Abstract
Purpose of Review
In this article, we summed up the different types of stem cells which are present in the skin and the role of adipose-derived stem cells (ADSCs) in providing various anti-aging properties.
Recent Findings
Adipose-derived stem cells (ADSCs) can be utilised in numerous unbelievable ways like in skin regeneration, soft tissue augmentation, anti-aging therapy, curing wrinkled skin and even in skin whitening. ADSCs prove to be useful even in cosmetic surgeries where it increases the success rate dramatically with the fusion of fat grafts and lipo-injections.
Summary
We sum up the various other related properties of ADSCs like angiogenesis, plastic surgery and cancer treatment and their applications in curing skin diseases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Skin, one of the largest organ, is always subjected to the outer environmental factors and has evolved rightly in order to respond to various stressors [1]. The aging of skin can be of various types from chronological to photoaging, where chronological refers to the physiological alterations of the skin functions and photoaging is related to light, radiations and UV rays [2]. There have been various studies focusing on the different factors that are responsible for the early skin aging and also on the changes that take place as skin and its cells gradually start to age. Apart from the scientists and researchers who are studying the aging of the skin and would thus even induce rapid aging in model skin tissues, nobody likes the skin to age and lose its young properties. Skin aging induction can be done by various ways like UV radiation-induced connective tissue damage [3].
There are numerous ways by which researchers have tried to bring back the aged skin to life and undoubtedly have even been successful in doing so. One still cannot deny the fact that there is lot to improve and would be so ahead as well, with this particular field always remaining a wide area of research for many researchers. One major way of healing the skin and regenerating it is doing it from the root source. This is where the stem cells come into account which due to their property of acting as a repair system of the body and replenishing other body cells always stays in the limelight [4]. One of the most easily accessible and most effective stem cells with diverse skin regeneration and rejuvenation properties is adipose stem cells (ADSCs) [5]. With its diverse properties, it can not only be used for skin regeneration and wound healing, but also for plastic surgery to even soft tissue augmentation. Its vibrant properties make it an interesting topic for the researchers to dwell deeper into it and find more uses.
Types of Skin Stem Cells
Skin is a simple-looking but a rather complex organ of the body. It is not a smooth surface as it tends to look like but a flat surface full of depths and troughs. As there are different types of cells and glands present on the skin, it is obvious that there are different types of stem cells also (Table 1). Stem cells are those distinct types of cells that can self-renew and differentiate into any type of adult stem cells and can replenish any lost cells during the course of time [6, 7]. These stem cells reside in a specific environment, known as “niche”, which is present in a tissue, for progeny production and self-renovation [8•].
Epidermal Stem Cells (ESC)
The epidermis is the outermost layer of the skin. Its major role is in protecting the tissues and cells from the unfavourable outer conditions. It is a tough layer with horny cells and comprises of stratum corneum [22, 23]. Epidermis also has some different sub layers in it, including the outer layers, mid layers and base layers, which all together form a proper epidermis. If epidermal layers also include the outer layer membranes, then the epidermis can be said to have 4 distinct sub divisions. The outermost membrane is the plasma membrane which consists of a lipid bilayer. Moreover, the outermost layers are protective layers which are categorised as the line barriers, further classified as the first line barrier and the second line barrier. Other sub layers are named as Granular layer, Spinous layer and Basal layer [23].
Epidermis mostly comprises keratinocytes and they account to more than 95% of the total cells present [24]. Apart from keratinocytes, there are 2 more type of dendritic cells; Langerhans cells and Melanocytes [25]. Their location too varies from one another, for an example, keratinocytes reside in the lower levels of epidermis [26], while Langerhans usually rest in the supra-basal position [27]. Epidermis is a type of tissue that continually renews itself (renewal turnover of epidermal cell is 7 days) and sheds of its unwanted dead cells [28]. The two upper layer of skin, the epidermis and the dermis, are filled with extracellular proteins in between them.
The particular interest is associated with epidermal stem cells (ESC) in skin repair as they are accessible and numerous in numbers. The ESC is highly proliferative and form large clones [29]. They are responsible for maintaining skin homeostasis and are widely used in clinical studies and regenerative medicine for skin repair [29]. Since 1970s, the regenerative potentials of ESCs have been explored to overcome the limitations associate with therapeutic conventional strategies and have been demonstrated to promote healing of wounds and replacement of irreversibly lost skin [30]. ESCs were found to get activated during wound healing. The lineage restriction of resident skin stem cells and spatial confinement led to the recruitment of ESCs from different regions of the skin and contribute to multiple ESCs during skin repair [31].
Follicular Stem Cells
A hair follicle comprises the whole structure from the outer strand of hair to the innermost bulb. In between this, there are many sub structures like Arrector pili muscle (APM), Bulb, Club hair, Dermal papilla (DP), Dermal Sheath (DS), Hair canal, Hair germ, Hair peg, Hair shaft, Infundibulum, Inner root sheath (IRS), Outer root sheath (ORS), Isthmus, Pelage hairs and Sebaceous gland [32]. All have various specific functions like dermal papilla regulates hair growth, arrector pili muscle controls piloerection and inner root sheath (IRS) expresses fibroblast growth factor mRNA (FGF-22 mRNA) [33].
It has been observed that the bulge region of the hair follicle is the most important niche where the stem cells reside [10]. Moreover, the bulge stem cells play a vital role in wound healing due to their regeneration potential, and their ability to migrate to the site of damaged epidermis [34]. The DP cells of the hair follicle have proven to produce many growth factors, matrix components and cytokines like fibroblast growth factor (bFGF), endothelin-1 (ET-1) and stem cell factor (SCF) [35]. Thus, proving their worth in skin repair and wound healing.
