Abstract
Background
Diabetes mellitus is a chronic metabolic disorder that affects 537 million of the population worldwide whereby continuous glucose monitoring (CGM) has been implemented in the management of diabetes.
Introduction
CGM tracks glucose levels for 24 h without interruption via sensor detection which provides a large data set for blood glucose prediction in diabetic patients. By incorporating the Internet-of-Things healthcare systems into wearable CGM devices, the artificial intelligence-based CGM models facilitate diabetes management by assisting with blood glucose trend analysis, blood glucose profile and diabetic risk prediction, early warning of the potential glycemic events predicted, and insulin dose optimization.
Conclusion
The development of AI-based technology has improved the overall outcome of diabetes management. The AI algorithms with different approaches are helpful in clinical decision-making and health-related data tracking, particularly in diabetes glucose management.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Background
Diabetes mellitus (DM) is a complicated long-term metabolic disorder that can be further categorised into type 1, type 2, and gestational diabetes according to the American Diabetes Association (ADA) classification [1]. However, it is suggested that an etiologic-based classification should be introduced due to the emergence of atypical DM phenotypes including monogenic diabetes and drug or chemical-induced diabetes [1]. According to the current ADA classification, type 1 diabetes can be further categorised into type 1A and type 1B based on their respective causes of pancreatic beta-cell destruction and dysfunction. Type 1A diabetes is an autoimmune disorder indicated by the occurrence of circulating islet cell antibodies targeting the pancreatic beta cell antigens (ICA or GAD65), whereas type IB diabetes is defined as idiopathic due to the unknown cause of the beta-cell destruction and dysfunction. On the other hand, T2D is characterised by absolute insulin deficiency and insulin resistance resulting from “glucotoxicity”, by which hyperglycaemia induces and exacerbates defective insulin secretion and insulin resistance.
Based on the International Diabetes Federation (IDF) Atlas Report [2], it is stated that the number of 20- to 79-year-old adults currently living with diabetes is estimated at 537 million, with a predicted increment of 11.3% to 643 million by 2030 and a 12.2% increment to 783 million by 2045. IDF estimates that 8.75 million individuals worldwide currently live with T1D, where individuals from low-income and lower-income countries account for 20% of the T1D population, represented by a number of 1.9 million [3]. An estimation performed by the IDF also states that newly diagnosed T1D cases in populations < 15 years worldwide contribute to a total number of > 108,000, and this rises to 150,000 with an age adjustment to a population of < 20 years [4]. On the other hand, T2D accounts for 90% of the diabetes cases reported, where the North African and Middle Eastern populations have the greatest prevalence, indicated by a rate of 16.2% [2].
Diabetes-related complications include macrovascular and microvascular events in which an elevated risk is observed in individuals with young-onset T2DM [5, 6]. Macrovascular (i.e., cardiovascular) complications, include stroke, fatal or non-fatal myocardial infarction, and hospitalised heart failure. On the other hand, microvascular complications mainly include nephropathy, neuropathy, retinopathy, and their associated outcomes [6]. A study conducted by He et al. [6] in China showed that the risks of all-cause and cardiovascular disease CVD mortality and macrovascular and microvascular complications were higher in T2D individuals presenting with metabolic syndrome (MetS) (i.e., higher blood pressure, BMI, and triglyceride level) in comparison with those without MetS. Furthermore, diabetic patients are subjected to a higher risk of foot complications including foot ulcerations and infections, peripheral artery disease (PAD) co-existing with ulceration, and Charcot’s arthropathy that are mostly attributable to diabetic polyneuropathy, affecting around one in three diabetic patients [7].
The assimilation of AI in the healthcare sector enhances productivity in healthcare services. AI acts like a human brain to find, filter, organise, interpret, and analyse various types of information. Healthcare practitioners can use the AI database to record and quickly track medical information quickly [8]. In other words, AI-based electronic data sources replace manpower in processing complex and tedious tasks [9]. As such, it has greatly reduced the workload and time in documenting the data with the intervention of AI, thus enhancing the efficiency of healthcare services. For example, the information in electronic medical records is useful in detecting 6.5% out of 54,652 patients who missed radiology examination appointments [10]. This allows healthcare providers to reflect on the reasons for missed appointments and instantly provide alternate solutions to enhance patient care. In brief, AI-based electronic health records support and leverage the efficiency of healthcare services and data tracking procedures.
On top of that, the AI system enables the personalisation of disease management strategy [11]. The applications of AI including wearables, mobile healthcare applications, and smart medical tools are good innovations that aid in formulating a patient’s care plan. These applications can monitor and record blood pressure, heart rate, blood glucose, active minutes, daily steps, calories burned, etc. AI systems can extract the raw data from these electronic health records and decipher the information through machine learning without human assistance [9, 12]. Doctors, nurses, and pharmacists can understand the needs of their patients through deep learning AI data analysis. Next, AI is employed to help doctors to predict the disease’s prognosis. The predictive model generated through an AI algorithm serves as a reference for the healthcare professional in making a clinical decision [13]. In a systematic review, Lee and Yoon [13] reported that AI-based technologies are superior to trained medical staff in terms of accuracy. Another study by Mitsala et al. [14] suggested that the integration of AI algorithms results in better clinical outcomes in terms of anticipation of colon cancer recurrence risk, detection of gene mutation, and screening of missed adenomas for colorectal cancer diagnosis. The adaptation of AI minimises human errors and improves the accuracy of the diagnoses of the patients. Now, healthcare professionals are able to make better judgements based on the statistics and algorithms predicted using AI. In short, AI enhances a comprehensive approach toward disease management by tailoring patients’ needs.
Moreover, the adoption of AI improves the quality of healthcare services. AI-based surgical robots assist surgeons in achieving high accuracy and precision around delicate organs or surgical sites such as the heart, lungs, and brain. The potential advantages of the surgical robots over the conventional approach include less post-operative discomfort, smaller surgical incisions, faster recovery time, and less scarring. For instance, the Retzius-sparing robotic-assisted radical prostatectomy (RARP) demonstrated a success rate of 23% in a clinical trial involving 17 patients [15]. In addition, the innovation of digital voice assistants such as Alexa (Amazon), Siri (Apple), and Google Assistant (Google) functions under voice commands allows easier access to medical information [13, 16]. Patients can utilise this virtual assistance to call for help during medical emergencies. The robots are employed to assist the administrative workflow.
Furthermore, AI facilitates the drug discovery processes which significantly shortens the time required for pre-clinical studies. AI-assisted drug development has accelerated the emergence of new formulations with improved physiochemical characteristics, such as greater biodegradability and drug release mechanisms. For instance, Deng et al. [17] constructed a prediction model by incorporating machine learning algorithms to predict the in vitro drug-release profiles of the microsphere-based drug delivery systems. Other than that, AI also contributed additional options for disease management apart from the standard therapies. For instance, the application of computer-aided design in 3D bioprinted scaffolds allows better diabetic wound healing in diabetic patients with foot ulceration [18]. In summary, AI-based technologies improve the quality of healthcare and increase the success rate of drug discovery processes.
The inception of AI in healthcare has automatically led to its significant involvement in the care of many medical conditions. Due to the vast improvements, AI can potentially contribute to many healthcare aspects such as diagnosing conditions, assessing risks, home monitoring, and managing lifestyle [19], along with the high prevalence of diabetes worldwide (463 million adults diagnosed in 2019) which is forecasted to increase if proper preventive-based intervention is not established [20], it is clear that the diabetic population and even the healthcare personnel would appreciate the immense help AI can provide to them. At the time of writing this paper, there have been applications and uses of AI in the management of diabetes.
