Abstract
We document patterns of fruit and vertebrate abundance within an extensive, virtually undisturbed mosaic of seasonally flooded (várzea and igapó) and unflooded (terra firme) forests of central Amazonia. Using phenological surveys and a standardised series of line-transect censuses we investigate the spatial and temporal patterns of immature and mature fruit availability and how this may affect patterns of habitat use by vertebrates in the landscape. All habitats showed marked peaks in fruiting activity, and vertebrate detection rates varied over time for most species both within and between forest types. Many arboreal and terrestrial vertebrates used both types of flooded forest on a seasonal basis, and fluctuations in the abundance of terrestrial species in várzea forest were correlated with fruit availability. Similarly, the abundance of arboreal seed predators such as buffy saki monkeys (Pithecia albicans) and macaws (Ara spp.) were closely linked with immature fruit availability in terra firme forest. We conclude that highly heterogeneous landscapes consisting of terra firme, várzea and igapó forest appear to play an important role in the dynamics of many vertebrate species in lowland Amazonia, but the extent to which different forest types are used is highly variable in both space and time.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Phenological studies of tropical forest plants have shown that reproductive plant parts are episodic, resulting in community-wide seasonal peaks in the availability of flowers and fruits (South-east Asia: Medway 1972; Hilty 1980; Leighton and Leighton 1983; van Schaik 1986; Wong 1986; South and Central America: Frankie et al. 1974; Opler et al. 1980; Foster 1982; Terborgh 1983; Peres 1994; mainland Africa: Lieberman 1982; Gautier-Hion et al. 1985; White 1994; Chapman et al. 1999). Likewise, several long-term studies have shown significant differences in flower and fruit availability between years (Foster 1982; Leighton and Leighton 1983; Corlett 1990).
Vertebrate frugivores are the dominant group of large animals in tropical forests. For example, approximately one-third of the resident avifauna in many neotropical forests are either frugivorous or granivorous (Terborgh 1980; Stiles 1985; Blake et al. 1990; Peres and Whittaker 1991), this proportion being even larger in mammals (Terborgh 1983, 1986; Haugaasen and Peres 2005a). The number of species that at least occasionally consume fruits is much larger. This is reflected in the role of frugivores as seed dispersal agents; some 90% of neotropical forest tree species, and up to 98% of small understorey plants are dispersed by birds and mammals (Gentry 1982; Howe and Smallwood 1982; Stiles 1985; van Roosmalen 1985; Peres and van Roosmalen 2002). Spatiotemporal variation in flowering and fruiting pulses in plant communities may therefore strongly influence vertebrates that rely wholly or partly on these resources (e.g. Moegenburg and Levey 2003). Seasonal shifts in fruit abundance thus appear to be an important component of vertebrate assemblage dynamics in tropical forests, although little is known about the ecological consequences of such seasonal cycles at large spatial scales. It is becoming increasingly clear, however, that changes in fruit and flower abundance influence the ranging and foraging behaviour of frugivorous and nectivorous birds, as non-demographic temporal changes in local population abundance appear to be common (e.g. Leck 1972; Greenberg 1981; Karr and Freemark 1983; Levey 1988; Blake and Loiselle 1991; Loiselle and Blake 1991). This supports the general hypothesis that seasonal movements may be linked to spatiotemporal changes in resource supply.
In this paper, we document the spatial and temporal patterns of immature and mature fruit availability in a natural mosaic of seasonally flooded and unflooded forest of central Amazonia, and how this may influence local vertebrate abundance. We examine food availability as a determinant of habitat use by 43 mid-sized to large-bodied mammal, bird and reptile species, and infer whether the dependence on spatiotemporally variable resources such as fruits necessitates seasonal movements. We thus attempt to pinpoint vertebrate species that respond to fruit lows by making seasonal movements to more favourable habitats. We work on the assumption that if a frugivorous species is affected by fruit abundance, we would observe concurrent changes in frugivore abundance and inferred movement patterns. In particular we address the following questions: (1) How seasonally variable is fruit production and the abundance of large-bodied vertebrates in the natural forest mosaic? (2) Are fluctuations in fruit availability and consumer abundance correlated? (3) How important is the landscape heterogeneity of the forest mosaic to the resident large vertebrate fauna? And (4) when do lateral movements of large vertebrates occur in a lowland tropical forest mosaic?
Study area
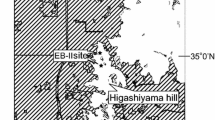
The study took place from February 2002 to November 2003 at Lago Uauaçú (04°20′S, 62°28′W) in the lower Rio Purús region of central Amazonia, about 350 km south-west of Manaus (Fig. 1). Lago Uauaçú is a 32-km long crescent-shaped black-water lake fed by rainfall collected in an internal catchment consisting primarily of terra firme forest. The study area consisted of a natural mosaic of unflooded upland (terra firme) and floodplain forests inundated on a seasonal basis by either white-water (várzea) or black-water (igapó; sensu Prance 1979) for as long as 6 months of the year (Fig. 1). The várzea forests trails F, G, I and J were unconnected from terra firme by a ∼50-m wide river channel, whereas trail K intersected a portion of várzea that merged into adjacent terra firme forest (Fig. 1). The study area retains a full complement of vertebrate species and remains relatively undisturbed, but was occupied by 30 extractive caboclo households located in a small village at the lake entrance (Fig. 1). These forest dwellers depended primarily on the collection of Brazil nuts from natural clusters of Brazil nut trees (Bertholletia excelsa Humboldt & Bonpland—Lecythidaceae) in addition to small-scale fishing, and highly selective logging and hunting. However, forest structure at the study site remains almost entirely undisturbed, and recent subsistence hunting has been restricted to very light harvest of large rodents and ungulates. Hunters at Lago Uauaçú did not take primates and several other game species harvested elsewhere in Amazonia, and hunting did not take place in the census areas. Small-scale logging by local people within the extended research area concentrated on a few timber species from terra firme [Angelím, Dinizia excelsa (Mimosaceae) and Bacurí, Clusia sp. (Clusiaceae)] and várzea forest [Assacú, Hura crepitans (Euphorbiaceae) and Maçaranduba, Manilkara sp. (Sapotaceae)]. We therefore assume that the structure of the vertebrate community is a response to intrinsic features of the habitat, rather than hunting pressure and habitat degradation. A more detailed description of the study area can be found elsewhere (Haugaasen and Peres 2006).