Melanocyte Stem Cells
White, pale, black and brown, this is how the colour of the skin is usually characterised. But what actually decides the colour of the skin is the total count of melanocyte present in it. More the number of melanocytes present, darker would the skin colour be. The melanocytes are not just confined to the skin but are present at other sites too. Residing in the follicles of hair, they tend to play a major role in hair pigmentation. They are even said to be phenotypically prominent but histologically inconspicuous skin cells [36].
Melanocyte stem cells have various functions and offer an advantageous model which helps us understand how stem cell behaviour is regulated by their surrounding microenvironment. They are fairly easy to isolate. The major hotspot for these stem cells is found out to be in the bulge region of the hair follicle [37]. Also, the epidermal cells and melanin form a functional unit which comprises of melanocytes and keratinocytes [38]. The melanin pigment is of immense importance to the skin as it has many useful functions like preventing the skin from harmful sun rays [39]. Moreover, it makes the skin less vulnerable to infections as compared to the skin with less melanin content thus helping in faster skin repair too.
Sebaceous Gland Stem Cells
Apart from the complexion, the skin can be oily, dry or normal. This texture of the skin is the result of different secretions that take place by different glands. Apart from its association which tends to be with hair follicles only, these glands are present on other sites as well [40•] and depending on the site, they are recognised by different names; for example, meibomian is the sebaceous gland of the eyelid [40•]. Sebaceous gland stem cells help in the renewal of the sebaceous gland. Many multiple stem and progenitor cell compartments have been identified [41] which contributes together to the regeneration and continuous renewal of hair follicles and sebaceous gland [42].
Mesenchymal Stem Cells
Be it bone, dermis or even smooth and rough muscles, the involvement of mesenchymal cells is everywhere. Talking about the migration of the mesenchymal cells, they tend to migrate in early embryonic development between the ectodermal and endodermal layers [43]. The most fascinating part of these cells is related with skin regeneration and wound healing, thanks to its ability of space filling and repair mechanisms [43]. A study has been performed where mesenchymal stem cells derived from the umbilical cord blood (UCB) proved to be extremely effective in cutaneous skin repair. The tests were periodically performed on rats [44]. Considering the source, there are different types of tissues derived from mesenchymal progenitor stem cells. Some of which are bone, cartilage, tendon, adipose, muscle and vascular derived [45].
Haematopoietic Stem Cells (HSC)
Haematopoietic stem cells are the stem cells from which all the different types of blood cells are derived. They were also the first tissue specific stem cells to be isolated [46]. There are many diseases like leukaemia and some autoimmune disorders in which the haematopoietic stem cells containing grafts prove to be extremely useful [47]. Because of the very short life span of most effector cells, mature blood cell production is an ongoing process with estimates suggesting the production of 1.5 × 106 blood cells every second in an adult human [48]. Haematopoietic stem cells have been demonstrated to restore the architecture of skin and hair follicles while enhancing the wound healing speed and quality [49].
Adipose Tissue and Cells
Adipose tissue or fat tissue is composed of lipid filled cells known as the lipocytes [50]. The location of this tissue is under the skin [50, 51]. Adipose cell consists of a large lipid droplet surrounded by a thin rim of cytoplasm [52]. There are usually 2 types of adipose tissues present in the body, white adipose tissue (WAT) and brown adipose tissue (BAT) [53]. White adipose tissues are the main supplier of lipid whereas the brown ones are more useful for fatty acids and heat production [54]. Adipose tissue provides thermal insulation, mechanical protection and is also known for its high expandable nature [55, 56]. Adipose tissue had also been observed to expand on the level of obesity [57], or shrink during weight loss [58].
Adipose-derived stem cells (ADSCs) are the mesenchymal cells that can be isolated from subcutaneous adipose tissue, are abundantly present in human body and can be isolated in large numbers [59]. Because of their multi-lineage capacity, the ADSCs potential had been extensively explored for tissue repair and maintenance [60]. The various applications of ADSCs in skin repair are discussed in the following sections.
Role and Applications of ADSCs
The ADSCs have immense capacity to differentiate into different types of cells, which is also responsible for their immense importance to skin regeneration and tissue augmentation. There are various factors that prove its worth in this very field. Some factors include [61] the following:
-
1.
They continue proliferating even after transplantation
-
2.
Differentiation of adipose-derived stem cells into endothelial cells and possible neovascularization
-
3.
Multipotent differentiation capacity
-
4.
Release of angiogenic growth factors
-
5.
Wound healing
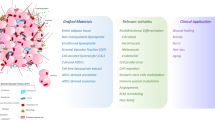
Thus, its multipotency and ability to release angiogenic growth factors can be useful in reducing the symptoms of many types of diseased patients, like diabetes and many other age-related complications [61]. ADSCs release various growth factors and cytokines which prove to be vital in skin repair and wound healing. They even have various other functions like storing fat and burning calories (Berry et al., 2015). Other vital applications include face reconstruction, skin whitening, skin rejuvenation and breast augmentation (Fig. 1).
ADSCs and Skin Whitening
According to a study, a conditioned medium of ADSCs was prepared and its skin lightening and effect on melanocytes was studied in melanoma B16 cells [62] ADSCs treatment inhibited the synthesis of melanin and the activity of tyrosinase in a dose dependent manner [62]. ADSCs also help in producing antioxidant cytokines which are helpful in giving a fair skin [63]. The melanin pigment forms a cap over the keratinocytes which protects it from the harmful rays of the sun like UV rays [39]. The main enzyme involved in the synthesis of melanin is tyrosinase [64], and the main site is melanosome [65]. Therefore, tyrosine inhibitors can be helpful in lightening the tone of the skin [62]. Human tyrosinase related protein-1(TRP1) have proved to be extremely important for the activation of tyrosinase enzyme. Moreover, it is stimulated and enhanced by cyclic AMP, ATP, magnesium, pH of around 6.2 and temperature of around 30 °C [66, 67].
