Abstract
Single-photon emission computed tomography with integrated computed tomography (SPECT/CT) systems has been applied in a wide range of clinical circumstances, and differentiated thyroid cancer (DTC) is one of the most important indications of SPECT/CT imaging. In the treatment of DTC, SPECT/CT images have been reported to have many advantages over conventional planar whole-body scintigraphy based on its precise localization and characterization of abnormal foci of radioactive iodine (RAI) accumulation, influencing the staging, risk stratification, and clinical management as well as reader confidence. On the other hand, SPECT/CT has limitations including additional radiation exposure from the CT component, additional imaging time, and cost-related issues. Each SPECT/CT image acquired at different time points throughout the management of DTC may have a different clinical meaning and significance. This review article addresses the clinical usefulness of RAI SPECT/CT images acquired during the pre-ablation period, post-therapy period, and long-term follow-up period, respectively.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The clinical use of hybrid imaging systems for the fusion of functional and anatomic images such as positron emission tomography (PET) with integrated computed tomography (CT) and single-photon emission computed tomography (SPECT) with integrated CT has become more prevalent over the past 2 decades. Like the PET/CT scanner, the integrated SPECT/CT system was developed in the early 1990s. The first clinical SPECT/CT system was introduced in 1999 using a low-output, slow-acquisition CT scanner with a dual-head gamma camera, and the second generation was introduced in 2004 using a commercially available diagnostic CT scanner [1]. With the technologic advances, integrated SPECT/CT systems have a wide range of clinical applications including endocrine and neuroendocrine diseases, infection/inflammation, benign and malignant bone diseases, radioguided surgery, lymphoscintigraphy for sentinel node biopsy, gastrointestinal and pulmonary circulation emergencies, coronary artery disease, and others [2].
Radioactive iodine (RAI) therapy has played an important role in the treatment of differentiated thyroid cancer (DTC), although this concept has been disputed mainly in low-risk patients, especially after the recent publication of the 2015 American Thyroid Association Management Guidelines [3]. In patients undergoing RAI therapy, whole-body scintigraphy (WBS) using RAI has been an essential imaging modality for the completion of staging, risk stratification, assessment of residual disease, therapeutic planning, and detection of recurrence. However, conventional planar WBS lacks anatomic information and has relatively poor image resolution, and its two-dimensional nature cannot provide depth information. Many advantages of SPECT/CT images over planar WBS have been reported in the management of DTC because of the precise localization and characterization of abnormal foci of RAI accumulation, which can lead to changes in staging, risk stratification, and clinical management and can increase reader confidence [4]. CT-based attenuation correction can be another advantage of the SPECT/CT system for improving image quality. Nonetheless, SPECT/CT has some limitations such as additional radiation exposure from the CT component (1–4 mSv with each acquisition), additional imaging time, and cost-related issues [5]. Occasionally, it might be difficult to establish a histologic diagnosis of abnormal foci of RAI accumulation as a reference standard, which can be a limitation of the interpretations of SPECT/CT images.
While conventional planar RAI WBS is usually acquired in the anterior and posterior projection using a dual-head gamma camera equipped with a high-energy collimator at a speed of 5–15 cm/min, SPECT images are obtained by a 64 × 64 or 128 × 128 matrix and 60, 64, or 72 frames with 20–60 s projections over 360° [4]. The SPECT volume session mostly includes the neck and thorax portion, and occasionally additional SPECT images were acquired at the regions of nonphysiologic foci of RAI uptake. After SPECT acquisition, a CT topogram was acquired, followed by a spiral CT acquisition with a variable voltage and tube current depending on the purpose and instrument.
Although SPECT/CT images have advantages over planar scans in staging, risk stratification, and clinical management because of the precise localization and characterization, each SPECT/CT image acquired at different time points throughout the management of DTC may have a different clinical meaning and significance. This review article addresses the clinical usefulness of RAI SPECT/CT images acquired during the pre-ablation period, post-therapy period, and long-term follow-up period, respectively (Table 1).
Pre-ablation Period
The clinical usefulness of pre-ablation RAI WBS has been a controversial issue because of the possibility of stunning effects caused by the pre-ablation RAI dose and physician’s preference for the use of an empirical fixed dose for RAI therapy based on the histopathologic results [6, 7]. Furthermore, a pre-ablation scan may cause patient inconvenience because of the prolonged hypothyroid period and additional low-iodine diet from the scan to subsequent RAI therapy. Some researchers have doubts about the utility of pre-ablation scans because they may give little additional information compared to the post-therapy scan and rarely influence the therapeutic decision [8].
