Abstract
The nutrient content of soils is considered a key productivity factor. Sediment input from Amazonian rivers is one of the natural sources of soil fertility for fluvial islands and riverbank flooded forests. Despite the importance of soil factors for ecosystems, few edaphic studies along fluvial islands sediment-gradients have been undertaken in tropical areas. The current study provides a step forward by describing a mixed water (sediment-poor black water with the input of sediment-rich white water) fluvial archipelago with an emphasis on soil characteristics of seasonally inundated forests. To investigate how geographic distance from a white-water river mouth affects island soil properties, soil chemical and physical attributes, we sampled 61 sites on 35 islands at Jaú and Anavilhanas archipelago. The studied Central Amazon fluvial islands showed high variability in hydromorphic soils properties. In general, the fluvial island soils were acid and with low fertility. Islands from Jaú and other sites closer to the sediment source (Branco River) had higher soil fertility than the Anavilhanas Archipelago islands, which are further away from the sediment source. Our results show that sediment inputs from the Branco River can play an important role in soil properties in a relatively nutrient-poor area, and shed light on the role of biogeochemical processes in the largest freshwater archipelago in the world. Given that soil fertility is often correlated with forest productivity, the results reported here may serve as a baseline to support conservation politics with scientific data for Central Amazonian fluvial island environments.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Floodplains represent 3–4% of the Amazon basin, covering more than 750,000 Km2 (Wittmann and Junk 2016). Such environments are subject to inundation for several months every year (Junk et al. 1989). The sediment input from large Amazonian rivers is one natural source of fertility for flooded forests growing on riverbanks (Junk et al. 2011). However, the volume of organic ions and sediments received vary with location. Flooded forests periodically inundated by sediment-rich white water (várzeas) grow on fertile soils, while those inundated by sediment-poor black and clearwater rivers (igapós) grows on oligotrophic and poorer soils (Prance 1979; Junk et al. 2011; Goulding et al. 2003). Although very useful, such categorical classification of water types simplifies a natural continuum. For example, the Branco River has an intermediate amount of organic ions, greater than that present in Guiana Shied-derived black water rivers, such as Negro River, but less than those found in rivers of Andean origin such as the Rio Amazonas, Rio Madeira, or Rio Solimões (Goulding et al. 2003). The Branco River is unusual because it is a sediment-rich white-water river where all neighboring rivers are sediment-poor (black-water) (Leenheer and Santos 1980). This spatial configuration may directly affect the Anavilhanas Archipelago, a system of fluvial islands within the nutrient-poor Negro River domain (Marinho et al. 2020).
Fluvial islands are subjected to similar environmental constraints imposed by their inundating rivers. Due to the seasonal influx of nutrients in sediment-rich white-water rivers, fluvial islands can be productive ecosystems that play an important role in biogeochemical cycles and geomorphological processes (Kalliola et al. 1991; Junk and Piedade 1993; Osterkamp 1998; Abril et al. 2013). The input of sediments from Amazonian rivers synchronizes with annual phenological patterning of leaf and fruit production (Montero et al. 2014), responsible for seasonal lateral movements of vertebrates between flooded and non-flooded environments (Haugaasen and Peres 2007; Costa et al. 2018). However, in black rivers, the input of sediment loading in islands is much lower (Marinho et al. 2020), leading to nutrient limitations that may scale up through the food chain. For instance, soil nutrient limitations may constrain the amount and quality of food sources available in forests growing on them, which may affect the terrestrial mammal’s species composition (Ferreira Neto et al. 2021).
The soil nutrient content is strongly linked to ecosystem productivity (Quesada et al. 2012; Grau et al. 2017). A global meta-analysis found that, as a general pattern, higher productivity (defined as the rate of carbon fixed through photosynthesis accessible to flora and fauna) can be a robust indicator of species diversity of several invertebrates, vertebrates and plant groups for most ecosystems (Mittelbach et al. 2001). For instance, more fertile soils provide more favorable conditions for plant establishment and growth (Malhi et al. 2004), investing more carbon in reproduction (i.e., flowers and fruits) than in secondary compounds or nutrient acquisition via root production and exudation (Vitousek 1984; Coley et al. 1985; Quesada et al. 2010, 2011). However, an increase in environmental productivity does not always mean an increase in species diversity. The relationship between environmental productivity and species diversity may be negative (Peres 1997), positive (Ferreira Neto et al. 2021), unimodal (Ganzhorn et al. 1997), U-shaped (Shepherd 1998), or inexistent (Williams and Marsh 1998). Despite the importance of soil factors for several groups of plants and animals, the importance of nutrients has been relatively overlooked and may vary between Amazonian flooded forests (Furch and Junk 1997; Piedade et al. 2005; Targhetta et al. 2015; Myster 2015).