Map of the Lago Uauaçú study area in the lower Rio Purús region of central-western Brazilian Amazonia. The main forest types occurring in the region included more extensive tracts of terra firme and várzea forest (open and shaded areas) and smaller patches of igapó forest (dotted areas). Transects A-M (lines) indicate the main focal areas censused in each forest type. Solid triangles indicate phenology plots
The total annual rainfall from February 2002 to January 2003 was 2,665 mm. Monthly precipitation showed a pronounced seasonal variation with a dry season lasting from July to October alternating with a rainy season from February to June (Fig. 2). The seasonal rainfall produced great fluctuations in the water level of the lake and surrounding rivers and streams (Fig. 2). In 2002, the maximum and minimum water level occurred in June and November, respectively, resulting in an annual fluctuation exceeding 10.5 m. Water level fluctuations were recorded using a 5-m long pole divided into 5-cm sections penetrating the lake bed vertically near the floating research station.
Methods
Forest types
Terra firme accounts for the most extensive forest type at Lago Uauaçú, whereas small expanses of igapó forest occur along the lake margins and perennial streams that drain the surrounding terra firme. The adjacent várzea forest was located north-east of the lake (Fig. 1). A detailed description of the floristic composition of the Lago Uauaçú region can be found elsewhere (Haugaasen 2004) so only a brief summary is included here. The floristic composition of the Uauaçú terra firme forest is representative of other central Amazonian forests, being dominated by the Lecythidaceae (e.g. Peres 1991; Amaral 1996; Lima Filho et al. 2001). Other dominant tree families were Chrysobalanaceae, Moraceae, Leguminosae, Sapotaceae and Myristicaceae. Emergent Brazil nut trees comprised the most important species as defined by their importance value index (Mori et al. 1983). Although little information is available on central Amazonian várzea forests, the floristic composition of the Uauaçú várzea forests was dominated by the Euphorbiaceae, Lecythidaceae, Annonaceae and Fabaceae, with Piranhea trifoliata (Euphorbiaceae) as the most important species, resembling those in the lower Rio Japurá (e.g. Mamirauá: Ayres 1986, 1993). The floristic composition of the Uauaçú igapó forests bears close resemblance to other central Amazonian igapó sites (e.g. Keel and Prance 1979; Ferreira 1997); typically, the super-family Leguminosae and the family Euphorbiaceae were very important, and Hevea spruceana (Euphorbiaceae) was the most important species.
Plant phenology
A random set of 1,200 phenology trees ≥ 20 cm in diameter at breast height (dbh) were sampled, including 400 trees in each forest type. All trees were aluminium-tagged and identified in the field by a highly experienced herbarium technician from Instituto Nacional de Pesquisas da Amazônia (INPA, Manaus). Phenological observations in terra firme, várzea and igapó forest were carried out over 40, 35 and 13 months, respectively, but only data collected during the period of vertebrate censuses (April 2002–November 2003) are presented here. Further details on phenological patterns can be found elsewhere (Haugaasen and Peres 2005b). The location of each phenology plot was chosen arbitrarily, and trees were selected randomly using a point-quadrant method following Peres (1994). We therefore used a proportionally represented community-wide sample to provide a realistic estimate of the overall fruit production across the entire tree assemblage in each forest type (Peres 1994).
Phenological data were obtained at monthly intervals during the last week of every month. All 1,200 trees were usually observed within 4 days, but sampling periods could be longer in the rainy season when frequent rain hindered data collection. Crown inspections were based on direct observations of plant parts with the help of a 10 × 40 pair of binoculars, as other techniques (e.g. fruit trapping, residual fruit-fall censuses) were less feasible in flooded forest. The abundance of immature and mature fruit was ranked based on a fruit abundance score (F), by visually scoring trees on a scale from 0 to 4. For example, trees were ranked 0 if fruits were conspicuously absent; 1, 2 and 3 if fruit loads were equivalent to ≤ 25, 25–50 and 50–75% of the maximum estimated crown capacity; and four if the crown capacity was filled to ≥ 75%. Only fruits still attached to parental plants were included. We then followed Develey and Peres (2000) in calculating a monthly index of fruit availability based on the sum of the basal area (BA) of all fruiting trees found in any given month times their fruit abundance scores (as a proportion of four: 0.0, 0.25, 0.5, 0.75 or 1.0) as in Σ (BA × F/4).
Line-transect censuses and species surveyed
All sites in terra firme, várzea and igapó forests were surveyed using a highly comparable line-transect census protocol described elsewhere (Peres 1999a). In summary, census walks were conducted from 07.00 to 11.00 hour and 13.00 to 17.00 hour, but not during rainy weather because this affected the intrinsic detectability of different species. Upon a detection event, the time, species identity, group size, perpendicular distance from the transect to the animal, sighting location along the transect and detection cue were recorded.
Survey walks were carried out in sampling blocks of 2 and 1 month off, coinciding with the late-wet, early dry, late-dry and early wet seasons. In total, this study is based on a two-way census effort of 2,026 km in terra firme, 2,309 km in várzea and 277 km in igapó forest, walked along 13 transects flagged every 50 m and ranging from 4,000 to 5,000 m in length (Table 1). This amounted to a total census effort of 4,612 km walked along 62.5 km of transects. Replicate census walks within each sampling block were systematically alternated between terra firme and floodplain forests in order to minimise any confounding effects of seasonality. In the high-water season, censuses along the same várzea and igapó transects were conducted by two observers using unmotorized dugout canoes. Because of changes in water level, canoe navigation along flooded forest transects was aided by flags that had to be frequently replaced. Data presented here focuses on diurnal vertebrates because night surveys were not carried out. This excludes only one primate species (night monkey, Aotus nigriceps) and a few other nocturnal mammals > 1 kg, including paca (Agouti paca), armadillos (Dasypus spp., Priodontes maximus) and kinkajou (Potos flavus).
We focus our analysis on 43 species (or species groupings) of mid-sized to large-bodied vertebrates, including primates, ungulates, squirrels, caviomorph rodents, coatis, xenarthrans, cracids, tinamids, trumpeters, wood-quails and tortoises (Appendix 1). Therefore, in addition to generalist frugivores, we also consider granivores (e.g. Ara macao, A. ararauna, Pithecia albicans), carnivores with a mixed diet (e.g. Nasua nasua), and terrestrial browsers (Mazama americana, M. gouazoupira) and facultative arboreal folivores (Alouatta seniculus) that consume at least some fruit. Information on diet and body mass was compiled from Peres (1999b) and Robinson and Redford (1986). Scientific nomenclature follows Wilson and Reeder (2005) for mammals, and Stotz et al. (1996) for birds.