However, in a retrospective review of 355 sets of scans, Van Nostrand et al. [9] reported that 53% of the patients had findings on pre-ablation RAI WBS that might have altered the management of DTC. Chen et al. [10] also reported that I-123 pre-ablation WBS was able to provide additional critical information in 25% of the patients requiring significant changes in the RAI therapy strategy. They supported the necessity of a routine pre-ablation scan and adjustment of the appropriate treatment dose of I-131 accordingly. A stunning effect can be reduced by the use of I-123 or a low activity (37–111 MBq) of I-131, and Yap et al. [11] reported that the use of a low diagnostic dose of I-131 (40 MBq) was safe and convenient without any adverse effect on the long-term clinical outcome.
A retrospective study on the feasibility of pre-ablation RAI SPECT/CT with planar WBS indicated that the addition of SPECT/CT reduced the number of equivocal cases on planar WBS by improvement of image interpretation and completed the post-surgical staging of DTC [12]. Recently, two impressive prospective studies were published on the utility of RAI SPECT/CT added to conventional planar WBS in the pre-ablation period. Avram et al. [13] demonstrated that pre-ablation SPECT/CT was able to detect residual or unsuspected regional metastases in 35% of the cases and distant metastases in 8% of the cases in 320 consecutive patients. This led to upstaging in 4% of the younger and 25% of the older patients compared to the initial staging based on clinical and histopathologic information. Even in a subanalysis of T1 tumors, SPECT/CT detected residual or unsuspected nodal and distant metastases in a significant number of patients. In their next study [14], pre-ablation SPECT/CT, combined with stimulated serum thyroglobulin (Tg) levels, altered the risk stratification in 15% of the patients by detecting residual nodal and distant metastases, which led to changes in the clinical management strategy in 29.4% of the patients.
The improved image quality of SPECT/CT can result in better performance to detect residual nodal and distant metastasis and to distinguish benign physiologic activity from metastatic disease, which can provide invaluable information on which patients will (or will not) benefit from RAI therapy and on the decision concerning therapeutic RAI activity regardless of the empirical fixed dose or dosimetry approach. In addition, SPECT/CT can provide patient-specific three-dimensional dosimetry data, which enable calculating the radiation absorbed doses to the tumor and to specific organs as well [15, 16].
Current clinical circumstances in the treatment of DTC are moving toward less aggressive initial surgery and less RAI therapy depending on the staging and risk stratification. The American Thyroid Association Management Guidelines [3] categorized the primary goal of the postoperative administration of RAI as the following: RAI remnant ablation, RAI adjuvant therapy, and RAI therapy based on postoperative risk stratification. In low-risk patients based on the histopathologic results without pre-ablation RAI SPECT/CT data, RAI ablation is not routinely recommended. For RAI remnant ablation, only low administered activity (approximately 1110 MBq) is recommended in patients with low or intermediate risk with lower risk features, while higher activity (up to 5550 MBq) is recommended for initial adjuvant therapy to treat suspected microscopic residual disease.
Thus, the re-assessment of the disease status during the postoperative/pre-ablation period may become an important issue for the decision-making in RAI therapy, and pre-ablation SPECT/CT combined with serum Tg levels can play a major role in defining the target of RAI therapy. However, there is still no specific consensus on the diagnostic strategy including RAI SPECT/CT and the optimal cutoff value of postoperative serum Tg levels for decision-making in RAI therapy [3].
Post-therapy Period
To complete staging and measure the RAI avidity of residual disease, a post-therapy WBS and/or SPECT/CT is strongly recommended, and the clinical role of SPECT/CT over conventional planar WBS has been widely investigated in the post-therapy period over the past decade.
In 2003, Yamamoto et al. [17] evaluated the potential clinical usefulness of SPECT/CT with the image fusion technique using an external marker from separately obtained SPECT and CT images in DTC patients during the post-therapy period. Subsequently, a series of studies was carried out using the integrated SPECT/CT system to evaluate the incremental value of SPECT/CT over planar WBS when the planar images were inconclusive [18–21]. They consistently reported that SPECT/CT images had an additional value over planar images in increasing diagnostic accuracy, reducing pitfalls, and modifying patient management by precise localization and improved characterization of equivocal foci of tracer uptake on planar images. Their studies were limited, however, to selected patients with inconclusive planar images.
