Summary
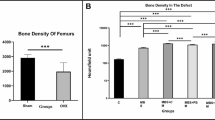
This study was designed to evaluate the effects of drilling through the growth plate and using adipose-derived stem cells (ADSCs) and bone morphogenetic protein-2 (BMP-2) to treat femoral head epiphyseal ischemic necrosis, which can be done in juvenile rabbits. Passagefour bromodeoxyuridine (BrdU)-labeled ADSCs were cultured, assayed with MTT to determine their viability and stained with alizarin red dye to determine their osteogenic ability. Two-month-old, healthy male rabbits (1.2 to 1.4 kg, n=45) underwent ischemic induction and were randomly divided into five groups (group A: animal model control; group B: drilling; group C: drilling & ADSCs; group D: drilling & BMP-2; and group E: drilling & ADSCs & BMP-2). Magnetic resonance imaging (MRI), X-ray imaging, hematoxylin and eosin staining and BrdU immunofluorescence detection were applied 4, 6 and 10 weeks after treatment. Approximately 90% of the ADSCs were labeled with BrdU and showed good viability and osteogenic ability. Similar results were observed in the rabbits in groups C and E at weeks 6 and 10. The animals of groups C and E demonstrated normal hip structure and improved femoral epiphyseal quotients and trabecular areas compared with those of the groups A and B (P<0.01). Group D demonstrated improved femoral epiphyseal quotients and trabecular areas compared with those of groups A and B (P<0.05). In summary, drilling through the growth plate combined with ADSC and BMP-2 treatments induced new bone formation and protected the femoral head epiphysis from collapsing in a juvenile rabbit model of femoral head epiphyseal ischemic necrosis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Maisuradze TG. Complex conservative treatment of legg-calve-perthes disease with dona-glucosamine sulfate (sachet). Georgian Med News (Russian), 2012,205(205):58–67
Johansson T, Lindblad M, Bladh M, et al. Incidence of Perthes’ disease in children born between 1973 and 1993. Acta Orthop, 2017,88(1):96–100
Guerado E, Caso E. The physiopathology of avascular necrosis of the femoral head: an update. Injury, 2016,47(Suppl 6):S16–S26
Salter RB. Legg-Perthes disease: the scientific basis for the methods of treatment and their indications. Clin Orthop Relat Res, 1980,150(150):8–11
Aruwajoye OO, Patel MK, Allen MR, et al. Microcrack density and nanomechanical properties in the subchondral region of the immature piglet femoral head following ischemic osteonecrosis. Bone, 2013,52(2):632–639
Stanitski CL. Hip range of motion in Perthes’ disease: comparison of pre-operative and intra-operative values. J Child Orthop, 2007,1(1):33–35
Mehta JS, Conybeare ME, Hinves BL, et al. Protein C levels in patients with Legg-Calve-Perthes disease: is it a true deficiency? J Pediatr Orthop, 2006,26(2):200–203
Nakamura N, Inaba Y, Machida J, et al. Rotational open-wedge osteotomy improves treatment outcomes for patients older than eight years with Legg-Calve-Perthes disease in the modified lateral pillar B/C border or C group. International Orthopaedics (SICOT), 2015,39(7):1359–1364
Young ML, Little DG, Kim HK. Evidence for using bisphosphonate to treat Legg-Calvé-Perthes disease. Clin Orthop Relat Res, 2012,470(9):2462–2475
Little DG, Kim HK. Future biologic treatments for Perthes disease. Orthop Clin North Am, 2011,42(3):423–427
Murphey MD, Roberts CC, Bencardino JT, et al. ACR appropriateness criteria osteonecrosis of the hip. J Am Coll Radiol, 2016,13(2):147–155
Kim SS, Lee CW, Kim HJ, et al. Treatment of lateonset Legg-Calve-Perthes disease by Arthrodiastasis. Clin Orthop Surg, 2016,8(4):452–457
