Abstract
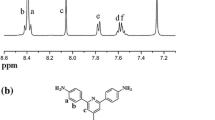
A new aromatic polyimides (PIs) having asymmetric aromatic diamine with methyl groups at the ortho position of amino group has been developed to prepare transparent film in the visible region. The all aromatic PIs presented here are derived from 4-(4-aminophenoxy)-2,6-dimethylaniline (APDMA) and aromatic dianhydride, 4,4'-(hexafluoroisopropylidene)diphthalic anhydride (6FDA), 4,4'-oxydiphthalic anhydride (ODPA), 4,4'-biphthalic anhydride (BPDA), and pyromellitic dianhydride (PMDA) via a two-step polycondensation. All PIs show good thermal properties: 5% weight loss at a temperature range of 510-529 °C and their glass transition temperature found above 290 °C. The PIs exhibit good optical transparency, such as cut-off wavelength in the region of 296-358 nm and above 92-99% at 550 nm. In addition, all PIs show low refractive indices in the range of 1.567-1.637 at wavelength of 637 nm and low birefringence.

Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
H. Choi, S.-J. Ko, Y. Choi, P. Joo, T. Kim, B. R. Lee, J.-W. Jung, H. J. Choi, M. Cha, and J.-R. Jeong, Nat. Photonics, 7, 732 (2013).
M. S. Jung, J. H. Seo, M. W. Moon, J. W. Choi, Y. C. Joo, and I. S. Choi, Adv. Energy Mater., 5, 1400611 (2015).
S. Park, B. J. Park, S. Yun, S. Nam, S. K. Park, and K.-U. Kyung, Opt. Express, 22, 23433 (2014).
K. I. Park, J. H. Son, G. T. Hwang, C. K. Jeong, J. Ryu, M. Koo, I. Choi, S. H. Lee, M. Byun, and Z. L. Wang, Adv. Mater., 26, 2514 (2014).
H. Souri, S. J. Yu, H. Yeo, M. Goh, J.-Y. Hwang, S. M. Kim, B.-C. Ku, Y. G. Jeong, and N.-H. You, RSC Adv., 6, 52509 (2016).
F. Di Giacomo, V. Zardetto, A. D'Epifanio, S. Pescetelli, F. Matteocci, S. Razza, A. Di Carlo, S. Licoccia, W. M. Kessels, and M. Creatore, Adv. Energy Mater., 5, 1401808 (2015).
N.-H. You, C.-C. Chueh, C.-L. Liu, M. Ueda, and W.-C. Chen, Macromolecules, 42, 4456 (2009).
N. H. You, N. Fukuzaki, Y. Suzuki, Y. Nakamura, T. Higashihara, S. Ando, and M. Ueda, J. Polym. Sci., Part A: Polym. Chem., 47, 4428 (2009).
N. H. You, Y. Nakamura, Y. Suzuki, T. Higashihara, S. Ando, and M. Ueda, J. Polym. Sci., Part A: Polym. Chem., 47, 4886 (2009).
N. H. You, T. Higashihara, S. Ando, and M. Ueda, J. Polym. Sci., Part A: Polym. Chem., 48, 2604 (2010).
N. H. You, N. Fukuzaki, Y. Suzuki, Y. Nakamura, T. Higashihara, S. Ando, and M. Ueda, J. Polym. Sci., Part A: Polym. Chem., 47, 4428 (2009).
H. Yeo, J. Lee, M. Goh, B. C. Ku, H. Sohn, M. Ueda, N. and H. You, J. Polym. Sci., Part A: Polym. Chem., 53, 944 (2015).
Y.-H. Chou, N.-H. You, T. Kurosawa, W.-Y. Lee, T. Higashihara, M. Ueda, and W.-C. Chen, Macromolecules, 45, 6946 (2012).
M. Nishihara, L. Christiani, A. Staykov, and K. Sasaki, J. Polym. Sci., Part B: Polym. Phys., 52, 293 (2014).
L. Christiani, S. Hilaire, K. Sasaki, and M. Nishihara, J. Polym. Sci., Part A: Polym. Chem., 52, 2991 (2014).
J. M. Salley and C. W. Frank, Plastics Engineering-New York, 36, 279 (1996).
H. Yeo, M. Goh, B.-C. Ku, and N.-H. You, Polymer, 76, 280 (2015).
S. D. Kim, S. Y. Kim, and I. S. Chung, J. Polym. Sci., Part A: Polym. Chem., 51, 4413 (2013).
P. K. Tapaswi, M. C. Choi, S. Nagappan, and C. S. Ha, J. Polym. Sci., Part A: Polym. Chem., 53, 479 (2015).
Y. Lu, G. Xiao, H. Chi, Y. Dong, and Z. Hu, High Perform. Polym., 25, 894 (2013).
Z. Huang and J. Zhao, RSC Adv., 6, 34825 (2016).
D. H. Wang, J. K. Riley, S. P. Fillery, M. F. Durstock, R. A. Vaia, and L. S. Tan, J. Polym. Sci., Part A: Polym. Chem., 51, 4998 (2013).
J. Zhao, L. Peng, Y.-L. Zhu, A.-M. Zheng, and Y.-Z. Shen, Polym. Chem., 7, 1765 (2016).
B. Ghanem, N. Alaslai, X. Miao, and I. Pinnau, Polymer, 96, 13 (2016).
Y. Guan, C. Wang, D. Wang, G. Dang, C. Chen, H. Zhou, and X. Zhao, Polymer, 62, 1 (2015).
J. Lim, H. Yeo, M. Goh, B.-C. Ku, S. G. Kim, H. S. Lee, B. Park, and N.-H. You, Chem. Mater., 27, 2040 (2015).
J. Lim, D. G. Shin, H. Yeo, M. Goh, B. C. Ku, C. M. Yang, D. S. Lee, J. Y. Hwang, B. Park, and N. H. You, J. Polym. Sci., Part B: Polym. Phys., 52, 960 (2014).
D.-J. Liaw, K.-L. Wang, Y.-C. Huang, K.-R. Lee, J.-Y. Lai, and C.-S. Ha, Prog. Polym. Sci., 37, 907 (2012).
M. J. Frisch, et al., Gaussian 03 (Revision C.01), Gaussian, Inc., Wallingford CT (2004).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bong, S., Yeo, H., Goh, M. et al. Synthesis and characterization of colorless polyimides derived from 4-(4-aminophenoxy)-2,6-dimethylaniline. Macromol. Res. 24, 1091–1097 (2016). https://doi.org/10.1007/s13233-016-4153-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-016-4153-y