Abstract
Long noncoding RNAs (lncRNAs), which refer to a group of RNAs with length more than 200 nucleotides and limited protein-coding potential, play a widespread role in regulating biological processes, such as cell differentiation, proliferation, apoptosis, and migration. LncRNAs are dysregulated in multiple cancers, playing an either oncogenic or tumor-suppressive role. LncRNA GAS5 is a recently identified tumor suppressor involved in several cancers, like breast cancer, prostate cancer, lung cancer, and colorectal cancer. The low-expression pattern confers tumor cells elevated capacity of proliferation and predicts poorer prognosis. Existing studies mirror that lncRNA GAS5 promises to be a novel diagnostic biomarker, therapy target, as well as prognostic biomarker. In this review, we will summarize the current knowledge about this vital lncRNA, from its discovery, characteristics, and biological function to molecular mechanism in various neoplasms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
For a long term, there had been a preconception on the primacy of the protein-coding RNAs over noncoding RNAs (ncRNAs). In the early years, on the one side, there was no suitable molecular technique to detect and analyze ncRNAs; on the other side, it was hardly conceivable that these transcriptional noises could play an active role in multibiological processes. Things have changed over the past several years; however, the advent of techniques such as microarrays and high-throughput sequencing has revolutionized our ability to uncover the widespread expression profile and crucial biological functions of ncRNAs [1]. The Encyclopedia of DNA Elements (ENCODE) project has demonstrated that the vast majority of the mamalian genome (up to 80 %) is transcribed into ncRNAs [2].
Noncoding RNAs are divided into two major classes based on transcript size: those under 200 nucleotides are usually referred to as short/small ncRNAs, and those over 200 nucleotides are known as long noncoding RNAs (lncRNAs) [3]. Studies have shown that small ncRNAs participate in the management of varieties of physiological and pathological pathways, and that abnormal expression is closely related to different human diseases [4, 5]. For example, many microRNAs (miRNAs) act as oncogenes or tumor suppressors through binding to mRNA targets and promoting their degradation and/or translational inhibition [5]. In contrast to small ncRNAs, much less is known regarding the role of the many thousands of lncRNAs and this is likely to provide an exciting and fertile new area of scientific discovery. Thus, scientists are now turning attention to the poorly understood transcriptional products of genomes: lncRNAs [6]. Emerging evidences increasingly support the view that lncRNAs play a pivotal role especially in human cancers [7]. Available researches show that a portion of lncRNAs function as chromatin modifier, transcriptional or post-transcriptional regulator of gene expression [8, 9].
Among all kinds of lncRNAs, the growth arrest-specific transcript 5 (GAS5) is a rising star among tumor-suppressive lncRNAs. Recent studies have brought GAS5 under the spotlight, suggesting pivotal roles in a variety of human diseases [10–13]. Next, we will introduce the biogenesis of GAS5 and summarize the current studies about its regulation mechanisms and functions in human cancer specially.
Biogenesis, characterizations, and regulation of GAS5
GAS5 was originally identified from a subtraction cDNA library and named after the finding that its expression level increased upon growth arrest in mammalian cells [14]. It is located at 1q25 and comprised of 12 exons which are alternatively spliced to yield two possible mature lncRNAs: GAS5a and GAS5b [14] and 11 introns which encode 10 box C/D small nucleolar RNAs (snoRNA) [15]. In spite of its short open reading frame, GAS5 does not possess protein-coding capacity and acts as a so-called host gene for snoRNAs.
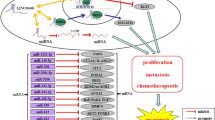
The abundance of GAS5 transcripts goes up upon cell growth arrest which induced by serum starvation or treatment with rapamycin [14, 16–18]. It is explained by the interplay between the mammalian target of rapamycin (mTOR) pathway [18] and the nonsense-mediated decay (NMD) pathway [19]. GAS5 is classified as a 5′-terminal oligopyrimidine RNA [15]; therefore, its translation is specifically controlled by the mTOR pathway [18, 20]. NMD is historically identified as a RNA quality control system to eliminate abnormal transcripts; however, increasing evidences have shown that NMD pathway could also regulate the enrichment of physiological transcripts [21–23] and the function of GAS5 in mammalian cells [19]. In short, the translation of GAS5 short reading frame is blocked when mTOR pathway activity is repressed, and nonsense-mediated decay pathway which depends on active translation no longer occurs [24]. GAS5 transcripts level therefore increases [18] (Fig. 1a).