Schmidt et al. [22] investigated the impact of SPECT/CT on nodal staging via a routine procedure in the first radioablation period and found that SPECT/CT altered the nodal stage in 35% of patients and changed risk stratification in 25% of patients compared with planar WBS. Wang et al. [23] reported that SPECT/CT detected new metastases that had been missed by planar WBS in 7% of the patients. The results of both studies showed the promising role of SPECT/CT as routine clinical practice in the post-therapy period. In addition, Mustafa et al. [24] reported that lymph node metastases occurred in approximately one quarter of patients with T1 papillary thyroid carcinoma (PTC) and in the microcarcinoma subgroup as well. They indicated that SPECT/CT provided more accurate nodal staging than planar imaging and suggested post-ablation SPECT/CT as a routine imaging modality in the management of T1 PTC patients who did not undergo lymph node dissection.
Therefore, is it necessary to obtain SPECT/CT imaging for every patient with DTC in the post-therapy period? Grewal et al. [25] performed SPECT/CT as a routine procedure in 148 consecutive patients and indicated that SPECT/CT reduced the need for additional cross-sectional imaging in 20% of the patients by lowering the equivocal findings on planar images and altered risk stratification in 6.4% of the patients. They recommended that SPECT/CT should be considered in intermediate and high-risk patients in the post-therapy period. Similarly, Maruoka et al. [26] reported that SPECT/CT improved the detection and localization in lymph node metastases and distant metastases, compared with planar WBS, and recommended that SPECT/CT should be performed routinely in high-risk DTC patients in the post-therapy period. Jeong et al. [27] demonstrated that SPECT/CT detected additional occult findings in 8.6% of the patients and that the serum Tg levels at the time of ablation were significantly higher in patients with additional SPECT/CT findings than in those without additional findings. They suggested that post-therapy SPECT/CT should be performed in patients with high serum Tg levels for the detection of hidden metastases even in patients with only bed uptake on planar WBS.
The prognostic values of SPECT/CT were also investigated in follow-up studies after RAI ablation. Aide et al. [28] conducted a prospective study to evaluate both the diagnostic and prognostic values of SPECT/CT after RAI ablation. They reported that SPECT/CT had a complementary role to WBS in diagnosis, and a positive finding on SPECT/CT was more predictive of treatment failure than a positive finding on WBS with a median follow-up of 21 months. In a retrospective study of 81 DTC patients, SPECT/CT at the first radioablation had a high negative predictive value for RAI-avid nodal metastases 5 months after initial therapy [29]. Ciappuccini et al. [30] further confirmed the prognostic value of post-ablation WBS with neck and thorax SPECT/CT by a prospective study of 170 consecutive patients. According to the univariate and multivariate analyses, both a positive finding on WBS with SPECT/CT and a high stimulated serum Tg level at the time of ablation were considered important factors related to an increased risk of persistent or recurrent disease.
In summary, it is evident that post-therapy SPECT/CT can offer plenty of valuable information on staging, risk stratification, patient management, and prognosis (Figs. 1 and 2), although there are no published data on the cost-effectiveness of RAI SPECT/CT in patients with DTC. Post-ablation SPECT/CT should be considered routinely, at least in patients who are at intermediate to high risk, or in those who had high serum Tg levels, or in those who did not undergo lymph node dissection, even in patients with small primary PTC, and in those with an inconclusive planar scan as well.