ADSCs culture mediums contain specific growth factors like TGF-β (Transforming growth factor-b1) which hinders the process of the melanin synthesis [68]. Anti TGF-β1 antibodies have also been tested and showed rapid re-stabilization of the dropped tyrosine levels of ADSC-CM treated melanoma B16 cells. This is the reason behind the ongoing research to bring in more effective tyrosine inhibitors that would prove useful in less melanin production. Among the numerous experiments, treatment of ADSCs in the culturing medium of B16 melanoma cells have been proven successful in reducing the tyrosinase activity in B16 cells (Fig. 2) [62].
Wrinkled Skin, ADSCs and Fat Grafts
Aging can be both intrinsic and extrinsic, where extrinsic factors are all the external environmental factors [69]. Studies have found various factors like extreme stress, ultraviolet rays and smoking intensively affecting the skin aging [70]. There are many changes that take place during the course of time in the skin structure and composition. For example, changes in the gene expression of skin fibroblasts led to a significant decrease in cell number [71] and skin collagen production also experiences a gradual decrease [72].
The skin also experiences a redistribution of fat after a certain age limit. The fat is redistributed in different areas of the body like the arms, thighs and buttocks; however, the fat consistency experiences a fair decrease in certain areas like face [69]. This tends to make the skin look loose and lack stiffness. This is where injections of ADSCs work. However, studies have proven that the effectiveness in skin rejuvenation is dramatically increased if the ADSCs injection is given in combination with the fat grafts [69].
Face Reconstruction
Face reconstruction is not only confined to the jawline repair and other lower jaw related corrections but also remains vibrant from lip augmentation to cheek lifting. There are various types of fillers used in fat grafting. The classical tools like the blunt cannula seems to be one of the easiest and safe fillers available [73]. Although, there have been significant successful improvements in the field of fat fillers but the use of old classical tools can never be neglected. More than the issue of filling the fat graft, the more concerning factor for the surgeons is the retention and viability of the same. This is the reason behind the continuous ongoing research for the improvement of fat graft viability. Studies have shown that there is a significant improvement in the retention of fat grafts with the combination of adipose-derived regenerative cells (ADRCs) [74].
There are many diseases whose side effects involved the loss of fat grafts in some parts of the face. Lipoinjection can prove to be divine in diseases like lipoatrophy which can even be associated with HIV infection [75]. However, it still needs further improvement as there are many loopholes which demand improvement. Although, autologous lipoinjection had proven to be one of the best treatment for soft tissue augmentation because there is no incision scar or complications associated [76].
Mechanism of ADSC Assisted Angiogenesis
Apart from the direct differentiation, ADSCs also triggers various paracrine functions like immunoregulation, vascularization, skin repair and others. Vascular endothelial growth factor (VEGF) released from the ADSCs promotes rapid growth of endothelial cells which thereafter helps in vessel formation. Moreover, hypoxic conditions have also turned out to be favourable for VEGF release and angiogenesis [77]. Also, the role of leptin cannot be left behind in enhancing the process of angiogenesis. The activation of specific receptor known as the leptin receptor (Ob-R) in the endothelial cells plays an important role in angiogenesis (Fig. 3) [78].
Angiogenesis and Wound Healing
Different phases of wound healing in the skin have been extensively documented where all involve dynamic interactions between epidermal cells, dermal cells and bone marrow-derived cells, leading to rapid wound closure and subsequent tissue repair [79]. The best available treatment for chronic wounds achieves only a 50% healing rate that is often temporary. Among the many factors contributing to non-healing wounds, impairment in the production of cytokines by local inflammatory cells and fibroblasts and reduced angiogenesis are crucial [80]. Tumours can develop, albeit infrequently at the site of chronic skin wounds or untreated mouth ulcers [81]. Wound healing response of fibroblasts has many similarities to the activation of fibroblasts in the tumour stroma—for example, the expression of markers of myofibroblasts used to predict the outcome of cancers of the lung, stomach and mammary gland [82].
One of the most important conclusions from recent research on the epidermis is that there are multiple populations of stem cells residing in different locations within the tissue [83]. Revascularization plays a critical role in wound healing and is regulated by a complex zone of growth factors and cytokines. Deficiencies in revascularization contribute to the development of chronic non-healing wounds [84]. According to a research done, the angiogenic capillaries are said to come in the fibronectin-rich wound clot and thereafter form a microvascular network throughout the granulation tissue [85]. Vascularization is the first step in the formation of blood vessels [86]. Endothelial cells which form the lining of the vessels have been proven to play a major role in angiogenesis [87•]. It had also been studied that wound angiogenesis seems to be regulated by endothelial cell interaction with the three-dimensional ECM environment in and around the wound area [85]. Apart from wound healing angiogenesis is also triggered in various other cases like female menstrual cycle, embryonic cycles and organ growth [88].
Angiogenesis and Aging
Skin aging is characterised by many factors from gradual timely aging of the various skin cells and reduced number of stem cells that are able to respond to proliferative signals [89]. Angiogenesis is the development of new blood vessels and can be a determinant factor in age-related changes. This is why we see more severity in vascular related disorders in older individuals [90]. Be it vascular sprouting, vessel maturing or any type of blood vessel formation, ECM (extracellular matrix) plays a very important role in it [83], like providing binding scaffold to various cytokines and helping in many signalling factors as well [91]. There is also a significant loss of vessels in aged individuals which are responsible for exchange of nutrients. These vessels are affected by both density and surface area [92].
Angiogenesis and Photoaging
Photoaging refers to the damage of skin which is caused by the over exposure to sunlight and other harmful rays of the sun [93, 94•]. UV radiation, infrared rays and even heat are the main age inducer trio of photoaging and reports have suggested that angiogenesis is induced by these stimuli [95]. However, although skin angiogenesis is induced by acute UV irradiation but a chorological decrease in cutaneous blood vessels was observed in photodamaged skin. The photoaged skin is characterised by coarse wrinkles, loss of elasticity, pigmented spots, dryness, verrucous papules and telangiectasia [96, 97].