a When mTOR pathway activity is repressed by serum starvation or rapamycin, translation of the GAS5 short reading frame is inhibited, and nonsense-mediated decay, which depends on active translation, no longer occurs. GAS5 transcript level therefore increases. b GAS5 could compete with DNA glucocorticoid response element (GRE) for binding to the DNA-binding domain (DBD) of the glucocorticoid receptor (GR), leading to the transcriptional repression of cIAP2 and SGK1. In addition, GAS5 is capable of suppressing miR-21 and miR-22 through lncRNA/miRNA interaction. Furthermore, GAS5 could directly regulate translation of target genes
What significance will increased GAS5 which accompanied with growth arrest bring? Mourtada-Maarabouni et al. firstly investigated its biological functions in human T cell lines [25]. The experimental results indicated that overexpression of GAS5 increased the rate of apoptosis and reduced the proportion of cells in S phase [25]. Correspondingly, GAS5 silencing caused the opposite outcomes [25]. Both demonstrated that GAS5 was sufficient and necessary for growth-retarding of T cell lines [25]. In addition, researchers reported a case that the GAS5 gene fused to the BCL6 gene in a patient diagnosed with diffuse large B cell lymphoma [26]. Considering the high morbidity and mortality of cancer, investigators have carried out many studies about the functional roles of GAS5 in such a life-threatening disease. They found that GAS5 expression level was reduced in several human solid tumors, and this abnormal expression had significant negative correlations with tumor size, disease stage, and patient prognosis [10, 11, 27], which will be described in detail below.
Given the recognition that apoptosis exerts important effect on many physiological and pathological processes and accumulation of GAS5 could induce apoptosis and growth arrest in several mammalian cell lines [28], there is mounting interest in uncovering the underlying molecular mechanisms of GAS5 action. It is well known that glucocorticoids exert potent and diverse effects on many aspects of biological processes including influence cell’s sensitivity to apoptosis through binding with glucocorticoid receptor (GR). Through a panel of experimental techniques such as LexA-based yeast two-hybrid screening assay, RNA/protein coprecipitation assay, and so on, Kino et al. confirmed that GAS5 could compete with DNA glucocorticoid response element (GRE) for binding to the DNA-binding domain (DBD) of the glucocorticoid receptor (GR) through mimicking GRE, thus, blocking glucocorticoid-GR-GRE interaction induced targeted gene transcription [29]. For example, apoptosis-suppressing genes cIAP2 and SGK1 whose promoters contain GREs were transcriptionally inhibited by GAS5 and then lead to increasing cell apoptosis [29]. Furthermore, in 2013, Zhang et al. showed that miR-21 was capable of suppressing GAS5 expression in a manner similar to the microRNA-mediated silencing of target mRNAs [30]. Interestingly, GAS5 itself could also inhibit miR-21 expression and then free miR-21 targeted tumor-suppressive genes (such as PTEN and PDCD4), implying a feedback loop between GAS5 and miR-21 [30]. They further quantified this inverse relationship in breast tumor specimens. In addition to the sponge role of miR-21, Zhao et al. discovered that miR-222 was another target of GAS5 recently [31]. GAS5 acted as a tumor suppressor by directly targeting and reducing miR-222 expression and then triggered tumor suppressor bmf and Plexin C1 (PLXN C1) transcription, suppressing cell growth, migration, and invasion, as well as promoting cell apoptosis in glioma [31]. Besides mimicking GREs and sponging miRNAs, it was said that GAS5 still played a role in mediating mRNA translation. Hu et al. found that GAS5 could directly bound to and repress c-Myc mRNA translation without influencing its transcription level as well as protein stability [32]. Anything else, GAS5 was proved to interact with transcriptional activator Y-box binding protein 1 (YBX1) [33]. Low-expression GAS5 increased the speed of YBX1 protein degradation instead of reducing YBX1 mRNA level, reducing p21 expression level and abolishing G1 phase arrest [33]. To sum up, lncRNA GAS5 exerts biological functions mainly through the above mentioned three mechanisms so far (Fig. 1b). However, a comprehensive and thorough understanding of the molecular mechanisms of GAS5 awaits much additional work.
GAS5 in human cancer
Considerable researches have rapidly increased the number of well-characterized lncRNAs and presented their important roles in several aspects of tumorigenesis, such as cancer cell proliferation, migration, invasion, and tumor-drug resistance [34–37]. Here, we review the expression profiling, biological function, and molecular mechanisms of GAS5 in various human cancers.
