have added value in special infection constellations with a lower bioburden (patients who have been pre-treated with antibiotics, late infections > 24 months after implantation). The study by Ribeiro also made it clear that the sensitivity and specificity of the microbiological examination methods were significantly dependent on the respective diagnostic criteria used for a PJI (IDSA [7], ICM [8] or EBJIS [29]). In the work by Rothenberg [17], sonication showed a comparable specificity compared to classical microbiology with high sensitivity; the high specificity was achieved here in particular in samples with a high bioburden of > 5 CFU/ml. In our opinion, the question of evaluating sonication by including a threshold value—as in Rothenberg's work—has not yet been sufficiently investigated. For these reasons, no threshold value was considered in the evaluation of the results in our study either. The definition of a threshold value could lead to an improvement in specificity, as sonication is more susceptible to contamination due to several diagnostic steps. The differences in sensitivity and specificity between microbiology and sonication are certainly also due to the fact that both procedures are not standardised.
Few studies on multiparameter diagnostics of PJI have so far included the significance of histology in the diagnosis of PJI [30, 31, 32]: our data show better sensitivity (0.95) and specificity (0.92) of histology in the diagnosis of PJI compared to sonication and microbiology. Also, our two logistic regression models including all investigated diagnostic parameters show that histology is a highly significant diagnostic parameter, regardless of whether conventional culture of biopsies was introduced as a major criterion or whether sonication was included in the model. In his publications [30, 31], Müller et al. also described a comparably high sensitivity and specificity for histology, which correlated well with the results of intraoperative bacterial detection (however, sonication of endoprosthesis material was not performed in the studies by Müller et al.) In the study by Tani et al. [32], the sensitivity of histopathological examination was comparable to that of sonication (72 vs. 77%), but significantly better than that of conventional culture from biopsy material (55%). Based on the current study and the studies described above, it could be concluded that the histological examination of periprosthetic biopsies should be given greater importance in the diagnosis of PJI. Particularly in view of the high rate of negative microbiological evidence, histology appears to be a reliable instrument in the multi-parameter diagnosis of PJI and clearly superior to the non-specific inflammatory parameters CRP and leukocytes.
The use of CRP and leukocytes from peripheral blood in the diagnosis of PJI would only play a subordinate role due to the sensitivities and specificities we have determined: Although both parameters are inexpensive in routine diagnostics and easy to determine due to their low invasiveness, they are very susceptible to interference in antibiotic-pretreated patients (44% of our patients were on antibiotics on admission) [33], depending on the type of infectious agent [34] and due to non-specific increases caused by co-morbidities such as autoimmune diseases or other acute/chronic inflammations. Leukocyte / CRP detection from synovial fluid [35, 36] or the use of new inflammatory parameters such as calprotectin [37] may be more suitable here.
Several studies have shown that patients with certain underlying diseases or risk factors (diabetes, obesity, renal insufficiency, nicotine) develop a higher risk of PJI after joint replacement surgery [38,39,40,41,42,43]. We were unable to determine this for individual risk factors in our study, but our patients with PJI also tended to have a more critical risk profile.
The added value of the present study certainly lies in the fact that the significance of histology was reassessed. One limitation for the interpretation of the data is certainly the limited number of patients analysed and the monocentric design. When evaluating the statistics, it must also be taken into account that parameters relating to the diagnosis of a PJI were included that are themselves part of the PJI definition. Our slightly modified criteria for histopathological diagnosis of the periprosthetic infection might represent further limitation of the current study. Although we use a more conservative citeria for the diagnosis of PJI in our institution (3 or more neutrophils/1 HPF or 5 and more neutrophils/5 HPF) when compared with the EBJIS 2021 criteria (5 or more neutrophils/1 HPF or 5 and more neutrophils/5 HPF [29] for routine diagnostics, we believe our data are comparable with other European groups. Even though there is some uncertainty about the exact number and localization of the histopathological specimens, we analyzed comparable numbers of specimens with other researchers [44] and believe that our data might help to better understanding of the pathogenesis and diagnostics of PJI.