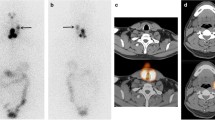
A 55-year-old female patient underwent total thyroidectomy with central neck dissection and was diagnosed as having papillary thyroid cancer (pT3N1aM0, stage III). a Post-therapy radioactive iodine (3.7 GBq of I-131) whole-body scan showed several foci of tracer uptake in the neck. b–d SPECT/CT images clearly demonstrated multilevel metastatic lymph nodes (level II–IV) in the right neck, which altered nodal stage from N1a to N1b. e–g CT component from SPECT/CT also showed small lymph nodes with slight high attenuation in the corresponding right neck (arrows). Eight months later, however, her diagnostic radioactive iodine whole-body scan and neck ultrasonography showed negative findings and stimulated serum thyroglobulin was less than 1 ng/ml. Post-therapy SPECT/CT findings altered nodal stage, but did not lead to changes in patient management
A 46-year-old female patient underwent rib excision for an incidentally found rib mass and was diagnosed as having metastatic thyroid cancer. Subsequently, she underwent total thyroidectomy with central neck dissection, and the pathologic result was intrathyroidal microscopic papillary carcinoma with one central neck node metastasis (pT1N1aM1, stage IV). a Post-therapy radioactive iodine (5.55 GBq of I-131) whole-body scan showed three unexpected foci of tracer uptake in the left shoulder and abdomen. b–d SPECT/CT images clearly demonstrated bone metastasis of the left scapula and two metastatic nodules in the omentum and retroperitoneum. e–g Retrospective review of preoperative F-18 FDG PET/CT showed two small high-attenuating nodules without significant FDG uptake in the corresponding abdominal areas (arrows), but no abnormal FDG uptake in the left scapula. Laparoscopic excision for the omental and retroperitoneal mass was done 1 month later, and metastatic thyroid cancer with cystic change was confirmed at pathologic findings. Her stimulated serum thyroglobulin gradually decreased from 270.2 ng/ml at the pre-ablation period to 26.1 ng/ml. Post-therapy SPECT/CT findings did not change the TNM staging, but contributed to changes in patient management by detection of unexpected metastatic lesions
Long-term Follow-up Period
Like in the post-therapy period, SPECT/CT is preferred over planar imaging for better localization and characterization of RAI uptake on planar imaging when diagnostic RAI WBS is performed in the long-term follow-up period, although a decreasing trend is observed in the primary indications of diagnostic RAI imaging after remnant ablation or adjuvant therapy, particularly in low-risk patients with an undetectable serum Tg level and negative neck ultrasonography [3].
In a study of patient populations at mixed periods, SPECT/CT showed incremental diagnostic value compared to planar imaging in 65% of the patients and changed the therapeutic approach in 41% of patients who underwent diagnostic WBS at the follow-up period after the initial therapy [19]. In a prospective study in consecutive patients, Spanu et al. [31] reported that SPECT/CT had an incremental value over planar imaging, modified therapeutic management, and avoided unnecessary treatment in a significant portion of the patients by precise localization and characterization of the thyroid tissue residue and RAI-avid metastases. They also identified additional occult foci of RAI uptake on SPECT/CT even in patients whose planar images were negative. They recommended the broad use of SPECT/CT in the follow-up of thyroidectomized DTC patients, particularly when planar WBS was not conclusive.
For diagnostic RAI imaging in the long-term follow-up period, the use of I-123 is recommended over I-131 because of its lower radiation doses and better quality images, but it has relatively limited availability. Barwick et al. [32] assessed the diagnostic performance of SPECT/CT using I-123 in 79 consecutive patients. Similar to I-131 SPECT/CT, I-123 SPECT/CT combined with WBS had significantly better diagnostic accuracy compared with just WBS and WBS plus SPECT alone.
The clinical usefulness of SPECT/CT in the follow-up period was further supported by other studies evaluating patient populations at mixed periods (post-therapy and follow-up period). Blum et al. [33] showed the superiority of SPECT/CT for elucidating cryptic findings on planar WBS from the head and neck to the pelvis, which could reduce unnecessary subsequent RAI therapy. There was no significant difference in successful interpretation between tracer dose images at the follow-up period and post-therapy images using a higher RAI dose. Menges et al. [34] showed that SPECT/CT had significantly higher specificity than planar imaging, but the sensitivities of SPECT/CT and planar imaging did not differ. They concluded that SPECT/CT provided superior diagnostic performance than planar WBS, but no incremental diagnostic value was observed in patients with negative planar imaging, unlike the findings of a previous report [31]. Thus, the issue of which patients will benefit from additional SPECT/CT over planar WBS in the long-term follow-up period should be further clarified.
Conclusion
SPECT/CT images provide additional information compared to conventional planar WBS by accurate localization, characterization, and differentiation of benign physiologic activity from metastatic disease, which can lead to a change in staging, risk stratification, and patient management and can give prognostic information concerning patients with DTC. Each SPECT/CT image acquired at different time points during the management of DTC may have a different clinical meaning and significance. Pre-ablation SPECT/CT images combined with serum Tg levels can contribute to the re-assessment of disease status after thyroidectomy for the decision-making of RAI therapy, although there is still no specific consensus on the diagnostic strategy and the optimal cutoff value for postoperative serum Tg levels. Post-therapy SPECT/CT images can clearly provide abundant, useful information on staging, risk stratification, patient management, and prognosis, which has been proved by many clinical studies over the last decade. SPECT/CT images at the long-term follow-up period using I-123 as well as a tracer dose of I-131 can also result in an incremental diagnostic value over planar WBS, modification of therapeutic management, and prevention of unnecessary treatment. Future studies are needed to confirm the relevance of the proper clinical indications and to analyze the cost-effectiveness issue of SPECT/CT acquired at different time points during the management of DTC.