Breast cancer
The involvement of GAS5 in human tumors was first investigated in breast cancer. In 2009, Mourtada-Maarabouni et al. showed that GAS5 transcript level was significantly reduced in breast cancer tissues relative to normal breast epithelial tissues, and overexpression of GAS5 could induce growth arrest and apoptosis of human breast cancer cell lines [28]. In addition, low expression level of GAS5 disadvantageously impacted patient prognosis, suggesting that GAS5 may act as a tumor suppressor in breast cancer [28]. GAS5 silencing attenuated cell apoptotic response to classical chemotherapeutic agents. However, imatinib’s curative effect was independent of GAS5 expression level [38]. These findings have clinical significance of enlightening us to use GAS5-independent drugs such as imatinib, or combined use of traditional chemotherapies and a dual PI3K/mTOR inhibitor to improve cell apoptotic response to cytotoxic therapy owning to restoration of GAS5 expression level. These studies stimulate further investigation of the possible roles and mechanisms of GAS5 in oncogenesis, tumor development, and conventional chemotherapy resistance involved with a wider range of malignant tumors.
Prostate cancer
Androgen plays a vital role in prostate cell survival and proliferation. Therefore, androgen deprivation therapy (ADT) is expected to achieve effective treatment of advanced prostate cancer by inhibiting cell growth and inducing apoptosis. Unfortunately, it is a palliative, not a cure, since prostate cancer inevitably progress to castrate-resistant prostate cancer (CRPC) with prolonged ADT [39]. Hence, there is an urgent need to develop novel therapies for CRPC. Study showed that GAS5 could bind the DBD of AR as it shared some of the same response sequence with the GR. Thus, GAS5 could also sequester the androgen/androgen receptor complex and prevented its binding to target DNA sequences in the prostate context [29]. Functional studies have already proved that GAS5 could promote the apoptosis of prostate cancer cells and demonstrated a positive correlation between the cellular GAS5 expression level and the extent of cell death [40]. Based on these experimental results, researchers hypothesized that enhancement of cellular GAS5 expression level coupled with conventional chemotherapy may therefore offer a reformative strategy for the treatment of advanced prostate cancer. Subsequent research proved this conjecture and found that mTOR inhibition displayed a selective enhancement effect of GAS5 transcript level since it failed to increase GAS5 level of PC-3 and DU 145 cell lines which exhibited a intrinsic low GAS5 expression level, which meant that GAS5 itself was necessary for mTOR inhibition action [12].
Lung cancer
The expression pattern and biological function of GAS5 were also investigated in human nonsmall cell lung cancer (NSCLC). Its expression level was downregulated in cancerous tissues and correlated with worse clinicopathological characteristics. Gain-of-function and loss-of-function experiments both demonstrated that GAS5 was indispensable for growth arrest and apoptosis of NSCLC cell lines [11]. To detect other mechanism leading to downregulation of GAS5 expression, bioinformatics techniques was for the first time used to analysis its promoter region and found there were CpG islands. That the upregulation of GAS5 after treatment with DNA demethylating agent 5-aza-2-deoxy-cytidine (5-aza-CdR) demonstrated that epigenetic modification be involved in the transcriptional inactivation of GAS5 [11].
The epidermal growth factor receptor tyrosine kinase inhibitors (EGFR-TKIs) are considered to be the standard first-line treatment for advanced NSCLC patients harboring activating EGFR mutations based on the IPASS and TORCH trials, which both demonstrated a prolonged survival with EGFR-TKI therapy compared to chemotherapy [41]. However, the inevitable emergence of primary and acquired drug resistance is an obstacle to clinical efficacy of EGRF-TKIs [42]. A ton of previous studies have demonstrated that lncRNAs are involved in tumorgenesis [43–46] and chemotherapy resistance [38, 47–49]; investigators therefore suppose whether lncRNAs play a novel role in the development of resistance to EGFR-TKIs. Cheng et al. firstly identified a total of 4667 differentially expressed (fold change >2) lncRNAs in gefitinib-sensitive and gefitinib-induced acquired-resistant lung cancer cells using lncRNA microarray [50]. Bioinformatic analysis revealed that many of them played crucial roles in regulating EGFR-TKIs resistance through proliferation and apoptosis-related signaling pathways. Subsequently, Dong et al. selected GAS5 as a target lncRNA to study its effects on EGFR-TKIs resistance [51]. In vitro and in vivo results showed that the upregulation of GAS5 could overcome the resistance of human adenocarcinoma A549 cell line to EGFR-TKIs, at least partially by insulin-like growth factor 1 receptor (IGF-1R) which has been connected with resistance to EGFR-TLIs and proved to be downregulated with dual GAS5 plus gefitinib treatment. In aggregate, these findings support a powerful role of lncRNAs in relation to many aspects of tumorgenesis, metastasis, and drug resistance, implying that these noncoding transcripts are hopeful to be potential biomarkers for the diagnosis and treatment of human cancers.