Amazonian archipelagos have been, and continue to be, threatened by anthropogenic disturbances, such as dams and logging (Macedo and Castello 2015; Flores et al. 2017). For example, in the Anavilhanas Archipelago, hunting, logging and sand extraction are quickly changing islands’ landscape (Souza Filho et al. 2006; Scabin et al. 2012; ICMBio 2017). These stressors should be considered since the increase of extreme climate events associated with the frequency and volume of precipitation is predicted to affect the flooding pulse in the Amazon and the contribution of sediments of Amazonian rivers (Filizola et al. 2011; Marengo and Espinoza 2016). The aim of this study was to provide information on how sediment inputs from a sediment-rich white-water river can affect soil fertility on downstream fluvial islands located in a sediment-poor black water system. The hypothesis is that, by supplying high volumes of sediments to the Negro River, the presence of the Branco River will favor variation in soil fertility in a mixed water riverine archipelago in the region. The main prediction is that soils closer to the Branco River (Jaú and adjacent sites) will show greater fertility than those further down the Negro River (Anavilhanas).
Methods
Study Area
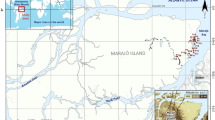
The study was carried out within two protected areas and surrounding sites on the Negro River, Central Amazonia, Brazil ─ Anavilhanas National Park (more distant from the Branco River)—(3.4168° S, 65.8561° W) and Jaú National Park close to Branco River—(2.2535° S, 62.6510° W) (Fig. 1). The annual flood pulse (the inundation of the floodplain and its subsequent dry phase) is seasonal and gradual (Junk et al. 1989). Fluvial islands and river margin forests are often flooded to depths of up to 15 m for more than seven months per year during peak water levels (Irion et al. 1997). Most fluvial islands are completely inundated during this period, so temporarily transforming terrestrial environments into aquatic ones (Junk 2013; Montero and Latrubesse 2013). Fluvial islands can be differentiated from terra-firme forests (not subject to seasonal flooding) also based on hydro-chemical (Sioli 1968; Marinho et al. 2020) and floristic differences (Prance 1979). At both Anavilhanas and Jaú National Parks, the water is at the lowest level between October and November (Montero and Latrubesse 2013) and at the maximum level between June and July (Filizola et al. 2011). The rainfall at both sites varies from 1750 to 2500 mm (ICMBio 2017). The lowest rainfall occurs between June and September, while October to May has the highest values (Salati and Marques 1984). The climate at the study sites is tropical rainy, characterized by high moisture and rainfall, with a mean monthly temperature of 25 °C (Salati and Marques 1984). All sampled islands had hydromorphic soils (ICMBio 2017).
Location of sampling sites of the study at the fluvial islands in the Negro River, Central Amazonia, Brazil. White dots indicate sites closer to the Branco River in front of Jaú National park. Black dots indicate more distant sites at Anavilhanas National park, where the soil chemical and physical attributes were collected
Sampling Design
We firstly established a 2 by 2 km virtual sampling grid in the entire study area and then placed sampling stations as close as logistically possible to the grid intercept points. Satellite images from Google Earth GeoEye were used, with decisive criteria based on river water color. Consequently, our sampling effort was concentrated on islands on the left side of Negro River. This design covered the majority of the fluvial islands within Jaú and Anavilhanas National Park, resulting in 61 sampling sites: 27 sites occurred on islands at vicinities of Jaú National Park, while 34 sites were placed on islands further from the Branco River in the Anavilhanas National Park (Fig. 1). At each site, we established a 0.1 ha plot (10 × 100 m). The maximum above-water area of the islands sampled ranged from 50 to 11,000 hectares (mean ± SD = 2.406 ha ± 2.766; Median = 962; Max = 11.064) based on satellite images. Almost 80% of the sites were placed on islands smaller than 2.000 hectares.
Soil Sampling and Analysis
Soil samples were collected at each sampling site, with a minimum distance of 10 m between samples. The vegetation of the sampling sites was composed of fluvial and lacustrine ecosystem elements, which in the Amazon is called igapó forests. The soils were collected at the margins of permanent natural fluvial islands. The natural islands of Anavilhanas and Jaú are not uniform in topography and geomorphology, which varies with the fluvial dynamics and the age and of the islands (Cunha and Sawakuchi 2017).
After leaf litter removal, the soil was collected to a depth of 20 cm using a Dutch auger brand PSP TRADOS & INOX, which allows us to collect soils with large volumes of roots. All samples within a plot were then combined in order to obtain one pooled sample per site. Soil sampling was conducted during the dry season of 2017. For each pooled soil sample, inorganic nitrogen—ammonium (NH4+) and nitrate (NO3−) were prevented from volatilizing by storing field samples individually in a styrofoam box and transferring them to the ICMBio base freezer at Novo Airão. Chemical analyses were carried out at the Thematic Laboratory of Soils and Plants at the National Institute of Amazonian Research (INPA, Manaus), with soils frozen upon arrival in the laboratory to avoid volatilization.