Data analysis
Detection data for sympatric congeners of three genera were pooled because they occurred in highly stable mixed-species groups (saddle-back and moustached tamarins, Saguinus fuscicollis and S. mystax) or could not always be unambiguously distinguished in the field (squirrels, Sciurus spp.; and red and grey brocket deer, M. americana and M. gouazoupira). Other congeners forming relatively cohesive functional groups (e.g. Ramphastos spp., Pteroglossus spp., Tinamus spp., Crypturellus spp.) were similarly pooled to increase sample sizes to allow more robust density estimates. Abundance estimates shown in Appendix 1 are expressed as the mean encounter rate (individuals per 10 km walked ±SE) across all seven census seasons in terra firme and várzea forest, and three seasons in igapó forest. Coefficients of variation (CV) for each species (or functional group) were calculated from the variance in mean encounter rate between seasons.
Due to the small number of observations in igapó forest and the unavailability of abundance estimates for some species occurring in a given forest type (Appendix 1), we constructed a presence-absence species-by-site matrix and used a non-metric multidimensional scaling (MDS) ordination technique to examine the cross-site variation in species composition. Using Primer v. 5.0 (Clarke 1993), all species were entered into the ordination as single entities. Forest sites are thus positioned in the ordination space according to their species composition. An index of multivariate dispersion (Warwick and Clarke 1993) was calculated to investigate the variability within terra firme, várzea and igapó samples. Using the presence-absence data, species similarity values between any given pair of transects were calculated using the Bray-Curtis similarity index following a log(x + 1) transformation, and average Bray-Curtis similarity values between transects in different forest types were also calculated.
To investigate to what degree local faunal assemblages were affected by the spatial structure of the overall data set, the physical distance between the end points of any given pairwise comparison of transects at all sites were measured. A Spearman correlation was then used to examine the relationships between geographical distance and assemblage similarity, considering all possible comparisons of transects both between and within the three forest types.
While investigating temporal profiles of the faunal assemblages, monthly sighting rates of species in all 2-month census blocks were used, but igapó forest was excluded from further analysis due to insufficient sample sizes. We also restricted our analysis to those species observed in both terra firme and varzea. To examine the temporal synchrony in abundance estimates between terra firme and várzea forest, we simply correlated the monthly encounter rates for each species in each forest type using Spearman correlations. To investigate the influence of fruit abundance in driving changes in species abundances, we correlated the Log10(x + 0.1) ratio of fruits (mature and immature fruits separately and combined) and species abundance between terra firme and varzea. Positive correlations suggest a concomitant increase in the abundance of both fruits and consumers from one forest type to another. This may imply between-habitat lateral movements and resource tracking by the species in question.
Results
Plant phenology and fruit availability
The 1,200 phenology trees monitored included 46 families and a minimum of 126 genera. A total of 35 families were represented in terra firme, 37 in várzea, and 27 in igapó forest. The most abundant family in the terra firme forest sample was the Chrysobalanaceae, whereas the Annonaceae and Euphorbiaceae were the most abundant families in the várzea and igapó forest samples, respectively.
The emergence of immature fruits commenced during the two driest months of the year (July–August). In terra firme forest, most trees bore immature fruits between October and January, whereas high levels of unripe fruit (and seed) availability extended from September to January in várzea, and September to March in igapó forest (Fig. 3a).
The abundance of mature fruits varied temporally both within and between forest types, with all samples showing marked mature fruit seasonality (Fig. 3b). However, there was a significant temporal overlap between the fruiting peaks in terra firme and várzea forests. This resulted in a positive correlation in monthly mature fruit availability between these forest types (r s = 0.572, n = 20, p = 0.008), although a substantial part of the fruiting seasons did not overlap. In terra firme, most trees bore ripe fruits from the onset through to the middle of the rainy season (December to March), whereas ripe fruit production in várzea and igapó forest started at the onset of the high-water season (January). In igapó, this extended until the middle of the high-water season (April), whereas fruit production continued in várzea until August, which marked the end of the flood pulse (Fig. 3b). Phenological monitoring in igapó covers too short a period to make conclusive comparisons, but monthly ripe fruit availability in this forest type was correlated with that of terra firme (r s = 0.283, n = 13, p = 0.349) and várzea forest (r s = 0.422, n = 13, p = 0.151), though not significantly so.
Patterns of vertebrate species abundance
Primates were the most abundant vertebrate group censused in all forest types (Appendix 1). On average across all census block, golden-backed squirrel monkeys comprised the most abundant primate species in várzea, whereas brown capuchins were the most abundant in terra firme and igapó forest (Appendix 1). Considering non-primate mammals, white-lipped peccaries were the most abundant species in terra firme and igapó forest. However, the sporadic appearance and low detection frequency of the large herds in which they travel ( > 250 peccaries), hinder reliable abundance estimates of this wide-ranging species through line-transect censuses, so these numbers should be interpreted with caution. The second most abundant non-primate mammal surveyed in terra firme and igapó forest was the black agouti, whereas the most frequently encountered species in várzea forest was the coati (Appendix 1). Considering large terrestrial and canopy birds, toucans represented the most numerically abundant genus in terra firme and igapó forests, whereas three species of macaws were the most abundant in várzea forest.
Spatial structure of the faunal assemblage
In terms of species composition, all terra firme transects were more similar to one another than any other transects surveyed. This was also the case of várzea and igapó forest as shown in the MDS ordination where terra firme and várzea forests formed two clearly distinct clusters (Fig. 4). Igapó forest sites fell between typical terra firme and seasonally flooded várzea sites (Fig. 4). In particular, the várzea site that was physically connected to terra firme (Jamarí) appears to fall between typical terra firme and all other várzea sites that were isolated from one another by a river channel, which prevented free exchange of several terrestrial and arboreal vertebrate taxa.
Non-metric multi-dimensional scaling (MDS) ordination of the large vertebrate fauna observed along the 13 transects in terra firme (triangles), várzea (squares) and igapó (circles) forest at Lago Uauaçú (Stress = 0.07). A brief description of each transect can be found in Table 1
The index of dispersion shows that there was little difference in variability between groups of terra firme (0.899) and várzea (1.059) forest samples, whereas igapó samples were more dispersed (1.926). This is also indicated by the MDS ordination, and further supported by the higher levels of similarity, using the Bray-Curtis measure, between samples within either várzea (87%) or terra firme forest (85%), whereas samples within igapó forest shared a lower mean similarity of 71%.