Defensins
Defensins are widely distributed and abundant antimicrobial peptides (3–4 KDa) that are variably cationic and contain six disulphide-paired cysteines [98]. They are broadly divided into three different types based on their structure; α-defensins, β-defensins and θ defensins [99]. The importance of these peptides in skin regeneration and wound healing cannot be ignored because of their ability to trigger many immunomodulatory and chemotactic functions. Some investigations are also done on the effects of human β-defensins (hBDs) on keratinocytes and fibroblasts, 2 major skin cell types involved in skin regeneration [100]. Not only skin regeneration and wound healing, these peptides also play an essential role in male fertility [101].
Other factors related to defensins and skin healing are acne and heat bumps. The basic mechanisms involved in the development of inflammation during acne vulgaris is a hot topic of research. There have been many experiments that have investigated the expression patterns of two antimicrobial peptides, human beta-defensin 1 (hBD1) and human beta-defensin 2 (hBD2), in healthy human hair follicles as well as in peri- and intralesional skin of acne vulgaris lesions such as comedones, papules and pustules. Strong hBD1 and hBD2 immunoreactivity have been found in all suprabasal layers of the epidermis, and all permanent compartments of the hair follicle including the distal ORS of the hair follicle and the pilosebaceous duct [102]. Moreover, due to their ability to trigger the activation of various cell types, along with playing a vital role in cytokine regulation, these defensins (human beta defensins in particular) also have been proven to be useful in the regulation of various allergic diseases [103].
Plastic Surgery, Tissue Augmentation, Skin Regeneration and Stem Cells
The idea of tissue augmentation and new skin regeneration is obviously a fascinating topic; however, the therapies involved are not that common in all laboratories and are done at a huge cost. These procedures tend to be even more complicated with the involvement of infection, surrounding fibrosis and contracture [104]. There are relatively cheaper alternatives like that of fat grafting which is also a rather easier treatment with relatively less complications. Fat grafting has been an active field of interest for many researchers where most of the observations include the deep survey of not less than 100 patients receiving minimum of at least two treatments [105]. As far as ADSCs are concerned, there have been many studies where their effectiveness in soft tissue augmentation has been evaluated [106•]. ADSCs appears to be more suitable for enhancement of fat graft as the self-renewal, proangiogenic and unlimited proliferation capacity of ADSCs have been known to promote the differentiation of epithelial cells, neovascularisation and increase proangiogenic factors concentration which ultimately improves the survival of fat grafts [107, 108]. A rapid development has been seen in soft tissue engineering based on ADSCs in past decades with ADSCs becoming gradually the main stem cells source for adipose engineering [109].
Tumour Growth, Angiogenesis and Cancer Treatment
Tumour is a rapid uncontrolled growth of cells and the factors required for cell growth, involves vessels formation and their critical role in supplying essential nutrients and other types of growth factors to the tumour site. Tumour growth requires formation of new blood vessels for continuous supply of oxygen and nutrients and the identification of chemical factors that mediate angiogenesis [110]. A study had demonstrated that the expression of various oncogenes, mainly the mutant ras, may lead to a marked induction of a potent paracrine stimulator of angiogenesis i.e. vascular endothelial growth factor (VEGF) [111]. Vascular endothelial growth factor (VEGF) is critical for angiogenesis [112].
There are various activating and inhibiting factors that play a major role in switching on or off the angiogenic state (Fig. 4). Talking about inhibitors in particular, although their main role is in the inhibition of new blood vessels (cytostatic nature), some direct inhibitors can even prove to be toxic too (cytotoxic) [113]. Apart from cytokines, there are various other factors that play a major role in angiogenesis and vasculogenesis, like macrophages (pro angiogenic cell), neutrophils (anti angiogenic factor), B & T cells (both angiogenic and anti-angiogenic), cancer-associated fibroblasts (pro angiogenic factor), adipocytes (promote angiogenesis by releasing pro angiogenic cytokines) and regulation of glucose availability [114].
Conclusion
There are various types of skin stem cells each having different useful properties. Among them, ADSCs tend to be extremely useful. Nor do they just have vibrant properties; they are even easy to isolate as compared to other skin stem cells. They are also useful in lightning the tone of the skin where these stem cells can be used in reducing the production of the melanin pigment. ADSCs have a vital role in tissue augmentation for which they become a wide area of interest to various plastic surgeons. It has even proved that with the combination of fat grafts, they can be extremely useful in reducing the wrinkles of the skin, be it due to the age or any external factor. Additionally, angiogenesis also plays a vital role in skin regeneration and rejuvenation. With angiogenesis providing vital help in triggering rapid cell growth, it tends to favour various oncogenic cells. Thus, as a counterblow, antiangiogenic factors may prove to be extremely useful in various fields like tumour suppression.
Abbreviations
- APM:
-
Arrector pili muscle
- ADSC:
-
Adipose stem cells
- ADSC-CM:
-
Adipose stem cells culture medium
- ADRC:
-
Adipose-derived regenerative cells
- ECM:
-
Extracellular matrix
- ESC:
-
Epidermal stem cells
- IRS:
-
Inner root sheath
- HSC:
-
Haematopoietic stem cells
- ORS:
-
Outer root sheath
- UV:
-
Ultraviolet
- WBC:
-
White blood cells
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Slominski A, Wortsman J, Paus R, Elias PM, Tobin DJ, Feingold KR. Skin as an endocrine organ: implications for its function. Drug Discov Today: Dis Mech. 2008;5(2):e137–44.
Hashizume H. Skin aging and dry skin. J Dermatol. 2004;31(8):603–9.
Hwang KA, Yi BR, Choi KC. Molecular mechanisms and in vivo mouse models of skin aging associated with dermal matrix alterations. Lab Anim Res. 2011;27(1):1–8.