The following soil characteristics were obtained: 1) pH, determined using 10 g of fine soil in 25 ml H2O for 1 min, with a mechanical stirrer; 2) concentrations of Na, K, Ca, Mg, Al, all determined with the Silver-Thiourea (Ag-TU, as described in Quesada et al. 2010); 3) Sum of bases was considered as the sum of concentrations of Na (sodium), K (potassium), Ca (calcium) and Mg (magnesium) (Pleysier and Juo 1980), while cation exchange capacity was calculated from the equation: Cation Exchange Capacity = (Sum of Bases + Aluminium) (Pleysier and Juo 1980). For this, cationic extraction was made in a 0.01 M Ag-TU solution, stirred together with the sample for 4 h (Anderson and Ingram 1989); 4) To estimate total phosphorus and total cations, soil samples were digested using sulfuric acid and hydrogen peroxide, with 0.25 g of soil added to 5 ml of distilled water and left to settle overnight. The following day, 5 ml of H2SO4 was added to the sample and heated to 360 ºC, and 0.5 ml of hydrogen peroxide (H2O2) was added after cooling the sample. This process was repeated 10 times. Total phosphorus reading was obtained by colorimetry in a Shimadzu Spectrophotometer, while total cations were read with atomic absorption via Flame Atomic Absorption Spectrometry (AAS) (Murphy and Riley 1962). Available organic (Po) and Inorganic phosphorus (Pi) were extracted with NaHCO3 (Sodium Bicarbonate) (Olsen 1954). 5) Preparation of extracts for inorganic nitrogen analysis used 20 g of soil and 40 mm of potassium sulfate (K2SO4) at 0.5 molar in falcon tubes for 15 min with centrifugation at 200 rpm. Extracts were then decanted for 30 min and filtered and frozen for further analysis of N-inorganic contents. Ammonium and nitrate were determined by colorimetry using a Shimadzu Spectrophotometer (UV mini 1240). Samples destined for N analysis were not oven-dried. For all other soil analyses, each soil sample was oven-dried at 65 ºC, and particles smaller than 2 mm removed with a soil sieve of 0.05 (Anderson and Ingram 1989); and 6) Soil particle size was estimated by the pipette method, using 10 g of fine soil pre-treated with physical and chemical dispersants. Then, silt percentage was separated according to their grain size, which was calculated from the difference between the total weight of sand and clay (Gee and Bauder 1986). For more details about soil attributes, see Online Resource 1.
Data Analysis
We used a Principal Component Analysis (PCA) to accommodate correlated soil variables and reduce data dimensionality. To standardize the data and build the PCA, we used the vegan package (Oksanen 2013). Data normality was checked before the PCA.
To describe how soil fertility is affected and change along geographic distance from the Branco River, we performed a linear regression between the first axis of PCA (PC1) and the geographical distance from the Branco River mouth. For this, the center of the closest sampled island to the Branco River mouth (S 01.36634º, W 061.79113º) was used to measure the distance to all other sampling sites. We checked model assumptions visually through graphics of residuals distribution. Additionally to the general soil nutrient patterns summarized by the PCA, we performed a linear regression for key soil factors such as phosphorus and sum of bases, soil texture factors (clay, sand and silt), and pH. These variables were chosen due to their role as key drivers of forest dynamics in terms of productivity, tree turnover, the soil’s capacity to hold essential nutrients (Quesada et al. 2010, 2012), and their relationship with arboreal and terrestrial mammals in Amazonia (Peres 2008; Ferreira Neto et al. 2021). Individual maps for the two important indicators of soil properties (sum of bases and total phosphorus) were analyzed. The regression coefficients and graphical plots can be found on Online Resource 2 and Online Resource 3. All statistical analyzes were performed in R version 3.3.1 (R Core Team 2019).
Results
Most soil nutrients were found at a higher concentration at the sites close to Branco River in relation to the other sites. For organic phosphorus (mean [SD] = 43.5[14.8], range = 24.8–86.3), more than half of the sites had less than 30 mg/kg, while less than a quarter had more than 40 mg/kg. A similar trend was found for inorganic phosphorus (mean [SD] = 30.3[12.8], range = 12.7–62.2), where more than half of the sites had less than 20 mg/kg, while a third had more than 25 mg/kg. The nitrate also varied along the river downstream (mean [SD] = 3.4[2.7], range = 0.5–11.2), only a sixth of the sites had more than 5 ug/g, while for ammonium (mean [SD] = 60.1[24.3], range = 36.7–144.6), a quarter of the sites had more than 50 ug/g and for inorganic nitrogen (mean [SD] = 63.5[24.2], range = 39.2–147.1), a sixth of the sites had more than 60 ug/g. The ratio between nitrate and ammonium decreases from 0.056ug/g islands close to Branco River to 0.049ug/g in the Anavilhanas islands (Table 1).
A diverse range of soil properties was found. In terms of soil particle size, Anavilhanas soils showed less variation than sites closer to the Branco River and could generally be classified as clay loam (average 40 ± 6.1; 36 ± 7.8 and 23 ± 13.1, for silt, clay and sand, respectively). On the other hand, island soils closer to the Branco River were classified as silty clay loam (average 46 ± 8.9; 36 ± 9.1 and 18 ± 14, for silt, clay and sand, respectively). In general, the percentage of silt was higher in sites closer to the Branco River, but there was no statistical difference between percentages of clay along the soil gradient. The fluvial islands’ soils were pretty acidic. In general, the highest pH values (> 5.0) occurred on Jaú islands, whereas the lowest (< 4.5) occurred in the Anavilhanas islands (Table 1).