Elzohairy MM. Short follow-up evaluation of proximal femoral varus osteotomy for treatment of Legg-Calvé-Perthes disease. J Orthop Traumatol, 2016,17(4):345–351
Ibrahim T, Little DG. The pathogenesis and treatment of Legg-Calvé-Perthes Disease. JBJS Rev, 2016,4(7):e4
Shohat N, Copeliovitch L, Smorgick Y, et al. The long-term outcome after varus derotational osteotomy for Legg-Calvé-Perthes disease: a mean follow-up of 42 years. J Bone Joint Surg Am, 2016,98(15):1277–1285
Kong SY, Gong SY, Kim HW, et al. Effects of multiple drilling on the ischemic capital femoral epiphysis of immature piglets. Yonsei Med J, 2011,52(5):809–817
Han N, Li Z, Cai Z, et al. P-glycoprotein overexpression in bone marrow-derived multipotent stromal cells decreases the risk of steroid-induced osteonecrosis in the femoral head. J Cell Mol Med, 2016,20(11):2173–2182
Rubessa M, Polkoff K, Bionaz M, et al. Use of pig as a model for mesenchymal stem cell therapies for bone regeneration. Anim Biotechnol, 2017(2):1–13
Guo SC, Tao SC, Yin WJ, et al. Exosomes from human synovial-derived mesenchymal stem cells prevent glucocorticoid-induced osteonecrosis of the femoral head in the rat. Int J Biol Sci, 2016,12(10):1262–1272
Zou Y, Fisher PD, Horstmann JK, et al. Synergistic local drug delivery in a piglet model of ischemic osteonecrosis. Journal of Pediatric Orthopaedics B, 2015,24(6):483–492
Yang XF, He X, He J, et al. High efficient isolation and systematic identification of human adipose-derived mesenchymal stem cells. J Biomed Sci, 2011,18(1):1–9
Xiao C, Zhou H, Liu G, et al. Bone marrow stromal cells with a combined expression of BMP-2 and VEGF-165 enhanced bone regeneration. Biomed Mater, 2011,6(1):015013
Tachi K, Takami M, Zhao B, et al. Bone morphogenetic protein 2 enhances mouse osteoclast differentiation via increased levels of receptor activator of NF-?B ligand expression in osteoblasts. Cell Tissue Res, 2010,342(2):213–220
Boss JH, Misselevich I. Osteonecrosis of the femoral head of laboratory animals: the lessons learned from a comparative study of osteonecrosis in man and experimental animals. Vet Pathol, 2003,40(4):345–354
Teves ME, Sundaresan G, Cohen DJ, et al. Spag17 deficiency results in skeletal malformations and bone abnormalities. PLoS One, 2015,10(5):e0125936
Yu Z, Zhu T, Li C, et al. Improvement of intertrochanteric bone quality in osteoporotic female rats after injection of polylactic acid-polyglycolic acid copolymer/collagen type I microspheres combined with bone mesenchymal stem cells. Int Orthop, 2012,36(10):2163–2171
Zhang P, Liang Y, Kim H, et al. Evaluation of a pig femoral head osteonecrosis model. J Orthop Surg Res, 2010,5(1):1–7
Martínez-Álvarez S, Epeldegui-Torre T, Manso-Díaz G, et al. Experimental induction of Perthes disease in lambs. Rev Esp Cir Ortop Traumatol, 2014,58(2):68–77
Norman D, Reis D, Zinman C, et al. Vascular deprivation-induced necrosis of the femoral head of the rat. An experimental model of avascular osteonecrosis in the skeletally immature individual or Legg-Perthes disease. Int J Exp Pathol, 1998,79(3):173–181
Kim HK, Su PH. Development of flattening and apparent fragmentation following ischemic necrosis of the capital femoral epiphysis in a piglet model. J Bone Joint Surg Am, 2002,84:1329–1334
Atsumi T, Yamano K, Muraki M, et al. The blood supply of the lateral epiphyseal arteries in Perthes’ disease. J Bone Joint Surg Br, 2000,82(3):392–398