Malignant pleural mesothelioma
To the best of our knowledge, Wright et al. firstly identified lncRNA expression patterns of malignant pleural mesothelioma (MPM) by using NCode long noncoding microarrays [52]. A panel of nine upregulated candidate lncRNAs were selected by analysis of differential expression and validated by RT-qPCR in independent MPM tissues and cell lines. Some of these lncRNAs have clinical significance, such as distinguishing normal mesothelium from MPM with high sensitivity and specificity and predicting nodal metastasis and overall survival [52]. A recent study mainly investigated whether GAS5 played a potential important role in MPM biology [53]. In line with what had been previously described, GAS5 expression was downregulated in MPM cell lines relative to normal mesothelial cells. Researchers observed that downregulated GAS5 could regulate cell proliferation by shortening the cell cycle length [53]. Given that MPM is a rare tumor and traditional combined chemotherapy contribute a median survival only around 12 months [13], identification of effective diagnostic and treatment biomarkers promises to increase the ratio of early diagnosis and work out the optimal individualized treatment regimen. Thus, GAS5 could be the right target and further studies are needed to unveil its mysterious yashmak.
Other human cancers
Lu et al. verified that GAS5 was decreased in pancreatic cancer tissues and overexpression of GAS5 could suppress pancreatic cancer cell proliferation. Furthermore, they demonstrated the negative relationship between GAS5 and cyclin-dependent kinase 6 (CDK6). GAS5 low-expression-induced cell proliferation was partially abrogated by the knockdown of CDK6, suggesting that CDK6 participated in this pathway as a downstream molecule [54]. Liu et al. uncovered that GAS5 inhibited bladder cancer cell proliferation, to a degree, by regulating CDK6 mRNA and protein expression levels [55], which was similar to previous study in pancreatic cancer [54].
It was revealed that the expression level of GAS5 was lower in gastric cancer tissues than in normal tissues and predicted poorer survival. GAS5 functioned as a tumor suppressor by regulating cell proliferation and apoptosis, which might be partially through posttranscriptional regulation of transcription factor E2F1 and P21 [10]. Recently, Liu et al. delineated another signal pathway of GAS5 in suppressing gastric cancer development. It was said that GAS5 could reduce YBX1 protein level rather than mRNA level, further caused P21 expression reduction and abolished G1 phase cell cycle arrest, driving tumorigenesis and development [33].
The expression of GAS5 in colorectal cancer tissues [27] and renal cell carcinoma (RCC) specimens [56] was lower than that in corresponding nontumor tissues. Researchers conducted preliminary functional exploration and validated that the proliferation, migration, and invasion abilities of cancer cells were inhibited by overexpression of GAS5. In addition, Tu et al. and Cao et al. proved for the first time that GAS5 was downregulated in liver cancer [57] and cervical cancer [58], respectively, and correlated with poorer prognosis.
To sum up, we have found the pervasively low-expression pattern (except for MPM patient tissues [53]) and tumor-suppressive role of GAS5 in many different types of human cancer, shown in Table 1. Besides mTOR and NMD pathways which could coregulate GAS5 abundance, hypermethylation of CpG islands in promoter regions also contributes to the downregulated expression of GAS5 [11]. Aberrant expression of GAS5 promotes cancer cell proliferation and suppresses apoptosis via different downstream signaling molecules in a variety of malignancy. Taken together, lncRNA GAS5 functions as a tumor suppressor and promises to be a potential therapy target or prognosis predictor. The precise mechanism of down-regulation of GAS5 and its central downstream signaling pathway deserves further exploration.
Conclusion
The last decade has witnessed an explosive expansion in exploring the biological functions and clinical significances of long noncoding RNAs. It is well documented that these noncoding transcripts are deregulated in cancers and can influence vital steps of tumor growth or metastasis, playing oncogenic or tumor-suppressive role. Some of which have obtained well interpretation, for example, hox transcript antisense intergenic RNA (HOTAIR), an oncogenic lncRNA, epigenetically regulates target genes expression via interacting with polycomb repressive complex2 (PRC2) and lysine-specific demethylase 1 (LSD1) [34, 35].