Perhaps one of the most impressive utilisations of AI in diabetes management is the monitoring of health complications that arise from diabetes, particularly diabetic retinopathy [11, 21, 22]. By using retinal fundus images from adult diabetic patients, AI can automatically diagnose and assess diabetic retinopathies in those patients via a deep learning algorithm [11, 23, 24]. The diagnosis of the eye condition using this AI-based method was found to have a sensitivity and specificity of more than 93% for both parameters [11]. Moreover, a new AI disease grading system for the classification of the severity of diabetic retinopathies based on deep learning was developed [11]. The high reliability of AI-based diagnosis for this diabetes complication together with the development of a potentially useful assessment tool can aid ophthalmologists in efficiently screening for diabetic retinopathy. Another important part of diabetes management is ensuring a daily diet is appropriate for controlling blood glucose levels, in which AI can play a huge role in assisting individuals with this non-pharmacological management. There are software applications under development that aim to analyse dietary content such as carbohydrate and calorie content directly and rapidly by utilising graphic analysis technologies, namely mobile phone cameras [11, 25]. By analysing pictures of meals driven by technology involving remote communications and deep learning, patients are able to acquire relevant nutrition information [11]. According to a 12-month randomised controlled experiment, nutritional intervention through the usage of AI had a similar average variation in HbA1c level (0.3%) with a standard deviation of 0.3% when compared with dietitians providing in-person guidance about nutritional intervention [11], indicating that AI can complement and reduce the burden of dietitians in giving dietary guidance to patients as well as potentially empowering patients to take charge of their diabetic condition.
Besides AI applications in diabetes, AI is also used in various metabolic disorders. For example, electronic health record (EHR) data can facilitate the identification of patients with familial hypercholesterolaemia (FH), which is another example of a metabolic disorder that affects lipid metabolism [26,27,28]. Banda et al. [27] developed a Random Forest classifier model derived from EHR data of FH patients and non-FH patients, and the authors concluded that this supervised machine learning approach is a practical strategy to detect patients that have the possibility of FH with an 88% positive predictive value, 99% specificity, and 75% sensitivity. In addition, the diagnosis aspect of hypertension, another type of metabolic disorder, has also seen significant improvements thanks to advancements in AI [26, 29, 30]. Moreover, AI approaches namely deep learning (DL), machine learning (ML), and artificial neural network (ANN) have been used in diagnosing hypertension by studying datasets containing specific variables from different human subjects [30].
Evidently, from the examples of AI applications in the management of metabolic disorders above, the application of AI technology can lead to significant improvement in the care of various medical conditions in patients. In diabetic care, frequent blood glucose monitoring is important not only to ensure that the blood sugar level does not fluctuate drastically and is within an acceptable limit but also to assess the therapeutic responses of glucose-lowering medications, especially insulin therapy, and non-pharmacological management and adjust them accordingly depending on the glycemic state of the patients [31]. In contrast to the usual self-monitoring blood glucose (SMBG) which requires frequent testing each day, CGM is a promising alternative blood glucose monitoring approach that can potentially solve some of the restrictions of SMBG, such as the incapability to predict imminent hypoglycaemia and/or hyperglycaemia [32, 33]. Similar to other management aspects of diabetic care mentioned above, it is postulated that incorporating AI into continuous glucose monitoring can drastically improve this aspect of diabetic care. This review is mainly focused on discussing AI-based prediction of glycemic events based on continuous glucose monitoring data to further improve diabetic care.
Role and application of AI in the healthcare sector
Digital healthcare
“AI-assisted diagnosis and treatment” of diseases in digital healthcare are defined as processes of diagnosing and treating illnesses with the utilisation of AI technology by healthcare providers in their decision-making [34]. These include diagnostic criteria, therapeutic care plan designation and execution, monitoring criteria, and treatment optimisation [34]. AI technology plays a crucial role in the emergence of digital healthcare by assisting and enabling the early disease diagnosis and prevention and early prediction of patient mortality through data-driven approaches including ML and DL [34, 35]. For instance, a study by Muscogiuri et al. [36] stated that the AI technology application in cardiovascular imaging for the diagnosis of cardiovascular diseases enables fast image analysis and high-quality image production. In addition, research by Chiou et al. [37] showed that AI-assisted echocardiography increases the outcome accuracy of the diagnosis of heart failure by minimising both intra- and inter-observer variation caused by clinicians with less experience. The AI-enabled echocardiography could also be used as a standalone test in the autodetection of pulmonary hypertension that exhibits high sensitivity and specificity suggested by Liu et al. [38].
Other than diagnosing use, AI application in smart health monitoring (SHM) plays a significant role in providing rapid, timely, economical, and dependable health monitoring services from distant locations [35]. SHM incorporates the Internet of Medical Things (IoMT) that allows data exchange through the interconnection of medical electronic devices, and the integration of AI enables the processing of data acquired, generating responses or solutions to the problem identified [39, 40]. For instance, AI-assisted wearable biosensors in various forms such as wristbands and e-patches detect health-related biochemical or electrophysiological signals through the body surface and convert the signals into the recognised form for data processing by machine learning (ML) [41, 42]. Besides health monitoring, AI is also extensively studied and utilised in epidemic monitoring including virus spreading patterns analysis, virus transmission prediction, and pandemic risk and severity assessments [43,44,45].
Healthcare supply chain
A healthcare supply chain (HCM) is a sophisticated network structure consisting of a series of components including manufacturers, suppliers, hospitals, pharmacies, and blood banks, comprising a key component of the healthcare sector aimed to provide high-quality healthcare service and satisfy the demand [46]. The function of AI in HCM management is to facilitate supply chain efficiency by guaranteeing a resilient and seamless flow of materials, inventories, and information throughout the processes [47]. AI technology is implemented in HCM restructuring to generate an omnichannel strategy which refers to a business tactic that provides a seamless customer purchasing experience across all online, mobile, and in-store platforms [48, 49]. Besides that, a study by Azadi et al. [46] showed that AI technologies such as network data envelopment analysis and deep learning could be implemented in HCM sustainability forecasting which predicts the future performance of the HCM and consequences of supply chain inefficiency by identifying the possible factors causing inefficient supply chain in the future. In addition to that, AI is applied in the innovation of AI-enhanced medical drones in the HCM sustainability approach in Ghana to promote the on-time delivery of emergency healthcare supplies and reduce the mortality rates in emergency clinical cases [50].
Current update on diabetes management and limitations
The management of diabetes mellitus is constantly evolving in accordance with the latest evidence-based clinical practice guidelines. Yu et al. [51] reviewed the recent updates made by the latest clinical guidelines on diabetic care published by the Korean Diabetes Association (KDA), Japan Diabetes Society (JDS), American Association of Clinical Endocrinology (AACE), American Diabetes Association (ADA), and European Association for the Study of Diabetes (EASD). From the review article [51], although the aforementioned scientific associations have slight variations in the recommendations regarding the target for glycemic control and initial glucose-lowering pharmacotherapy with or without existing comorbidities, there is a consensus made by scientific organisations on other aspects of diabetes management. Firstly, the individualisation of glycemic control targets is strongly advocated by most of the clinical guidelines [51]. In other words, each diabetic patient should follow their target blood glucose or glycosylated haemoglobin (HbA1c) instructed by their physicians to prevent complications associated with diabetes due to differences in clinical characteristics including the risk of extreme glycemic state, comorbidities, and behavioural factors [51,52,53]. Besides, the patient-centred approach should also be applied to glucose-lowering pharmacotherapy and prevention of diabetes-associated complications [51, 52]. Due to the inherent heterogeneous nature of the diabetes condition, individualisation of the management of this metabolic disorder should be implemented to enhance compliance and treatment outcomes [52, 54]. However, perhaps the most remarkable update to the current guidelines on diabetes management is the recommendation for CGM application in the management of diabetes, which is also the focus of this literature review. The updates on the new monitoring modality in the aforementioned clinical guidelines include an extension of the recommendation of CGM for different groups of diabetic patients such as diabetic children, patients on intensive insulin therapy (which is defined as greater or equal to three daily injections or usage of an insulin pump) and long-acting (basal) insulin, and the preference of real-time CGM over intermittent CGM in the case of problematic hypoglycaemia requiring anticipating alert [51]. Most of the latest clinical guidelines acknowledge the advancement in technology to benefit diabetes management, particularly glycemic control by using CGM.
However, despite constant updates on diabetes management, some limitations still exist. Although improvements in current diabetes management guidelines are made, the attainment of treatment outcomes ultimately depends on the patients’ initiatives to improve their health. In addition, there are limitations and challenges pertaining to the clinical implementation of CGM. Firstly, the implementation of a CGM device can be very expensive. Although Wan et al. [55] demonstrated that CGM is cost-effective in addition to improved glycemic control, the noticeably higher cost of CGM when compared to SMBG may impede a patient’s decision to use CGM. Besides, as CGM measures the glucose level in the interstitial fluid instead of directly measuring the glucose level in the blood capillary, CGM can sometimes be unreliable as there is a lag time before the glucose levels in the interstitial fluid and blood capillary equilibrate [32, 56, 57]. This problem can be clinically significant if there is a high fluctuation in blood glucose levels, which can delay the treatment of hyperglycaemia or hypoglycaemia [56]. Nonetheless, as CGM can improve the quality of life in patients and also provide better glycemic control to prevent hyperglycaemia and hypoglycaemia, the benefits should therefore outweigh the limitations.
