Abstract
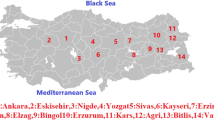
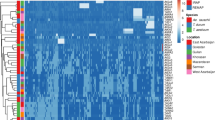
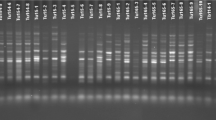
Analysis of genetic diversity and population structure in bread wheat is an essential step in their conservation, utilization, and breeding. Retrotransposons are ubiquitous and abundant a throughout the plant genomes, therefore extensively used as ideal molecular markers for genetic variability, DNA fingerprinting and genetic mapping studies in plant species. In the current research, we used two retrotransposon-based marker systems, inter-retrotransposon amplified polymorphisms (IRAPs), and the retrotransposon-microsatellite amplified polymorphisms (REMAPs) markers to evaluate the genetic diversity and survey activity of long terminal repeat retrotransposon (LTR-retrotransposon) elements in a collection of 49 bread wheat (Triticum aestivum L.) cultivars that mainly bred in Iran. In general, 90 and 126 loci were amplified using 9 IRAP and 20 REMAP primers, respectively. Both techniques produced a satisfactory number of bands for cultivar analysis; however, the technique IRAP, particularly single primer Nikita generated a large number of bands, indicating the wide activity of Nikita family under various environmental conditions of bread wheat. The percentage of polymorphic loci (PPL) in the studied collection for IRAP and REMAP markers was 81.78 and 86.40%, respectively. A model-based Bayesian method, Principal coordinate analysis (PCoA) and cluster analysis using Minimum Evolution (ME) algorithm hinted of the existence of two groups. This grouping was in agreement with the growing season and conformed by the high within-group bootstrap value. These results demonstrated that these markers developed using transpositionally active retrotransposons (RTNs) are efficient and reliable markers in determining level of genetic diversity and population structure in bread wheat in breeding programs.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Abbasi Holasou H, Abdollahi Mandoulakani B, Jafari M, Bernousi I. 2016. Use of IRAP and REMAP markers to interpret the population structure of Linum usitatissimum from Iran. Biolojia https://doi.org/10.1515/biolog-2016-0042
Abdollahi Mandoulakani B, Nasri Sh, Dashchi S, Arzhang S, Bernousi I, Abbasi Holasou H. 2017. Preliminary evidence for associations between molecular markers and quantitative traits in a set of bread wheat (Triticum aestivum L.) cultivars and breeding lines. CR Biol. 340: 307–313
Abdollahi Mandoulakani B, Yaniv E, Kalendar R, Raats D, Bariana HS, Bihamta MR, Schulman AH. 2014. Development of IRAP- and REMAP-derived SCAR markers for markerassisted selection of the stripe rust resistance gene Yr15 derived from wild emmer wheat. Theor. Appl. Genet. 128: 211–219
Abouzied HM, Eldemery SMM, Abdellatif KF. 2013. SSR-based genetic diversity assessement in tetraploid and hexaploid wheat populations. Brit. Biotechnol. J. 3: 390–404
Aremu CO, Adebayo MA, Ariyo OJ, Adewale BB. 2007. Classification of genetic diversity and choice of parents for hybridization in cowpea vigna unguiculata (L.) walp for humid savanna ecology. Afr. J. Biotechnol. 6: 2333–2339
Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K, Albright LM, Coen DM, Varki A. 1995. Current Protocols in Molecular Biology. John Wiley, New York
Bento M, Pereira HS, Rocheta M, Gustafson P, Viegas W, Silva M. 2008. Polyploidization as a retraction force in plant genome evolution: sequence rearrangements in Triticale. PLoSONE 3, e1402. https://doi.org/10.1371/journal.pone.0001402
Carvalho A, Guedes-Pinto H, Martins-Lopes P, Lima-Brito J. 2010. Genetic variability of old Portuguese bread wheat cultivars assayed by IRAP and REMAP markers. Ann. Appl. Biol. 3: 337–345