References
Patton JA, Townsend DW, Hutton BF. Hybrid imaging technology: from dreams and vision to clinical devices. Semin Nucl Med. 2009;39:247–63.
Mariani G, Bruselli L, Kuwert T, Kim EE, Flotats A, Israel O, et al. A review on the clinical uses of SPECT/CT. Eur J Nucl Med Mol Imaging. 2010;37:1959–85.
Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, et al. 2015 American Thyroid Association Management Guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American Thyroid Association Guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid. 2016;26:1–133.
Xue YL, Qiu ZL, Song HJ, Luo QY. Value of (1)(3)(1)I SPECT/CT for the evaluation of differentiated thyroid cancer: a systematic review of the literature. Eur J Nucl Med Mol Imaging. 2013;40:768–78.
Avram AM. Radioiodine scintigraphy with SPECT/CT: an important diagnostic tool for thyroid cancer staging and risk stratification. J Nucl Med. 2012;53:754–64.
McDougall IR, Iagaru A. Thyroid stunning: fact or fiction? Semin Nucl Med. 2011;41:105–12.
Pacini F, Schlumberger M, Dralle H, Elisei R, Smit JW, Wiersinga W, et al. European consensus for the management of patients with differentiated thyroid carcinoma of the follicular epithelium. Eur J Endocrinol. 2006;154:787–803.
Schlumberger MJ, Pacini F. The low utility of pretherapy scans in thyroid cancer patients. Thyroid. 2009;19:815–6.
Van Nostrand D, Aiken M, Atkins F, Moreau S, Garcia C, Acio E, et al. The utility of radioiodine scans prior to iodine 131 ablation in patients with well-differentiated thyroid cancer. Thyroid. 2009;19:849–55.
Chen MK, Yasrebi M, Samii J, Staib LH, Doddamane I, Cheng DW. The utility of I-123 pretherapy scan in I-131 radioiodine therapy for thyroid cancer. Thyroid. 2012;22:304–9.
Yap BK, Murby B. No adverse affect in clinical outcome using low preablation diagnostic (131)i activity in differentiated thyroid cancer: refuting thyroid-stunning effect. J Clin Endocrinol Metab. 2014;99:2433–40.
Wong KK, Sisson JC, Koral KF, Frey KA, Avram AM. Staging of differentiated thyroid carcinoma using diagnostic 131I SPECT/CT. AJR Am J Roentgenol. 2010;195:730–6.
Avram AM, Fig LM, Frey KA, Gross MD, Wong KK. Preablation 131-I scans with SPECT/CT in postoperative thyroid cancer patients: what is the impact on staging? J Clin Endocrinol Metab. 2013;98:1163–71.
Avram AM, Esfandiari NH, Wong KK. Preablation 131-I scans with SPECT/CT contribute to thyroid cancer risk stratification and 131-I therapy planning. J Clin Endocrinol Metab. 2015;100:1895–902.
Prideaux AR, Song H, Hobbs RF, He B, Frey EC, Ladenson PW, et al. Three-dimensional radiobiologic dosimetry: application of radiobiologic modeling to patient-specific 3-dimensional imaging-based internal dosimetry. J Nucl Med. 2007;48:1008–16.
Sisson JC, Dewaraja YK, Wizauer EJ, Giordano TJ, Avram AM. Thyroid carcinoma metastasis to skull with infringement of brain: treatment with radioiodine. Thyroid. 2009;19:297–303.
Yamamoto Y, Nishiyama Y, Monden T, Matsumura Y, Satoh K, Ohkawa M. Clinical usefulness of fusion of 131I SPECT and CT images in patients with differentiated thyroid carcinoma. J Nucl Med. 2003;44:1905–10.
Ruf J, Lehmkuhl L, Bertram H, Sandrock D, Amthauer H, Humplik B, et al. Impact of SPECT and integrated low-dose CT after radioiodine therapy on the management of patients with thyroid carcinoma. Nucl Med Commun. 2004;25:1177–82.
Tharp K, Israel O, Hausmann J, Bettman L, Martin WH, Daitzchman M, et al. Impact of 131I-SPECT/CT images obtained with an integrated system in the follow-up of patients with thyroid carcinoma. Eur J Nucl Med Mol Imaging. 2004;31:1435–42.
Chen L, Luo Q, Shen Y, Yu Y, Yuan Z, Lu H, et al. Incremental value of 131I SPECT/CT in the management of patients with differentiated thyroid carcinoma. J Nucl Med. 2008;49:1952–7.