In this review, we emphatically introduce long noncoding RNA GAS5. It is not difficult to draw a conclusion that downregulated GAS5 in cancerous tissues is capable of promoting cell proliferation, reducing apoptosis and as such leads to cancer development. Therefore, forced expression of GAS5 in cancer tissues in vivo would be an attractive strategy for cancer treatment. That mTOR pathway control the cellular GAS5 expression level [18] prompts us to use mTOR inhibitors as an adjuvant therapy. It turns out to be feasible because mTOR targeting therapy have been improved and proved to be useful in anticancer treatment [59, 60]. It is noteworthy that this enhancement relies on inherent GAS5 expression which has been mentioned in prostate cancer context [12]. In addition to the use of mTOR inhibitors, designing a vector that would induce the expression of GAS5 when injected into the tumor may provide another attractive therapeutic approach. As already mentioned above, DNA methylation modification is another mechanism decreasing GAS5 expression level. Thus, we can achieve GAS5 enhancement by using DNA methyltransferase inhibitor, which is independent of inherent GAS5 expression level. Realizing the translation from basic researches to clinical practice needs more exploration and well-founded experiments. Only with full understanding of the mechanisms contribute to GAS5 downregulation can we work out pointed implement strategies.
It has been found by analyzing GAS5 level in tumor tissues and clinical parameters that GAS5 correlates with advanced tumor stage and worse prognosis. Based on this, GAS5 could be utilized as a diagnostic and prognostic biomarker. Nevertheless, the minimal invasiveness and modest effectiveness need to be taken into account. There is no doubt that blood biopsy is the optimal method. A study has proved that GAS5 was elevated in secreted exosomes, and hence, we can isolate exosomes from blood to analysis and monitor GAS5 level dynamically [61]. Besides, as far as we know, an experimental team has observed a decline of GAS5 in patients who have received completed tumor resection and validated its specificity and sensitivity in differentiating benign and malignant NSCLC. However, the practicality of GAS5 as well as other lncRNAs in distinguishing benign lesions and malignancies, monitoring early malignant recurrence and guiding tumor therapy needs further exploration and validation.
The molecular mechanism of a certain disease is a complicated biological process, which may involve in stepwise accumulation of gene mutations, genomic instability, epigenetic changes, and aberrant expression of protein-coding and noncoding genes. Instead of the traditional concepts that protein-coding genes play important roles in biological processes, lncRNAs provide us a novel insight into the understanding of disease occurrence and development. From a clinical perspective, the targeting of lncRNAs as a novel diagnosis biomarker and therapeutic approach will require a wider and deeper understanding of their biological functions and molecular mechanisms of action.
Abbreviations
- GAS5:
-
Growth arrest-specific transcript 5
- lncRNAs:
-
Long noncoding RNAs
- miRNAs:
-
MicroRNAs
- snoRNA:
-
Small nucleolar RNA
- m-TOR:
-
Mammalian target of rapamycin
- NMD:
-
Nonsense-mediated decay
- GR:
-
Glucocorticoid receptor
- GRE:
-
Glucocorticoid response element
- DBD:
-
DNA-binding domain
- PLXN C1:
-
Plexin C1
- YBX1:
-
Y-box binding protein 1
- ADT:
-
Androgen deprivation therapy
- CRPC:
-
Castrate-resistant prostate cancer
- NSCLC:
-
Nonsmall-cell lung cancer
- 5-aza-CdR:
-
5-Aza-2-deoxy-cytidine
- EGFR-TKIs:
-
Epidermal growth factor receptor tyrosine kinase inhibitors
- IGF-1R:
-
Insulin-like growth factor 1 receptor
- MPM:
-
Malignant pleural mesothelioma
- CDK6:
-
Cyclin-dependent kinase 6
- RCC:
-
Renal cell carcinoma
- HOTAIR:
-
Hox transcript antisense intergenic RNA
- PRC2:
-
Polycomb repressive complex 2
- LSD1:
-
Lysine-specific demethylase 1
References
Booton R, Lindsay MA. Emerging role of MicroRNAs and long noncoding RNAs in respiratory disease. Chest. 2014;146(1):193–204.
Consortium TEP. An integrated encyclopedia of DNA elements in the human genome. Nature. 2012;489(7414):57–74.
Nagano T, Fraser P. No-nonsense functions for long noncoding RNAs. Cell. 2011;145(2):178–81.
Hammond SM. An overview of microRNAs. Adv Drug Deliv Rev. 2015;87:3–14.