Kim HK, Morgan-Bagley S, Kostenuik P. RANKL inhibition: a novel strategy to decrease femoral head deformity after ischemic osteonecrosis. J Bone Miner Res, 2006,21(12):1946–1954
Dahners LE, Hillsgrove DC. The effects of drilling on revascularization and new bone formation in canine femoral heads with avascular necrosis: an initial study. J Orthop Trauma, 1989,3(4):309–312
Shi F, Chen Q, Liu W. Different diameters injury of epiphyseal plate impact on the development of growth. Chin J Pediatic Surg (Chinese), 2002,23:53–55
Houdek MT, Wyles CC, Martin JR, et al. Stem cell treatment for avascular necrosis of the femoral head: current perspectives. Stem Cells Cloning, 2014,7:65–70
Tanzer M, Bobyn JD, Krygier JJ, et al. Histopathologic retrieval analysis of clinically failed porous tantalum osteonecrosis implants. J Bone Joint Surg Am, 2008,90(6):1282–1289
Fennema EM, Tchang LAH, Yuan H, et al. Ectopic bone formation by aggregated mesenchymal stem cells from bone marrow and adipose tissue: a comparative study. J Tissue Eng Regen Med, 2017
Hacobian AR, Posa-Markaryan K, Sperger S, et al. Improved osteogenic vector for non-viral gene therapy. Eur Cells Mater, 2016,31(31):191–204
Vanhatupa S, Ojansivu M, Autio R, et al. Bone morphogenetic protein-2 induces donor-dependent osteogenic and adipogenic differentiation in human adipose stem cells. Stem Cells Transl Med, 2015,4(12):1391–1402
Qing W, Guang-Xing C, Lin G, et al. The osteogenic study of tissue engineering bone with BMP2 and BMP7 gene-modified rat adipose-derived stem cell. J Biomed Biotechnol, 2012,2012:410879
Jain A, Kebaish KM, Sponseller PD. Factors associated with use of bone morphogenetic protein during pediatric spinal fusion surgery: an analysis of 4817 patients. J Bone Joint Surg Am, 2013,95(14):1265–1270
Gressot LV, Patel AJ, Hwang SW, et al. Rh-BMP-2 for L5-S1 arthrodesis in long fusions to the pelvis for neuromuscular spinal deformity in the pediatric age group: analysis of 11 patients. Childs Nerv Syst, 2014,30(2):249–255
Vandermeer JS, Kamiya N, Aya-ay J, et al. Local administration of ibandronate and bone morphogenetic protein-2 after ischemic osteonecrosis of the immature femoral head: a combined therapy that stimulates bone formation and decreases femoral head deformity. J Bone Joint Surg Am, 2011,93(10):905–913
Kim HK, Aruwajoye O, Du J, et al. Local administration of bone morphogenetic protein-2 and bisphosphonate during non-weight-bearing treatment of ischemic osteonecrosis of the femoral head: an experimental investigation in immature pigs. J Bone Joint Surg Am, 2014,96(18):1515–1524
Açil Y, Zhang X, Nitsche T, et al. Effects of different scaffolds on rat adipose tissue derived stroma cells. J Craniomaxillofac Surg, 2014,42(6):825–834
Acknowledgements
We thank the Third Xiangya Hospital of Central South University for supporting our research.
Author information
Authors and Affiliations
Corresponding authors
Additional information
This project was supported by the National Natural Science Foundation of China (No. 81572150, No. 81571939, No. 81301636 and No. 81772134), the Natural Science Foundation of Hunan Province (No. 13JJ2013 and No.2015JJ2187), and the Wu Jie-Ping Medical Foundation of the Minister of Health of China (No. 320.6750.14118).
Rights and permissions
About this article
Cite this article
Wang, Zl., He, Rz., Tu, B. et al. Drilling Combined with Adipose-derived Stem Cells and Bone Morphogenetic Protein-2 to Treat Femoral Head Epiphyseal Necrosis in Juvenile Rabbits. CURR MED SCI 38, 277–288 (2018). https://doi.org/10.1007/s11596-018-1876-3
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-018-1876-3