Ciaffi M, Dominici L, Lafiandra D, Porceddu E. 1992. Seed storage proteins of wild wheat progenitors and their relationships with technological properties. Hereditas 116: 315–322
Donini P, Law JR, Koebner RMD, Reeves JC, Cooke RJ. 2000. Temporal trends in the diversity of UK wheat. Theor. Appl. Genet. 100: 912–917
Earl DA, Von Holdt BM. 2012. STRUCTURE HARVESTER: a website and program for visualizing structure output and implementing the Evanno method. Conserv. Genet. Resour. 4: 359–361
Evanno G, Regnaut S, Goudet J. 2005. Detecting the number of clusters of individuals using the software STRUCTURE: A simulation study. Mol. Ecol. 14: 2611–2620
Galindo-Gonzalez L, Mhiri C, Deyholos MK, Grandbastien MA. 2017. LTR-retrotransposons in plants: Engines of evolution, Gene 626: 14–25
Grandbastien MA, Audeon C, Bonnivard E, Casacuberta JM, Chalhoub B, Costa AP, Le QH, Melayah D, Petit M, Poncet C, Tam SM, Van Sluys MA, Mhiri C. 2005. Stress activation and genomic impact of Tnt1 retrotransposons in Solanaceae. Cytogenet. Genome Res. 110: 229–241
Gupta PK, Rustgi S, Kulwal PL. 2005. Linkage disequilibrium and association studies in higher plants: present status and future prospects, Plant Mol. Biol. 57: 461–485
Jaaskelainen M, Chang W, Moisy C, Schulman AH. 2013. Retrotransposon BARE displays strong tissue-specific differences in expression. New Phytol. 200: 1000–1008
Kabbaj H, Sall AT, Al-Abdallat A, Geleta M, Amri A, Filali-Maltouf A, Belkadi B, Ortiz R, diversity within a global panel of Durum Wheat (Triticum durum) landraces and modern germpalsm reveals the history of alleles exchange. Front. Plant Sci. 8: 1277
Kalendar R, Flavell AJ, Ellis THN, Sjakste T, Moisy C, Schulman AH. 2011. Analysis of plant diversity with retrotransposonbased molecular markers. Heredity 106: 520–530
Kalendar R, Grab T, Regina M, Souniemi A, Schulman AH. 1999. IRAP and REMAP: two new retrotransposon-based DNA fingerprinting techniques. Theor. Appl. Genet. 98: 704–711
Kalendar R, Tanskanen J, Immonen S, Nevo E, Schulman AH. 2000. Genome evolution of wild barley (Hordeum spontaneum) by BARE-1 retrotransposon dynamics in response to sharp microclimatic divergence. Proc. Natl. Acad. Sci. 97: 6603–6607
Kumar J, Agrawal V. 2017. Analysis of genetic diversity and population genetic structure in Simarouba glauca DC. (an important bio-energy crop) employing ISSR and SRAP markers. Ind. Crops Prod. 100: 198–207
Laurentin H. 2009. Data analysis for molecular characterization of plant genetic resources. Genet. Resour. Crop Evol. 56: 277–292
Liu L, Chen W, Zheng X, Li J, Yan DT, Liu L, Liu X, Wang YL. 2016. Genetic diversity of Ulmus lamellosa by morphological traits and sequence-related amplified polymorphism (SRAP) markers. Biochem. Syst. Ecol. 66: 272–280
Lou Q, Chen J. 2007. Ty1-copia retrotransposon-based SSAP marker development and its potential in the genetic study of cucurbits. Genome 50: 802–810
Manifesto MM, Schlatter AR, Hopp HE, Suarez EY, Dubcovky J. 2001. Quantitative evaluation of genetic diversity germplasm using molecular markers. Crop Sci. 41: 682–690
Mohammadi SA, Khodarahmi M, Jamalirad S, Jalal Kamali MR. 2009. Genetic diversity in a collection of old and new bread wheat cultivars from Iran as revealed by simple sequence repeat-based analysis. Ann. Appl. Biol. 154: 67–76
Nasri Sh, Abdollahi Mandoulakani B, Darvishzadeh R, Bernoosi I. 2013. Retrotransposon insertional polymorphism in Iranian bread wheat cultivars and breeding lines revealed by IRAP and REMAP markers. Biochem. Genet. 51: 927–943
Nozawa K, Kawagishi Y, Kawabe A, Sato M, Masuta Y, Kato A, Ito H. 2017. Epigenetic regulation of a heat-activated retrotransposon in cruciferous vegetables. Epigenomes 1: 7