The first component of the PCA explained ≅ 55%, and the second component explained ≅ 14% of the total variance in soil properties of the fluvial islands (Fig. 2). Jaú sites had more variability (mean [SD] = 4.81 [5.44] – range 0.34 to 19.1), while Anavilhanas sites varied between 0.1 to 10.21 (mean [SD] = 5.005 [2.46] in the PC1. Mobile rock-derived elements were the soil factors that correlated strongest with the PC1, with their influence on the PCA loadings declining in the order SB > ECEC > Mg > Ca > Na > PT. Aluminum was the dominant cation in the studied sites, with magnesium being the second and calcium the third. Non-mobile total phosphorus and available organic P also followed mobile cations (Online Resource 4), which together with the silt of the islands could suggest the recent entry of elements in the islands closer to Branco River and the high weathering of other islands in Anavilhanas. Therefore, mobile elements account for most of the variation in island soil properties.
The distance to the Branco River was a strong driver for island soil fertility (R2 = 0.57; p < 0.05) (Fig. 3). The distance to the Branco River also explains over 30% of the variation in the availability of SB, ECEC, Na, organic P, inorganic P, total P and inorganic N, on the fluvial islands.
Relationship between the geographic distance from the Branco River and soil chemical and physical variables (first axis of principal component analysis). Each point represents one sampling station (n = 61). Lines represent model predictions of statistically supported effects; grey bands indicate 95% confidence intervals
Individual maps for the two important indicators of soil properties (sum of bases and total phosphorus) showed a similar pattern along the edaphic gradient (Fig. 4). A consistent trend across the soil gradient showed that the sum of bases and total phosphorus vary on average more than five or six times, respectively, concerning the lowest values generally found in igapó sites (Fig. 4). The sum of bases concentration increases in Jaú islands, located close to Branco River (mean [SD] = 1.0 [0.5], range = 0.2–2.3), more than a third of the sites ranged between 0.391–0.650 cmolc/kg and about a fifth of the sites had more than 1 cmolc/Kg. Also, the total phosphorus concentration increases Jaú islands (mean [SD] = 350.4 [97.4], range = 142.5–549.8), almost half of the sites had less than 250 mg/kg. However, a third of the sites had more than 300 mg/Kg. Overall, from the 17 soil factors analyzed, 14 soil factors (SB, cation exchange capacity (ECEC), ph (H20), Ca, K, Mg, Na, Porg (organic phosphorus), Pinor (inorganic phosphorus), Ptot (total phosphorus), Ninor (Inorganic nitrogen), Ammonium, Nitrate and Silt) had the highest concentrations close to Branco River. None of the edaphic factors analyzed had the highest concentrations downstream in the Anavilhanas archipelago (Table 1).
Discussion
There was considerable variation in soil fertility and physical properties observed along the fluvial islands investigated. The distance from the mouth of the Branco River was an important predictor of the nutrients concentrations, soil texture and other soil attributes on the fluvial islands downstream. Due to differences in temperature, pH and volume of suspended sediments, the Branco River sediment-rich white water is not rapidly mixed in Negro River sediment-poor black water (Goulding et al. 2003; Marinho et al. 2020). As the Negro River is wide and relatively slow, the suspended nutrients are gradually deposited on the fluvial islands (Latrubesse and Stevaux 2015). Thus, our results indicated that islands closer to the mouth of the Branco River have soils that are richer in nutrients, while islands more distant from the mouth of the Branco River, have on average, poorer soils. The effects of water chemistry and sediment inputs on edaphic factors are widely known, with pertinent examples from many other parts of the world, such as riverine wetlands in northern Minnesota and Wisconsin (Johnston et al. 2001), wetlands in southern Ontario (Rutledge and Chow-Fraser 2019), Alpine lakes in Italy (Tiberti et al. 2010), boreal rivers in Finland (Varanka et al. 2015) and wetland soils of northeast China (Zhang et al. 2013). Our results align with this trend, even though the Branco River is not as rich as other white-water rivers in the Amazon, such as Rio Madeira or Solimões (Goulding et al. 2003).
The differences between sediment inputs of black and white rivers affect not only the forest soil composition of igapó and várzea forests, but it is also applied to other features of plant and animal groups. Overall, soil nutrients such as the sum of bases was highly variable across our longitudinal sample of islands (ranging from 0.18 to 2.34 cmolc/kg). However, soil nutrients (mean = 0.73 cmolc/kg) were generally more fertile than typical igapó soils from Uatumã Reserve (Targhetta et al. 2015). This difference in the nutrient imbalance between igapó, várzea and terra-firme forests can also explain the divergence in plant functional trait diversity in the Colombian Orinoquia flooded forests (Bonilla et al. 2020), species richness and abundance of galling insects at Mamirauá and Amanã Reserves (Julião et al. 2018), and bird species composition in Central Amazonian forests (Beja et al. 2010). This variation in soil nutrients also impacts the animal community at the top of the food chain, as shown on the Anavilhanas and Jaú sites, where the species composition of mammals varies along the same soil gradient described here (Ferreira Neto et al. 2021).