Importance of continuous glucose monitoring in diabetic management
Continuous glucose monitoring (CGM) tracks glucose levels under the skin for 24 h without interruption via sensor detection. Unlike blood glucose monitoring, a CGM device involves the insertion of a small sensor under the skin to detect the presence of glucose in the interstitial fluids and transmit signals to a monitor or smartphone directly every 5 min [58,59,60]. The use of microneedle-based systems for diabetes has led to the emergence of painless drug delivery and monitoring whereby the frequent invasive blood sampling procedures for BG monitoring could be avoided, significantly contributing to an increase in patients’ compliance towards the management of the diseases [61,62,63].
The CGM system is able to present a robust diabetes patient glucose profile. It provides useful information about the glycemic status and displays the trend of glucose fluctuation on the monitor screen [64, 65]. Hence, diabetic patients can observe and review their glycemic variability. The glucose fluctuation might be due to changes in medications, food intake, exercise, and stress level. The device will signal the patients when the glucose trend reaches the minimum or maximum baselines [66]. As such, patients can change their habits accordingly and improve their glycemic control. It has been proven that CGM significantly impacts the reduction of hypoglycaemia incidents by 17% (from 33 to 50%) [65]. Therefore, CGM is very suitable for diabetic patients prone to hyperglycaemia or hypoglycaemia as it offers a bigger picture of the glucose profile.
Additionally, CGM data backs up other glucose measurements. A study showed that CGM data is helpful when SMBG and HbA1c testing are insufficient to forecast glucose level changes. A few studies reported some improvement in HbA1c values for T1D patients, whereas some found that T2D patients have better glycemic management with the help of CGM [58, 66, 67]. In recent research, the use of flash CGM accounted for 1.1% and 1.6% of HbA1c reduction for the insulin group and non-insulin group, respectively, for T2D patients with a baseline of > 8% [68]. Although CGM is superior to SMBG, CGM should be carried out along with self-monitored blood glucose to ensure a more accurate glucose reading.
Application of AI in diabetes management through continuous glucose monitoring data
Types of continuous glucose monitoring (CGM) systems
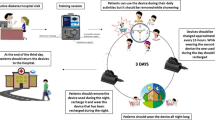
There are various types of CGM systems on the market. However, each CGM system requires a doctor’s prescription to purchase. Each CGM device has a different working mechanism, durability, and special features as shown in Table 1. Therefore, the users should choose the CGM systems based on their preferences. An example of a CGM system that detects interstitial fluid glucose levels is illustrated in Fig. 1.
Illustration of a continuous glucose monitoring (CGM) system. The sensor of the CGM system measures the interstitial fluid glucose concentration and transmits the signals to a device. Figure comes under CC-BY licence [119]
Dexcom®
Dexcom® is one of the integrated CGM systems to measure real-time glucose readings [69]. The function of the needle in the applicator of Dexcom® is to ease the insertion of the applicator through the skin in the interstitial fluid. Then, the needle will retract into the applicator and the sensor will remain on top of the skin. The user will experience a slight pinch during the insertion of this minimally invasive device. Upon insertion, the sensor detects the fluctuation in glucose levels within the interstitial fluid continuously and sends the real-time reading to the monitoring devices through signal transmission. Users are able to check their glucose data through smartphones at any time. Thus, users and healthcare professionals are able to interpret the glucose trends and adjust the acute and long-term treatment accordingly. As such, this helps detect hypoglycaemia and hyperglycaemia incidence. The sensor is advised to be placed on the upper buttocks, abdomen, and back of the upper arm. Moreover, the Dexcom® system can communicate with digital devices autonomously [69]. Dexcom® CGM is suitable to be used in persons aged ≥ 2 years. Those users who are afraid of needles are encouraged to use Dexcom® CGM as it does not require fingersticks or calibrations in diabetes management [69]. As such, the users have the freedom to take control of their diabetes management. The device will alert the users 20 min in advance when their glucose level reaches the maximum or minimum baselines. Pregnant women with T1D or gestational diabetes can also benefit from using the Dexcom ® CGM system [69]. However, the Dexcom® CGM is contraindicated with computed tomography (CT) scan, magnetic resonance imaging (MRI), or high-frequency electrical heat treatment as the magnetic field and heat may disturb the sensor readings, thus reducing the accuracy of glucose measurement. Not only that, the administration of acetaminophen (> 1000 mg) will affect the Dexcom® CGM reading [70, 71].
The overall performance of the Dexcom® CGM system has a remarkable enhancement from Dexcom® 5th generation (G5) to 7th generation (G7). The Dexcom® G7 is the easiest to insert, requires the least warm-up interval, covers the most data storage, and has the longest life span compared to other Dexcom® systems. In comparison with other systems that are placed on the arm and abdomen, the arm-placed Dexcom® G7 showed the lowest mean absolute relative differences (MARD) and the highest unadjusted and adjusted accuracy metrics [72]. Besides, the Dexcom® G7 system demonstrated the lowest missed hypoglycaemia detection rate than earlier systems at 55 mg/dL glycemic profile threshold [72]. However, all three systems did not show prominent differences in terms of hypoglycaemia detection rate at 70 mg/dL glycemic profile threshold and false hypoglycaemia alert rate at 55 mg/dL glycemic profile threshold [72]. The improvement and usability over different generations of Dexcom® CGM systems offer sustainable glycemic control and thus improve clinical outcomes. The addition of a permselective membrane in the Dexcom® G6 resolves the interference of acetaminophen [71]. Moreover, Chellan et al. [73] summarised that the implementation of the Dexcom® G6 can reduce HbA1C in T1D patients with comorbidities by 2% and reduce the development risk of cataracts and heart failure by 19% and 16%, respectively. In other words, the CGM system allows the patients to have better glycemic control and thus improve their overall well-being and quality of life. Oppel et al. [74] suggested that the Dexcom® sensor is suitable for diabetic patients with allergic contact dermatitis to isobornyl acrylate found in Freestyle Libre™ for glucose monitoring.
Freestyle Libre™
Freestyle Libre™ is another type of CGM system that measures Flash Glucose Monitoring (FGM). In contrast with the CGM system’s real-time glucose monitoring, FGM requires manual scanning with the sensor to retrieve glucose readings [75]. In 2017, the FDA approved the Freestyle Libre™ as a CGM system for diabetes management [69]. Similar to the Dexcom® system, FreeStyle Libre™ offers a complete picture of the glucose trend and patterns as it measures the interstitial glucose levels for 24 h. This FreeStyle Libre™ is suitable to be used for aged ≥ 18 years diabetic patients [69]. The features of FreeStyle Libre™ include a scannable sensor that can last up to 14 days wear time, a handheld reader, and a trend arrow indicating the glucose change [75]. The trend arrow icons on the reader act as visual aids to allow the users to have an insight into their glucose change. Notably, an upward trend arrow indicates a rise in glucose concentration, a downward trend arrow refers to glucose reduction, and a flat trend arrow indicates slight changes in glucose. The directionality of the trend arrows reminds the patients who use multiple daily insulin injections to adjust their insulin therapy according to their glycemic variation to prevent hypoglycaemia or hyperglycaemia incidents [75]. This FreeStyle Libre™ system should not be used in pregnant ladies and dialysis patients. Diabetic patients may need to take note of certain active ingredients such as salicylic acid (≥ 650 mg) and ascorbic acid (≥ 500 mg) that may affect the FreeStyle Libre™ system’s glucose readings [75]. Fortunately, the administration of acetaminophen will not interfere with the Freestyle libre™ CGM system reading [76].