We also tested the degree to which vertebrate species diversity was affected by the spatial independence between transects. There was a significant negative relationship between Bray-Curtis similarity values and the straight-line distance between transects when all combinations were included in the analysis (r s = −0.359, n = 78, p = 0.001). This is expected due to the greater number of pairwise combinations between different forest types. However, physical distance between any two sites within the same forest type had no significant effect on their species composition (r s = 0.104, n = 26, p = 0.615) indicating a low degree of within-habitat spatial autocorrelation in the assemblage data.
Temporal changes in faunal assemblages
Encounter rates varied over time for all species both within and between forest types (Appendix 1; Fig. 5a–h). Still, no synchronous changes in abundance in different directions were found between várzea and terra firme. In other words, monthly encounter rates for all species present in these two forest types did not portray any significant negative correlations, which could indicate synchronous compensatory shifts in abundance as animals moved from terra firme to várzea or vice-versa. However, this appears to be the case for macaws, as they became more abundant in terra firme in October–November 2002 following a simultaneous decrease in their várzea abundance. This appears to occur in the opposite direction in the next two census blocks (Fig. 5a). It is also apparent that some species such as buffy saki monkeys only appeared in várzea forest at certain times of the year (Fig. 5b). Likewise, mixed-species groups of tamarins showed only sporadic appearances in this forest type (Fig. 5e). However, these primate species were only observed along transect K (Jamarí) which intersected the várzea forest interfacing abutting terra firme forest. Indeed, the intermediate position of this site along MDS axis 1 (Fig. 4) is best explained by the seasonal influx of saki monkeys, tamarins, white-fronted capuchins and spider monkeys. Other relatively common arboreal bird (Fig. 5c, g, h) and mammal species (Fig. 5d, f) occurring in both terra firme and várzea forest could be encountered on a year-round basis but showed highly variable abundances within each forest type.
We were unable to statistically detect a significant role of fruit abundance in determining temporal shifts in species abundance. However, it appears that monthly changes in encounter rates of some species are positively affected by concomitant changes in immature and mature fruit production. For example, fluctuations in the abundance of macaws and saki monkeys appear to be closely linked with immature fruit abundance in terra firme forest (Fig. 5a, b). Similarly, there appears to be an association between monthly encounter rates of Ramphastos toucans with mature fruit availability in várzea, and razor-billed curassows and immature fruit availability in terra firme (Fig. 5c, h).
A select set of terrestrial vertebrates used várzea at certain times of the year (Fig. 6a). Changes in terrestrial vertebrate numbers in várzea forest were, however, tightly governed by the seasonal fluctuation in water level. The aggregate number of terrestrial vertebrates in várzea during any given census month was negatively correlated with water level in that same month (r s = −0.615, n = 12, p = 0.033), whereas this was not the case for arboreal taxa (r s = 0.042, n = 12, p = 0.897). This is not surprising since the várzea forest canopy can be used by volant and non-volant arboreal vertebrates all year-round (Fig. 6b), whereas várzea incursions by strictly terrestrial taxa are intimately tied to the availability of dry land. It is also clear that terrestrial vertebrates moved into várzea as soon as dry land appeared, then remained in this forest type for the entire dry period, and moved out only when the last patches of dry land were submerged (Fig. 6a). This resulted in significant positive relationships between the total monthly number of terrestrial vertebrates and the availability of immature (r s = 0.612, n = 14, p = 0.02) and mature fruit (r s = 0.564, n = 14, p = 0.036) abundance, since várzea trees were observed bearing immature fruits throughout the dry season (Fig. 3a) and fruits started to mature before the onset of the floods (Fig. 3b).
Aggregate abundance of arboreal and terrestrial vertebrates in várzea forest during seven bimonthly census blocks (grey bars) and monthly water level (grey shading). Terrestrial vertebrates included in the total are: Dasyprocta fuliginosa, Pecari tajacu, Mazama spp., Tapirus terrestris, Geochelone spp., Tinamus spp., Crypturellus undulatus, and Psophia leucoptera
Discussion
Fruiting phenology
The availability of immature and mature fruit was clearly episodic in all three forest types. The tendency towards high levels of fruit production in terra firme forest in the early wet season is similar to those noted in other neotropical forests (Janzen 1967; Smythe 1970; Alencar et al. 1979; Foster 1982; ter Steege and Persaud 1991; Peres 1994; Baider 2000). The clearly demarcated fruiting peak during the inundation period in both types of seasonally flooded forest is also consistent with what few plant phenology studies are available for other Amazonian várzea (Ayres 1993) and igapó forests (Adis et al. 1979). Fruiting during the high-water season appears to be a homogenous trait among floodplain forest trees (Parolin et al. 2002), a phenological adaptation consistent with the high proportion of hydrochorous trees and lianas in seasonally inundated forests (Kubitzki and Ziburski 1994; Boedeltje et al. 2004). This is especially the case of large, water-dispersed seeds, characterized by high buoyancy, that are gradually deposited at high densities on the várzea and igapó forest floor as the water level recedes.
Spatiotemporal dynamics of faunal assemblages
The spatial organization of Uauaçú vertebrate assemblages was a function of intrinsic environmental differences between forest types rather than other factors. The forest structure and composition in the Uauaçú region remained relatively undisturbed, and recent selective harvest has been restricted to very low-subsistence hunting pressure on high-fecundity terrestrial game species that are not markedly affected by hunting in other Amazonian forests (Peres 2000). This is corroborated by the fact that no local extinctions or population declines were detected for harvest-sensitive species compared with other non-hunted sites in central-western Amazonia (Haugaasen and Peres 2005a). Moreover, distance between transects within the same forest type had no influence on species composition, but there were considerable differences in species composition between transects in different forest types as shown by the MDS ordination (Fig. 4) and the similarity analysis.