Pritchard CC, Cheng HH, Tewari M. MicroRNA profiling: approaches and considerations. Nat Rev Genet. 2012;13(5):358–69.
Derrien T, Johnson R, Bussotti G, Tanzer A, Djebali S, Tilgner H, et al. The GENCODE v7 catalog of human long noncoding RNAs: Analysis of their gene structure, evolution, and expression. Genome Res. 2012;22(9):1775–89.
Wapinski O, Chang HY. Long noncoding RNAs and human disease. Trends Cell Biol. 2011;21(6):354–61.
Gibb EA, Brown CJ, Lam WL. The functional role of long non-coding RNA in human carcinomas. Mol Cancer. 2011;10:38.
Shi X, Sun M, Wu Y, Yao Y, Liu H, Wu G, et al. Post-transcriptional regulation of long noncoding RNAs in cancer. Tumour Biol. 2015;36(2):503–13.
Sun M, Jin FY, Xia R, Kong R, Li JH, Xu TP, et al. Decreased expression of long noncoding RNA GAS5 indicates a poor prognosis and promotes cell proliferation in gastric cancer. BMC Cancer. 2014;14:319.
Shi X, Sun M, Liu H, Yao Y, Kong R, Chen F, et al. A critical role for the long non-coding RNA GAS5 in proliferation and apoptosis in non-small-cell lung cancer. Mol Carcinog. 2015;54 Suppl 1:E1–E12.
Yacqub-Usman K, Pickard MR, Williams GT. Reciprocal regulation of GAS5 lncRNA levels and mTOR inhibitor action in prostate cancer cells. Prostate. 2015;75(7):693–705.
Stahel RA, Weder W, Felip E. Malignant pleural mesothelioma: ESMO clinical recommendations for diagnosis, treatment and follow-up. Ann Oncol. 2009;20 Suppl 4:73–5.
Schneider C, King RM, Philipson L. Genes specifically expressed at growth arrest of mammalian cells. Cell. 1988;54:787–93.
SMITH CM, STEITZ JA. Classification of gas5 as a multi-small-nucleolar-RNA (snoRNA) host gene and a member of the 5′terminal oligopyrimidine gene family reveals common features of snoRNA host genes. Mol Cell Biol. 1998;18(12):6897–909.
Williams GT, Farzaneh F. Are snoRNAs and snoRNA host genes new players in cancer? Nat Rev Cancer. 2012;12(2):84–8.
COCCIA EM, Cicala C, Charlesworth A, Ciccarelli C, Rossi GB, Philipson L, et al. Regulation and expression of a growth arrest-specific gene (gas5) during growth, differentiation, and development. Mol Cell Biol. 1992;12(8):3514–21.
Mourtada-Maarabouni M, Hasan AM, Farzaneh F, Williams GT. Inhibition of human T-cell proliferation by mammalian target of rapamycin (mTOR) antagonists requires noncoding RNA growth-arrest-specific transcript 5 (GAS5). Mol Pharmaco. 2010;78(1):19–28.
Tani H, Torimura M, Akimitsu N. The RNA degradation pathway regulates the function of GAS5 a non-coding RNA in mammalian cells. PLoS One. 2013;8(1):e55684.
Meyuhas O. Synthesis of the translational apparatus is regulated at the translational level. Eur J Biochem. 2000;267(21):6321–30.
Tani H, Imamachi N, Salam KA, Mizutani R, Ijiri K, Irie T, et al. Identification of hundreds of novel UPF1 target transcripts by direct determination of whole transcriptome stability. RNA Biol. 2012;9(11):1370–9.
Imamachi N. Up-frameshift protein 1 (UPF1): multitalented entertainer in RNA decay. Drug Discov Ther. 2012;6(2):55–61.
Mendell JT, Sharifi NA, Meyers JL, Martinez-Murillo F, Dietz HC. Nonsense surveillance regulates expression of diverse classes of mammalian transcripts and mutes genomic noise. Nat Genet. 2004;36(10):1073–8.
Isken O, Maquat LE. Quality control of eukaryotic mRNA: safeguarding cells from abnormal mRNA function. Genes Dev. 2007;21(15):1833–56.
Mourtada-Maarabouni M, Hedge VL, Kirkham L, Farzaneh F, Williams GT. Growth arrest in human T-cells is controlled by the non-coding RNA growth-arrest-specific transcript 5 (GAS5). J Cell Sci. 2008;121(Pt 7):939–46.