Yaron et al. [77] reported that the HbA1c value of T2D patients on multiple daily insulin injections was reduced by 0.82% with the aid of the FGM system for 10 weeks. This FGM technology also gained high satisfaction among diabetic patients [77]. Miller et al. [78] found that the intervention of the FreeStyle Libre™ system has shown a significant risk reduction of 0.019 acute diabetes events per patient-years in T2D patients who are not receiving bolus insulin treatment for at least 6 months. The evidence above has proven the intervention of the FreeStyle Libre™ system to have better clinical outcomes in diabetes management. Evans et al. [79] demonstrated that the use of the FreeStyle Libre™ system shows a sustained HbA1c reduction of 0.53% and 0.45% in T1D and T2D patients, respectively.
DBLG Diabeloop ©
Diabeloop Generation 1 (DBLG1©) is another CGM creation that measures the real-time glucose reading at 5-min intervals. This DBLG1© technology works in a hybrid closed-loop system by pairing a CGM device with an AID pump and a digital device through a Bluetooth® connection [80]. The interesting traits of the DBLG1© system include flexible adjustment of the parameters such as target and hypoglycaemia or hyperglycaemia thresholds, automated optimisation of weight, insulin dose, basal rate profile, and meal ratios, as well as a comprehensive history of user’s medical, medication, meal, and exercise inputs [80]. DBLG1© has an algorithm to analyse the glycemic readings from the CGM device and transfer the updated data to the insulin pump. Then, the AID pump will administer the calculated dose of insulin into the user’s body. The insulin pump is suitable for users aged 8 years and above. This DBLG1© offers a seamless insulin delivery process in diabetes management.
Amadou et al. [81] revealed that the DBLG1© system showed a mean HbA1c reduction of 0.8% and a mean TIR increment of 33% over 6 months. This indicates that the DBLG1© system can improve the users’ glycemic control. In comparison with the conventional sensor-assisted insulin pumps, the DBLG1© system presented a better outcome in terms of reducing the mean HbA1c, increasing the proportion of time spent at desired glucose concentration range, and decreasing the duration of hypoglycaemia incidence in T1D adults for 12 weeks [82]. In short, the DBLG1© system can facilitate diabetes therapeutic management and ease the mental burden of the users.
Application of AI in diabetes management through continuous glucose monitoring data
CGM in the management of diabetes allows remote blood glucose level monitoring by incorporating IoT healthcare systems into wearable CGM devices [83]. These AI-based CGM models facilitate diabetes management through an increased understanding of the disease progression by interpreting the large data sets and transforming the predicted blood glucose profiles into therapeutic actions [84]. For instance, CGM system-integrated applications apply computer vision to assist blood glucose trend analysis, blood glucose profile and diabetic risk prediction, early warning of the potential glycemic events predicted, and insulin dose optimisation [85].
Blood glucose trend and glycemic variability prediction
AI-based algorithms incorporated into CGM systems can be trained to forecast future blood glucose (BG) profiles through the interpretation of the prior and current CGM data. For instance, machine learning (ML) is a group of AI algorithms that can self-learn and recognise patterns of the BG data, increasing its intelligence in BG prediction over time without human intervention [86]. The ML algorithms that can be utilised for BG prediction include linear regression, decision trees, neural networks, random forests, XGBoost, support vector machines, and more, whereby their high prediction accuracy has been widely reported [86,87,88]. In a study by Ahmed et al. [86], high BG estimation accuracy from the wearable device CGM data was reported by incorporating a combination of traditional and deep learning ML models, in which the accuracy was validated by a root mean square error (RMSE) range of 0.099 to 0.197. Besides that, a study by Doorn et al. [88] proposed ML models for BG prediction at 15- and 60-min intervals in T1DM individuals and reported a 5–10% deviation from the actual BG values, indicating the high accuracy of the ML models. Among the ML algorithms available, ANNs and DL are the most extensively applied techniques for blood glucose prediction [89].
Artificial neural network (ANN) is an ML algorithm-based AI inspired by the biological ability of the human brain to execute computations, which functions by simulating the neuronal activity in the human brain [90, 91]. The application of ANN in diabetes management has been extensively studied, such as diabetes prediction, diabetes diagnosis and prognosis, and BG level prediction [91,92,93]. Studies on the application of ANN in BG-level prediction have reported promising results in which high prediction accuracy was demonstrated. For instance, a study by Ali et al. [91] suggested that the ANN-predicted blood glucose values mostly aligned with the actual blood glucose values. This result was represented by the line graphs generated with minimal deviation occurring between peaks and troughs. The adaptive ANN model proposed in this study using CGM data as the single input outperformed other methods discussed in the literature as lower RMSE values were determined at each prediction horizon of 15 min (RMSE, 6.43 mg/dL), 30 min (RMSE, 7.45 mg/dL), 45 min (RMSE, 8.13 mg/dL), and 60 min (RMSE, 9.03 mg/dL) [91]. In addition to that, Alfian et al. [94] reported a higher degree of BG prediction accuracy achieved by the proposed multi-layer perceptron (MLP) model, which is a type of ANN, by incorporating time-domain features in the model. The greater prediction accuracy, especially for long-term prediction (PH, 60 min), was represented by the higher reduction in the RMSE generated [94].
On the other hand, deep neural networks (DNNs) have also been incorporated in studies to perform BG prediction where successful outcomes have been reported. DNN is a subset of ANN that involves multiple hidden layers between the output and input layers that are trained with high-performance computational resources in which the transparency of the computational processes is still in the midst of finding [95]. In a study by Dudukcu et. al [96], the decision-level fusion method encompassing three different DNNs including gated recurrent unit (GRU), long short-term memory (LSTM), and WaveNet were applied for BG prediction. Each DNN was tested individually, followed by the fusion of two networks (LSTM + GRU; LSTM + WaveNet; GRU + WaveNet), and lastly the fusion of all three networks (LSTM + GRU + WaveNet) [14]. The average prediction performance metrics obtained from different PHs in the study suggested the fusion of three networks as the most successful method [96]. Besides that, a BG forecasting model involving large heterogeneous data obtained from a total of 158 T1DM individuals was proposed by Cichosz et al. [97] to conquer the sample size limitation observed in published studies performed by other researchers. However, the NN model proposed failed to demonstrate the potential for a generalised approach due to limitations such as variations in CGM sensors and unidentical study cohorts [97].
Deep learning (DL) is a subset of ML that involves DNN as the primary algorithm, encompassing various neural network architectures including recurrent neural networks (RNNs), convolutional neural networks (CNNs), and multi-layer perceptron (MLP) to perform different tasks [98]. DL has also been extensively studied for its application in BG prediction where promising results have been shown. For instance, a study by Zhu et al. [99] applied a dilated CNN model using four inputs, including CGM data, carbohydrate intake, insulin event, and time index for BG prediction. High prediction accuracy was shown whereby the proposed model outperformed the published simple autoregressive models utilising the same CGM data as the only input. Besides that, Zhang et al. [100] also studied two separate DL models, including a DCNN model and a sequence-to-sequence long short-term memory (Seq-to-Seq LSTM) model for BG forecasting in T1DM patients. The results showed that the Seq-to-Seq LSTM model generated the greatest performance for BG prediction at the PH of 30 min [100]. Other than that, five RNN-based algorithms were applied in a study to generate two types of BG prediction models: univariate models and multivariate models, in which the former used CGM data as a single input, whereas the latter included carbohydrates and insulin information to the CGM data [101]. In this study, individualised BG prediction with high accuracy was investigated. Additionally, another study used an LSTM-based deep RNN model which generated BG forecasting data with fewer fluctuations in the BG prediction curve, through pre-processing of the CGM data with the Kaman smoothing technique [102].