Kohlfuerst S, Igerc I, Lobnig M, Gallowitsch HJ, Gomez-Segovia I, Matschnig S, et al. Posttherapeutic (131)I SPECT-CT offers high diagnostic accuracy when the findings on conventional planar imaging are inconclusive and allows a tailored patient treatment regimen. Eur J Nucl Med Mol Imaging. 2009;36:886–93.
Schmidt D, Szikszai A, Linke R, Bautz W, Kuwert T. Impact of 131I SPECT/spiral CT on nodal staging of differentiated thyroid carcinoma at the first radioablation. J Nucl Med. 2009;50:18–23.
Wang H, Fu HL, Li JN, Zou RJ, Gu ZH, Wu JC. The role of single-photon emission computed tomography/computed tomography for precise localization of metastases in patients with differentiated thyroid cancer. Clin Imaging. 2009;33:49–54.
Mustafa M, Kuwert T, Weber K, Knesewitsch P, Negele T, Haug A, et al. Regional lymph node involvement in T1 papillary thyroid carcinoma: a bicentric prospective SPECT/CT study. Eur J Nucl Med Mol Imaging. 2010;37:1462–6.
Grewal RK, Tuttle RM, Fox J, Borkar S, Chou JF, Gonen M, et al. The effect of posttherapy 131I SPECT/CT on risk classification and management of patients with differentiated thyroid cancer. J Nucl Med. 2010;51:1361–7.
Maruoka Y, Abe K, Baba S, Isoda T, Sawamoto H, Tanabe Y, et al. Incremental diagnostic value of SPECT/CT with 131I scintigraphy after radioiodine therapy in patients with well-differentiated thyroid carcinoma. Radiology. 2012;265:902–9.
Jeong SY, Lee SW, Kim HW, Song BI, Ahn BC, Lee J. Clinical applications of SPECT/CT after first I-131 ablation in patients with differentiated thyroid cancer. Clin Endocrinol (Oxf). 2014;81:445–51.
Aide N, Heutte N, Rame JP, Rousseau E, Loiseau C, Henry-Amar M, et al. Clinical relevance of single-photon emission computed tomography/computed tomography of the neck and thorax in postablation (131)I scintigraphy for thyroid cancer. J Clin Endocrinol Metab. 2009;94:2075–84.
Schmidt D, Linke R, Uder M, Kuwert T. Five months’ follow-up of patients with and without iodine-positive lymph node metastases of thyroid carcinoma as disclosed by (131)I-SPECT/CT at the first radioablation. Eur J Nucl Med Mol Imaging. 2010;37:699–705.
Ciappuccini R, Heutte N, Trzepla G, Rame JP, Vaur D, Aide N, et al. Postablation (131)I scintigraphy with neck and thorax SPECT-CT and stimulated serum thyroglobulin level predict the outcome of patients with differentiated thyroid cancer. Eur J Endocrinol. 2011;164:961–9.
Spanu A, Solinas ME, Chessa F, Sanna D, Nuvoli S, Madeddu G. 131I SPECT/CT in the follow-up of differentiated thyroid carcinoma: incremental value versus planar imaging. J Nucl Med. 2009;50:184–90.
Barwick T, Murray I, Megadmi H, Drake WM, Plowman PN, Akker SA, et al. Single photon emission computed tomography (SPECT)/computed tomography using iodine-123 in patients with differentiated thyroid cancer: additional value over whole body planar imaging and SPECT. Eur J Endocrinol. 2010;162:1131–9.
Blum M, Tiu S, Chu M, Goel S, Friedman K. I-131 SPECT/CT elucidates cryptic findings on planar whole-body scans and can reduce needless therapy with I-131 in post-thyroidectomy thyroid cancer patients. Thyroid. 2011;21:1235–47.
Menges M, Uder M, Kuwert T, Schmidt D. 131I SPECT/CT in the follow-up of patients with differentiated thyroid carcinoma. Clin Nucl Med. 2012;37:555–60.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Sang-Woo Lee declares that he has no conflict of interest.
Ethical Statement
No research interventions were done in this review (no ethical approval or informed consent needed). The manuscript has not been published before and is not under consideration for publication anywhere else.
Rights and permissions
About this article
Cite this article
Lee, SW. SPECT/CT in the Treatment of Differentiated Thyroid Cancer. Nucl Med Mol Imaging 51, 297–303 (2017). https://doi.org/10.1007/s13139-017-0473-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13139-017-0473-x