The spatial characteristics of the vertebrate assemblages was largely a function of physical connectivity and proximity to adjacent terra firme forests. There was a marked loss of typical terra firme species across the river channel from the várzea transect at Jamarí, which abutted terra firme terraces, to the unconnected várzea transects (F, G, I, J). At least five primate species observed at Jamarí (saddle-back and moustached tamarins, buffy saki, white-fronted capuchin and spider monkey) were never found in any patch of várzea “island” completely cut-off from terra firme. This suggests that the ∼50-m wide paranã channel served as an effective fluvial barrier to dispersal and movements by many species although most terrestrial species are adept swimmers (e.g. tapir, red brocket deer and jaguar), and could be observed across the river channel at unconnected várzea sites and other várzea “islands”. This fluvial barrier was therefore a less effective deterrent to terrestrial vertebrates. As in Jamarí, the intermediate position of the two igapó forest transects was due to the composition of the assemblages which included both flooded and unflooded forest species as a result of juxtaposed terra firme forest. The igapo sites were also visited by two additional terra firme primate species that were never observed in várzea forest: woolly monkeys and yellow-handed titi monkeys. Many terrestrial vertebrates detected during census walks were also attracted to igapó streams as a drinking source—particularly during the dry season—further adding to the faunal similarity with terra firme forest.
It is surprising that vertebrate encounter rates in terra firme forest were as variable as those in várzea and igapó (Appendix 1) given the less extreme seasonal changes experienced in this environment. This is particularly the case for species occupying relatively small home ranges such as agoutis and tamarins, which one would expect to encounter at similar monthly rates along all transects. It is possible that differences in detectability due to unknown behavioural traits or climatic variables have played a role. For example, some species could have been more vocal and more easily detected during the breeding season, or wind background noise could have hindered detection (although censuses were normally suspended in sub-optimal conditions). The reasons for the large variation in detection rates in terra firme forest thus remain unclear.
Prolonged seasonal inundation prohibits strictly terrestrial and understorey species from using flooded forest for much of the year, and this is the simplest explanation for the temporal variation in species abundance in igapó and várzea forest. However, the appearance of terrestrial species in várzea closely tracks the residual fruit and seed availability on the forest floor as seen in the positive correlation between terrestrial vertebrate abundance and immature and mature fruit availability. Várzea forest trees bear immature fruits throughout the dry period and clearly begin fruiting at the onset of the rainy season, immediately prior to the inundation period (Fig. 3a, b). It is therefore likely that terrestrial vertebrates take advantage of these resources until the forest is completely inundated. Similarly, many fruits and seeds available on the crowns during the high-water fruiting season were deposited on the ground after the inundation period. Terrestrial frugivores will then presumably track the receding water level and move in from upland habitat to take advantage of these resources. Additionally, new growth from young seedlings and new leaves on larger trees and saplings also starts to emerge when water levels drop and dry land becomes exposed (T. Haugaasen personal observation). This further invited predominantly herbivorous species into these areas, since young foliage is a preferred food due to a more favourable nutrient level and lower concentrations of chemical defences than mature foliage (Klinge et al. 1983). Consequently, we also detected ample evidence of an influx of large terrestrial carnivores, such as puma, jaguar and ocelot, tracking primary consumers during this period.
Although we were unable to gather any statistical support for the influence of fruit availability on temporal changes in the abundance of arboreal vertebrates, some observations suggest that fruit may play an important role in shaping the temporal structure of arboreal faunal assemblages. For example, the connection between the density of standing immature fruit crops and sighting rates of saki monkeys and macaws in terra firme, and mature fruit and large toucans in várzea forest provide substance to this hypothesis. Buffy saki monkeys and all macaw species are highly granivorous and consume a large number of young seeds in immature fruit (Peres 1993a; T. Haugaasen, personal observation); indeed, unripe fruit were almost always taken by arboreal granivores consuming immature endosperms, rather than generalist frugivores consuming unripe mesocarps. Maximum encounter rates of these species coincided with the peak of immature fruit availability in terra firme forest (Fig. 5a, b). It is not immediately clear why detection rates of saki monkeys were higher at this time. We found no significant reverse shifts in saki abundance between these two forest types and this species is a year-round resident in terra firme forest. Increased detection probability during this period could be due to greater levels of group cohesiveness in P. albicans (Peres 1993a) and higher frequency of discarded fruits dropping to the forest floor.
What is clear, however, is that saki monkeys appeared in várzea forest only during the end of the várzea fruiting season, and this was consistent in both years of the study. Observations suggest that these incursions may be connected to the fruiting of one or more Lauraceae tree species (T. Haugaasen, personal observation). The same pattern was observed for white-face capuchins and spider monekys which were only observed at the connected várzea site during the height of the várzea fruiting season. This was confirmed by information gathered during informal local interviews, which consistently described these species as seasonal visitors of várzea forest. The lack of clear compensatory shifts in abundance between terra firme and várzea forest, and the sporadic appearance of some species (e.g. Saguinus spp.) in the latter, suggest that the groups encountered had home ranges bordering or straddling the terra firme–várzea interface. Since most terra firme transects were located away from the terra firme–várzea interface (Fig. 1), such movements would be difficult to detect using the current sampling design.
Macaws were the only species in which there may be a suggestion of simultaneous, reverse shifts in abundance between terra firme and várzea forest (Fig. 5a), symptomatic of long-distance movements between these forest types. Macaw abundance was also apparently linked to immature fruit availability in terra firme, and increased in numbers in várzea during its fruiting pulse (Fig. 5a). These observations suggest that local fruit availability may drive lateral seasonal movements by macaws in the study region. This is supported by surveys along the Rio Manu, southern Peru, showing that large macaws may alter their use of forest mosaics and exhibit seasonal variation in abundance (Renton 2002). In the case of toucans, Graham (2001) noted that fruit abundance influenced toucan movements in Mexico when habitat connectivity was high.
Although the igapó sample was insufficient to be included in the monthly analysis, this forest type behaved in a similar fashion to várzea. Igapó experiences the same water level fluctuations, and produces mature fruits at the same time of year as várzea forest—when water levels are rising. That this forest is used on a seasonal basis by primarily terra firme forest species is supported by observations of some species (e.g. L. lagotricha and C. torquatus) that enter igapó to take advantage of the fruit resources at the height of the fruiting season (T. Haugaasen, personal observation). The importance of igapó in the overall forest landscape matrix is reinforced by the heavy reliance of both Scarlet macaw (A. macao) and the Blue-and-yellow macaw (A. ararauna) on immature seeds of H. spruceana (Euphorbiaceae) during months of fruit scarcity. This tree species was one of the most abundant in both várzea and igapó forest (Haugaasen 2004).