Hypoglycemic risk prediction
The ability of AI-based algorithms to forecast blood glucose levels from CGM data also facilitates the early warning of potential hypoglycemic or hyperglycemic episodes in patients. In a study by Elhadd et al. [103], five distinct ML techniques were applied to predict the glycemic variability in T2DM patients who are fasting during Ramadan while on multiple diabetic drug therapies. The proposed machine learning models suggested an increased hypoglycaemia risk in patients who include sulphonylurea or SGLT-2 inhibitors in their complex diabetic regimen [103]. In contrast, Motaib et al. [104] applied ML models to predict poor glycemic control in non-fasting patients during Ramadan, whereby the study suggested that non-fasting women have a greater hypoglycaemia risk. Although CGM data were not used as input, the study provides fundamental knowledge for hypoglycaemia risk prediction using ML models in individuals who perform Ramadan. On the other hand, another study by Mosquera-Lopez [105] investigated the hypoglycaemia risk during and post-physical activities using mixed-effects linear regression and mixed-effects random forest, in which high prediction accuracy was reported. The applied ML model identified that post-exercise hypoglycaemia risk was associated with several factors, including insulin exposure before exercise, intensity and timing of the physical activity, and low blood glucose index before exercise [105]. Besides that, Tyler et al. [106] also predicted the BG change during physical activity by utilising ML models with CGM data as the input. Consequently, ML-based models can significantly contribute to accurate BG prediction, facilitating optimal diabetes management tailored to the patient’s blood glucose condition, especially in those who are on a multidrug regimen. The comparisons of the proposed models that are in the literature discussed are shown in Table 2. The illustrations of the specific machine learning model known as random forest and the prediction of hypoglycemia by using machine learning are displayed in Figs. 2 and 3, respectively.
Illustration of random forest classifier model. It uses CGM datasets from diabetic patients to train the model to predict hypoglycemic events. Figure comes under CC-BY licence [120]
Illustration of patient glucose level after a meal at different time points. a CGM data point showed no peaks due to the small carbohydrate intake by the patient. b Rapid decrease in glucose showed low peak after meal. c Rapid glucose depletion after a carbohydrate-rich diet showed a steep peak. d No peak and rapid fall after a meal after injecting insulin before a meal. Figure comes under CC-BY licence [121]
Technical and social challenges
Any implementation of novel ideas comes with inherent challenges, and AI-based continuous glucose monitoring is no exception. The application of AI/ML itself into continuous glucose monitoring certainly has challenges before it is available commercially and becomes a norm in diabetes management. Firstly, it is important to ensure that the vast amount of data used to train and programme the system is accurate and of top quality [107]. This can be a concern if the data collected for AI/ML training is heterogeneous, which is highly variable data resulting in inaccurate, low quality, and ambiguous data [107, 108]. Furthermore, bias and noise in the data collected, especially from health records from different clinics, must be taken into consideration as they can result in poor prediction results [107, 109]. Another factor that makes the implementation of AI-based CGM difficult is the high-cost burden that patients need to bear. Wan et al. [55] reported that the cost of using CGM was very expensive, where the total average cost for using CGM for 6 months was US$ 11,032, and the CGM device alone cost US$ 2554. Indeed, the implementation of AI into CGM will surely cost significantly more, further contributing to patients’ economic burden. The real-time monitoring of AI-based CGM requires internet connectivity, which is similar to any Internet of Medical Things (IoMT) device [107]. This can become an issue if patients live in remote areas where the lack of internet access can impede the utilisation of AI-based CGM. As implicated in the cost and requirement of internet connectivity to use AI-based CGM for better glycemic control, there will be inequality in access to this monitoring device due to socio-economic factors. In other words, wealthy patients who live in urban settings are more likely to have access to AI-based CGM compared with poorer patients living in rural areas. To expand the usage of AI-based CGM, doctors, who are usually the first healthcare personnel, need to implement this glycemic prediction tool for diabetic patients and trust that the device itself functions properly. It can be challenging for doctors to implement AI-based CGM if they do not trust the efficacy of the device, which could arise due to possible false-positive prediction of hyper-/hypoglycaemia; ensuring minimal false-positive occurrence is a challenge in AI-based CGM [110].
Privacy and security concerns
Generally, one of the primary reasons for patients’ willingness to utilise a medical service is due to their trust towards that particular service. Any factors that can diminish patients’ confidence in that specific service can compromise patients’ trust, leading to the unwillingness to use the particular medical service and subsequently lesser usage. In the case of AI-based CGM, patient data is the main concern. Despite the numerous benefits provided by implementing AI in healthcare, its implementation can also lead to challenges regarding patients’ data privacy. Normally, AI-based devices utilise the cloud to store patients’ data collected, and this can lead to the leaking and misuse of patients’ data [107, 111, 112]. The leaking and misuse of data can be apparent if the security of the system is not robust enough to prevent cyberattacks; current healthcare-related AI services are generally vulnerable to cyber threats, which can be exploited to steal patients’ data for various purposes, leading to a breach in patient confidentiality [113]. Breaching of data can result in severe consequences ranging from healthcare data being stolen or disclosed to even the death of the patients should data tampering occur [114]. The acquisition of datasets from patients to train the AI/ML models itself is also an issue of data privacy [114].
Patency
A patent is a legal right designed to protect the original inventor’s invention that is novel, non-obvious, and useful, while also prohibiting the making or usage of the specific invention by others within a stipulated period [115, 116]. Although the utilisation of CGM data for AI-based prediction of glycemic events is certainly useful as evidenced by the promising results shown in the applications of AI for CGM, the idea is not technically a novel and non-obvious invention, as there is a patent published regarding the application of neural network system, a type of machine learning, in CGM for predicting glycemic events. There is a patent with ongoing exclusive rights published in 2015 by Cameron et al. [117] pertaining to the neural network system, in which one of its uses consists of the prediction of glycemic events using CGM data (US9076107B2). Besides, Mikhno et al. [118] have published a patent that is currently awaiting an application grant regarding the usage of an ML-based system to estimate blood glucose (US20220039755A1).
Conclusion
In short, the development of AI-based technology has improved the overall outcome of diabetes management. The AI algorithms with ML, ANN, and DL approaches are helpful in clinical decision-making and health-related data tracking, particularly in diabetes glucose management. The innovation of CGM optimises the glucose control of diabetes patients without the need for human monitoring. However, high cost, biased data, false alarms, and patients’ data security remain the main challenges of the usage of AI-based CGM. Therefore, the continuous development of AI-based healthcare technology serves as a foundation for sustainable and comprehensive diabetes care.
Data availability
Not applicable.
Abbreviations
- CGM:
-
Continuous glucose monitoring
- AI:
-
Artificial intelligence
- ML:
-
Machine learning
- T2DM:
-
Type 2 diabetes mellitus
- ADA:
-
American Diabetes Association
- IDF:
-
International Diabetes Federation
- CVD:
-
Cardiovascular disease
- Mets:
-
Metabolic syndrome
- PAD:
-
Peripheral artery disease
- RARP:
-
Robotic-assisted radical prostatectomy
- EHR:
-
Electronic health record
- FH:
-
Hypercholesterolaemia
- DL:
-
Deep learning
- ANN:
-
Artificial neural network
- SMBG:
-
Self-monitoring blood glucose
- SHM:
-
Smart health monitoring
- HCM:
-
Healthcare supply chain
- KDA:
-
Korean Diabetes Association
- JDS:
-
Japan Diabetes Society
- AACE:
-
American Association of Clinical Endocrinology
- EASD:
-
European Association for the Study of Diabetes
- BG:
-
Blood glucose
- DNN:
-
Deep neural networks
- GRU:
-
Gated recurrent unit
- LSTM:
-
Long short-term memory
References
American Diabetes Association. Classification and diagnosis of diabetes. Standards of medical care in diabetes-2021. Diabetes care. 2021;44:S15-33.
International Diabetes Federation. IDF diabetes atlas tenth edition. 2021. Available at: https://diabetesatlas.org/atlas/tenth-edition/, cited on: 05-08-2023.
Ogle GD, Wang F, Gregory GA, Maniam J. IDF Atlas reports: Type 1 diabetes numbers in children and adults. 2022. Available at: https://diabetesatlas.org/idfawp/resource-files/2022/12/IDF-T1D-Index-Report.pdf, cited on: 05-08-2023.
Gandhi N, Nicholas F, Wareham J. Epidemiology of diabetes. Medicine. 2022;50(1):638–43.
Cho Y, Park H-S, Huh BW, Seo SH, Seo DH, Ahn SH, Hong S, Suh YJ, Kim SH. Prevalence and risk of diabetic complications in young-onset versus late-onset type 2 diabetes mellitus. Diabetes Metab. 2022;48(6):101389.
He S, Wang J, Zhang X, Qian X, Yan S, Wang W, Zhang B, et al. Long-term influence of type 2 diabetes and metabolic syndrome on all-cause and cardiovascular death, and microvascular and macrovascular complications in Chinese adults—a 30-year follow-up of the Da Qing diabetes study. Diabetes Res Clin Pract. 2022;191:110048.