Concluding from our data, histology seems to have the most significant role among the multiparameter diagnostic of PJI and should be reevaluated particularly in view of the high rate of negative microbiological finding. Sonication lacks standardization, is labor-intensive and in our study showed no advantage over the microbiological culture of biopsies. It should therefore by no means replace conventional microbiology, but in individual cases it may represent a supplementary diagnostic method. In our view, CRP and leukocytes in peripheral blood as criteria are dispensable when histology is available.
Data availability
No datasets were generated or analysed during the current study
References
Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Jt Surg Am. 2007;89(4):780–5.
Etkin CD, Springer BD. The American Joint Replacement Registry—the first 5 years. Arthroplasty Today. 2017;3(2):67–9.
Bozic KJ, Kurtz SM, Lau E, Ong K, Vail TP, Berry DJ. The epidemiology of revision total hip arthroplasty in the United States. J Bone Jt Surg Am. 2009;91(1):128–33.
Bozic KJ, Kurtz SM, Lau E, Ong K, Chiu V, Vail TP. The epidemiology of revision total knee arthroplasty in the United States. Clin Orthop. 2010;468(1):45–51.
Vanhegan IS, Malik AK, Jayakumar P, Ul Islam S, Haddad FS. A financial analysis of revision hip arthroplasty: the economic burden in relation to the national tariff. J Bone Jt Surg Br. 2012;94(5):619–23.
Müller M, Trampuz A, Winkler T, Perka C. Die ökonomische herausforderung der zentralisierten behandlung von patienten mit periprothetischen infektionen. Z Für Orthop Unfallchirurgie. 2018;156(04):407–13.
Osmon DR, Berbari EF, Berendt AR, Lew D, Zimmerli W, Steckelberg JM. Executive summary diagnosis and management of prosthetic joint infection: clinical practice guidelines by the infectious diseases society of americaa. Clin Infect Dis. 2013;56(1):1–10.
Parvizi J, et al. The 2018 Definition of Periprosthetic Hip and Knee Infection: An Evidence-Based and Validated Criteria. J Arthroplasty. 2018;33(5):1309-1314.e2. https://doi.org/10.1016/j.arth.2018.02.078.
Izakovicova P, Borens O, Trampuz A. Periprosthetic joint infection: current concepts and outlook. EFORT Open Rev. 2019;4(7):482–94.
Mühlhofer H, Renz N, Zahar A, Lüdemann M, Rudert M, Hube R. Diagnostik der periprothetischen infektion. Orthopade. 2021;50(4):312–25.
Li C, Renz N, Trampuz A, Ojeda-Thies C. Twenty common errors in the diagnosis and treatment of periprosthetic joint infection. Int Orthop. 2020;44(1):3–14.
Zimmerli W. Prosthetic-Joint Infections. N Engl J Med. 2004;315(16):1645–54.
Pandey R, Drakoulakis E, Athanasou NA. An assessment of the histological criteria used to diagnose infection in hip revision arthroplasty tissues. J Clin Pathol. 1999;52(2):118–23.
Trampuz A, Piper KE, Jacobson MJ, Hanssen AD, Unni KK, Osmon DR. Sonication of removed hip and knee prostheses for diagnosis of infection. N Engl J Med. 2007;357(7):654–63.
Mirra JM, Amstutz HC, Matos M, Gold R. The pathology of the joint tissues and its clinical relevance in prosthesis failure. Clin Orthop Relat Res. 1976;117:221–40.
Portillo ME, Salvadó M, Alier A, Martínez S, Sorli L, Horcajada JP. Advantages of sonication fluid culture for the diagnosis of prosthetic joint infection. J Infect. 2014;69(1):35–41.
Rothenberg AC, Wilson AE, Hayes JP, O’Malley MJ, Klatt BA. Sonication of arthroplasty implants improves accuracy of periprosthetic joint infection cultures. Clin Orthop. 2017;475(7):1827–36.