Given the above scenarios, it appears that populations of different vertebrate species at Uauaçú adopt three broad strategies regarding the spatial integration of the forest mosaic: (a) year-round residents that never move between different forest types and are able to secure their annual metabolic requirements within a single forest type, and therefore rely on forest heterogeneity at the within-habitat scale; (b) those that occupy home ranges bordering or straddling the interface between two forest types and use at least two habitats on a daily, weekly, monthly or seasonal basis. These populations will either include a portion of two or more forest types within their home ranges to acquire sufficient resources to meet their metabolic demands, or sporadically use a second forest type for any activity due to its proximity to their “normal” home range; and (c) wide-ranging populations that on a seasonal basis occupy large home ranges in different forest types depending on the local resource supply, thereby exhibiting long-distance movements between the two. These will take advantage of their efficient mobility that is often, but not always, aided by flight, and shift their home ranges according to the abundance of resources crucial to survival. Future research should investigate how far some individuals may travel to gain the benefits of adjacent forest types.
In summary, there appear to be two patterns of vertebrate incursions into floodplain forests at Uauaçú. One by a suite of arboreal species taking advantage of newly available immature or mature fruit patches in the canopy, and one by terrestrial vertebrates when the waters recede, taking advantage of fruits and seeds deposited on the ground and new vegetative growth. However, seasonal movements are not only exhibited by terrestrial primary consumers, as large carnivores show similar movement patterns in response to their prey. The macro-mosaic of terra firme, várzea and igapó forest thus appear to play an important role in the dynamics and survival of some vertebrate species at Uauaçú, but the extent to which different forest types are used is highly variable in both space and time.
Flooded forests as keystone habitats?
On a pan-Amazonian scale, evidence suggesting that várzea and igapó forests become seasonally important to terra firme forest vertebrates remains patchy and anecdotal as there have been few explicit attempts to document the use of várzea or igapó forest by a single species or suite of species. However, in Peru, Remsen and Parker (1983) showed seasonal use of várzea by birds and Bodmer (1990) found igapó to be seasonally important to ungulates. Similarly, primates along the Rio Tapajós of Brazilian Amazonia (Branch 1983) and at least two of its tributaries (C. Peres, unpublished data) use flooded forest on a seasonal basis. Trolle (2003) also observed several primates entering igapó forest in the Rio Jauaperí and lateral movements by primates into várzea forest occur along the entire length of the Rio Juruá (Peres 1993b). Fragoso (1998) and Peres (1996) report seasonal use of várzea forest by peccaries, Boubli (1999) observed extensive use of igapó forest by black-headed uakaris, and woolly monkeys use igapó forest in the Colombian Amazon on a seasonal basis (Defler 1996). Extensive seasonal use of igapó by woolly monkeys and bearded saki monkeys has also been observed in the lower Rio Aripuanã (M. G. M van Roosmalen, personal communication).
There was a considerable overlap in the fruiting seasons of the three forest types studied here, but it is clear that fruiting peaks in várzea and igapó diverged considerably from those in terra firme forest. For animals that are capable of moving long distances and taking advantage of fruit crops as they ripen in the canopy, or fruits and seeds deposited on the ground during the low-water season, floodplain forests can provide seasonally important resources at a time when fruit production in terra firme is low. Since such resources can limit forest carrying capacity to frugivores, the proximity of flooded forest may boost the overall biomass and density of primary consumers relying heavily on spatially staggered resources. For example, spider monkeys (Ateles belzebuth) occupying a home range divided equally between flooded and unflooded forest can live at higher densities than those in other areas where a single forest type predominates (Ahumada et al. 1998). It is therefore likely that the large-scale mosaic of igapó, várzea and terra firme forest in our study region has a positive impact on the landscape-wide resource abundance for primary consumers such as ripe fruit specialists and seed predators. Furthermore, this can have a positive knock-on effect on the abundance of large predators. Therefore, although information is scarce, it appears that Amazonian flooded forest is seasonally important to a large number of vertebrate species. The combination of different juxtaposed forest types exhibiting staggered resource pulses augment the year-round resource supply, thereby contributing to the landscape-scale viability of many wide-ranging habitat generalists. It may thus be argued that, in this respect, Amazonian seasonally flooded forests can be considered as keystone habitats for the predominantly terra firme vertebrate fauna. However, more work is needed to further map the extent to which flooded forest resources complement those of adjacent terra firme forests.
Implications for reserve design
Despite recent discussions on conservation planning and reserve design criteria in tropical forests, the role of faunal dynamics at the landscape scale has been largely overlooked. We have shown that fruit abundance in a highly heterogeneous Amazonian forest landscape varies in complex spatial and temporal patterns, and how this may affect resident frugivore populations. From a conservation perspective, this is important because a landscape including a structurally and floristically diverse habitat mosaic encompassing asynchronous resource patches is more likely to offer suitable resources in at least one habitat type when supplies are low in another. This emphasises the importance of including várzea and igapó forest in regional scale conservation planning to sustain healthy frugivorous vertebrate assemblages, as floodplain forests can provide an alternative habitat for many wide-ranging species in times of fruit scarcity in terra firme forest. It also indicates that loss of habitat area and landcsape heterogeneity may result in population declines, if not species loss. Preventing this with appropriate reserve design criteria is particularly important given the vital role that frugivores play in tropical forests dynamics by providing dispersal services to shrubs and trees and thereby securing successful regeneration. Conservation of frugivores and consequently their seed dispersal services thus require an understanding of large-scale linkages between food resources and frugivore activity.
Although the forest types were moderately similar in terms of vertebrate species composition, our results show that incorporating different lowland habitats into Amazonian reserves can maximise species diversity. Many habitat-generalist species may be ubiquitous throughout the landscape but occur at high densities in only one habitat (Haugaasen and Peres 2005a, c). This calls for the protection of adjacent habitats to safeguard numerically viable (meta)populations. Information on the extent of seasonal movements is also needed to ensure that forest reserves are sufficiently large to protect all habitats required by wide-ranging and migratory species on an annual basis.
In conclusion, our results imply that the persistence of many species and the biotic integrity of entire tropical forest landscapes will require conservation of large, connected tracts of suitable habitat mosaics. The intimate juxtaposition of the three adjacent forest types at Lago Uauaçú greatly contributes to the high conservation value of this area, which represents a unique system in which to study landscape-scale processes and interactions.