McCulloch EA, Till JE. Perspectives on the properties of stem cells. Nat Med. 2005;11(10):1026–8.
Konno M, Hamabe A, Hasegawa S, Ogawa H, Fukusumi T, Nishikawa S, Ohta K, Kano Y, Ozaki M, Noguchi Y, Sakai D. Adipose-derived mesenchymal stem cells and regenerative medicine. Dev Growth Differ. 2013;55(3):309–18.
Li L, Xie T. Stem cell niche: structure and function. Annu Rev Cell Dev Biol. 2005;10(21):605–31.
Ohlstein B, Kai T, Decotto E, Spradling A. The stem cell niche: theme and variations. Curr Opin Cell Biol. 2004;16(6):693–9.
Yang R, Liu F, Wang J, Chen X, Xie J, Xiong K. Epidermal stem cells in wound healing and their clinical applications. Stem Cell Res Ther. 2019;10(1):1–4. In this paper, the authors discussed different characteristics of epidermal stem cell, their functions and the underlying mechanism of wound healing. The clinical potential of epidermal stem cell in improving wound healing is also discussed in this paper.
Lavker RM, Sun TT. Epidermal stem cells: properties, markers, and location. Proc Natl Acad Sci. 2000;97(25):13473–5.
Waters JM, Richardson GD, Jahoda CA. Hair follicle stem cells. Semin Cell Dev Biol. 2007;18(2):245–54.
Nowak JA, Polak L, Pasolli HA, Fuchs E. Hair follicle stem cells are specified and function in early skin morphogenesis. Cell Stem Cell. 2008;3(1):33–43.
Mull AN, Zolekar A, Wang YC. Understanding melanocyte stem cells for disease modeling and regenerative medicine applications. Int J Mol Sci. 2015;16(12):30458–69.
Nishimura EK, Jordan SA, Oshima H, Yoshida H, Osawa M, Moriyama M, Jackson IJ, Barrandon Y, Miyachi Y, Nishikawa SI. Dominant role of the niche in melanocyte stem-cell fate determination. Nature. 2002;416(6883):854–60.
Nguyen DT, Orgill DP, Murphy GF. The pathophysiologic basis for wound healing and cutaneous regeneration. In Biomaterials for treating skin loss. 2009;(pp. 25–57). Woodhead Publishing.
Geueke A, Niemann C. Stem and progenitor cells in sebaceous gland development, homeostasis and pathologies. Exp Dermatol. 2021;30(4):588–97.
Niemann C, Horsley V. Development and homeostasis of the sebaceous gland. Semin Cell Dev Biol. 2012;23:928–36.
Oh M, Nör JE. The perivascular niche and self-renewal of stem cells. Front Physiol. 2015;2(6):367.
Li Z, Hu X, Zhong JF. Mesenchymal stem cells: characteristics, function, and application. 2019. In this paper, the various clinical applications of mesenchymal stem cells in skin wound healing, cartilage and bone repair, neuronal regeneration and immune disorder treatment have been discussed.
Caplan AI. Mesenchymal stem cells in regenerative medicine. In Principles of Regenerative Medicine 2019;(pp. 219–227). Academic Press. In this paper, the author describes the different extraction methods for mesenchymal stem cells. The author also describes the various preclinical and clinical applications of these stem cells in regenerative medicine.
Bydlowski SP, Levy D, Ruiz JM, Pereira J. Hematopoietic stem cell niche: role in normal and malignant hematopoiesis. Stem Cell Biology in Normal Life and Diseases. 2013;8:17.
Hawley RG, Ramezani A, Hawley TS. Hematopoietic stem cells. Methods Enzymol. 2006;1(419):149–79.
Lavker RM, Sun TT. Epidermal stem cells. J Investig Dermatol. 1983;81(1):S121–7.
Baroni A, Buommino E, De Gregorio V, Ruocco E, Ruocco V, Wolf R. Structure and function of the epidermis related to barrier properties. Clin Dermatol. 2012;30(3):257–62.
McGowan JE. A Journal of the Pathological Society of Great Britain and Ireland. 1993.
Regnier M, Patwardhan A, Scheynius A, Schmidt R. Reconstructed human epidermis composed of keratinocytes, melanocytes and Langerhans cells. Med Biol Eng Compu. 1998;36(6):821–4.
Zelickson AS, Mottaz JH. Epidermal dendritic cells: a quantitative study. Arch Dermatol. 1968;98(6):652–9.
Romani N, Holzmann S, Tripp CH, Koch F, Stoitzner P. Langerhans cells–dendritic cells of the epidermis. APMIS. 2003;111(7–8):725–40.
Potten CS. Cell replacement in epidermis (keratopoiesis) via discrete units of proliferation. Int Rev Cytol. 1981;1(69):271–318.
Wang ZL, He RZ, Tu B, He JS, Cao X, Xia HS, Ba HL, Wu S, Peng C, Xiong K. Drilling combined with adipose-derived stem cells and bone morphogenetic protein-2 to treat femoral head epiphyseal necrosis in juvenile rabbits. Current Med Sci. 2018;38(2):277–88.
Teng M, Huang Y, Zhang H. Application of stems cells in wound healing—an update. Wound Repair Regen. 2014;22(2):151–60.
Mascre G, Dekoninck S, Drogat B, Youssef KK, Brohee S, Sotiropoulou PA, Simons BD, Blanpain C. Distinct contribution of stem and progenitor cells to epidermal maintenance. Nature. 2012;489:257–62.
Schneider MR, Schmidt-Ullrich R, Paus R. The hair follicle as a dynamic miniorgan. Curr Biol. 2009;19(3):R132–42.