Flurin L, Greenwood-Quaintance KE, Patel R. Microbiology of polymicrobial prosthetic joint infection. Diagn Microbiol Infect Dis. 2019;94(3):255–9.
Peel TN, Cheng AC, Buising KL, Choong PFM. Microbiological aetiology, epidemiology, and clinical profile of prosthetic joint infections: are current antibiotic prophylaxis guidelines effective? Antimicrob Agents Chemother. 2012;56(5):2386–91.
Benito N, Mur I, Ribera A, Soriano A, Rodríguez-Pardo D, Sorlí L. The different microbial etiology of prosthetic joint infections according to route of acquisition and time after prosthesis implantation, including the role of multidrug-resistant organisms. J Clin Med. 2019;8(5):673.
Puig-Verdié L, Alentorn-Geli E, González-Cuevas A, Sorlí L, Salvadó M, Alier A. Implant sonication increases the diagnostic accuracy of infection in patients with delayed but not early orthopaedic implant failure. Bone Jt J. 2013;95(2):244–9.
Janz V, Wassilew GI, Kribus M, Trampuz A, Perka C. Improved identification of polymicrobial infection in total knee arthroplasty through sonicate fluid cultures. Arch Orthop Trauma Surg. 2015;135(10):1453–7.
Shen H, Tang J, Wang Q, Jiang Y, Zhang X. Sonication of explanted prosthesis combined with incubation in bd bactec bottles for pathogen-based diagnosis of prosthetic joint infection. J Clin Microbiol. 2015;53(3):777–81.
Ribeiro TC, Honda EK, Daniachi D, de Cury RPL, da Silva CB, Klautau, GB. The impact of sonication cultures when the diagnosis of prosthetic joint infection is inconclusive. PLoS ONE. 2021. https://doi.org/10.1371/journal.pone.0252322.
Grosso MJ, Frangiamore SJ, Yakubek G, Bauer TW, Iannotti JP, Ricchetti ET. Performance of implant sonication culture for the diagnosis of periprosthetic shoulder infection. J Shoulder Elbow Surg. 2018;27(2):211–6.
Van Diek FM, Albers CGM, Van Hooff ML, Meis JF, Goosen JHM. Low sensitivity of implant sonication when screening for infection in revision surgery. Acta Orthop. 2017;88(3):294–9.
Puchner SE, Döring K, Staats K, Böhler C, Lass R, Hirschl AM. Sonication culture improves microbiological diagnosis of modular megaprostheses. J Orthop Res. 2017;35(7):1383–7.
Dudareva M, Barrett L, Figtree M, Scarborough M, Watanabe M, Newnham R. Sonication versus tissue sampling for diagnosis of prosthetic joint and other orthopedic device-related infections. J Clin Microbiol. 2018;56(12):e00688-e718.
McNally M, Sousa R, Wouthuyzen-Bakker M, Chen AF, Soriano A, Vogely HC, Clauss M, Higuera CA, Trebše R. The EBJIS definition of periprosthetic joint infection. Bone Jt J. 2021;103(1):18–25. https://doi.org/10.1302/0301-620X.103B1.BJJ-2020-1381.R1.
Müller M, Morawietz L, Hasart O, Strube P, Perka C, Tohtz S. Histopathological diagnosis of periprosthetic joint infection following total hip arthroplasty : use of a standardized classification system of the periprosthetic interface membrane. Orthopade. 2009;38(11):1087–96.
Müller M, Morawietz L, Hasart O, Strube P, Perka C, Tohtz S. Diagnosis of periprosthetic infection following total hip arthroplasty – evaluation of the diagnostic values of pre- and intraoperative parameters and the associated strategy to preoperatively select patients with a high probability of joint infection. J Orthop Surg. 2008;3:31.
Tani S, Lepetsos P, Stylianakis A, Vlamis J, Birbas K, Kaklamanos I. Superiority of the sonication method against conventional periprosthetic tissue cultures for diagnosis of prosthetic joint infections. Eur J Orthop Surg Traumatol Orthop Traumatol. 2018;28(1):51–7.