Dewi F, Hinchliffe RJ. Foot complications in patients with diabetes. Surgery (Oxford). 2020;38(2):108–13.
Barlow C. Oncology Research: Clinical trial management systems, electronic medical record, and artificial intelligence. Semin Oncol Nurs. 2020;36(2):151005.
Kellar E, Bornstein S, Caban A, Crouthamel M, Celingant C, McIntire PA, et al. Optimizing the use of electronic data sources in clinical trials: the technology landscape. Ther Innov Regul Sci. 2017;51(5):551–67.
Harvey HB, Liu C, Ai J, Jaworsky C, Guerrier CE, Flores E, et al. Predicting no-shows in radiology using regression modeling of data available in the electronic medical record. JACR. 2017;14(10):1303–9.
Li J, Huang J, Zheng L, Li X. Application of artificial intelligence in diabetes education and management: present status and promising prospect. Front Public Health. 2020;8:173.
Jiang F, Jiang Y, Zhi H, Dong Y, Li H, Ma S, et al. Artificial intelligence in healthcare: past, present and future. Stroke Vasc Neurol. 2017;2(4):230–43.
Lee D, Yoon SN. Application of artificial intelligence-based technologies in the healthcare industry: opportunities and challenges. Int J Environ Res Public Health. 2021;18(1):271.
Mitsala A, Tsalikidis C, Pitiakoudis M, Simopoulos C, Tsaroucha AK. Artificial intelligence in colorectal cancer screening, diagnosis and treatment. A new era Curr Oncol. 2021;28(3):1581.
Chang KD, Abdel Raheem A, Choi YD, Chung BH, Rha KH. Retzius-sparing robot-assisted radical prostatectomy using the Revo-i robotic surgical system: surgical technique and results of the first human trial. BJU Int. 2018;122(3):441–8.
Clipper B, Batcheller J, Thomaz AL, Rozga A. Artificial intelligence and robotics: a nurse leader primer. Nurse Lead. 2018;16(6):379–84.
Deng J, Ye Z, Zheng W, Chen J, Gao H, Wu Z, et al. Machine learning in accelerating microsphere formulation development. Drug Deliv Transl Res. 2022;13(4):966–82.
Glover K, Mathew E, Pitzanti G, Magee E, Lamprou DA. 3D bioprinted scaffolds for diabetic wound-healing applications. Drug Deliv Transl Res. 2023;13(8):2096–109.
Tarumi S, Takeuchi W, Chalkidis G, Rodriguez-Loya S, Kuwata J, Flynn M, et al. Leveraging artificial intelligence to improve chronic disease care: methods and application to pharmacotherapy decision support for type-2 diabetes mellitus. Methods Inf Med. 2021;60:e32-43.
Akhtar S, Nasir JA, Ali A, Asghar M, Majeed R, Sarwar A. Prevalence of type-2 diabetes and prediabetes in Malaysia: a systematic review and meta-analysis. PLoS ONE. 2022;17(1):e0263139.
Padhy SK, Takkar B, Chawla R, Kumar A. Artificial intelligence in diabetic retinopathy: a natural step to the future. Indian J Ophthalmol. 2019;67:1004–9.
Huang X, Wang H, She C, Feng J, Liu X, Hu X, Chen L, Tao Y. Artificial intelligence promotes the diagnosis and screening of diabetic retinopathy. Front Endocrinol. 2022;13:946915.
Ellahham S. Artificial intelligence: the future for diabetes care. Am J Med. 2020;133(8):895–900.
Singla R, Singla A, Gupta Y, Kalra S. Artificial intelligence/machine learning in diabetes care. Indian J Endocrinol Metab. 2019;23(4):495–7.
Doupis J, Festas G, Tsilivigos C, Efthymiou V, Kokkinos A. Smartphone-based technology in diabetes management. Diabetes Ther. 2020;11(3):607–19.
Stephens CR, Easton JF, Robles-Cabrera A, Fossion R, De la Cruz L, Martínez-Tapia R, Barajas-Martínez A, Hernández-Chávez A, López-Rivera JA, Rivera AL. The impact of education and age on metabolic disorders. Front Public Health. 2020;8:485859.
Banda JM, Sarraju A, Abbasi F, Parizo J, Pariani M, Ison H, et al. Finding missed cases of familial hypercholesterolemia in health systems using machine learning. NPJ Digit Med. 2019;2(1):23.
Dritsas E, Trigka M. Machine learning methods for hypercholesterolemia long-term risk prediction. Sensors. 2022;22(14):5365.
Tanaka M, Itoh H. Hypertension as a metabolic disorder and the novel role of the gut. Curr Hypertens Rep. 2019;21:63.
Chaikijurajai T, Laffin LJ, Tang WHW. Artificial intelligence and hypertension: recent advances and future outlook. Am J Hypertens. 2020;33:967–74.
Kirk JK, Stegner J. Self-monitoring of blood glucose: practical aspects. J Diabetes Sci Technol. 2010;4(2):435–9.
Rodbard D. Continuous glucose monitoring: a review of successes, challenges, and opportunities. Diabetes Technol Ther. 2016;18(S2):S2–3.
Khadilkar KS, Bandgar T, Shivane V, Lila A, Shah N. Current concepts in blood glucose monitoring. Indian J Endocrinol Metab. 2013;17:S643–9.
Tian X, Tang H, Cheng L, Liao Z, Li Y, He J, et al. Evaluation system framework of artificial intelligence applications in medical diagnosis and treatment. Procedia Comput Sci. 2022;214:495–502.
Bordoloi D, Singh V, Sanober S, Buhari SM, Ujjan JA, Boddu R. Deep learning in healthcare system for quality of service. J Healthc Eng. 2022;2022.
Muscogiuri G, Volpato V, Cau R, Chiesa M, Saba L, Guglielmo M, Senatieri A, et al. Application of AI in cardiovascular multimodality imaging. Heliyon. 2022;8(10).
Chiou YA, Hung CL, Lin SF. AI-assisted echocardiographic prescreening of heart failure with preserved ejection fraction on the basis of intrabeat dynamics. JACC Cardiovasc Imaging. 2021;14(11):2091–104.
Liu CM, Shih ESC, Chen JY, Huang CH, Wu IC, Chen PF, et al. Artificial intelligence enabled electrocardiogram improves the diagnosis and prediction of mortality in patients 678 with pulmonary hypertension. JACC: Asia. 2022;2(3):258–70.
Sujith AVLN, Sajja GS, Mahalakshmi V, Nuhmani S, Prasanalakshmi B. Systematic review of smart health monitoring using deep learning and artificial intelligence. Neuroscience informatics. 2022;2(3):100028.
Ganesh GS, Kolusu AS, Prasad K, Samudrala PK, Nemmani KVS. Advancing health care via artificial intelligence: from concept to clinic. Eur J Pharmacol. 2022;934:175320.
Zhang Y, Hu Y, Jiang N, Yetisen AK. Wearable artificial intelligence biosensor networks. Biosens Bioelectron. 2023;219:114825.
Nahavandi D, Alizadehsani R, Khosravi A, Acharya UR. Application of artificial intelligence in wearable devices: opportunities and challenges. Comput Methods Prog Biomed. 2022;213:106541.
Sarker S, Jamal L, Ahmed SF, Irtisam N. Robotics and artificial intelligence in healthcare during COVID-19 pandemic: a systematic review. Robot Auton Syst. 2021;146:103902.
Yi J, Zhang H, Mao J, Chen Y, Zhong H, Wang Y. Review on the COVID-19 pandemic prevention and control system based on AI. Eng Appl Artif Intell. 2022;114:105184.
Tanwar S, Kumari A, Vekaria D, Kumar N, Sharma R. An AI-based disease detection and prevention scheme for COVID-19. Comput Electr Eng. 2022;103:108352.
Azadi M, Yousefi S, Saen RF, Shabanpour H, Jabeen F. Forecasting sustainability of healthcare supply chains using deep learning and network data envelopment analysis. J Bus Res. 2023;154:113357.
Ganesh AD, Kalpana P. Future of artificial intelligence and its influence on supply chain risk management–a systematic review. Comput Ind Eng. 2022;169:108206.