Nakatake Y, Hoshikawa M, Asaki T, Kassai Y, Itoh N. Identification of a novel fibroblast growth factor, FGF-22, preferentially expressed in the inner root sheath of the hair follicle. Biochimica et Biophysica Acta (BBA)-Gene Structure and Expression. 2001;1517(3):460–3.
Garcin CL, Ansell DM, Headon DJ, Paus R. Hardman MJ Hair follicle bulge stem cells appear dispensable for the acute phase of wound re-epithelialization. Stem Cells. 2016;34(5):1377–85.
Zhang CP, Fu XB. Therapeutic potential of stem cells in skin repair and regeneration. Chinese Journal of Traumatology (English Edition). 2008;11(4):209–21.
Lin JY, Fisher DE. Melanocyte biology and skin pigmentation. Nature. 2007;445(7130):843–50.
Lang D, Mascarenhas JB, Shea CR. Melanocytes, melanocyte stem cells, and melanoma stem cells. Clin Dermatol. 2013;31(2):166–78.
Seiberg M, Paine C, Sharlow E, Andrade-Gordon P, Costanzo M, Eisinger M, Shapiro SS. The protease-activated receptor 2 regulates pigmentation via keratinocyte-melanocyte interactions. Exp Cell Res. 2000;254(1):25–32.
Singh SK, Nizard C, Kurfurst R, Bonte F, Schnebert S, Tobin DJ. The silver locus product (Silv/gp100/Pmel17) as a new tool for the analysis of melanosome transfer in human melanocyte–keratinocyte co-culture. Exp Dermatol. 2008;17(5):418–26.
Veniaminova NA, Grachtchouk M, Doane OJ, Peterson JK, Quigley DA, Lull MV, Pyrozhenko DV, Nair RR, Patrick MT, Balmain A, Dlugosz AA. Niche-specific factors dynamically regulate sebaceous gland stem cells in the skin. Dev Cell. 2019;4;51(3):326–40. The study defines different factors which act together for modulating oil-secreting sebaceous glands (SGs) stem cells and thus critically regulate skin functions. The study also demonstrated the integration of diverse niche signals by sebaceous glands.
Frances D, Niemann C. Stem cell dynamics in sebaceous gland morphogenesis in mouse skin. Dev Biol. 2012;363(1):138–46.
Jaks V, Kasper M, Toftgård R. The hair follicle—a stem cell zoo. Exp Cell Res. 2010;316(8):1422–8.
Sohni A, Verfaillie CM. Mesenchymal stem cells migration homing and tracking. Stem Cells Int. 2013.
Luo G, Cheng W, He W, Wang X, Tan J, Fitzgerald M, Li X, Wu J. Promotion of cutaneous wound healing by local application of mesenchymal stem cells derived from human umbilical cord blood. Wound repair and regeneration. 2010;18(5):506–13.
Minguell JJ, Erices A, Conget P. Mesenchymal stem cells. Exp Biol Med. 2001;226(6):507–20.
Spangrude GJ, Heimfeld S, Weissman IL. Purification and characterization of mouse hematopoietic stem cells. Science. 1988;241:58–62.
Weissman IL. Translating stem and progenitor cell biology to the clinic: barriers and opportunities. Science. 2000;287:1442–6.
Bryder D, Rossi DJ, Weissman IL. Hematopoietic stem cells: the paradigmatic tissue-specific stem cell. Am J Pathol. 2006;169(2):338–46.
Tolar J, McGrath JA. Augmentation of cutaneous wound healing by pharmacologic mobilization of endogenous bone marrow stem cells. J Investig Dermatol. 2014;134(9):2312–4.
Ahima RS, Flier JS. Adipose tissue as an endocrine organ. Trends Endocrinol Metab. 2000;11(8):327–32.
Choi JS, Choi YC, Kim JD, Kim EJ, Lee HY, Kwon IC, Cho YW. Adipose tissue: a valuable resource of biomaterials for soft tissue engineering. Macromol Res. 2014;22(9):932–47.
Cushman SW. Structure-function relationships in the adipose cell I. Ultrastructure of the isolated adipose cell. J Cell Biol. 1970;46(2):326–41.
Pénicaud L, Cousin B, Leloup C, Lorsignol A, Casteilla L. The autonomic nervous system, adipose tissue plasticity, and energy balance. Nutrition. 2000;16(10):903–8.
Casteilla L, Pénicaud L, Cousin B, Calise D. Choosing an adipose tissue depot for sampling. Methods in molecular biology (Clifton, NJ). 2008;456:23–38.
Trayhurn P, Beattie JH. Physiological role of adipose tissue: white adipose tissue as an endocrine and secretory organ. Proceedings of the Nutrition Society. 2001;60(3):329–39.
Sethi JK, Vidal-Puig AJ. Thematic review series: adipocyte biology. Adipose tissue function and plasticity orchestrate nutritional adaptation. J Lipid Res. 2007;48(6):1253–62.
Alkhouli N, Mansfield J, Green E, Bell J, Knight B, Liversedge N, Tham JC, Welbourn R, Shore AC, Kos K, Winlove CP. The mechanical properties of human adipose tissues and their relationships to the structure and composition of the extracellular matrix. Am J Physiol Endocrinol Metab. 2013;305(12):E1427–35.
Mariman EC, Wang P. Adipocyte extracellular matrix composition, dynamics and role in obesity. Cell Mol Life Sci. 2010;67(8):1277–92.
Zuk PA, Zhu MI, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001;7(2):211–28.
Zuk P. Adipose-derived stem cells in tissue regeneration: a review. International Scholarly Res Notices. 2013.
Brown SA, Levi B, Lequex C, Wong VW, Mojallal A, Longaker MT. Basic science review on adipose tissue for clinicians. Plast Reconstr Surg. 2010;126(6):1936–46.
Kim WS, Park SH, Ahn SJ, Kim HK, Park JS, Lee GY, Kim KJ, Whang KK, Kang SH, Park BS, Sung JH. Whitening effect of adipose-derived stem cells: a critical role of TGF-β1. Biol Pharm Bull. 2008;31(4):606–10.