The Branco River is the only major sediment-rich tributary of the Negro River in the archipelago region (Leenheer and Santos 1980; Latrubesse and Franzinelli 2005). The other rivers and water courses that reach the Negro river near the Jaú and Anavilhanas Archipelagos have similar chemical and sedimentological characteristics of the Negro River (Leenheer and Santos 1980; Latrubesse and Franzinelli 2005). Therefore, the concentrations of nutrients and the sediment load at the mouth of the Branco River is the primary source for nutrients and sediments in the downstream archipelago. The variation in soil fertility is usually related to suspended sediments but can also be associated with island age and elevation (Radambrasil 1978). The natural islands of Anavilhanas vary greatly in age, as these features have been forming over hundreds or even thousands of years (Cunha and Sawakuchi 2017). Nutrient concentrations and soil physical conditions are interrelated because both are affected by pedogenetic development and geology. However, soils with longer pedogenetic developments are often less fertile (Quesada et al. 2010, 2012). This relationship may be blurred in these systems, as fluvial islands at the Jaú National Park may harbor the largest variation in soil attributes resulting from Branco River sediments input rather than their pedogenetic development. These sites are located in a different geomorphological reach of the Negro River than Anavilhanas. While the mouth of the Jaú National Park area has huge, muddy islands, the Anavilhanas area contains more compact and wider islands (Latrubesse and Stevaux 2015).
The sedimentation dynamics responsible for the architecture of the archipelago’s fluvial islands could result from fluctuations in climatic conditions in the Branco River basin. In this context, the island’s formation is attributed to the flocculation of fine sediments from the Branco River and variations in sedimentary contribution controlled by precipitation in the Branco River basin (Leenheer and Santos 1980). The flocculation of fine sediments occurs because of the low concentration of suspended sediments in the Negro River (~ 5 mg / l) (Meade et al. 1979) compared to the concentration of suspended sediments in the Branco River (50 to 300 mg / l), near its confluence with the Negro River. In terms of soil granulometry, island soils became less silty and contained low nutrient content with increasing distance from the Branco River. It was expected that the coarser sediments (sands) would be deposited first; thus, the sand content should decrease downstream from the Branco River. However, surprisingly, there was no difference between sand and clay content between Jaú and Anavilhanas islands. One possible explanation is that other smaller water courses with characteristics of black water rivers inserted in the archipelago could also be transporting sand and clay from erosion of the Guyana Shield (Ríos-Villamizar et al. 2020). Another explanation involves the constant and growing commercial exploitation of sand within the archipelago. This sand extraction is illegal and can cause erosion and affect the soil composition and dynamics of the flooded forests, as it tends to leach heavily, altering the alluvial sediment (ICMBio 2017).
We reinforced the importance of sediment inputs to soil fertility in a mixed-water fluvial archipelago, embedded in a nutrient-poor black water river. Other factors such as the local geological matrix, landscape heterogeneity, microtopography, age of sedimentation, and drainage conditions could also account for the soil nutrient variability. Most nutrients used here can be recommended for researchers to quickly assess the relationship between soil fertility and species composition of animals and plants. Additionally, knowledge of spatial variation in soil fertility can improve our understanding of islands’ resilience concerning current and future impacts. For instance, several dams are planned for the Branco River in the near future (Finer and Jenkins 2012; Forsberg et al. 2017). Such water retention could decrease the suspended sediments inputs of the Branco River on the Negro Basin, affecting flora and fauna downstream (Finer and Jenkins 2012; Forsberg et al. 2017). The results reported here can serve as a baseline for conservation policies, to assess relationships involving edaphic factors and sediment inputs from rivers, and their relationship with plant and animal species composition and turnover along a soil gradient.
Data and Availability (data transparency)
All data produced from this study are provided in this manuscript.
Code Availability (software application or custom code)
Not applicable.
References
Abril G, Martinez J, Artigas L, Moreira-Turcq P, Benedetti M, Vidal L, Meziane T, Kim J, Bernardes M, Savoye N, Deborde J, Souza E, Albéric P, Landim Souza M, Roland F (2013) Amazon River carbon dioxide outgassing fuelled by wetlands. Nature 505:395–398. https://doi.org/10.1038/nature12797