Dugdale EM, Uvodich ME, Osmon DR, Pagnano MW, Berry DJ, Abdel MP. Recent antibiotic treatment impacts serum and synovial laboratory values in early periprosthetic joint infection workup. J Arthroplasty. 2022;37(6S):S286–90. https://doi.org/10.1016/j.arth.2022.02.064.
Unter Ecker N, Suero EM, Gehrke T, Haasper C, Zahar A, Lausmann C, Hawi N, Citak M. Serum C-reactive protein relationship in high- versus low-virulence pathogens in the diagnosis of periprosthetic joint infection. J Med Microbiol. 2019;68(6):910–7. https://doi.org/10.1099/jmm.0.000958.
Buttaro MA, Martorell G, Quinteros M, Comba F, Zanotti G, Piccaluga F. Intraoperative synovial C-reactive protein is as useful as frozen section to detect periprosthetic hip infection. Clin Orthop Relat Res. 2015;473(12):3876–81. https://doi.org/10.1007/s11999-015-4340-8.
Shahi A, Tan TL, Kheir MM, Tan DD, Parvizi J. Diagnosing Periprosthetic Joint Infection: And the Winner Is? J Arthroplasty. 2017;32(9S):S232–5. https://doi.org/10.1016/j.arth.2017.06.005.
Schindler M, Walter N, Maderbacher G, Sigmund IK, Alt V, Rupp M. Novel diagnostic markers for periprosthetic joint infection: a systematic review. Front Cell Infect Microbiol. 2023;13:1210345. https://doi.org/10.3389/fcimb.2023.1210345.
Peersman G, Laskin R, Davis J, Peterson M. Infection in total knee replacement: a retrospective review of 6489 total knee replacements. Clin Orthop. 2001;392:15–23.
Namba RS, Inacio MCS, Paxton EW. Risk factors associated with deep surgical site infections after primary total knee arthroplasty: an analysis of 56,216 knees. J Bone Joint Surg Am. 2013;95(9):775–82.
Malinzak RA, Ritter MA, Berend ME, Meding JB, Olberding EM, Davis KE. Morbidly obese, diabetic, younger, and unilateral joint arthroplasty patients have elevated total joint arthroplasty infection rates. J Arthroplasty. 2009;24(6):84–8.
Dowsey MM, Choong PFM. obese diabetic patients are at substantial risk for deep infection after primary TKA. Clin Orthop. 2009;467(6):1577–81.
Dowsey MM, Choong PFM. Obesity is a major risk factor for prosthetic infection after primary hip arthroplasty. Clin Orthop. 2008;466(1):153–8.
Singh JA, Schleck C, Harmsen WS, Jacob AK, Warner DO, Lewallen DG. Current tobacco use is associated with higher rates of implant revision and deep infection after total hip or knee arthroplasty: a prospective cohort study. BMC Med. 2015;13:283.
Sigmund IK, Yeghiazaryan L, Luger M, Windhager R, Sulzbacher I, McNally MA. Three to six tissue specimens for histopathological analysis are most accurate for diagnosing periprosthetic joint infection. Bone Jt J. 2023;105(2):158–65. https://doi.org/10.1302/0301-620X.105B2.BJJ-2022-0859.R1.
Funding
No funds, grants, or other support was received.
Author information
Authors and Affiliations
Contributions
A.R. and A.A. responsible for study design, wrote manuscript, prepared figures 1 and tables F.K. responsible for statistics, prepared figure 2, reviewed mansucript B.F. responsible for study design C.W. reviewed manuscript J.Z. responsible for histology, reviewed manuscript A.G. resposible for microbiological data
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Röhrl, A., Klawonn, F., Füchtmeier, B. et al. Results of a monocentric field study: value of histology compared to sonication method and conventional tissue culture in the diagnosis of periprosthetic joint infection (PJI). Infection (2024). https://doi.org/10.1007/s15010-024-02278-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s15010-024-02278-x