Bag S, Dhamija P, Singh RK, Rahman MS, Sreedharan VR. Big data analytics and artificial intelligence technologies based collaborative platform empowering absorptive capacity in health care supply chain: an empirical study. J Bus Res. 2023;154:113315.
Chang V, Xu QA, Hall K, Wang YA, Kamal MM. Digitalization in omnichannel healthcare supply chain businesses: the role of smart wearable devices. J Bus Res. 2023;156:113369.
Damoah IS, Ayakwah A, Tingbani I. Artificial intelligence (AI)-enhanced medical drones in the healthcare supply chain (HSC) for sustainability development: A case study. J Clean Prod 328(2021):129598.
Yu J, Lee SH, Kim MK. Recent updates to clinical practice guidelines for diabetes mellitus. Endocrinol Metab. 2022;37(1):26–37.
Williams DM, Jones H, Stephens JW. Personalized type 2 diabetes management: an update on recent advances and recommendations. Diabetes Metab Syndr Obes. 2022;15:281–95.
Westall SJ, Narayanan RP, Watmough S, Irving G, Furlong N, McNulty S, et al. The individualisation of glycaemic targets in response to patient characteristics in type 2 diabetes: a scoping review. Clin Med. 2022;22(3):257–65.
Saucier AN, Ansa B, Coffin J, Akhtar M, Miller A, Mahoney H, et al. Patient perspectives of an individualized diabetes care management plan. Eur J Pers Cent Healthc. 2017;5(2):213–9.
Wan W, Skandari MR, Minc A, Nathan AG, Winn A, Zarei P, et al. Cost-effectiveness of continuous glucose monitoring for adults with type 1 diabetes compared with self-monitoring of blood glucose: the DIAMOND randomized trial. Diabetes Care. 2018;41(6):1227–34.
Sun MT, Li IC, Lin WS, Lin GM. Pros and cons of continuous glucose monitoring in the intensive care unit. World J Clin Cases. 2021;9(29):8666–70.
Janapala RN, Jayaraj JS, Fathima N, Kashif T, Usman N, Dasari A, Jahan N, Sachmechi I. Continuous glucose monitoring versus self-monitoring of blood glucose in type 2 diabetes mellitus: a systematic review with meta-analysis. Cureus. 2019;11(9).
Poolsup N, Suksomboon N, Kyaw AM. Systematic review and meta-analysis of the effectiveness of continuous glucose monitoring (CGM) on glucose control in diabetes. Diabetol Metab Syndr. 2013;5.
Olczuk D, Priefer R. A history of continuous glucose monitors (CGMs) in self-monitoring of diabetes mellitus. Diabetes Metab Syndr: Clin Res Rev. 2018;12(2):181–7.
Araki E, Goto A, Kondo T, Noda M, Noto H, Origasa H, Osawa H, et al. Japanese clinical practice guideline for diabetes 2019. Diabetol Int. 2020;11:165–223.
Li WX, Zhang XP, Chen BZ, Fei WM, Cui Y, Zhang CY, et al. An update on microneedle-based systems for diabetes. Drug Deliv Transl Res. 2022;12(10):2275–86.
Zahoor I, Singh S, Behl T, Sharma N, Naved T, Subramaniyan V, et al. Emergence of microneedles as a potential therapeutics in diabetes mellitus. Environ Sci Pollut Res. 2021;29:3302–22.
Zhao J, Xu G, Yao X, Zhou H, Lyu B, Pei S, Wen P. Microneedlebased insulin transdermal delivery system: current status and translation challenges. Drug Deliv Transl Res. 2022:1–25.
Lu J, Ma X, Zhou J, Zhang L, Mo Y, Ying L, et al. Association of time in range, as assessed by continuous glucose monitoring, with diabetic retinopathy in type 2 diabetes. Diabetes Care. 2018;41:2370–6.
Rodbard D. Continuous glucose monitoring: a review of recent studies demonstrating improved glycemic outcomes. Diabetes Technol Ther. 2017;19:S25-37.
Smith MB, Albanese-O’Neill A, Macieira TGR, Yao Y, Abbatematteo JM, Lyon D, et al. Human factors associated with continuous glucose monitor use in patients with diabetes: a systematic review. Diabetes Technol Ther. 2019;21:575–80.
Addala A, Maahs DM, Scheinker D, Chertow S, Leverenz B, Prahalad P. Uninterrupted continuous glucose monitoring access is associated with a decrease in HbA1c in youth with type 1 diabetes and public insurance. Pediatr Diabetes. 2020;21:1301–9.
Wright EE, Kerr MSD, Reyes IJ, Nabutovsky Y, Miller E. Use of flash continuous glucose monitoring is associated with A1C reduction in people with type 2 diabetes treated with basal insulin or noninsulin therapy. Diab Spectr. 2021;34:184–9.
Funtanilla VD, Caliendo T, Hilas O. Continuous glucose monitoring: a review of available systems. Pharm Ther. 2019;44(9):550.
Maahs DM, DeSalvo D, Pyle L, Ly T, Messer L, Clinton P, et al. Effect of acetaminophen on CGM glucose in an outpatient setting. Diabetes Care. 2015;38:e158–9.
Denham D. Effect of repeated doses of acetaminophen on a continuous glucose monitoring system with permselective membrane. J Diabetes Sci Technol. 2021;15:517–8.
Welsh JB, Psavko S, Zhang X, Gao P, Balo AK. Comparisons of fifth-, sixth-, and seventh-generation continuous glucose monitoring systems. J Diabetes Sci Technol. 2024;18(1):143–7.
Chellan N, Muller CJF. Running with type 1 diabetes: a case report on the benefit of sensor technology. Diabetology. 2022;3:310–4.
Oppel E, Kamann S, Reichl F, Högg C. The Dexcom glucose monitoring system—an isobornyl acrylate-free alternative for diabetic patients. Contact Dermatitis. 2019;81:32–6.
Kudva YC, Ahmann AJ, Bergenstal RM, Gavin JR, Kruger DF, Midyett LK, et al. Approach to using trend arrows in the Freestyle Libre Flash glucose monitoring systems in adults. J Endocr Soc. 2018;2:1320–37.
Blum A. Freestyle Libre glucose monitoring system. Clinical diabetes. 2018;36:203–4.
Yaron M, Roitman E, Aharon-Hananel G, Landau Z, Ganz T, Yanuv I, et al. Effect of flash glucose monitoring technology on glycemic control and treatment satisfaction in patients with type 2 diabetes. Diabetes Care. 2019;42:1178–84.
Miller E, Kerr MSD, Roberts GJ, Souto D, Nabutovsky Y, Wright E. 85-LB: Freestyle libre system use associated with reduction in acute diabetes events and all-cause hospitalizations in patients with type 2 diabetes without bolus insulin. Diabetes. 2020;69(Supplement_1).
Evans M, Welsh Z, Seibold A. Reductions in HbA1C with flash glucose monitoring are sustained for up to 24 months: a meta-analysis of 75 real-world observational studies. Diabetes Ther. 2022;13:1175–85.
Domingo-Lopez DA, Lattanzi G, H. J. Schreiber L, Wallace EJ, Wylie R, O’Sullivan J, et al. Medical devices, smart drug delivery, wearables and technology for the treatment of diabetes mellitus. Adv Drug Deliv Rev. 2022;185:114280.
Amadou C, Franc S, Benhamou PY, Lablanche S, Huneker E, Charpentier G, et al. Diabeloop DBLG1 closed-loop system enables patients with type 1 diabetes to significantly improve their glycemic control in real-life situations without serious adverse events: 6-month follow-up. Diabetes Care. 2021;44:844–6.
Benhamou PY, Franc S, Reznik Y, Thivolet C, Schaepelynck P, Renard E, et al. Closed loop insulin delivery in adults with type 1 diabetes in real-life conditions: a 12-week multicentre, open-label randomised controlled crossover trial. Lancet Digit Health. 2019;1:e17-25.
Hossain MI, Yusof AF, Sadiq AS. Factors influencing adoption model of continuous glucose monitoring devices for internet of things healthcare. Internet of Things. 2021;15:100353.
Gautier T, Ziegler LB, Gerber MS, Campos-Náñez E, Patek SD. Artificial intelligence and diabetes technology: a review. Metab. 2021;124.