Anderson JM, Ingram JSI (1989) Tropical Soil Biology and Fertility. Soil Science 157-265. https://doi.org/10.1097/00010694-199404000-00012
Beja P, Santos CD, Santana J, Pereira MJ, Marques JT, Queiroz HL, Palmeirim JM (2010) Seasonal patterns of spatial variation in understory bird assemblages across a mosaic of flooded and unflooded Amazonian forests. Biodivers Conserv 19:129–152
Bonilla D, Aldana AM, Cárdenas S, Sanchez A (2020) Functional divergence between várzea and igapó forests: A study of functional trait diversity in the Colombian Orinoco Basin. Forests 11:11–1172. https://doi.org/10.3390/f11111172
Coley P, Bryant J, Chapin F (1985) Resource Availability and Plant Antiherbivore Defense. Science 230:895–899. https://doi.org/10.1126/science.230.4728.895
Costa HC, Peres CA, Abrahams MI (2018) Seasonal dynamics of terrestrial vertebrate abundance between Amazonian flooded and unflooded forests. PeerJ 6:5058. https://doi.org/10.7717/peerj.5058
Cunha DFD, Sawakuchi AO (2017) Evolução sedimentar do Arquipélago de Anavilhanas no baixo Rio Negro, Amazônia Central. Master’s Thesis, Universidade de São Paulo, São Paulo, Brazil
Ferreira Neto GDS, Baccaro FB, Spironello WR, Benchimol M, Fleischer K, Quesada CA, Sousa Gonçalves AL, Pequeno PAL, Barnett APA (2021) Soil fertility and anthropogenic disturbances drive mammal species richness and assemblage composition on tropical fluvial islands. Austral Ecol. https://doi.org/10.1111/aec.13023
Finer M, Jenkins CN (2012) Proliferation of hydroelectric dams in the Andean Amazon and implications for Andes-Amazon connectivity. PLoS ONE 7:4–35126. https://doi.org/10.1371/journal.pone.0035126
Flores BM, Holmgren M, Xu C, Van Nes EH, Jakovac CC, Mesquita RC, Scheffer M (2017) Floodplains as an Achilles heel of Amazonian forest resilience. Proc Natl Acad Sci 114:4442–4446. https://doi.org/10.1073/pnas.1617988114
Filizola N, Guyot JL, Wittmann Martinez H, Oliveira JM (2011) The significance of suspended sediment transport determination on the Amazonian hydrological scenario. Sediment Transport in Aquatic Environments 45:64. https://doi.org/10.5772/19948
Forsberg B, Melack JM, Dunne T, Barthem RB, Goulding M, Paiva RC, Weisser S (2017) The potential impact of new Andean dams on Amazon fluvial ecosystems. PLoS ONE 12(8):e0182254. https://doi.org/10.1371/journal.pone.0182254
Furch K, Junk WJ (1997) Physicochemical conditions in the floodplain. The Central Amazonian Floodplain, Floodplain: Ecology of a Pulsing System. Ecological Studies Vol. 126, Springer, Berlin 69–108, 1997
Ganzhorn JU, Malcomber S, Andrianantoanina O, Goodman SM (1997) Habitat characteristics and Lemur species richness in Madagascar. Biotropica 29:331–343. https://doi.org/10.1111/j.1744-7429.1997.tb00434.x
Gee GW, Bauder JW (1986) Particle size analysis. Methods of soil analysis: Part 1 Physical and mineralogical methods 5:383–411. https://doi.org/10.2136/sssabookser5.1.2ed.c15
Goulding M, Barthem RB, Ferreira EJG (2003) The Smithsonian Atlas of the Amazon. Smithsonian Institution, Washington, p 253
Grau O, Peñuelas J, Ferry B, Freycon V, Blanc L, Desprez M, Guitet S (2017) Nutrient-cycling mechanisms other than the direct absorption from soil may control forest structure and dynamics in poor Amazonian soils. Sci Rep 45017:23–27. https://doi.org/10.1038/srep45017
Haugaasen T, Peres CA (2007) Vertebrate responses to fruit production in Amazonian flooded and unflooded forests. Biodivers Conserv 16:4165–4190. https://doi.org/10.1007/s10531-007-9217-z
ICMBio (2017) Plano de Manejo do Parque Nacional de Anavilhanas. Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis. Ministério do Meio Ambiente, ICMBio/MMA, Brasília, p 401
Irion G, Junk WJ, Mello JASN (1997) The large central Amazonian river floodplains near and geomorphological aspects. In: Junk WJ (ed) The Central Amazon Floodplain: Ecology of a Pulsing System. Springer-Verlag, Berlin pp. 23–46. https://doi.org/10.1007/978-3-662-03416-3_2
Johnston CA, Bridgham SD, Schubauer-Berigan JP (2001) Nutrient dynamics in relation to geomorphology of riverine wetlands. Soil Sci Soc Am J 65:557–577
Julião GR, Venticinque EM, Fernandes GW (2018) Influence of Flood Levels on the Richness and Abundance of Galling Insects Associated with Trees from Seasonally Flooded Forests of Central Amazonia, Brazil. Springer, Cham, In Igapó (Black-water flooded forests) of the Amazon Basin (pp. 99–117)
Junk WJ, Bayley PB, Sparks RE (1989) The floodpulse concept in river-floodplain systems. Can Spec Publ Fish Aqu Sci 106:110–127
Junk WJ, Piedade MTF (1993) Herbaceous plants of the flood plain near Manaus: species diversity and adaptations to the flood pulse. Amazoniana 12:467–484
Junk WJ, Piedade MT, Wittmann F, Schöngart J, Parolin P (2010) Amazonian floodplain forests: ecophysiology, biodiversity and sustainable management (Vol. 210). Springer Science & Business Media
Junk WJ, Piedade MTF, Schongart J, Cohn-Haft M, Adeney JM, Wittmann F (2011) A classification of major naturally-occurring Amazonian lowland wetlands. Wetlands 31:623–640. https://doi.org/10.1007/s13157-011-0190-7