Peakall R, Smouse PE. 2006. GenAlEx 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol. Ecol. Notes. 6: 288–295
Pritchard JK, Stephens M, Donnelly P. 2000. Inference of population structure using multilocus genotype data. Genetics 155: 945–959
Queen RA, Gribbon BM, James C, Jack P, Falvell AJ. 2004. Retrotransposon-based molecular markers for linkage and genetic diversity analysis in wheat. Mol. Genet. Genomics 271: 91–97
Rao NK. 2004. Plant genetic resources: Advancing conservation and use through biotechnology. Afr. J. Biotechnol. 3: 136–145
Rohlf FJ. 2000. NTSYS-pc: Numerical Taxonomy and Multivariate Analysis System, Version 2.1. Exeter Software, New York
Roussel V, Koenig J, Beckert M, Balfourier F. 2004. Molecular diversity in French bread wheat accessions related to temporal trends and breeding programs. Theor. Appl. Genet. 108: 920–930
Roussel V, Leisova L, Exbrayat F, Stehno Z, Balfourier F. 2005. SSR allelic diversity changes in 480 European bread wheat varieties released from 1840 to 2000. Theor. Appl. Genet. 111: 162–170
Saeidi H, Rahiminejad MR, Heslop-Harrison JS. 2008. Retroelement insertional polymorphisms, diversity and phylogeography within diploid, D-genome Aegilops tauschii (Triticeae, Poaceae) sub taxa in Iran. Ann. Bot. 101: 855–861
Salamini F, Ozkan H, Brandolini A, Schafer-Pregl R, Martin W. 2002. Genetics and geography of wild cereal domestication in the near east. Nat. Rev. Genet. 3: 429–441
Schulman A, Flavell A, Paux E, Ellis THN. 2012. The application of LTR retrotransposons as molecular markers in plants. Meth. Mol. Biol. 859: 115–153
Shimamura M, Yasue H, Ohshima K, Abe H, Kato H, Kishiro T, Goto M, Munechika I, Okada N. 1997. Molecular evidence from retroposons that whales form a clade within even-toed ungulates. Nature 388: 666–670
Tabrizivad Taheri M, Alavi-Kia SS, Mohammadi SA, Moghaddam Vahed M. 2018. Assessment of genetic diversity and relationships among Triticum urartu and Triticum boeoticum populations from Iran using IRAP and REMAP markers. Genet. Resour. Crop Evol. https://doi.org/10.1007/s10722-018-0660-3
Tamura K, Dudley J, Nei M, Kumar S. 2007. MEGA4: Molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol. Biol. Evol. 24: 1596–1599
Uddin MS, Boerner A. 2008. Genetic diversity in hexaploid and tetraploid wheat genotypes using microsatellite markers. Plant Tiss. Cult. Biotech. 18: 65–73
Vicient CM, Kalendar R, Anamthawat-Jonnson K, Suoniemi A, Schulman AH. 1999. Structure, functionality, and evolution of the BARE-1 retrotransposon of barley. Genetica 107: 53–63
Vuorinen AL, Kalendar R, Fahima T, Korpelainen H, Nevo E, Schulman AH. 2018. Retrotransposon-based genetic diversity assessment in wild emmer wheat (Triticum turgidum ssp. Dicoccoides). Agronomy https://doi.org/10.3390/agronomy8070107
Wang J, Luo MC, Chen Z, You FM, Wei Y, Zheng Y, Dvorak J. 2013. Aegilops tauschii single nucleotide polymorphisms shed light on the origins of wheat D-genome genetic diversity and pinpoint the geographic origin of hexaploid wheat. New Phytol. 198: 925–937
Zarei Abbasabbad E, Mohammadi SA, Moghadam M, Jalal Kamali MR. 2016. Analysis of genetic diversity, population structure and linkage disequilibrium in Iranian wheat landraces using SSR markers. Plant Genet. Resour. 15: 327–334
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abbasi Holasou, H., Rahmati, F., Rahmani, F. et al. Elucidate Genetic Diversity and Population Structure of Bread Wheat (Triticum Aestivum L.) Cultivars Using IRAP and REMAP Markers. J. Crop Sci. Biotechnol. 22, 139–151 (2019). https://doi.org/10.1007/s12892-018-0211-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12892-018-0211-0