Nakamura Y, Takahashi N, Kakegawa E, Yoshida K, Ito Y, Kayano H, et al. The GAS5 (growth arrest-specific transcript 5) gene fuses to BCL6 as a result of t(1;3)(q25;q27) in a patient with B-cell lymphoma. Cancer Genet Cytogenet. 2008;182(2):144–9.
Yin D, He X, Zhang E, Kong R, De W, Zhang Z. Long noncoding RNA GAS5 affects cell proliferation and predicts a poor prognosis in patients with colorectal cancer. Med Oncol. 2014;31(11):253.
Mourtada-Maarabouni M, Pickard MR, Hedge VL, Farzaneh F, Williams GT. GAS5, a non-protein-coding RNA, controls apoptosis and is downregulated in breast cancer. Oncogene. 2009;28(2):195–208.
Kino T, Hurt DE, Ichijo T, Nader N, Chrousos GP. Noncoding RNA gas5 is a growth arrest- and starvation-associated repressor of the glucocorticoid receptor. Sci Signal. 2010;3(107):ra8.
Zhang Z, Zhu Z, Watabe K, Zhang X, Bai C, Xu M, et al. Negative regulation of lncRNA GAS5 by miR-21. Cell Death Differ. 2013;20(11):1558–68.
Zhao X, Wang P, Liu J, Zheng J, Liu Y, Chen J, et al. Gas5 exerts tumor-suppressive functions in human glioma cells by targeting miR-222. Mol Ther. 2015. doi:10.1038/mt.2015.170
Hu G, Lou Z, Gupta M. The long non-coding RNA GAS5 cooperates with the eukaryotic translation initiation factor 4E to regulate c-Myc translation. PLoS One. 2014;9(9):e107016.
Liu Y, Zhao J, Zhang W, Gan J, Hu C, Huang G, et al. LncRNA GAS5 enhances G1 cell cycle arrest via binding to YBX1 to regulate p21 expression in stomach cancer. Sci Rep. 2015;5:10159.
Hajjari M, Salavaty A. HOTAIR: an oncogenic long non-coding RNA in different cancers. Cancer Biol Med. 2015;12(1):1–9.
Wu Y, Zhang L, Wang Y, Li H, Ren X, Wei F, et al. Long noncoding RNA HOTAIR involvement in cancer. Tumour Biol. 2014;35(10):9531–8.
Yan L, Zhou J, Gao Y, Ghazal S, Lu L, Bellone S, et al. Regulation of tumor cell migration and invasion by the H19/let-7 axis is antagonized by metformin-induced DNA methylation. Oncogene. 2014;34(23):3076–84.
Li S, Wang Q, Qiang Q, Shan H, Shi M, Chen B, et al. Sp1-mediated transcriptional regulation of MALAT1 plays a critical role in tumor. J Cancer Res Clin Oncol. 2015;141(11):1909–20.
Pickard MR, Williams GT. Regulation of apoptosis by long non-coding RNA GAS5 in breast cancer cells: implications for chemotherapy. Breast Cancer Res Treat. 2014;145(2):359–70.
Tamburrino L, Salvianti F, Marchiani S, Pinzani P, Nesi G, Serni S, et al. Androgen receptor (AR) expression in prostate cancer and progression of the tumor: Lessons from cell lines, animal models and human specimens. Steroids. 2012;77(10):996–1001.
Pickard MR, Mourtada-Maarabouni M, Williams GT. Long non-coding RNA GAS5 regulates apoptosis in prostate cancer cell lines. Biochim Biophys Acta. 2013;1832(10):1613–23.
Chan BA, Hughes BG. Targeted therapy for non-small cell lung cancer: current standards and the promise of the future. Transl Lung Cancer Res. 2015;4(1):36–54.
Perez-Soler R, Chachoua A, Hammond LA, Rowinsky EK, Huberman M, Karp D, et al. Determinants of tumor response and survival with erlotinib in patients with non--small-cell lung cancer. J Clin Oncol. 2004;22(16):3238–47.
Takahashi K, Yan I, Haga H, Patel T. Long noncoding RNA in liver diseases. Hepatology. 2014;60(2):744–53.
Svoboda M, Slyskova J, Schneiderova M, Makovicky P, Bielik L, Levy M, et al. HOTAIR long non-coding RNA is a negative prognostic factor not only in primary tumors, but also in the blood of colorectal cancer patients. Carcinogenesis. 2014;35(7):1510–5.
Yuan JH, Yang F, Wang F, Ma JZ, Guo YJ, Tao QF, et al. A long noncoding RNA activated by TGF-beta promotes the invasion-metastasis cascade in hepatocellular carcinoma. Cancer Cell. 2014;25(5):666–81.