References
Adis J, Furch K, Irmler U (1979) Litter production of a central Amazonian blackwater inundation forest. Trop Ecol 20:236–245
Ahumada JA, Stevenson PR, Quinones MJ (1998) Ecological response of spider monkeys to temporal variation in fruit abundance: the importance of flooded forest as a keystone habitat. Primate Conserv 18:10–14
Alencar JC, de Almeida RA, Fernandes NP (1979) Fenologia de espécies florestais em floresta tropical úmida de terra firme na Amazônia Central. Acta Amazonica 9:163–198
Amaral IL (1996) Diversidade florística em floresta terra firme, na região do Rio Urucú—AM. MSc Thesis. Instituto Nacional de Pesquisas da Amazônia, Universidade do Amazonas, Manaus
Ayres JM (1986) Uakaris and Amazonian flooded forest. Ph.D. Thesis. University of Cambridge, Cambridge
Ayres JM (1993) As matas de várzea do Mamirauá. Sociedade Civil Mamiraua, Belém
Baider C (2000) Demografia e ecologia de dispersão de frutos de Bertholletia excelsa Humb. & Bonpl. (Lecythidaceae) em castanhais silvestres da Amazônia oriental. Ph.D. Thesis. University of São Paulo, São Paulo
Blake JG, Loiselle BA (1991) Variation in resource abundance affects capture rates of birds in three lowland habitats in Costa-Rica. Auk 108:114–130
Blake JG, Stiles FG, Loiselle BA (1990) Birds of La Selva Biological Station: habitat use, trophic composition, and migrants. In: Gentry AH (ed) Four neotropical forests. Yale University Press, New Haven, pp 161–182
Bodmer RE (1990) Responses of ungulates to seasonal inundations in the Amazon floodplain. J Trop Ecol 6:191–201
Boedeltje G, Bakker JP, Ten Brinke AJ, Van Groenendael M, Soesbergen M (2004) Dispersal phenology of hydrochorous plants in relation to discharge, seed release time and buoyancy of seeds: the flood pulse concept supported. J Ecol 92:786–796
Boubli JP (1999) Feeding ecology of black-headed uacaris (Cacajao melanocephalus melanocephalus) in Pico da Neblina National Park, Brazil. Int J Primatol 20:719–749
Branch LC (1983) Seasonal and habitat differences in the abundance of primates in the Amazon (Tapajós) National Park, Brazil. Primates 24:424–431
Chapman CA, Wrangham RW, Chapman LJ, Kennard DK, Zanne AE (1999) Fruit and flower phenology at two sites in Kibale National Park, Uganda. J Trop Ecol 15:189–211
Clarke KR (1993) Nonparametric multivariate analyses of changes in community structure. Aust J Ecol 18:117–143
Corlett RT (1990) Flora and reproductive phenology of the rain forest at Bukit Timah, Singapore. J Trop Ecol 6:55–63
Defler TR (1996) Aspects of the ranging pattern in a group of wild woolly monkeys (Lagothrix lagothricha). Am J Primatol 38:289–302
Develey PF, Peres CA (2000) Resource seasonality and the structure of mixed species bird flocks in a coastal Atlantic forest of southeastern Brazil. J Trop Ecol 16:33–53
Ferreira LV (1997) Effects of the duration of flooding on species richness and floristic composition in three hectares in the Jaú National Park in floodplain forests in central Amazonia. Biodivers Conserv 6:1353–1363
Foster RB (1982) The seasonal rhythm of fruit fall on Barro Colorado Island. In: Leigh EG, Rand AS, Windsor DM (ed) The ecology of a tropical forest: seasonal rhythms and long-term changes. Smithsonian Institutional Press, Washington DC, pp 151–172
Fragoso JMV (1998) Home range and movement patterns of white-lipped peccary (Tayassu pecari) herds in the northern Brazilian Amazon. Biotropica 30:458–469
Frankie GW, Bake HG, Opler PA (1974) Comparative phenological studies of trees in tropical wet and dry forests in the lowlands of Costa Rica. J Ecol 62:881–919
Gautier-Hion A, Duplantier JM, Emmons L, Feer F, Heckestweiler P, Moungazi A, Quris R, Sourd C (1985) Coadaptation between fruiting periodicity and frugivory in a Gabon rain-forest—myth or reality. Rev D Ecologie-La Terre Et La Vie 40:405–434
Gentry AH (1982) Patterns of Neotropical plant-species diversity. Evol Biol 15:1–85
Graham CH (2001) Factors influencing movement patterns of keel-billed toucans in a fragmented tropical landscape in southern Mexico. Conserv Biol 15:1789–1798
Greenberg R (1981) The abundance and seasonality of forest canopy birds on Barro Colorado Island. Biotropica 13:241–251
Haugaasen T (2004) Structure, composition and dynamics of a central Amazonian forest landscape: a conservation perspective. Ph.D. Thesis. University of East Anglia, Norwich
Haugaasen T, Peres CA (2005a) Mammal assemblage structure in Amazonian flooded and unflooded forests. J Trop Ecol 21:1–13
Haugaasen T, Peres CA (2005b) Patterns of tree phenology in adjacent Amazonian flooded and unflooded forests. Biotropica 37:620–630
Haugaasen T, Peres CA (2005c) Primate assemblage structure in Amazonian flooded and unflooded forests. Am J Primatol 67:243–258
Haugaasen T, Peres CA (2006) Floristic, edaphic and structural characteristics of flooded and unflooded forests in the lower Purús region of central Amazonia, Brazil. Acta Amazonica 36:25–36
Hilty SL (1980) Flowering and fruiting periodicity in a premontane rain-forest in Pacific Colombia. Biotropica 12:292–306
Howe HF, Smallwood J (1982) Ecology of seed dispersal. Ann Rev Ecol Syst 13:201–228
Janzen DH (1967) Synchronization of sexual reproduction of trees within dry season in Central America. Evolution 21:620–637
Karr JR, Freemark KE (1983) Habitat selection and environmental gradients—dynamics in the stable tropics. Ecology 64:1481–1494
Keel SH, Prance GT (1979) Studies of the vegetation of a white-sand black-water igapó (Rio Negro, Brazil). Acta Amazonica 9:645–655
Klinge H, Furch B, Harms E, Revilla JD (1983) Foliar nutrient levels of native tree species from Central Amazonia. 1. Inundation forest. Amazoniana 8:19–45
Kubitzki K, Ziburski A (1994) Seed dispersal in flood-plain forests of Amazonia. Biotropica 26:30–43
Leck CF (1972) Seasonal changes in feeding pressures of fruit- and nectar-eating birds in Panama. Condor 74:54–60