Amorim D, Miranda F, Ferreira L, Abreu C. Data-driven carbohydrate counting accuracy monitoring: a personalized approach. Procedia Comput Sci. 2022;204:900–6.
Ahmed A, Aziz S, Qidwai U, Abd-Alrazaq A, Sheikh J. Performance of artificial intelligence models in estimating blood glucose level among diabetic patients using non-invasive wearable device data. Comput Methods Programs Biomed. 2023;3:100094.
Agrawal H, Jain P, Joshi AM. Machine learning models for non-invasive glucose measurement: towards diabetes management in smart healthcare. Health Technol (Berl). 2022;12(5):955–70.
Van Doorn WPTM, Foreman YD, Schaper NC, Savelberg HHCM, Koster A, van der Kallen CJH, et al. Machine learning-based glucose prediction with use of continuous glucose and physical activity monitoring data: the Maastricht study. PLoS One. 2021;16.
Wadghiri MZ, Idri A, el Idrissi T, Hakkoum H. Ensemble blood glucose prediction in diabetes mellitus: a review. Comput Biol Med. 2022;147:105674.
Sinha K, Uddin Z, Kawsar HI, Islam S, Deen MJ, Howlader MMR. Analyzing chronic disease biomarkers using electrochemical sensors and artificial neural networks. TrAC, Trends Anal Chem. 2023;158:116861.
Ben Ali J, Hamdi T, Fnaiech N, di Costanzo V, Fnaiech F, Ginoux JM. Continuous blood glucose level prediction of type 1 diabetes based on artificial neural network. Biocybern Biomed Eng. 2018;38(4):828–40.
Bukhari MM, Alkhamees BF, Hussain S, Gumaei A, Assiri A, Ullah SS. An improved artificial neural network model for effective diabetes prediction. Complexity. 2021;2021:1–10.
Poradzka AA, Czupryniak L. The use of the artificial neural network for three-month prognosis in diabetic foot syndrome. J Diabetes Complications. 2023;37.
Alfian G, Syafrudin M, Anshari M, Benes F, Atmaji FTD, Fahrurrozi I, et al. Blood glucose prediction model for type 1 diabetes based on artificial neural network with time-domain features. Biocybern Biomed Eng. 2020;40:1586–99.
Saleem R, Yuan B, Kurugollu F, Anjum A, Liu L. Explaining deep neural networks: a survey on the global interpretation methods. Neurocomputing. 2022;513:165–80.
Dudukcu HV, Taskiran M, Yildirim T. Blood glucose prediction with deep neural networks using weighted decision level fusion. Biocybern Biomed Eng. 2021;41(3):1208–23.
Cichosz SL, Jensen MH, Hejlesen O. Short-term prediction of future continuous glucose monitoring readings in type 1 diabetes: development and validation of a neural network regression model. Int J Med Inform. 2021;151:104472.
Mohammed A, Kora R. A comprehensive review on ensemble deep learning: opportunities and challenges. J King Saud Univ Comput Inf Sci. 2023;35(2):757–74.
Zhu T, Li K, Herrero P, Chen J, Georgiou P. A deep learning algorithm for personalized blood glucose prediction. In: KHD@ IJCAI; 2018. p. 64–78.
Zhang M, Flores KB, Tran HT. Deep learning and regression approaches to forecasting blood glucose levels for type 1 diabetes. Biomed signal process control. 2021;69.
Kim DY, Choi DS, Kang AR, Woo J, Han Y, Chun SW, et al. Intelligent ensemble deep learning system for blood glucose prediction using genetic algorithms. Complexity. 2022;2022.
Rabby MF, Tu Y, Hossen MI, Lee I, Maida AS, Hei X. Stacked LSTM based deep recurrent neural network with kalman smoothing for blood glucose prediction. BMC Med Inform Decis Mak. 2021;21:1–15.
Elhadd T, Mall R, Bashir M, Palotti J, Fernandez-Luque L, Farooq F, Al Mohanadi D, et al. Artificial intelligence (AI) based machine learning models predict glucose variability and hypoglycaemia risk in patients with type 2 diabetes on a multiple drug regimen who fast during ramadan (the PROFAST–IT Ramadan study). Diabetes Res Clin Pract. 2020;169:108388.
Motaib I, Aitlahbib F, Fadil A, Z.Rhmari Tlemcani F, Elamari S, Laidi S, et al. Predicting poor glycemic control during Ramadan among non-fasting patients with diabetes using artificial intelligence based machine learning models. Diabetes Res Clin Pract. 2022;190.
Mosquera-Lopez C, Ramsey KL, Roquemen-Echeverri V, Jacobs PG. Modeling risk of hypoglycemia during and following physical activity in people with type 1 diabetes using explainable mixed-effects machine learning. Comput Biol Med. 2023;155:106670.
Tyler NS, Mosquera-Lopez C, Young GM, el Youssef J, Castle JR, Jacobs PG. Quantifying the impact of physical activity on future glucose trends using machine learning. IScience. 2022;25.
Manickam P, Mariappan SA, Murugesan SM, Hansda S, Kaushik A, Shinde R, et al. Artificial intelligence (AI) and internet of medical things (IoMT) assisted biomedical systems for intelligent healthcare. Biosensors (Basel). 2022;12:562.
Wang L. Heterogeneous data and big data analytics. Autom Contr Info Sci. 2017;3:8–15.
Gupta S, Gupta A. Dealing with noise problem in machine learning data-sets: a systematic review. Procedia Comput Sci. 2019;161:466–74.
Kakhi K, Alizadehsani R, Kabir HMD, Khosravi A, Nahavandi S, Acharya UR. The internet of medical things and artificial intelligence: trends, challenges, and opportunities. Biocybern Biomed Eng. 2022;42(3):749–71.
Boulemtafes A, Derhab A, Challal Y. Privacy-preserving deep learning for pervasive health monitoring: a study of environment requirements and existing solutions adequacy. Health Technol (Berl). 2022;12(2):285–304.
Boulemtafes A, Derhab A, Challal Y. A review of privacy-preserving techniques for deep learning. Neurocomputing. 2020;384:21–45.
Gerke S, Minssen T, Cohen G. Ethical and legal challenges of artificial intelligence-driven healthcare. Artif Intell Healthcare. 2020;295–336.
Shaheen MY. AI in healthcare: medical and socio-economic benefits and challenges (September 25, 2021). Available at SSRN: https://ssrn.com/abstract=3932277 or https://doi.org/10.2139/ssrn.393227.
Hall BH. Patents and patent policy. Oxf Rev Econ Policy. 2007;23:568–87.
Tidwell JL, Liotta LA. Inventions and patents: a practical tutorial. Methods Mol Biol. 2012;823:391–408.
Cameron BD, Pappada SM. Neural network system and uses thereof (patent no. US20140304204A1); 2015. https://patents.google.com/patent/US20140304204A1/en.
Agrawal P, Mcmahon CM, Neemuchwala HF, Zhong Y, Hoebing J, Mikhno A. Machine learning-based system for estimating glucose values. US20220039755A1, 2020.
Freckmann G. Basics and use of continuous glucose monitoring (CGM) in diabetes therapy. J Lab Med. 2020;44(2):71–9.
Khan MY, Qayoom A, Nizami MS, Siddiqui MS, Wasi S, Raazi SMK ur R. Automated prediction of good dictionary EXamples (GDEX): a comprehensive experiment with distant supervision, machine learning, and word embedding-based deep learning techniques. Complexity. 2021;2021:1–18.
Seo W, Lee YB, Lee S, Jin SM, Park SM. A machine-learning approach to predict postprandial hypoglycemia. BMC Med Inform Decis Mak. 2019;19:210.
Author information
Authors and Affiliations
Contributions
LPY, OXY, and OWQ: data collection and manuscript writing. NJ and BP: data collection and manuscript editing. JM: data collection and manuscript writing. MP and MC: manuscript structure, conceptualization, administration, supervision, editing, and coordination. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ying, L.P., Yin, O.X., Quan, O.W. et al. Continuous glucose monitoring data for artificial intelligence-based predictive glycemic event: A potential aspect for diabetic care. Int J Diabetes Dev Ctries (2024). https://doi.org/10.1007/s13410-024-01349-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13410-024-01349-x