Junk WJ (2013) The central Amazon floodplain: ecology of a pulsing system 126. Springer Science & Business Media
Kalliola R, Salo J, Puhakka M, Rajasilta M (1991) New Site Formation and Colonizing Vegetation in Primary Succession on the Western Amazon Floodplains. The Journal of Ecology 877-901. https://doi.org/10.2307/2261087
Latrubesse EM, Stevaux JC (2015) The Anavilhanas and Mariuá Archipelagos: Fluvial Wonders from the Rio Negro, Amazon Basin. In: Vieira BC, Salgado AAR, Santos LJC. Landscapes and Landforms of Brazil. Dordrecht: Springer Netherlands 157–169. https://doi.org/10.1007/978-94-017-8023-0_14
Latrubesse EM, Franzinelli E (2005) The late Quaternary evolution of the Negro River, Amazon, Brazil: Implications for island and floodplain formation in large anabranching tropical systems. Geomorphology 70:372–397
Leenheer JA, Santos UDM (1980) Considerações sobre os processos de sedimentação na água preta ácida do Rio Negro (Amazônia Central). Acta Amazon 10:343–357. https://doi.org/10.1590/1809-43921980102343
Macedo M, Castello L (2015) State of the Amazon: Freshwater connectivity and ecosystem health. WWF Living Amazon Initiative, Brasilia
Malhi Y, Baker T, Phillips O, Almeida S, Alvarez E, Arroyo L, Chave J, Czimczik C, Fiore A, Higuchi N, Killeen T, Laurance S, Laurance W, Lewis S, Montoya L, Monteagudo A, Neill D, Vargas P, Patino S, Pitman N, Quesada C, Salomao R, Silva J, Lezama A, Martinez R, Terborgh J, Vinceti B, Lloyd J (2004) The above-ground coarse wood productivity of 104 Neotropical forest plots. Glob Change Biol 10:563–591. https://doi.org/10.1111/j.1529-8817.2003.00778.x
Marengo JA, Espinoza JC (2016) Extreme seasonal droughts and floods in Amazonia: causes, trends and impacts. Int J Climatol 36:1033–1050. https://doi.org/10.1002/joc.4420
Marinho RR, Filizola Jr, Cremon NP (2020) Analysis of Suspended Sediment in the Anavilhanas Archipelago, Rio Negro, Amazon Basin. Water 1073:4-12. https://doi.org/10.3390/w12041073
Meade RH, Nordin CF, Curtis WF, Costa Rodrigues FM, Edmond JM (1979) Transporte de sedimentos no rio Amazonas. Acta Amazon 9:529–547
Mittelbach GG, Steiner CF, Scheiner SM, Gross KL, Reynolds HL, Waide RB, Willig MR, Dodson SI, Gough L (2001) What is the observed relationship between species richness and productivity? Ecology 82:2381–2396. https://doi.org/10.1890/02-3128
Myster RW (2015) Black-water forests (igapó) vs. white-water forests (várzea) in the Amazon: floristics and physical structure. The Biologist (lima) 13:391–406
Montero JC, Latrubesse EM (2013) The igapó of the Rio Negro in central Amazonia: linking late-successional inundation forest with fluvial geomorphology. J S Am Earth Sci 46:137–149. https://doi.org/10.1016/j.jsames.2013.05.009
Montero JC, Piedade MTF, Wittmann F (2014) Floristic variation across 600 km of inundation forests (igapó) along the Rio Negro, Central Amazonia. Hydrobiologia 729:229–246. https://doi.org/10.1007/s10750-012-1381-9
Murphy J, Riley J (1962) A modified single solution method for the determination of phosphate in natural waters. Anal Chim Acta 27:31–36. https://doi.org/10.1016/s0003-2670(00)88444-5
Oksanen J (2013) Multivariate Analysis of Ecological Communities in R: Vegan Tutorial. http://vegan.r-forge.r-project.org. Accessed 13 Aug 2020
Olsen RS (1954) Estimation of available phosphorus in soils by extraction with sodium bicarbonate 939. US Dept. Agric Stat
Osterkamp W (1998) Processes of fluvial island formation, with examples from Plum Creek, Colorado and Snake river, Idaho. Wetlands 18:530–545. https://doi.org/10.1007/bf03161670
Peres CA (1997) Primate community structure at twenty western Amazonian flooded and unflooded forests. J Trop Ecol 13:381–405. https://doi.org/10.1017/s0266467400010580
Peres CA (2008) Soil fertility and arboreal mammal biomass in tropical forests. Trop for Commun Ecol 349:364
Piedade MTF, Junk WJ, Adis J, Parolin P (2005) Ecologia, zonação e colonização da vegetação arbórea das Ilhas Anavilhanas. Pesquisas, Botânica 56:117–143
Pleysier JL, Juo ASR (1980) A single-extraction method using silver-thiourea for measuring exchangeable cations and effective CEC in soils with variable charges. Soil Sci 129:205–211. https://doi.org/10.1097/00010694-198004000-00002
Prance G (1979) Notes on the Vegetation of Amazonia III. The Terminology of Amazonian Forest Types Subject to Inundation. Brittonia 31:26–38. https://doi.org/10.2307/2806669
Quesada C, Lloyd J, Schwarz M, Patiño S, Baker T, Czimczik C, Fyllas N, Martinelli L, Nardoto G, Schmerler J, Santos A, Hodnett M, Herrera R, Luizão F, Arneth A, Lloyd G, Dezzeo N, Hilke I, Kuhlmann I, Raessler M, Brand W, Geilmann H, Moraes Filho J, Carvalho F, Araujo Filho R, Chaves J, Cruz Junior O, Pimentel T, Paiva R (2010) Variations in chemical and physical properties of Amazon forest soils in relation to their genesis. Biogeosciences 7:1515–1541. https://doi.org/10.5194/soil-6-53-2020