Leighton M, Leighton DR (1983) Vertebrate responses to fruiting seasonality within a Bornean rain forest. In: Sutton SL, Whitmore TC, Chadwick AC (eds) Tropical rain forest: ecology and management. Blackwell Scientific, London, pp 181–196
Levey DJ (1988) Spatial and temporal variation in Costa Rican fruit and fruit-eating bird abundance. Ecol Monogr 58:251–269
Lieberman D (1982) Seasonality and phenology in a dry tropical forest in Ghana. J Ecol 70:791–806
Lima Filho DA, Matos FDA, Amaral L, Revilla JD, Coelho LS, Ramos JF, Santos JL (2001) Inventário florístico de floresta ombrófila densa de terra firme, na região do Rio Urucu—Amazonas, Brasil. Acta Amazonica 31:565–579
Loiselle BA, Blake JG (1991) Temporal variation in birds and fruits along an elevational gradient in Costa Rica. Ecology 72:180–193
Medway L (1972) Phenology of a tropical rain forest in Malaya. Biol J Linn Soc 4:117–146
Moegenburg SM, Levey DJ (2003) Do frugivores respond to fruit harvest? An experimental study of short-term responses. Ecology 84:2600–2612
Mori SA, Boom BM, Carvalho AM, Santos TS (1983) Ecological importance of Myrtaceae in Eastern Brazilian wet forest. Biotropica 15:68–70
Opler PA, Frankie GW, Baker HG (1980) Comparative phenological studies of treelet and shrub species in tropical wet and dry forests in the lowlands of Costa Rica. J Ecol 68:167–188
Parolin P, Armbruester N, Wittmann F, Ferreira LV, Piedade MTF, Junk WJ (2002) A review of tree phenology in central Amazonian floodplains. Pesqui Botânica 52:195–222
Peres CA (1991) Ecology of mixed-species groups of tamarins in Amazonian terra firme forests. Ph.D. Thesis. University of Cambridge, Cambridge
Peres CA (1993a) Notes on the ecology of Buffy Saki Monkeys (Pithecia albicans, Gray 1860)—a canopy seed predator. Am J Primatol 31:129–140
Peres CA (1993b) Structure and spatial organization of an Amazonian terra firme forest primate community. J Trop Ecol 9:259–276
Peres CA (1994) Primate responses to phenological changes in an Amazonian terra firme forest. Biotropica 26:98–112
Peres CA (1996) Population status of white-lipped Tayassu pecari and collared peccaries T. tajacu in hunted and unhunted Amazonian forests. Biol Conserv 77:115–123
Peres CA (1999a) General guidelines for standardising line-transect surverys of tropical forest primates. Neotrop Primates 7:11–16
Peres CA (1999b) The structure of nonvolant mammal communities in different Amazonian forest types. In: Eisenberg JF, Redford KH (eds) Mammals of the neotropics: the central neotropics. University of Chicago Press, Chicago, pp 564–578
Peres CA (2000) Effects of subsistence hunting on vertebrate community structure in Amazonian forests. Conserv Biol 14:240–253
Peres CA, Whittaker A (1991) Annotated checklist of bird species of the upper Rio Urucú, Amazonas, Brazil. Bull Br Ornithological Club 111:156–171
Peres CA, van Roosmalen MGM (2002) Patterns of primate frugivory in Amazonia and the Guianan shield: implications to the demography of large-seeded plants in overhunted tropical forests. In: Levey W, Silva D, Galetti M (eds) Frugivory and seed dispersal: ecological, evolutionary and conservation issues. CAB International, Oxford, pp 407–423
Prance GT (1979) Notes on the vegetation of Amazonia III. The terminology of Amazonian forest types subject to inundation. Brittonia 31:26–38
Remsen JV, Parker TA (1983) Contribution of river-created habitats to bird species richness in Amazonia. Biotropica 15:223–231
Renton K (2002) Seasonal variation in occurrence of macaws along a rainforest river. J Field Ornithol 73:15–19
Robinson JG, Redford KH (1986) Body size, diet, and population density of Neotropical forest mammals. Am Nat 128:665–680
Smythe N (1970) Relationship between fruiting seasons and seed dispersal methods in a neotropical forest. Am Nat 104:25–35
Stiles FG (1985) On the role of birds in the dynamics of neotropical forests. ICBP Tech Publ 4:49–59
Stotz DF, Fitzpatrick JW, Parker TA, Moskovits DK (1996) Neotropical birds. Ecology and conservation. University of Chicago Press, Chicago
ter Steege H, Persaud CA (1991) The phenology of Guyanese timber species—a compilation of a century of observations. Vegetatio 95:177–198
Terborgh J (1980) Causes of tropical species diversity. Proceedings XVII. Int Ornithological Cong (1978):955–961
Terborgh J (1983) Five new world primates—a study in comparative ecology. Princeton University Press, Princeton
Terborgh J (1986) Community aspects of frugivory in tropical forests. In: Estrada A, Fleming TH (eds) Frugivores and seed dispersal. Dr. W. Junk Publishers, Dordecht, The Netherlands, pp 371–384
Trolle M (2003) Mammal survey in the Rio Jauaperi region, Rio Negro Basin, the Amazon, Brazil. Mammalia 67:75–83
van Roosmalen MGM (1985) Fruits of the Guianan Flora. Institute of Systematic Botany, Utrecht
van Schaik CP (1986) Phenological changes in a Sumatran rain forest. J Trop Ecol 2:327–347
Warwick RM, Clarke KR (1993) Increased variability as a symptom of stress in marine communities. J Exp Mar Biol Ecol 172:215–226
White LJT (1994) Patterns of fruit-fall phenology in the Lope Reserve, Gabon. J Trop Ecol 10:289–312
Wilson DE, Reeder DM (eds) (2005) Mammal species of the world. Smithsonian Institution Press, Washington
Wong M (1986) Trophic organization of understory birds in a Malaysian Dipterocarp forest. Auk 103:100–116
Acknowledgements
This study was supported by small grants from the Wildlife Conservation Society, WWF-US and the Amazon Conservation Team (ACT). Logistical support was provided by the Amazon Association for the Preservation of High Biodiversity Areas (AAPA). Our special thanks go to Marilene, Edivar, Zé and Evineu for their help with field work. P. Judge prepared Fig. 1.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Rights and permissions
About this article
Cite this article
Haugaasen, T., Peres, C.A. Vertebrate responses to fruit production in Amazonian flooded and unflooded forests. Biodivers Conserv 16, 4165–4190 (2007). https://doi.org/10.1007/s10531-007-9217-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-007-9217-z