Maeda K, Okubo K, Shimomura I, Mizuno K, Matsuzawa Y, Matsubara K. Analysis of an expression profile of genes in the human adipose tissue. Gene. 1997;190(2):227–35.
Parvez S, Kang M, Chung HS, Cho C, Hong MC, Shin MK, Bae H. Survey and mechanism of skin depigmenting and lightening agents. Phytotherapy Research: an International Journal Devoted to Pharmacological and Toxicological Evaluation of Natural Product Derivatives. 2006;20(11):921–34.
Yoshimura K, Sato K, Aoi N, Kurita M, Inoue K, Suga H, Eto H, Kato H, Hirohi T, Harii K. Cell-assisted lipotransfer for facial lipoatrophy: efficacy of clinical use of adipose-derived stem cells. Dermatol Surg. 2008;34(9):1178–85.
Körner A, Pawelek J. Activation of melanoma tyrosinase by a cyclic AMP-dependent protein kinase in a cell-free system. Nature. 1977;267(5610):444–7.
Rzepka Z, Buszman E, Beberok A, Wrześniok D. From tyrosine to melanin: signaling pathways and factors regulating melanogenesis. Postepy Hig Med Dosw(Online). 2016;30(70):695–708.
Kim DW, Jeon BJ, Hwang NH, Kim MS, Park SH, Dhong ES, Yoon ES, Lee BI. Adipose-derived stem cells inhibit epidermal melanocytes through an interleukin-6–mediated mechanism. Plast Reconstr Surg. 2014;134(3):470–80.
Sjerobabski-Masnec I, Šitum M. Skin aging. Acta Clin Croat. 2010;49(4):515–8.
Fisher GJ, Kang S, Varani J, Bata-Csorgo Z, Wan Y, Datta S, Voorhees JJ. Mechanisms of photoaging and chronological skin aging. Arch Dermatol. 2002;138(11):1462–70.
Kim HH, Lee MJ, Lee SR, Kim KH, Cho KH, Eun HC, Chung JH. Augmentation of UV-induced skin wrinkling by infrared irradiation in hairless mice. Mech Ageing Dev. 2005;126(11):1170–7.
Rittié L, Fisher GJ. UV-light-induced signal cascades and skin aging. Ageing Res Rev. 2002;1(4):705–20.
Coleman SR. Structural fat grafts: the ideal filler? Clin Plast Surg. 2001;28(1):111–9.
Zhu M, Zhou Z, Chen Y, Schreiber R, Ransom JT, Fraser JK, Hedrick MH, Pinkernell K, Kuo HC. Supplementation of fat grafts with adipose-derived regenerative cells improves long-term graft retention. Ann Plast Surg. 2010;64(2):222–8.
Domergue S, Psomas C, Yachouh J, Lesnik A, Reynes J, Goudot P, Jammet P. Fat microinfiltration autografting for facial restructuring in HIV patients. J Cranio-Maxillofac Surg. 2006;34(8):484–8.
Carpaneda CA, Ribeiro MT. Percentage of graft viability versus injected volume in adipose autotransplants. Aesthetic Plast Surg. 1994;18(1):17–9.
Hassan WU, Greiser U, Wang W. Role of adipose-derived stem cells in wound healing. Wound Repair Regen. 2014;22(3):313–25.
Park HY, Kwon HM, Lim HJ, Hong BK, Lee JY, Park BE, Jang YS, Cho SY, Kim HS. Potential role of leptin in angiogenesis: leptin induces endothelial cell proliferation and expression of matrix metalloproteinases in vivo and in vitro. Exp Mol Med. 2001;33(2):95–102.
Shaw TJ, Martin P. Wound repair at a glance. J Cell Sci. 2009;122(18):3209–13.
Falanga V. Wound healing and its impairment in the diabetic foot. Lancet. 2005;366:1736–43.
Dunham LJ. Cancer in man at site of prior benign lesion of skin or mucous membrane: a review. Can Res. 1972;32(7):1359–74.
Chang HY, Sneddon JB, Alizadeh AA, Sood R, West RB, Montgomery K, Chi JT, Rijn MV, Botstein D, Brown PO, Liu ET. Gene expression signature of fibroblast serum response predicts human cancer progression: similarities between tumors and wounds. PLoS Biology. 2004;2(2):e7.
Arwert EN, Hoste E, Watt FM. Epithelial stem cells, wound healing and cancer. Nat Rev Cancer. 2012;12(3):170–80.
King A, Balaji S, Keswani SG, Crombleholme TM. The role of stem cells in wound angiogenesis. Adv Wound Care. 2014;3(10):614–25.
Tonnesen MG, Feng X, Clark RA. Angiogenesis in wound healing. In J Investig Dermatol Symp Proc. 2000;(Vol. 5, No. 1, pp. 40–46).
Jussila L, Alitalo K. Vascular growth factors and lymphangiogenesis. Physiol Rev. 2002;82(3):673–700.
Fatima F, Garrett J. Resolvin D1 interferes with several of the required steps for angiogenesis. The FASEB J. 2019;33(S1):527–1. In this paper, the authors demonstrated that the treatment of endothelium with Resolvin D1 slows the migration of endothelial cells and also decreased endothelial cell proliferation. It inhibits the essential steps of angiogenesis and thus have therapeutic potential for cancer treatment.
Mundel TM, Kalluri R. Type IV collagen-derived angiogenesis inhibitors. Microvasc Res. 2007;74(2–3):85–9.
Zouboulis CC, Adjaye J, Akamatsu H, Moe-Behrens G, Niemann C. Human skin stem cells and the ageing process. Exp Gerontol. 2008;43(11):986–97.