Ling H, Vincent K, Pichler M, Fodde R, Berindan-Neagoe I, Slack FJ, et al. Junk DNA and the long non-coding RNA twist in cancer genetics. Oncogene. 2015;34(39):5003–11.
Tsang WP, Wong TW, Cheung AH, Co CN, Kwok TT. Induction of drug resistance and transformation in human cancer cells by the noncoding RNA CUDR. RNA. 2007;13(6):890–8.
Jiang M, Huang O, Xie Z, Wu S, Zhang X, Shen A, et al. A novel long non-coding RNA-ARA: adriamycin resistance-associated. Biochem Pharmacol. 2014;87(2):254–83.
Yang Y, Li H, Hou S, Hu B, Liu J, Wang J. The noncoding RNA expression profile and the effect of lncRNA AK126698 on cisplatin resistance in non-small-cell lung cancer cell. PLoS One. 2013;8(5):e65309.
Cheng N, Li X, Zhao C, Ren S, Chen X, Cai W, et al. Microarray expression profile of long non-coding RNAs in EGFR-TKIs resistance of human non-small cell lung cancer. Oncol Rep. 2015;33(2):833–9.
Dong S, Qu X, Li W, Zhong X, Li P, Yang S, et al. The long non-coding RNA, GAS5, enhances gefitinib-induced cell death in innate EGFR tyrosine kinase inhibitor-resistant lung adenocarcinoma cells with wide-type EGFR via downregulation of the IGF-1R expression. J Hematol Oncol. 2015;8(1):43.
Wright CM, Kirschner MB, Cheng YY, O’Byrne KJ, Gray SG, Schelch K, et al. Long non coding RNAs (lncRNAs) are dysregulated in malignant pleural mesothelioma (MPM). PLoS One. 2013;8:e70940.
Renganathan A, Kresoja-Rakic J, Echeverry N, Ziltener G, Vrugt B, Opitz I, et al. GAS5 long non-coding RNA in malignant pleural mesothelioma. Mol Cancer. 2014;13:119.
Lu X, Fang Y, Wang Z, Xie J, Zhan Q, Deng X, et al. Downregulation of gas5 increases pancreatic cancer cell proliferation by regulating CDK6. Cell Tissue Res. 2013;354(3):891–6.
Liu Z, Wang W, Jiang J, Bao E, Xu D, Zeng Y, et al. Downregulation of GAS5 promotes bladder cancer cell proliferation, partly by regulating CDK6. PLoS ONE. 2013;8(9):e73991.
Qiao HP, Gao WS, Huo JX, Yang ZS. Long non-coding RNA GAS5 functions as a tumor suppressor in renal cell carcinoma. Asian Pac J Cancer Prev. 2013;14(2):1077–82.
Tu ZQ, Li RJ, Mei JZ, Li XH. Down-regulation of long non-coding RNA GAS5 is associated with the prognosis of hepatocellular carcinoma. Int J Clin Exp Pathol. 2014;7(7):4303–9.
Cao S, Liu W, Li F, Zhao W, Qin C. Decreased expression of lncRNA GAS5 predicts a poor prognosis in cervical cancer. Int J Clin Exp Pathol. 2014;7(10):6776–83.
Dufour M, Dormond-Meuwly A, Demartines N, Dormond O. Targeting the mammalian target of rapamycin (mTOR) in cancer therapy: lessons from past and future perspectives. Cancers (Basel). 2011;3(2):2478–500.
Zaytseva YY, Valentino JD, Gulhati P, Evers BM. mTOR inhibitors in cancer therapy. Cancer Lett. 2012;319(1):1–7.
Gezer U, Özgür E, Cetinkaya M, Isin M, Dalay N. Long non-coding RNAs with low expression levels in cells are enriched in secreted exosomes. Cell Biol Int. 2014;38(9):1076–9.
Acknowledgments
This work was supported by grants from the Natural Science Foundation of Jiangsu Province (no. BK2011658), Clinical Science and Technology Project of Jiangsu Province (no. BL2013026), and the National Natural Science Foundation of China (no. 81401903). We apologize to all researchers whose relevant contributions were not cited due to space limitations.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Rights and permissions
About this article
Cite this article
Ma, C., Shi, X., Zhu, Q. et al. The growth arrest-specific transcript 5 (GAS5): a pivotal tumor suppressor long noncoding RNA in human cancers. Tumor Biol. 37, 1437–1444 (2016). https://doi.org/10.1007/s13277-015-4521-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-4521-9