Quesada CA, Lloyd J, Anderson LO, Fyllas NM, Schwarz M, Czimczik CI (2011) Soils of Amazonia with particular reference to the RAINFOR sites. Biogeosciences 8:1415–1440. https://doi.org/10.5194/bg-8-1415-2011
Quesada CA, Phillips OL, Schwarz M, Czimczik CI, Baker TR, Patiño S, Fyllas NM, Hodnett MG, Herrera Almeida R (2012) Basin-wide variations in Amazon forest structure and function are mediated by both soils and climate. Biogeosciences 9:2203–2246. https://doi.org/10.5194/soil-6-53-2020
Radambrasil (1978) Levantamento dos Recursos Naturais, Ministério das minas e energia, Departamento nacional da produção mineral. Folha SA-20 Manaus. DNPM/Projeto Radambrasil, Rio de Janeiro
R Core Team (2019) R : A language and environment for statistical computing. R foundation for statistical computing, Vienna, Austria. https://www.R-project.org. Accessed 13 Aug 2020
Ríos-Villamizar EA, Adeney JM, Piedade MTF, Junk WJ (2020) New insights on the classification of major Amazonian river water types. Sustainable Water Resources Management 6:1–16. https://doi.org/10.1007/s40899-020-00440-5
Rutledge JM, Chow-Fraser P (2019) Landscape characteristics driving spatial variation in total phosphorus and sediment loading from sub-watersheds of the Nottawasaga River, Ontario. J Environ Manage 234:357–366
Salati E, Marques J (1984) Climatology of the Amazon region. In The Amazon, pp. 85–126. Springer, Dordrecht. https://doi.org/10.1007/978-94-009-6542-3_4
Scabin AB, Costa FRC, Schöngart J (2012) The spatial distribution of illegal logging in the Anavilhanas archipelago (Central Amazonia) and logging impacts on species. Environ Conserv 39:111–121. https://doi.org/10.1017/s0376892911000610
Shepherd UL (1998) A Comparison of Species Diversity and Morphological Diversity across the North American Latitudinal Gradient 25:19–29. https://doi.org/10.1046/j.1365-2699.1998.251172.x
Sioli H (1968) Hydrochemistry and geology in the Brazilian Amazon region. Amazoniana 1:267–277
Souza Filho PWM, Paradella WR, Souza Jr, Valeriano C, Miranda FP (2006) Sensoriamento Remoto e recursos naturais da Amazônia. Ciência e Cultura. 58:37-41
Targhetta N, Kesselmeier J, Wittmann F (2015) Effects of the hydroedaphic gradient on tree species composition and aboveground wood biomass of oligotrophic forest ecosystems in the central Amazon basin. Folia Geobot 50:185–205. https://doi.org/10.1007/s12224-015-9225-9
Tiberti R, Tartari GA, Marchetto A (2010) Geomorphology and hydrochemistry of 12 Alpine lakes in the Gran Paradiso National Park, Italy. J Limnol 69:242–256
Varanka S, Hjort J, Luoto M (2015) Geomorphological factors predict water quality in boreal rivers. Earth Surf Proc Land 40:1989–1999
Vitousek PM (1984) Litterfall, nutrient cycling, and nutrient limitation in tropical forests. Ecology 65:285–298. https://doi.org/10.2307/1939481
Williams SE, Marsh H (1998) Changes in small mammal assemblage structure across a rain forest/open forest ecotone. J Trop Ecol 14:187–198. https://doi.org/10.1017/s0266467498000157
Wittmann F, Junk WJ (2016) The Amazon river basin. The Wetland book II: Distribution, description and conservation 1-16. https://doi.org/10.1007/978-94-007-6173-5_83-1
Zhang ZS, Song XL, Lu XG, Xue ZS (2013) Ecological stoichiometry of carbon, nitrogen, and phosphorus in estuarine wetland soils: influences of vegetation coverage, plant communities, geomorphology, and seawalls. J Soils Sediments 13:1043–1051
Acknowledgements
We had the material support from the Thematic Laboratory of Soils and Plants (INPA) and the logistic support from ICMBio (permit numbers 55180-1 and 59367-1). Special thanks to Dr. Orlando Cruz and Jonas Souza Filho for instructions to performing soil analysis. We acknowledge the Amazonian Mammal Research Group. GDSFN thanks the National Council for Scientific and Technological Development [130817/2016-3], CAPES for graduate program support and The National Institute of Amazonian Research. FBB is continuously supported by a CNPq grant (313986/2020-7).
Funding
This study was funded by The Rufford Foundation (grant number 20754- 1 to GSFN) and Idea Wild.
Author information
Authors and Affiliations
Contributions
GDSFN wrote the first draft of the manuscript. All authors read and approved the final manuscript. GDSFN conducted field sampling. GDSFN, AB and FBB conceived the study. GDSFN, ASF, and EG performed laboratory analysis. GDSFN, CAQ, and WRS supported the study. RO helped with methodology. GDSFN and FBB performed data analysis.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no conflict of interest.
Ethics Approval
This study was approved by the Chico Mendes Institute for Biodiversity Conservation (ICMBio), (permit numbers 55180–1 and 59367–1).
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
de Souza Ferreira Neto, G., Baccaro, F.B., Quesada, C.A.N. et al. Sedimental Journey: Soil Fertility of Fluvial Islands Increases with Proximity to An Amazonian White-Water River. Wetlands 41, 104 (2021). https://doi.org/10.1007/s13157-021-01506-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13157-021-01506-6