Moriya J, Minamino T. Angiogenesis, cancer, and vascular aging. Frontiers in Cardiovascular Medicine. 2017;24(4):65.
Senger DR, Davis GE. Angiogenesis. Cold Spring Harbor perspectives in biology. 2011;3(8):a005090.
Kelly RI, Pearse R, Bull RH, Leveque JL, de Rigal J, Mortimer PS. The effects of aging on the cutaneous microvasculature. J Am Acad Dermatol. 1995;33(5):749–56.
Rabe JH, Mamelak AJ, McElgunn PJ, Morison WL, Sauder DN. Photoaging: mechanisms and repair. J Am Acad Dermatol. 2006;55(1):1–9.
Worrede A, Douglass SM, Weeraratna AT. The dark side of daylight: photoaging and the tumor microenvironment in melanoma progression. J Clin Invest. 2021;131(6). In this paper, the authors provide an overview of UVR impact on the skin microenvironment and its effect on intrinsic and extrinsic aging altering fundamental processes such as extracellular matrix deposition, melanogenesis, inflammation and immune response.
Chung JH, Eun HC. Angiogenesis in skin aging and photoaging. J Dermatol. 2007;34(9):593–600.
Ichihashi M, Ando H, Yoshida M, Niki Y, Matsui M. Photoaging of the skin. Anti-aging medicine. 2009;6(6):46–59.
Bernstein EF, Brown DB, Schwartz MD, Kaidbey K, Ksenzenko SM. The polyhydroxy acid gluconolactone protects against ultraviolet radiation in an in vitro model of cutaneous photoaging. Dermatol Surg. 2004;30(2):189–96.
Ganz T, Lehrer RI. Defensins. Curr Opin Immunol. 1994;6(4):584–9.
Schneider JJ, Unholzer A, Schaller M, Schäfer-Korting M, Korting HC. Human defensins. J Mol Med. 2005;83(8):587–95.
Van Kilsdonk JW, Jansen PA, Van Den Bogaard EH, Bos C, Bergers M, Zeeuwen PL, Schalkwijk J. The effects of human beta-defensins on skin cells in vitro. Dermatology. 2017;233(2–3):155–63.
Suarez-Carmona M, Hubert P, Delvenne P, Herfs M. Defensins: “Simple” antimicrobial peptides or broad-spectrum molecules? Cytokine Growth Factor Rev. 2015;26(3):361–70.
Philpott MP. Defensins and acne. Mol Immunol. 2003;40(7):457–62.
Niyonsaba F, Kiatsurayanon C, Ogawa H. The role of human β-defensins in allergic diseases. Clin Exp Allergy. 2016;46(12):1522–30.
Salibian AA, Widgerow AD, Abrouk M, Evans GR. Stem cells in plastic surgery: a review of current clinical and translational applications. Arch Plast Surg. 2013;40(6):666.
Eremia S, Newman N. Long-term follow-up after autologous fat grafting: analysis of results from 116 patients followed at least 12 months after receiving the last of a minimum of two treatments. Dermatol Surg. 2000;26(12):1150–8.
Yoshimura K, Sato K, Aoi N, Kurita M, Hirohi T, Harii K. Cell-assisted lipotransfer for cosmetic breast augmentation: supportive use of adipose-derived stem/stromal cells. Aesthet Plast Surg. 2020;44(4):1258–65. In this paper, the authors developed a novel strategy known as cell-assisted lipotransfer (CAL) in which autologous adipose-derived stem cells (ASCs) were used in combination with lipoinjection. CAL was found to be effective and safe for soft tissue augmentation and superior to conventional lipoinjection.
Gir P, Oni G, Brown SA, Mojallal A, Rohrich RJ. Human adipose stem cells: current clinical applications. Plast Reconstr Surg. 2012;129:1277–90.
Zhu M, Dong Z, Gao J, Liao Y, Xue J, Yuan Y, Liu L, Chang Q, Lu F. Adipocyte regeneration after free fat transplantation: promotion by stromal vascular fraction cells. Cell Transplant. 2015;24:49–62.
Combellack EJ, Jessop ZM, Naderi N, Griffin M, Dobbs T, Ibrahim A, Evans S, Burnell S, Doak SH, Whitaker IS. Adipose regeneration and implications for breast reconstruction: update and the future. Gland Surg. 2016;5:227–41.
Folkman J. Clinical applications of research on angiogenesis. N Engl J Med. 1995;333(26):1757–63.
Rak J, Mitsuhashi Y, Sheehan C, Tamir A, Viloria-Petit A, Filmus J, Mansour SJ, Ahn NG, Kerbel RS. Oncogenes and tumor angiogenesis: differential modes of vascular endothelial growth factor up-regulation in ras-transformed epithelial cells and fibroblasts. Can Res. 2000;60(2):490–8.
Bagchi M, Kim LA, Boucher J, Walshe TE, Kahn CR, D’Amore PA. Vascular endothelial growth factor is important for brown adipose tissue development and maintenance. FASEB J. 2013;27(8):3257–71.
d’Onofrio A, Cerrai P. A bi-parametric model for the tumour angiogenesis and antiangiogenesis therapy. Math Comput Model. 2009;49(5–6):1156–63.
De Palma M, Biziato D, Petrova TV. Microenvironmental regulation of tumour angiogenesis. Nat Rev Cancer. 2017;17(8):457–74.
Acknowledgements
The authors would like to thank Dr. Uma Kabra for reviewing their manuscript.
Author information
Authors and Affiliations
Contributions
Araiz Ali has done literature search, analyse and wrote manuscript. Jeena Gupta has reviewed, formatted and approve the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors have no conflicts of interest to declare.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ali, A., Gupta, J. Applications of Stem Cell Therapy and Adipose-Derived Stem Cells for Skin Repair. Curr Derm Rep 11, 120–130 (2022). https://doi.org/10.1007/s13671-022-00357-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13671-022-00357-6